Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sebastian Ciężki | -- | 10467 | 2024-02-27 12:21:56 | | | |
| 2 | Jason Zhu | Meta information modification | 10467 | 2024-02-28 07:13:07 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Ciężki, S.; Kurpiewska, E.; Bossowski, A.; Głowińska-Olszewska, B. Complications of Obesity in Children. Encyclopedia. Available online: https://encyclopedia.pub/entry/55540 (accessed on 29 March 2026).
Ciężki S, Kurpiewska E, Bossowski A, Głowińska-Olszewska B. Complications of Obesity in Children. Encyclopedia. Available at: https://encyclopedia.pub/entry/55540. Accessed March 29, 2026.
Ciężki, Sebastian, Emilia Kurpiewska, Artur Bossowski, Barbara Głowińska-Olszewska. "Complications of Obesity in Children" Encyclopedia, https://encyclopedia.pub/entry/55540 (accessed March 29, 2026).
Ciężki, S., Kurpiewska, E., Bossowski, A., & Głowińska-Olszewska, B. (2024, February 27). Complications of Obesity in Children. In Encyclopedia. https://encyclopedia.pub/entry/55540
Ciężki, Sebastian, et al. "Complications of Obesity in Children." Encyclopedia. Web. 27 February, 2024.
Copy Citation
The increasing incidence of obesity in the pediatric population requires attention to its serious complications. It turns out that in addition to typical, well-known metabolic complications, obesity as a systemic disease carries the risk of equally serious, although less obvious, non-metabolic complications, such as cardiovascular diseases, polycystic ovary syndrome, chronic kidney disease, asthma, thyroid dysfunction, immunologic and dermatologic conditions, and mental health problems. They can affect almost all systems of the young body and also leave their mark in adulthood. In addition, obesity also contributes to the exacerbation of existing childhood diseases.
childhood obesity
obesity complications
non-metabolic complications
1. Introduction
Over the last few decades, obesity has become a huge challenge for the healthcare system on a global scale [1][2]. The latest WHO report on data collected in 2018–2020 in European countries reported that approximately 29% of children aged 7–9 were overweight or obese, while another showed that this problem affected more than 17% of children worldwide [3].
Obesity and overweight in childhood may result in problems with maintaining a normal body weight in adulthood. It is a complex, multifactorial disease characterized by abnormal or excessive accumulation of fat tissue, which is associated with an increased risk of many serious diseases as well as its premature onset if obesity occurs from an early age. Numerous complications of obesity, previously most often described in relation to adults, such as cardiovascular diseases (CVD), left ventricular hypertrophy, or hypertension, are increasingly observed in children and adolescents. The incidence of these complications increases with the prevalence of obesity itself. Problems affect both physical and mental wellness [4]. Numerous studies have shown the metabolic complications of obesity, which have been widely discussed in previous works, such as insulin resistance, hyperglycemia, dyslipidemia, hypertension, and type 2 diabetes, negatively affect physical health and are even associated with higher overall mortality in the population [5][6][7][8].
Recently, the concept of distinguishing between metabolically healthy obesity (MHO) and metabolically unhealthy obesity (MUO) has been developed. MHO is characterized by the absence of cardiom etabolic disorders, including insulin resistance, impaired glucose tolerance, dyslipidemia, and hypertension, despite excessive accumulation of adipose tissue. However, this is most likely temporary and the risk of developing cardiometabolic diseases is still higher than in healthy slim people [9].
2. Metabolic Complications of Obesity in Children
2.1. Insulin Resistance
By definition, insulin resistance (IR) is a state of reduced response to insulin of target tissues with normal or elevated serum insulin levels and is the reciprocal of insulin sensitivity [7]. It is one of the earliest and most serious changes in hormonal balance in obesity, regardless of the age at which overweight occurs, and its prevalence in the pediatric population is estimated at 11.5% (according to some studies, even up to 44%) [10]. The consequence of IR is chronic, compensatory hyperinsulinemia allowing for maintenance of the required biological effect in the form of obtaining proper glucose levels while reducing the suppression of glucose production in the liver [11]. When insulin release is no longer able to compensate for IR, it leads to disorders of carbohydrate metabolism, such as type 2 diabetes mellitus (T2DM). Typical markers of the state of IR are increased levels of insulin, which is mainly responsible for the adverse effects of this phenomenon, glucose, and free fatty acids (FFA) in the bloodstream [12]. The occurrence of IR is primarily associated with visceral adipose tissue and, as a consequence of its excess, abdominal (visceral) obesity. Therefore, a greater degree of IR and metabolic disorders are also observed in children with a greater share of visceral fat in the child’s overall BMI [13]. Progression of decrease in tissue insulin sensitivity may lead not only to T2DM but also to dyslipidemia, metabolic syndrome (MetS), or non-alcoholic fatty liver disease (NAFLD).
2.2. Diabetes Mellitus
Until recently, type 2 diabetes was diagnosed almost exclusively in adults, but over the past few decades, the incidence of T2DM among children and adolescents worldwide has increased several fold [14], interestingly coinciding with a parallel increase in childhood obesity [1].
The primary pathology here is insulin resistance, impaired glucose tolerance, and hyperinsulinemia [15]. The first detected biochemical abnormalities indicate the development of so-called prediabetes: abnormal fasting glucose or impaired glucose tolerance in the oral glucose tolerance test (OGTT). Sinha et al. found impaired glucose tolerance in 21% of adolescents aged 11–18 and 25% of children aged 4–10, with both groups affected by obesity [16]. Furthermore, children with obesity are four times more likely to develop T2DM compared to children with normal BMI values [17]. Compared to adults with T2DM, in whom secondary complications develop later in life despite long-term insulin resistance, clinical diabetes and its complications develop faster and more aggressively in children. In addition, children show an earlier need for insulin treatment [18].
Obesity is suggested to be associated with an increased incidence of type 1 diabetes mellitus (T1DM) [19]. The “accelerator hypothesis” posits that obesity-induced insulin resistance in predisposed individuals accelerates the development of clinically overt T1DM [20]. Additionally, obesity is also believed to be connected with the evolution of an autoimmune response against multiple organs, together with pancreatic β-cells. Inflammation induced by obesity appears to increase the risk of developing clinical T1DM in the presence of only one type of autoantibodies—islet cell antibodies (ICA) [15]. The appearance of autoimmune response against pancreatic islet antigens is associated with lower insulin sensitivity and occurs more often in obese patients with impaired carbohydrate metabolism [21]. Moreover, long before T1DM, obesity is clinically evident and increases the switch to two autoantibodies in childhood and adolescence, although only in specific genetic groups [22].
2.3. Dyslipidemia
In the general pediatric population, the incidence of dyslipidemia is estimated at 8–20% [23], while data show that, in the population of children with obesity, abnormal lipid values range from 20% to over 40% [24][25]. The paper by Nielsen et al. showed that the risk of lipid disorders is 2.8 times higher in children with BMI > 90th percentile than in children with body weights within normal limits [26]. Obesity-linked dyslipidemia is primarily characterized by elevated plasma FFA and triglyceride levels, decreased high-density lipoprotein (HDL) levels, and abnormal low-density lipoprotein (LDL) composition [27]. Elevated FFA levels appear to play a significant role as a cause of insulin resistance. It has been suggested that increased levels of FFA and intracellular lipids inhibit the insulin signaling mechanism, leading to reduced glucose transport into muscle [10]. FFAs also play a mediating role between insulin resistance and β-cell dysfunction, indicating that reducing FFA levels may be a desirable therapeutic target [28].
Adipose tissue directly drives the development of many risk factors through its endocrine role of secreting inflammatory cytokines, proteins of the renin-angiotensin system, or metabolizing sex steroids and glucocorticoids [29]. It also influences reduced secretion of adiponectin—an adipose tissue-specific circulating protein known to be a homeostatic factor for regulating glucose levels and lipid metabolism as well as an insulin-sensitizing hormone affecting a wide range of tissues [6].
The body fat mass and body mass index have emerged to be the most important variables that determine circulating leptin concentrations. Obesity is associated with increased levels of leptin, which has only very limited effects due to tissue resistance to leptin associated with insulin resistance and abdominal obesity. In addition to the regulatory role of leptin in energy intake and expenditure, the independent involvement of leptin in maintaining insulin and glucose homeostasis has been documented. Leptin inhibits insulin secretion from the pancreas and mobilizes peripheral organs to increase glucose uptake. It follows that both hypothalamic leptin deficiency due to leptinopenia and reduced leptin transport across the blood–brain barrier due to obesity-related hyperleptinemia precede the pathophysiological consequences of diabetes types 1 and 2 [30].
2.4. Hypertension
Obesity is the most common risk factor for hypertension in today’s children. As many as 39% of those with severe obesity, 26% of children affected by obesity, and 20% of minors who are overweight suffer from hypertension, but its prevalence among children of normal body weight is only 5% [31]. Being overweight especially increases systolic blood pressure, and this relationship is even stronger in children than in adults. Systolic blood pressure in children is highly dependent on BMI, skinfold thickness, and waist circumference, and it increases with these variables [32]. Additionally, previously mentioned leptin, which is excessively secreted from adipose tissue, causes systemic vasoconstriction, sodium retention, and also affects the synthesis of nitric oxide and activation of the sympathetic nervous system, which leads to an increase in blood pressure [33].
2.5. Metabolic Syndrome
Metabolic syndrome has not yet been assigned a specific definition. Notwithstanding, it is generally considered as a coexistence of abdominal obesity, high insulin resistance, hyperglycemia, atherogenic dyslipidemia, and elevated blood pressure [34]. Reinehr et al. compared different definitions of MetS in a cohort of 1205 children and adolescents and found a wide range of prevalence ranging from 6 to 39%, depending on the proposed definition [35]. Therefore, there is a crucial need to establish a detailed definition and diagnostic criteria of MetS for the pediatric population in order to enable prevention, early detection, and treatment of this condition.
Even though MetS manifests itself more often in people with diabetes than in the general population, a noticeably higher prevalence of its criteria was identified in the group of obese and overweight patients in comparison to patients with a normal body weight range [36]. Many studies have identified a sedentary lifestyle and unbalanced dietary patterns resulting in excess body weight as the main pathological factors inducing risk factors belonging to the MetS cluster [34].
By far the most effective strategy for preventing the occurrence of MetS in the future is to proactively prevent obesity among children and adolescents. MetS significantly increases the risk of CVD and other mortality causes, like kidney failure, by 1.5–2-fold in the pediatric age group [34]. Identifying children at risk of developing MetS could enable pediatricians to quickly recognize and treat children and remains an important task that may contribute to reducing the incidence of cardiovascular disease and T2DM in later life [37]. This involves the need for multi-aspect medical intervention, both non-pharmacological and often pharmacological. Moreover, MetS is also associated with other disease processes associated with childhood obesity, including NAFLD [38] and renal dysfunction, with implications for chronic kidney disease [39].
2.6. NAFLD/MAFLD
Abdominal obesity (especially a high percentage of visceral fat accumulation and increased waist circumference), insulin resistance, and hypertension are typical features of children with NAFLD—the presence of hepatic steatosis [40]—which is considered as the most common liver disease during childhood [41]. The pathogenesis of pediatric NAFLD is not fully elucidated; however, several studies have reported elevation of BMI as an independent risk factor for fatty liver disease [42][43][44]. An excessive amount of energy is routinely stored in adipose tissue, causing adipocyte hyperplasia; unfortunately, if the buffering capacity of adipose tissue is impaired, ectopic fat aggregation may occur in various organs, along with the liver [45]. Results from a cohort of obese Taiwanese children showed that BMI z-score independently predicted both the occurrence and remission of NAFLD. Individuals with an increased BMI had a higher risk of developing NAFLD, and those with a lower BMI had a higher chance of NAFLD remission [43]. Moreover, a study conducted on obese Danish children showed that a 10% weight reduction was correlated with a reduction in liver steatosis (assessed by ultrasound) by almost 45–30% [46], while a different study showed that, after a year of weight loss, serum ALT levels were significantly reduced and the incidence of fatty liver disease decreased from 100% to 33% [47].
NAFLD can present in three stages of severity, namely steatosis, non-alcoholic steatohepatitis (NASH), where the accumulation of toxic fat in the steatosis stage is accompanied by inflammation, and finally cirrhosis, where liver damage begins to cause fibrosis leading to organ failure. NAFLD in children is associated with hepatic and extrahepatic morbidity and mortality. Children with NAFLD often have comorbidities such as CVD, obstructive sleep apnea, polycystic ovary syndrome (PCOS), and osteopenia [48][49]. Studies show that weight gain in childhood and adolescence is associated with an increased risk of not only NAFLD but also end-stage liver disease and hepatocellular carcinoma later in life [50][51].
A new terminology for NAFLD has recently been updated based on international consensus—metabolic (dysfunction) associated fatty liver disease (MAFLD) [52]. The global pervasiveness of MAFLD, estimated from existing fatty liver disease data, is 33.78% in the general pediatric population and 44.94% in obesity clinics [53]. The diagnosis of MAFLD is based on the accumulation of fat in the liver and one of the following three criteria: overweight/obesity, T2DM, or symptoms of metabolic dysregulation [54].
3. Non-Metabolic Complications of Obesity in Children
3.1. Cardiovascular Consequences
The important contributions of intra-abdominal adiposity (IAA) to dyslipidemia and insulin resistance provide an indirect, though clinically important, link to the genesis and progression of atherosclerosis and cardiovascular diseases [55]. Excess fat tissue is usually accompanied by elevated levels of high-sensitivity C-reactive protein (hsCRP), a marker of inflammation [56]. Inflammation is triggered by immune cells that attack dysfunctional fat tissue. As obesity progresses, inflammatory cells infiltrate the adipose tissue, pancreas, and other tissues. The obesity-related phase of atherosclerotic inflammation causes coronary artery calcification [57]. Additionally, obesity itself in young children is associated with endothelial cell dysfunction [58] and increased carotid intima media thickness (c-IMT) [59].
There is a strong link between childhood obesity and CVD in adulthood. However, normalization of weight status before puberty appears to eliminate this risk.
3.2. Cardiomyopathy
A recent prospective cohort study of 1.7 million subjects showed that an increased BMI in adolescence is associated with the development of cardiomyopathy in adulthood and an up to 8-fold increase in the risk of dilated cardiomyopathy if BMI ≥ 35 kg/m2 [60]. The severity and duration of obesity are considered important factors affecting changes in cardiac performance and morphology. Multiple mechanisms determine the progression of myocardial remodeling in people with obesity, including hemodynamic changes such as cardiac output and increased blood volume, leading to left ventricular hypertrophy and dilation [61]. Abnormal lipid metabolism, including excessive activity of adipocyte cell signaling molecules, as well as oxidative stress and inflammation, can potentially cause cardiac fibrosis and impaired systolic and diastolic function [62]. The manifestation of particular types of cardiomyopathy is related to body weight, although it is strongest in the case of dilated cardiomyopathy [60]. These findings are disturbing due to the high mortality rate of patients with this type of cardiomyopathy [63].
3.3. Endocrine and Gynecological Consequences
The growing incidence of obesity in the childhood population carries the risk of endocrine and gynecological consequences such as central precocious puberty [64], earlier attainment of pubertal milestones [65], menstrual disturbances [66], the occurrence of polycystic ovary syndrome [64], and alterations in thyroid hormonal status [67].
3.3.1. Precocious Puberty
Recently, an interesting link between overweight and the occurrence of central precocious puberty (CPP) has been noted, particularly in girls [68]. These findings appear to be supported by a Chinese population-based study by Liu et al., which found that the incidence of precocious puberty was higher among obese (48.00%) and overweight (27.94%) girls compared to their normal-weight peers (8.73%) [69]. In turn, among boys, precocious puberty prevalence was greater in the group of obese subjects (6.78%) than in the normal-weight group (2.86%) [69]. Interestingly, the overweight group of boys presented lower prevalence of precocious puberty than the normal-weight group (p > 0.05) [69]. Thus, obesity rather than overweight has been shown to be a risk factor for the occurrence of precocious puberty in boys [70]. In addition, a study conducted on a group of Chinese children by Chen et al. also demonstrated that obesity has an apparent impact on the occurrence of precocious puberty in both sexes [71]. What is more, reduction in BMI-SDS has been previously shown to be related to earlier gonadotropin-dependent onset of puberty in boys and delayed attainment of puberty in girls, which may imply that girls dealing with obesity experience earlier puberty and boys with obesity demonstrate delayed puberty [72]. It is important to remember that children with excessive weight are more likely to present isolated, mild manifestations of precocious puberty, such as precocious pubarche (the most common form), axillarche, and thelarche [66]. It appears that precocious pubarche and thelarche may result in a modest acceleration of skeletal growth and maturation [73][74]. The results of a Chinese study indicated that the bone age of girls aged 4–8 years with isolated premature thelarche may be substantially advanced [75]. Moreover, in the same group of subjects, BMI-SDS, serum IGF-1 SDS, and DHEAS SDS were identified as factors associated with an increased risk for advanced bone age [75]. The majority of sources in the literature assert that these mild forms of precocious puberty are self-limited [74] and usually do not need any treatment [66][76]. Nevertheless, follow-up is recommended [77] as they could potentially be a manifestation of central precocious puberty that may manifest in later years [74]. However, taking the above into account, further research into the treatment of mild forms of precocious puberty and their implications is needed.
There are several possible explanations for the cause of precocious puberty in children with obesity. Firstly, it may be a result of increased levels of leptin (a hormone secreted by adipocytes that is a permissive factor in the onset and progression of puberty) in both obese genders and kisspeptin (a peptide that is crucial in the initiation and development of puberty) in girls suffering from obesity [78]. Furthermore, in the increased amount of adipose tissue in the course of obesity occurs an intense aromatization of adrenal and ovarian androgens to estrogen, which is reflected in earlier adrenarche and thelarche in girls [79]. In addition, it was shown that obese prepubertal girls had significantly higher BMI-SDS, leptin, insulin, and HOMA-IR values than the control group, and that girls with obesity in early puberty presented higher BMI-SDS, leptin, IGF-1, IGFBP3, insulin, HOMA-IR, LH, LH/FSH ratio, ACTH, DHEA-S, androstenedione, testosterone, and free testosterone values than the control group [80]. Consequently, the hyperinsulinemia that occurs in girls experiencing obesity is considered to play a facilitating role in early puberty by leading to stimulation of adrenal and ovarian androgen production, decreased hepatic synthesis of sex hormone-binding globulin (SHBG), and increased aromatase activity in adipocytes [78][81][82]. It was observed that an excess of insulin is connected with the occurrence of precocious pubarche [66]. Furthermore, stimulation of adrenal androgen production by hyperinsulinemia may present in the form of precocious pubic and axillary hair appearance before the age of 8 years in girls and 9 in boys [66]. It may occur together with mild acne, pubertal sweat odor, and moderately accelerated growth and bone age [66].
Interestingly, previous studies have shown that earlier puberty may be a consequence of early adiposity rebound [83]. Throughout a child’s development, typically there is an initial rise in BMI during the first year of life, followed by its decline, reaching a nadir around the age of 6 [83]. Thereafter, an increase in BMI occurs again, which is commonly known as adiposity rebound (AR) [83]. In their study, German et al. demonstrated that, among children who experienced an earlier adiposity rebound, boys exhibited earlier pubarche and faster pubertal progression, while girls showed an earlier ages of menarche and thelarche compared to their peers who underwent AR later [84]. Furthermore, evidence suggests that early adiposity rebound is a contributing factor to the increased risk of polycystic ovary syndrome (PCOS) and elevated BMI in adulthood [85]. Very early (<42 months) and early (42–59 months) AR have also been shown to be associated with the deterioration of cardiac and metabolic parameters, including lipid parameters, total and central adiposity, blood pressure, and HOMA-IR, in children at the age of 10 years [86].
It turns out that excessive body weight not only leads to the emergence of precocious puberty in children but also to the earlier achievement of pubertal milestones. It was found that both girls and boys with higher BMI reached all of the pubertal milestones earlier, in a dose-dependent manner [65]. Moreover, it has been demonstrated that overweight and obesity contribute to earlier breast development and menarche in females, which can have detrimental effects on their mental health, social functioning, and physical health in adulthood, like increased risk of developing type 2 diabetes mellitus, cardiovascular diseases, shorter adult height, or post-menopausal breast cancer [68][78]. The results of the study by Li et al. are in line with these findings and clearly indicate that higher BMI before puberty is connected with earlier breast development and the appearance of menstruation in girls and with earlier first spermatorrhea (which indicates late-stage puberty in boys) and testicular development among boys [87]. Interestingly, studies are inconclusive regarding the impact of overweight and obesity on puberty in boys. Some findings show that overweight boys reach puberty faster, while boys with obesity experience a delay in pubertal development [88]. The results of the study by Lee et al. demonstrated that boys dealing with overweight experienced early puberty for Tanner scale GD2 or above compared to their normal-weight peers (9.3 years for overweight vs. 10.0 years for normal-weight boys) and Tanner GD5 or above (14.5 years for overweight vs. 15.2 years for normal-weight boys). Boys suffering from obesity were shown to have delayed puberty for GD5 and above compared to their overweight and normal-weight counterparts (15.4 years for obese vs. 14.5 years for overweight and 15.2 years for normal-weight boys) [88]. A potential explanation for such correlations may be that adipose tissue-derived aromatase activity can lead to excessive estrogen production in boys, and it is hypothesized that increased estrogen production inhibits puberty in boys with obesity, while no such effect occurs in those who are overweight [88]. Nevertheless, the influence of overweight and obesity on puberty timing in boys as well as the mechanisms that affect this process require further investigation.
It is worth adding that, among girls experiencing idiopathic precocious puberty, the pattern of constitutional advancement of growth is commonly observed [89]. It is associated with early puberty and marked by accelerated early-stage growth, peaking in the initial 2–4 years of life [90]. Intriguingly, this pattern is also correlated with an increased incidence of childhood obesity, and the precise causes of this condition are not yet known [90]. Furthermore, in a multicohort study conducted on American children, it was observed that girls who experienced an accelerated BMI gain in late infancy tended to reach menarche earlier, whereas those with rapid BMI growth during early childhood achieved pubic hair stage greater than 1 more quickly [91]. Therefore, the occurrence of accelerated growth during the first few years of life can help identify children who are likely to start puberty earlier [91].
Remarkably, insulin-like growth factor-1 (IGF-1) has been identified as a very prominent factor accelerating the onset of puberty through its inhibitory effect on dynorphin, a neuropeptide that suppresses the pre-pubertal secretion of GnRH [92]. Findings suggest that IGF-1 may also contribute to the occurrence of a pattern of constitutional advancement of growth and puberty (CAGP), as IGF-1 has been found to be increased in this condition [92]. This pattern is more typically seen in girls [92]. It appears that, in the course of obesity, there occurs an increase in free IGF-1 levels, predominantly through a decrease in IGF-1-binding protein levels caused by obesity-related hyperinsulinemia [93]. On the other hand, GH secretion in individuals with obesity is reduced [93]. Interestingly, greater levels of IGF-1 before puberty were demonstrated to be associated with an earlier onset of thelarche and menarche among girls [94]. Similarly, higher growth before puberty was linked to lower ages of thelarche and menarche [94]. In the case of boys, higher pre-pubertal IGF-1 levels were associated with earlier gonadarche and increased height [94]. A German study found that children with obesity showed higher growth at birth than their normal-weight counterparts and had an accelerated growth rate thereafter, which was associated with increased levels of IGF-1, insulin, and leptin in these children [95]. By contrast, during puberty, children suffering from obesity exhibited a decline in growth rate, which was correlated with decreases in IGF-1 and testosterone levels in boys and estradiol levels in girls [95]. The exact mechanism for the drop in IGF-1 levels during puberty remains unclear; however, it is hypothesized that low testosterone levels in boys with obesity may be a contributing factor, although this hypothesis does not seem to apply to girls [95]. In the same study, it was observed that, during the onset of puberty, boys struggling with obesity were 6.6 months older while girls affected by obesity were 9.1 months younger than normal-weight subjects [95]. Additionally, menarche occurred 8.9 months earlier in girls experiencing obesity compared to their normal-weight peers [95].
What is more, adolescents who are overweight or obese have been shown to be more likely to experience gynecological and obstetric complications, both in adolescence and adulthood [79]. An interesting link has been found between obesity and menstrual disorders, such as irregular and infrequent menstrual cycles or even amenorrhea, lack of ovulation, PCOS, and heavy menstrual bleeding in both adolescence and adulthood [78]. Recent studies indicate that having a higher BMI in childhood is related to menstrual irregularity, and that a greater childhood BMI and waist/height ratio (WHtR) is associated with the development of PCOS in adulthood among white but not black participants [96]. Moreover, pregnant adolescents with obesity are predisposed to the occurrence of perinatal complications such as preeclampsia, gestational hypertension, gestational diabetes, cesarean and instrumental delivery, and induction of labor [79]. For instance, the results of a recent study by Ambia et al. showed that young adolescents struggling with obesity (≤15 years old) were at higher risk of developing pre-eclampsia and delivering by cesarean section than those who were non-obese [97]. The prevalence of overweight and obesity in childhood and adolescence may also lead to macromastia and a greater likelihood of developing breast and endometrial cancer [79].
3.3.2. Polycystic Ovary Syndrome
As mentioned earlier, obesity in girls contributes to a greater likelihood of developing PCOS. It should be added that PCOS is the most frequent endocrinopathy in young adult females and the number one cause of non-ovulatory infertility [78]. The diagnosis of PCOS in adolescent girls can be made after meeting both criteria: (1) the occurrence of menstrual disturbances (irregular menstruation, oligomenorrhea, or secondary amenorrhea) and (2) the presence of clinical (in the form of hirsutism or moderate to severe acne) or biochemical hyperandrogenism [66][98]. It is challenging to diagnose PCOS in adolescent girls, as the key features of a PCOS diagnosis, like irregular menstrual cycles, acne, and polycystic ovarian morphology on pelvic ultrasound, coincide with the physiological changes seen during puberty [98]. Furthermore, it was demonstrated that PCOS coexists with a number of metabolic disorders, such as the occurrence of both insulin resistance and hyperinsulinemia [99]. Also, adolescents with PCOS are widely overweight and obese, and they more often develop metabolic syndrome, impaired glucose tolerance, and type 2 diabetes, so patients with PCOS should undergo screening for the presence of these conditions [99]. According to a meta-analysis describing the impact of obesity on metabolic disturbances in adolescent PCOS, adolescents with obesity and PCOS exhibited significantly reduced levels of SHBG and HDL-C and substantially elevated levels of triglycerides, leptin, fasting insulin, LDL-C, and free testosterone when compared to normal-weight adolescents with PCOS [100]. In addition, they showed significantly elevated fasting insulin, LDL-C, free testosterone, and 2 h OGTT levels in comparison to non-PCOS adolescents affected by obesity [100]. It has been demonstrated that the underlying cause of PCOS in adolescents with obesity actually lies in hyperinsulinemia, which, as mentioned earlier is able to promote androgen production in the ovaries and adrenal glands as well as decrease the hepatic synthesis of sex hormone-binding globulin (SHBG), thus leading to an excessive amount of androgens [66]. The interesting study by Jobira et al. indicated that there exists an alteration in the composition of the intestinal microbiota among adolescents experiencing PCOS and obesity, although further investigation is needed to understand the relationship between the microbiome and androgens, which has the potential to provide a new therapeutic pathway [62].
3.3.3. Thyroid Function in Childhood Obesity
Recently, there has been increased medical interest in thyroid function in obesity [67]. Individuals with obesity often exhibit alterations in thyroid function and thyroid structure, which is similar to that of Hashimoto thyroiditis, yet in the absence of disease [101]. It was previously demonstrated that TSH and total T4 levels were substantially elevated in children affected by obesity in comparison to their non-obese peers, and that subclinical hypothyroidism was significantly more prevalent in children with obesity (14%) than in healthy individuals (6.8%) [67]. These findings seem to be in line with the results of the study by Rumińska et al., which revealed that children and teenagers dealing with obesity presented higher TSH levels, even when they were within the normal range compared to normal-weight group, and the concentration of TSH was positively correlated with parameters characteristic of abdominal obesity [102]. However, a study conducted by Krause et al. found that obesity in the pediatric population is linked to increased TSH levels, decreased FT4 concentrations, and higher incidence of abnormally elevated TSH levels [103]. Also, moderate elevation of total T3 and FT3 levels (within the upper normal range or slightly above) is also reported as a hormonal impairment in childhood obesity [104]. Interestingly, a relevant association between the occurrence of hepatic steatosis and TSH levels in children and adolescents experiencing obesity has been also demonstrated [105]. Licenziati et al., in their study, showed that changes in thyroid function and structure in children with obesity tend to be reversed after weight loss [101].
Subclinical hypothyroidism, with a prevalence of up to 10–23% in the obese pediatric population, is a consequence rather than a causative factor of increased body weight [106]. While its mild form is relatively benign (if not connected to Hashimoto’s thyroiditis) in the pediatric population, it has been shown that isolated elevation of TSH levels may be related to an increased risk of developing or progressing cardiovascular disease and non-alcoholic fatty liver disease (NAFLD) in children and adolescents suffering from obesity [107]. Furthermore, it has been also demonstrated that overweight and obesity in children carry a risk of progression from subclinical to overt hypothyroidism [108]. In spite of this, there is limited information regarding the treatment of thyroid disorders co-occurring with obesity in children [107]. It is established that children with subclinical hypothyroidism and serum TSH levels above 10 mU/L are treated, regardless of the underlying cause [106]. However, for lower concentrations, the decision to initiate treatment must be individualized [106]. As obesity is a potentially reversible factor contributing to thyroid dysfunction, the emphasis should be set on diet and changes in lifestyle [109]. It is advisable to reassess thyroid function after losing weight [109] because previous reports indicate that weight loss might lead to the normalization of TSH levels [67][106]. Treatment of subclinical hypothyroidism with levothyroxine has not been proven to contribute to weight loss or normalization of lipid and thyroid parameters in children dealing with obesity [107][108]. Thus, if there are no clinical and laboratory indications of overt hypothyroidism, levothyroxine treatment is not required in such a group of patients [108].
There are several hypotheses that could potentially explain changes in thyroid endocrine status. Firstly, it may originate from a disruption in the hypothalamic-pituitary axis, and it is thought that leptin may be responsible for the alteration of hypothalamic thyrotropin-releasing hormone (TRH) production [110]. Secondly, thyroid hormone resistance caused by a decrease in the amount of TSH or peripheral thyroid hormone receptors may also be one of the explanations for elevated TSH and T3 levels [110]. Furthermore, it was demonstrated that individuals with obesity secrete a high amount of inflammatory cytokines, which could negatively affect the function of the sodium/iodide symporter, ultimately leading to compensatory rise in TSH levels [101]. Finally, thyroid disorders may be the consequence of an adaptation process aimed at increasing energy expenditure [110].
In conclusion, the incidence of obesity in the pediatric population raises the risk of developing endocrine and gynecological disorders, which may also have negative consequences on their adulthood health, but the underlying causes of these disturbances are not fully understood and require further research.
3.3.4. Obesity-Induced Hypogonadism
Adult males who are obese present an increased risk of developing hypogonadism compared to those of normal weight [111]. However, the data indicate that the association between obesity and hypogonadism in children and adolescents is less well studied [111]. Nevertheless, the incidence of hypogonadism in male adolescents is on the rise in parallel with the increasing prevalence of obesity [111]. The diagnosis of hypogonadism in boys can be difficult, and symptoms may not appear until puberty [112]. At that time, the development of sexual characteristics may be absent or interrupted [112]. The interactions between sex hormones, excessive aromatase activity in adipose tissue, hormone production by fat cells, and the presence of inflammatory markers occurring in the course of obesity lead to reduced testosterone production and thus contribute to hypogonadism [111]. It has been shown that normal-weight adolescents have higher levels of free testosterone, total testosterone, FSH, and inhibin B and exhibit lower levels of estradiol than their obese counterparts [113]. Thus, an obesity–hypogonadism relationship has been demonstrated and may be driven by increased estradiol levels and a reduced testosterone/estradiol ratio [113].
3.4. Other Non-Metabolic Consequences
It is well known that obesity is a systemic disease that potentially affects almost all organ systems [12]. Therefore, obesity occurring during childhood and adolescence can induce or exacerbate multiple diseases and conditions, including those that were not mentioned above, such as respiratory and neurological complications, oral health disorders, immunologic diseases, and renal, gastrointestinal, musculoskeletal, and dermatologic disturbances [12][66].
3.4.1. Respiratory Complications
Asthma
Among children, obesity is associated with an increased risk of developing asthma [114], and this risk is more pronounced in girls than boys [115]. According to a meta-analysis based on 18 articles, children dealing with overweight or obesity had 1.23 and 1.40 times higher risks of asthma, respectively [115]. These results seem to be in line with a meta-analysis of case–control studies by Azizpour et al., which demonstrated that children and adolescents who are overweight or obese are more likely to have asthma (1.64 and 1.92 times, respectively) than their normal weight/underweight peers [116]. Moreover, it has been shown that children who gain weight rapidly during the first 2 years of life are more prone to developing asthma in the first 6 years of life than those who gained less weight at an early age [117]. Interestingly, a recent study by Pavlidou et al. found that, among preschool children, not only the prevalence of overweight and obesity but also the presence of abnormal birth anthropometric measures, such as length, birth weight, and head circumference, may increase the likelihood of developing asthma in preschool-aged children [118]. Therefore, an increase in BMI in infancy may be more important in predicting the occurrence of asthma in childhood than being overweight at any specific age [117].
What is more, for children already suffering from asthma, coexisting obesity contributes to worse control of symptoms, more frequent and less treatment-responsive exacerbations, poorer response to inhaled corticosteroids (possibly to systemic corticosteroids as well), and decreased quality of life [114]. In addition, children with asthma and obesity are more likely to be unresponsive to bronchodilators and require controlling medications, but on the other hand, they are better responders to leukotriene receptor inhibitors [119]. Thus, apart from the simple co-occurrence of asthma and obesity in certain children, there is increasing evidence of an “obese asthma” phenotype, where increased body weight influences and alters asthma features [119]. In contrast to the “classic” asthma phenotype, the “obese asthma” phenotype can be identified by a low Th2 profile with prevalent infiltration of neutrophils in the bronchial mucosa along with poor infiltration of eosinophils and low levels of IgE [120]. Taken together, these differences in inflammatory response between asthma phenotypes may consequently account for the poorer response to typical asthma treatment in individuals with obesity [121].
There are several mechanisms that may potentially account for the association between childhood obesity and asthma, such as immunological, mechanical [122], dietary, and metabolic factors or changes in the gut microbiota [114] (Figure 1).
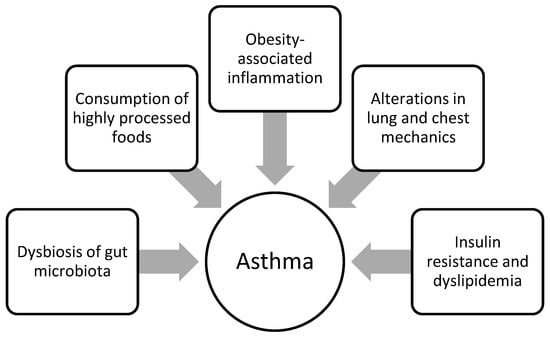
Figure 1. Obesity-related factors contributing to the development of asthma or worsening control of pre-existing disease in children with obesity.
As excessive amounts of adipose tissue in the course of obesity are associated with increased secretion of pro-inflammatory cytokines and leptin and decreased secretion of the anti-inflammatory adipokine, adiponectin [123], it has been demonstrated that pro-inflammatory factors may act as a mediator in the relationship between obesity and the development of asthma [120]. The study by Ma et al. discovered that children with asthma in the observation group with a BMI > 28 kg/m2 presented substantially elevated leptin levels and, at the same time, significantly lower adiponectin levels in comparison to their counterparts with a normal BMI. Furthermore, the number of children with severe disease was greater in the observation group, which may indicate that BMI is associated with greater disease severity.
Obesity has also been shown to adversely affect lung and chest mechanics, as well as cause a reduction in lung compliance [123]. The fat load on the chest wall and abdomen in turn reduces the mobility of the thorax and diaphragm, which ultimately leads to reduced respiratory compliance and increased breathing effort [124]. A meta-analysis that assessed the association between overweight or obesity and pulmonary function in both children and adults revealed that all lung function parameters were reduced in individuals suffering from obesity; however, adults with obesity exhibited lower FEV1 (forced expiratory volume in 1 s), FVC (forced vital capacity), TLC (total lung capacity), and RV (residual volume), whereas children affected by obesity were characterized more by a reduction in FEV1/FVC with intact FEV1 or FVC [125]. This may be related to the finding that obesity in children is associated with airway dysanapsis, defined as a mismatch between lung and airway growth, and more specifically in a faster increase in lung volume and airway length and a concomitant slower increase in airway caliber [126]. Airway dysanapsis associated with obesity is related to adverse clinical outcomes for children with asthma that include a higher risk of severe asthma exacerbations requiring the use of systemic corticosteroids and, in part, their worsened response to treatment [126].
Metabolic dysregulation in the form of insulin resistance and dyslipidemia has previously been shown to likely contribute to asthma in children suffering from obesity [114]. A recent study of children from two independent cohorts, the Tucson Children’s Respiratory Study (TCRS) and the Avon Longitudinal Study of Parents and Children (ALSPAC), found that elevated serum insulin levels in early childhood correspond with higher risks of developing asthma in childhood, adolescence, and adulthood, regardless of BMI, although the processes explaining this phenomenon have not been fully explained [127]. A cross-sectional study aimed at examining the association between obesity, insulin sensitivity, metabolic syndrome, and lung function among American adolescents found that insulin resistance was negatively correlated with FEV1 and FVC in subjects with and without asthma [128]. In addition, it was demonstrated that, among Korean adolescents, subjects with high serum total cholesterol (TC) levels and a high triglyceride (TG)/high-density lipoprotein cholesterol (HDL-C) ratio presented a higher prevalence of asthma [129]. It is likely that dyslipidemia, by activating innate and acquired immunity and enhancing airway inflammatory pathway signaling, consequently increases bronchial smooth muscle tone and airway inflammation and hyperresponsiveness [130].
As a parallel increase in the occurrence of asthma and consumption of highly processed foods, which are poor in antioxidants and rich in saturated fatty acids, has been observed, this may indicate that nutritional habits could play an important role in the development of asthma [131]. Interestingly, a cross-sectional observational study by Tobias et al. found that poor nutritional status (which was characterized by low levels of carotenoids and n-3 fatty acids) in adolescent asthmatics with obesity was correlated with airway obstruction and metabolic disturbances [132]. These findings may indicate that carotenoids (antioxidants that are mainly found in fruits and vegetables and have the potential to reduce the risk of many chronic diseases, including asthma) and n-3 PUFA (polyunsaturated fatty acids with anti-inflammatory properties) may protect against airway obstruction in adolescents with asthma related to obesity [132]. Furthermore, obese asthmatic children with low vitamin D levels showed worsened lung function in the form of reduced FEV1 and FRC (functional residual capacity) than their normal-weight but also asthmatic counterparts [133]. Hence, changes in diet can induce beneficial effects on obesity-related immune and metabolic impairments that concomitantly contribute to the development of asthma [114].
Recent research suggests that the gut microbiota may play a mediating role in the association between obesity and asthma [134]. Dysbiosis of the gut microbiota, which has been observed in both obesity and asthma, negatively affects lipid metabolism and immune response and induces chronic low-grade inflammation. This may indicate that it is a contributing factor in the pathogenesis of obesity-associated asthma, although the underlying mechanisms have not yet been fully investigated [134]. Figure 1 summarises the previously mentioned factors involved in the development of asthma or deterioration of control of pre-existing disease in children affected by obesity.
Obstructive Sleep Apnea
Obesity is also a significant factor in the development of sleep-related breathing disorders [135], especially obstructive sleep apnea (OSA), in the pediatric population and it also plays a leading role in the emergence of this condition during adolescence [136]. OSA is manifested by recurrent episodes of partial or complete upper airway obstruction, which disrupts normal sleep and ventilation, eventually causing intermittent hypoxia and/or increased frequency of arousals [137]. The prevalence of OSA in children is estimated to be around 2–3%, with adenotonsillar tissue hypertrophy being the main risk factor determining the development of this condition, especially among young children [138]. However, the rise in the prevalence of obesity in the pediatric population has increased the incidence of OSA to up to 6% of all children [138]. In the course of obesity, fat in the soft tissue of the throat reduces the size of the lumen in the upper airway, leading to increased structural collapse [139]. In addition, increased amounts of fat in the abdominal region decrease lung function, and both of these mechanisms contribute to the occurrence of obstructive sleep apnea [139]. OSA occurring in the pediatric population carries the risk of long-term complications, such as impaired growth and development, hypertension, pulmonary hypertension, endocrine and metabolic disorders, neurocognitive impairment, maxillofacial dysplasia, or a higher risk of cardiovascular episodes if it is not diagnosed and treated in a timely manner [140]. It should be noted that treatment of obesity is associated with improvements in OSA in children and adolescents [141]. A decrease in SDS-BMI has been shown to be strongly associated with a decrease in the apnea-hypopnea index (the sum of the number of apneas and hypopneas divided by the total recording time) after about six months of therapy, indicating that treatment targeting weight loss should be considered as a first-line treatment for OSA in children and adolescents dealing with overweight or obesity [141].
3.4.2. Other Allergic Diseases
In addition to asthma, obesity has been shown to affect the development and course of such allergic diseases as atopic dermatitis, allergic rhinitis, and food allergies [142]. Obesity, due to its inflammatory effects causing deterioration of the epidermal barrier function and changes in the microbiome, has been shown to contribute to the development of atopic dermatitis, worsening of its course, and poorer results of its treatment [142]. On the other hand, the effectiveness of treatment and alleviation of symptoms may be attributed to weight loss [142]. Findings indicate that children with atopic dermatitis demonstrate a greater frequency of being overweight, obese, and dyslipidemic than their healthy peers [143]. According to a meta-analysis by Zhou et al., BMI is associated with an increased likelihood of allergic rhinitis in children rather than adults [144]. The inflammatory milieu often found in obesity or vitamin D deficiency, which increases the risk of atopic diseases, may be responsible for this link [144]. In turn, a Japanese cross-sectional study aimed at examining the relationship between being overweight and the incidence of food allergies among children found a significant correlation between these two conditions. However, this association was observed specifically in girls and not in boys [145]. A possible explanation underlying this relationship may be that obesity and a high-fat diet induce intestinal inflammation, weakening the barrier function of the intestines and leading to diminished immunological tolerance to allergens. Additionally, obesity and a high-fat diet contribute to a shift in the composition of the gut microbiota [142]. It is also worth adding that, concerning another condition, children who are overweight or obese tend to be older at the time of diagnosis of eosinophilic esophagitis than those of normal weight, and abdominal pain is their main symptom [146].
3.4.3. Neurological Complications
Pseudotumor Cerebri
Among pubertal children, in contrast to those before puberty, obesity acts as a risk factor for pseudotumor cerebri, a condition in which increased intracranial pressure is observed, although with the absence of a mass [147]. The main symptoms of pseudotumor cerebri include headache, nausea, vomiting, retroocular pain, and visual disturbances [66]; however, it should be noted that the clinical picture in children differs depending on age, and younger children may present less prominent symptoms [148]. Many cases of pseudotumor cerebri are idiopathic, and in such situations the condition is termed primary [149]. Nevertheless, its association with many medications, such as tetracyclines, growth hormones, or retinoids, and medical conditions like cerebral venous abnormalities leads to its classification as secondary [148][149]. A prospective population-based cohort study of British and Irish children with newly diagnosed pseudotumor cerebri syndrome (PTCS) showed that only a minority of cases developed PTCS before the age of 7 years, usually without identified risk factors, whereas the majority of cases occurred from the age of 7 years [150]. The prevalence was twice as frequent in girls and increased with age and degree of overweight [150]. Interestingly, it has been shown that over 80% of PTCS in children aged 12–15 can be attributed to obesity [150]. Potential explanations for the link between obesity and pseudotumor cerebri may be increased intrathoracic and intraabdominal pressures occurring in the course of obesity, which lead to decreased intracranial venous drainage and, consequently, decreased cerebrospinal fluid absorption along with increased intracranial pressure [151]. Also, increased leptin levels in the cerebrospinal fluid of obese patients may account for the occurrence of pseudotumor cerebri in these individuals [151]. Therefore, weight loss is considered to be the only treatment that modifies the course of PTCS [148].
Migraine
Additionally, obesity appears to contribute to an increased risk of migraine progression and incidence [66]. The pathogenesis of both obesity and migraine is multifactorial and may be shared between both conditions [152]. It has been shown that neurotransmitters like serotonin, peptides like orexin, inflammatory mediators like calcitonin gene-related protein (CGRP), and adipocytokines such as adiponectin or leptin may have an impact on the common pathophysiology [152]. An Italian study by Tarantino et al. conducted on children and adolescents suffering from migraine found that children who are overweight experienced more frequent migraine attacks than those of normal weight [153]. Thus, the treatment of children affected by obesity and migraine should be focused on weight loss therapies that include exercise, diet, and behavioral interventions [154].
3.4.4. Oral Health Disorders
The incidence of oral health conditions is higher among children and adolescents with obesity [12]. For instance, children experiencing overweight or obesity are more likely to develop dental caries [155]. Obese schoolchildren have been shown to be more likely to develop gingivitis than their normal-weight counterparts, whereas this relationship is modified by gender [156]. It was demonstrated that girls suffering from obesity tend to have a higher likelihood of gingivitis; however, no such relationship was observed among boys [156]. A study of children suffering from both gingivitis and obesity found that obesity amplifies gingivitis [157]. Interestingly, gingivitis through a mechanism involving neutrophil activation can also exacerbate obesity, which is known as disease reciprocity [157]. In a cross-sectional study, it was shown that children and adolescents who were overweight or obese, in contrast to those of normal weight, presented more caries in permanent teeth, increased plaque accumulation and gingivitis, and reduced saliva secretion and buffer capacity [158]. Potential causes linking obesity and dental caries include dietary and lifestyle factors, particularly frequent and excessive consumption of fermentable sugars, changes in saliva composition, and its lower secretion in individuals with obesity [159]. In turn, an impaired immune response and enhanced chronic systemic inflammation may be responsible for the association between obesity and gingivitis, but the underlying factors are not well investigated [156].
On the other hand, studies that aim to investigate the impact of obesity on oral disorders are not entirely consistent. In contrast to the aforementioned results, a cross-sectional study of the pediatric population by Guaré et al. found that individuals experiencing overweight or obesity presented a lower risk of having dental caries, which may be potentially explained by higher levels of protective IgA-s in the saliva of children with excess body mass [160]. These findings seem to be in line with those of the study by García Pérez et al. conducted on Mexican schoolchildren [161]. It has also been shown that, in children aged 6–11 years, BMI was not an independent factor for worse oral hygiene and gingivitis [162]. Taking this into account, the link between obesity and oral diseases should be the subject of further research as evidence is inconclusive.
3.4.5. Immunologic Diseases
The excessive aggregation of adipose tissue present in obesity is associated with subsequent metabolic dysregulation of adipocytes [163]. Adipokines secreted by adipocytes can influence immune cell activity and trigger pro-inflammatory signals, thereby resulting in a chronic, low-grade systemic inflammatory state [163]. Therefore, pediatric obesity has been demonstrated to increase the incidence or severity of immune-mediated or autoimmune diseases, with obesity-related low-grade inflammation as a contributor [12].
Interestingly, central obesity in school-aged children emerged as a risk factor for the development of autoimmune diseases, including type 1 diabetes, autoimmune thyroiditis, juvenile idiopathic arthritis, and inflammatory bowel disease in the adolescent period [164].
Moreover, being obese, particularly during adolescence, is commonly related to an increased risk of both adult as well as pediatric multiple sclerosis (MS) [165]. According to the results of a study by Huppke et al. conducted on a group of 453 German children with multiple sclerosis, obesity was associated with a doubled risk of developing MS and a poorer response to first-line treatment [166]. In addition, Harroud et al., in their study, seemed to confirm the association between higher BMI and susceptibility to MS in the pediatric population [167]. It is expected that childhood obesity will contribute to 14% of the total risk of multiple sclerosis in 2035 [168].
Notably, studies indicate that children experiencing overweight or obesity have a high prevalence of vitamin D deficiency [169]. They are at risk because obesity has a major impact on the status of this lipophilic vitamin, negatively affecting its concentration [170]. In a study by Tayde et al., children with obesity and those of normal weight were administered the same dose of vitamin D (a single oral dose of 150.000 IU) [171]. Their 25(OH)D levels were assessed one week and one month after administration, revealing that participants suffering from obesity exhibited a 2.2 times poorer increase in 25(OH)D compared to their lean counterparts, both after one week and one month [171]. Therefore, it is suggested that children with obesity might require increased doses of vitamin D or repeated administration of a dose [171]. Vitamin D has been proven to exert a wide variety of immunomodulatory, anti-inflammatory, antioxidant, and antifibrotic functions and has been shown to be deficient in numerous patients with autoimmune diseases [172]. A deficiency in vitamin D concentrations among pediatric patients is associated with a higher susceptibility to immunological conditions, including an increased risk of infections, as it was observed during the COVID-19 pandemic and autoimmune diseases such as type 1 diabetes mellitus and multiple sclerosis [173][174]. Research indicates that vitamin D, during the early stages of life, may provide protection against the development of type 1 diabetes in children genetically susceptible to the disease [175]. This is supported by the observation of lower vitamin D concentrations 18 months prior to the first seroconversion in children with islet autoimmunity having two or more pancreatic antibodies or already existing disease compared to controls who did not show positivity for two or more antibodies [175]. Intriguingly, in Finland, a nation previously marked by a high incidence of vitamin D deficiency, an increase in its concentration among children led to a subsequent plateau in the rising prevalence of type 1 diabetes [176].
Taking the above into consideration, it is apparent that a deficiency in vitamin D3 concentrations can play a significant role in the development of children affected by obesity. Therefore, there is urgent need to focus on maintaining its proper serum levels.
3.4.6. Renal Consequences
It was previously demonstrated that children struggling with obesity present a higher risk of chronic kidney disease (CKD) or end-stage renal disease (ESRD) than those having a normal BMI [177]. What is more, obesity in the pediatric population is also a factor that increases the likelihood for disease progression when renal impairment is already present, subsequently raising the mortality risk of children with ESRD [178]. Furthermore, children with obesity may experience a syndrome known as obesity-related glomerulopathy (ORG) [177]. ORG is a key factor contributing to CKD and is characterized by proteinuria that increases over time in many patients [177]. It differs from nephrotic syndrome by the absence of edema, hypoalbuminemia, and hyperlipidemia [177]. It was revealed that kidneys of children experiencing obesity are larger and exhibit higher renal blood flow compared to those of children of normal weight, suggesting that kidney alterations are induced by obesity in early life [178]. A retrospective cross-sectional study by Marzuillo et al., which was conducted on 2957 children and adolescents affected by obesity, showed an interesting positive correlation between eGFR (estimated glomerular filtration rate) and BMI-SDS and a negative correlation between eGFR and SBP-SDS (systolic blood pressure), HOMA-IR, and duration of obesity [179]. This can be explained by the fact that there is an evolution of obesity-associated nephropathy in which a progressive increase in eGFR levels is observed initially (hyperfiltration), with a subsequent decrease in eGFR levels [179]. In contrast, HOMA-IR increases its effect over the course of the disease [179]. Moreover, a large Israeli study of 1,194,704 adolescents aged 17 years found that adolescents dealing with overweight or obesity presented higher future risk for treated ESRD than those of normal weight [180]. It is unclear what exactly accounts for the association between obesity and CKD; however, hemodynamic (glomerular hyperfiltration, glomerulomegaly, glomerulosclerosis, or increased podocyte injury) and metabolic (adipokine dysregulation, increased insulin resistance, abnormal lipid metabolism, or enhanced oxidative stress) alterations as well as lipid nephrotoxicity (excessive renal fat accumulation) may cause or worsen CKD in individuals affected by obesity [181]. On the other hand, a recent Polish study investigating the link between the function of kidneys and lipid metabolism among children with obesity aged 11–17 years revealed normal renal function in subjects, with a weak association with the parameters of lipid metabolism [182]. Intriguingly, it has also been shown that children of obese mothers are more predisposed to CKD and obesity [181].
3.4.7. Gastrointestinal Disturbances
Gastrointestinal diseases such as MAFLD (metabolic dysfunction-associated fatty liver disease, mentioned earlier in the section concerning metabolic complications of obesity) [183], cholelithiasis [184], GERD (gastroesophageal reflux disease) [185], or functional disorders [186] are described in the literature as coexisting with obesity in children.
Biliary Duct Diseases
An increasing incidence of gallstones in children has been observed over the last two decades, with a potential cause including a parallel increased prevalence of obesity in the pediatric group [187]. This is an interesting link because cholelithiasis in children has usually been characterized as a rare condition with the main causes including hemolytic diseases, prolonged parenteral nutrition, and prematurity; however, over the last 20 years, the incidence of cholelithiasis unrelated to hemolytic causes has increased from 1.9 to 4% [188]. It has been found that obesity contributes to a higher risk of developing gallstones through excessive liver secretion and bile saturation with cholesterol or impaired cholecyst mobility [184]. Recently, it has also been shown that normal-weight children and adolescents with gallstones and also those who are obese present significantly higher levels of chemerin, an adipokine, than their healthy counterparts [189]. Interestingly, it was also demonstrated that chemerin levels positively correlate with BMI and HOMA-IR in the pediatric population [190]. Hence, the role of chemerin and other adipokines in the development of cholelithiasis needs further investigation. Taken together, children and adolescents whose BMI exceeds the 85th percentile and who experience right upper quadrant or epigastric pain should be suspected of gallbladder disease [191]. A recent multicenter Polish study investigating the relationship between body weight and gallstone disease risk among children revealed that subjects with gallstones presented a substantially higher BMI in comparison to their healthy counterparts [187]. The results of this study are in line with the findings of the Czech study by Frybova et al., which additionally indicated that children with a higher BMI are at a higher risk of developing choledocholithiasis [192]. Furthermore, subjects suffering from choledocholithiasis presented a higher BMI than children with cholelithiasis [192]. As obesity among children increases the likelihood of the occurrence of complications of cholelithiasis, like pancreatitis, and is also associated with a higher rate of hospitalization due to gallstones, it is crucial to reduce the prevalence of obesity in the pediatric population [184]. It should be noted, however, that rapid weight loss by accelerating the elimination of cholesterol that saturates bile has been shown to contribute to the formation of gallstones [193]. The results of an interesting prospective study by Heida et al. showed that 5.9% of children with severe obesity at baseline developed gallstones during a 6-month lifestyle intervention such as exercise or diet [194]. This occurred after losing > 10% of their initial body weight, whereas the highest prevalence of gallstones was observed in the group that lost > 25% of their initial weight [194]. It is worth mentioning that both cholelithiasis and the resulting cholecystitis are important risk factors for gallbladder cancer in later years [195]. The onset of obesity in early adulthood, regardless of prior diagnosis of primary sclerosing cholangitis and cirrhosis, was found to be notably associated with cholangiocarcinoma and may anticipate onset at a younger age [196]. Recently, the influence of obesity on the increased incidence of biliary tract cancer, both with and without previously diagnosed cholelithiasis, has been demonstrated in the adult Asian population [197]. Unfortunately, there is no such information for the pediatric population, which would require further research.
Gastroesophageal Reflux
As mentioned earlier, children with obesity often suffer from gastroesophageal reflux disease (GERD), but they are also at higher risk of esophageal adenocarcinoma, even though they may develop this cancer later in life [185]. Causes of reflux in individuals with obesity include increased intragastric pressure along with lower esophageal sphincter tone and more frequent episodes of its relaxation [198]. A study by Lang et al. found that children affected by obesity and suffering from asthma are more than 7 times more likely to experience multiple symptoms of gastroesophageal reflux (of which the most common were belching, nausea, and swallowing problems) than lean children [199]. In addition, it has also been shown that, among juveniles with obesity, reflux manifestations are associated with worse asthma symptoms, whereas this association was not apparent in the group of their lean counterparts [199]. Further research is needed regarding the association between obesity and GERD in the pediatric population, as specific data on children are rather scarce and most of the literature refers to the adult population.
An interesting correlation was also found between the higher incidence of functional gastrointestinal disorders (FGIDs), such as functional constipation, functional dyspepsia, and irritable bowel syndrome, among children and adolescents struggling with obesity [186]. The mediating role in the link between obesity and the occurrence of functional gastrointestinal disorders may be represented by low fiber intake (which stimulates stool osmolarity), high saturated fat content (which promotes motility), and increased fermentable carbohydrate consumption [200].
3.4.8. Musculoskeletal Disorders
Children and adolescents who are overweight or obese are more likely to develop short- and long-term musculoskeletal disorders, such as pain, injuries, and fractures, due to obesity-induced adverse biomechanical changes in their gait pattern and greater joint burdens [201]. What is more, overweight and obesity occurring in the pediatric population are also associated with impaired postural control, enhanced forces affecting the lower limbs, malalignment of lower extremities, and diminished bone mineral density [202]. The results of the Merder–Coşkun study proved a higher prevalence of musculoskeletal disorders among children dealing with overweight or obesity compared to lean ones [203]. Furthermore, a Polish study investigating the prevalence of postural defects among children and adolescents with excessive body weight found that 69.2% of overweight individuals and 78.6% of individuals with obesity presented postural defects, of which valgus knees and flat feet were the most common [204]. Ferrer et al. demonstrated in their study involving 553 children that body weight has an impact on bone mineralization [205]. The bone mineral content (BMC) and bone mineral density (BMD) measured by bone densitometry (DXA) were highest among those of normal weight and, interestingly, also among those who were overweight [205]. On the other hand, those subjects who were obese or very obese as well as those who were underweight showed lower BMC and BMD values [205]. The increase in BMD and BMC in children dealing with overweight may be the consequence of compensation in response to weight gain; however, this mechanism breaks down among children with obesity, resulting in a decrease in BMD and BMC [205]. Previous studies have demonstrated that children suffering from obesity are more susceptible to extremity fractures, including those in the forearm, and have a higher risk of death due to trauma-related injuries than their non-obese counterparts [206][207]. Musculoskeletal complaints can reduce the determination to exercise and limit the physical capacity of children affected by obesity, potentially contributing to the perpetuation of a sedentary lifestyle and, consequently, obesity [202]. Taken together, it is essential to assess the musculoskeletal system of children who are overweight or obese at every primary care visit, even in the absence of complaints [203]. Intervention aimed at weight loss may help protect them from serious musculoskeletal diseases [203].
3.4.9. Dermatologic Disturbances
As mentioned earlier, obesity is a disease that affects many systems and therefore does not spare the skin either. It has been established that obesity negatively affects the integrity of the skin barrier, alters the structure of collagen, increases the production of sebum and sweat, impairs skin circulation, promotes inflammation, and alters the immune response [208]. Thus, it may contribute to the increased incidence of cutaneous manifestations and disorders, such as acanthosis nigricans, hirsutism, striae distensae, acrochordons, keratosis pilaris, chronic venous insufficiency, plantar hyperkeratosis, cellulitis, psoriasis, skin infections, and hidradenitis suppurativa [209]. Numerous studies regarding the link between obesity and dermatological diseases among adults can be found in the literature; however, evidence for the pediatric population is scarce [210]. Nevertheless, a recent study of 103 children and adolescents with excessive body weight showed a high incidence of skin abnormalities occurring in this group [210]. Skin alterations were associated with increasing BMI and age, and the most prevalent were striae distensae (71%), keratosis pilaris (64.7%), acanthosis nigricans (45%), acne vulgaris (39.2%), acrochordons (25.5%), and plantar hyperkeratosis (17.6%) [210]. In addition, 38.9% of subjects showed reduced dermatological quality of life [210]. Striae distensae are also the most frequently observed dermatologic condition among children with obesity, according to the study by Güven et al. [211].
It has also been shown that children and adolescents with hidradenitis suppurativa (HS) have a 2.48 times higher risk of obesity compared to children without HS [212], and, what is more, an increased BMI during childhood is positively correlated with the likelihood of HS occurrence in adulthood [213]. It should also be mentioned that Phan et al. showed an interesting statistically significant association between the incidence of psoriasis in the pediatric population, overweight/obesity, and waist/height ratio > 0.5 [214]. Furthermore, this relationship varies according to the severity of obesity, as patients with moderately severe psoriasis are more likely to be obese in comparison to those with mild psoriasis [214]. A large cross-sectional Israeli study also identified a link between higher BMI and psoriasis among adolescents [215].
Moreover, the presence of acanthosis nigricans, keratosis pilaris, and acne vulgaris is associated with increased HOMA-IR (an index of insulin resistance) among children and adolescents with obesity, which may suggest a role for skin lesions as a potential marker of insulin resistance [210]. Insulin resistance has been shown to promote proliferation of fibroblasts and keratinocytes by insulin, insulin-like growth factor receptor-1 (IGFR1), and fibroblast and epidermal growth factors, resulting in the development of acanthosis nigricans [216]. Increased levels of insulin also stimulate androgen production and can reduce serum levels of sex hormone-binding globulin (SHBG), consequently strongly enhancing androgen activity and favoring acne development [217]. Hyperandrogenemia resulting from elevated insulin levels contributes to excessive keratinocyte proliferation of the pilosebacious unit of hair follicles, which may account for the increased incidence of keratosis pilaris in individuals with insulin resistance [218].
References
- (NCD-RisC). Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet 2017, 390, 2627–2642.
- Fabin-Czepiel, K.; Pieczyńska-Chapuła, K.; Deja, G. “The obesity pandemic” in the COVID-19 pandemic—New treatment for an old problem. Pediatr. Endocrinol. Diabetes Metab. 2023, 29, 104–111.
- World Health Organization. Report on the Fifth Round of Data Collection, 2018–2020; WHO Regional Office for Europe: Copenhagen, Denmark, 2022.
- Davies, S.C.; Department of Health and Social Care, Great Britain. Time to Solve Childhood Obesity: An Independent Report by the Chief Medical Officer, 2019; Department of Health and Social Care, APS Group: Cheshire, UK, 2019.
- Powell-Wiley, T.M.; Poirier, P.; Burke, L.E.; Després, J.P.; Gordon-Larsen, P.; Lavie, C.J.; Lear, S.A.; Ndumele, C.E.; Neeland, I.J.; Sanders, P.; et al. Obesity and Cardiovascular Disease: A Scientific Statement From the American Heart Association. Circulation 2021, 143, e984–e1010.
- Chait, A.; den Hartigh, L.J. Adipose Tissue Distribution, Inflammation and Its Metabolic Consequences, Including Diabetes and Cardiovascular Disease. Front. Cardiovasc. Med. 2020, 7, 22.
- Tagi, V.M.; Giannini, C.; Chiarelli, F. Insulin Resistance in Children. Front. Endocrinol. 2019, 10, 342.
- Redondo, M.J.; Foster, N.C.; Libman, I.M.; Mehta, S.N.; Hathway, J.M.; Bethin, K.E.; Nathan, B.M.; Ecker, M.A.; Shah, A.C.; DuBose, S.N.; et al. Prevalence of cardiovascular risk factors in youth with type 1 diabetes and elevated body mass index. Acta Diabetol. 2016, 53, 271–277.
- Blüher, M. Metabolically Healthy Obesity. Endocr. Rev. 2020, 41, bnaa004.
- Al-Beltagi, M.; Bediwy, A.S.; Saeed, N.K. Insulin-resistance in paediatric age: Its magnitude and implications. World J. Diabetes 2022, 13, 282–307.
- Tagi, V.M.; Samvelyan, S.; Chiarelli, F. An update of the consensus statement on insulin resistance in children 2010. Front. Endocrinol. 2022, 13, 1061524.
- Marcus, C.; Danielsson, P.; Hagman, E. Pediatric obesity-Long-term consequences and effect of weight loss. J. Intern. Med. 2022, 292, 870–891.
- Huang, H.; Zheng, X.; Wen, X.; Zhong, J.; Zhou, Y.; Xu, L. Visceral fat correlates with insulin secretion and sensitivity independent of BMI and subcutaneous fat in Chinese with type 2 diabetes. Front. Endocrinol. 2023, 14, 1144834.
- Xu, H.; Verre, M.C. Type 2 Diabetes Mellitus in Children. Am. Fam. Physician 2018, 98, 590–594.
- Chylińska-Frątczak, A.; Michalak, A.; Baranowska-Jaźwiecka, A.; Mianowska, B.; Szadkowska, A. Incidence of hyperglycaemic disorders in children and adolescents with obesity. Pediatr. Endocrinol. Diabetes Metab. 2022, 28, 274–280.
- Sinha, R.; Fisch, G.; Teague, B.; Tamborlane, W.V.; Banyas, B.; Allen, K.; Savoye, M.; Rieger, V.; Taksali, S.; Barbetta, G.; et al. Prevalence of impaired glucose tolerance among children and adolescents with marked obesity. N. Engl. J. Med. 2002, 346, 802–810.
- Abbasi, A.; Juszczyk, D.; van Jaarsveld, C.H.M.; Gulliford, M.C. Body Mass Index and Incident Type 1 and Type 2 Diabetes in Children and Young Adults: A Retrospective Cohort Study. J. Endocr. Soc. 2017, 1, 524–537.
- Pinhas-Hamiel, O.; Zeitler, P. Acute and chronic complications of type 2 diabetes mellitus in children and adolescents. Lancet 2007, 369, 1823–1831.
- Maffeis, C.; Birkebaek, N.H.; Konstantinova, M.; Schwandt, A.; Vazeou, A.; Casteels, K.; Jali, S.; Limbert, C.; Pundziute-Lycka, A.; Toth-Heyn, P.; et al. Prevalence of underweight, overweight, and obesity in children and adolescents with type 1 diabetes: Data from the international SWEET registry. Pediatr. Diabetes 2018, 19, 1211–1220.
- Abela, A.G.; Fava, S. Why is the Incidence of Type 1 Diabetes Increasing? Curr. Diabetes Rev. 2021, 17, e030521193110.
- Chylińska-Frątczak, A.; Pietrzak, I.; Michalak, A.; Wyka, K.; Szadkowska, A. Autoimmune reaction against pancreatic beta cells in children and adolescents with simple obesity. Front. Endocrinol. 2022, 13, 1061671.
- Ferrara-Cook, C.; Geyer, S.M.; Evans-Molina, C.; Libman, I.M.; Becker, D.J.; Gitelman, S.E.; Redondo, M.J.; Group, T.D.T.S. Excess BMI Accelerates Islet Autoimmunity in Older Children and Adolescents. Diabetes Care 2020, 43, 580–587.
- Dathan-Stumpf, A.; Vogel, M.; Hiemisch, A.; Thiery, J.; Burkhardt, R.; Kratzsch, J.; Kiess, W. Pediatric reference data of serum lipids and prevalence of dyslipidemia: Results from a population-based cohort in Germany. Clin. Biochem. 2016, 49, 740–749.
- Brzeziński, M.; Metelska, P.; Myśliwiec, M.; Szlagatys-Sidorkiewicz, A. Lipid disorders in children living with overweight and obesity- large cohort study from Poland. Lipids Health Dis. 2020, 19, 47.
- Umer, A.; Kelley, G.A.; Cottrell, L.E.; Giacobbi, P.; Innes, K.E.; Lilly, C.L. Childhood obesity and adult cardiovascular disease risk factors: A systematic review with meta-analysis. BMC Public Health 2017, 17, 683.
- Nielsen, T.R.H.; Lausten-Thomsen, U.; Fonvig, C.E.; Bøjsøe, C.; Pedersen, L.; Bratholm, P.S.; Hansen, T.; Pedersen, O.; Holm, J.C. Dyslipidemia and reference values for fasting plasma lipid concentrations in Danish/North-European White children and adolescents. BMC Pediatr. 2017, 17, 116.
- Jung, U.J.; Choi, M.S. Obesity and its metabolic complications: The role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia and nonalcoholic fatty liver disease. Int. J. Mol. Sci. 2014, 15, 6184–6223.
- Zhao, X.; An, X.; Yang, C.; Sun, W.; Ji, H.; Lian, F. The crucial role and mechanism of insulin resistance in metabolic disease. Front. Endocrinol. 2023, 14, 1149239.
- Kershaw, E.E.; Flier, J.S. Adipose tissue as an endocrine organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556.
- Soliman, A.T.; Yasin, M.; Kassem, A. Leptin in pediatrics: A hormone from adipocyte that wheels several functions in children. Indian J. Endocrinol. Metab. 2012, 16 (Suppl. 3), S577–S587.
- Litwin, M.; Kułaga, Z. Obesity, metabolic syndrome, and primary hypertension. Pediatr. Nephrol. 2021, 36, 825–837.
- Maximova, K.; O’Loughlin, J.; Paradis, G.; Hanley, J.A.; Lynch, J. Changes in anthropometric characteristics and blood pressure during adolescence. Epidemiology 2010, 21, 324–331.
- Sabbatini, A.R.; Faria, A.P.; Barbaro, N.R.; Gordo, W.M.; Modolo, R.G.; Pinho, C.; Fontana, V.; Moreno, H. Deregulation of adipokines related to target organ damage on resistant hypertension. J. Hum. Hypertens 2014, 28, 388–392.
- Christian Flemming, G.M.; Bussler, S.; Körner, A.; Kiess, W. Definition and early diagnosis of metabolic syndrome in children. J. Pediatr. Endocrinol. Metab. 2020, 33, 821–833.
- Reinehr, T.; de Sousa, G.; Toschke, A.M.; Andler, W. Comparison of metabolic syndrome prevalence using eight different definitions: A critical approach. Arch. Dis. Child. 2007, 92, 1067–1072.
- Pinhas-Hamiel, O.; Levek-Motola, N.; Kaidar, K.; Boyko, V.; Tisch, E.; Mazor-Aronovitch, K.; Graf-Barel, C.; Landau, Z.; Lerner-Geva, L.; Frumkin Ben-David, R. Prevalence of overweight, obesity and metabolic syndrome components in children, adolescents and young adults with type 1 diabetes mellitus. Diabetes Metab. Res. Rev. 2015, 31, 76–84.
- Tropeano, A.; Corica, D.; Li Pomi, A.; Pepe, G.; Morabito, L.A.; Curatola, S.L.; Casto, C.; Aversa, T.; Wasniewska, M. The metabolic syndrome in pediatrics: Do we have a reliable definition? A systematic review. Eur. J. Endocrinol. 2021, 185, 265–278.
- Yki-Järvinen, H. Non-alcoholic fatty liver disease as a cause and a consequence of metabolic syndrome. Lancet Diabetes Endocrinol. 2014, 2, 901–910.
- Nehus, E.; Mitsnefes, M. Childhood Obesity and the Metabolic Syndrome. Pediatr. Clin. N. Am. 2019, 66, 31–43.
- Nobili, V.; Alisi, A.; Valenti, L.; Miele, L.; Feldstein, A.E.; Alkhouri, N. NAFLD in children: New genes, new diagnostic modalities and new drugs. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 517–530.
- Loomba, R.; Sanyal, A.J. The global NAFLD epidemic. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 686–690.
- Fan, R.; Wang, J.; Du, J. Association between body mass index and fatty liver risk: A dose-response analysis. Sci. Rep. 2018, 8, 15273.
- Lin, Y.C.; Chang, P.F.; Liu, K.; Chang, M.H.; Ni, Y.H. Predictors for incidence and remission of nonalcoholic fatty liver disease in obese children and adolescents. J. Formos. Med. Assoc. 2022, 121 Pt 1, 36–42.
- Loomis, A.K.; Kabadi, S.; Preiss, D.; Hyde, C.; Bonato, V.; St Louis, M.; Desai, J.; Gill, J.M.; Welsh, P.; Waterworth, D.; et al. Body Mass Index and Risk of Nonalcoholic Fatty Liver Disease: Two Electronic Health Record Prospective Studies. J. Clin. Endocrinol. Metab. 2016, 101, 945–952.
- Haczeyni, F.; Bell-Anderson, K.S.; Farrell, G.C. Causes and mechanisms of adipocyte enlargement and adipose expansion. Obes. Rev. 2018, 19, 406–420.
- Grønbæk, H.; Lange, A.; Birkebæk, N.H.; Holland-Fischer, P.; Solvig, J.; Hørlyck, A.; Kristensen, K.; Rittig, S.; Vilstrup, H. Effect of a 10-week weight loss camp on fatty liver disease and insulin sensitivity in obese Danish children. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 223–228.
- Campos, R.M.; de Piano, A.; da Silva, P.L.; Carnier, J.; Sanches, P.L.; Corgosinho, F.C.; Masquio, D.C.; Lazaretti-Castro, M.; Oyama, L.M.; Nascimento, C.M.; et al. The role of pro/anti-inflammatory adipokines on bone metabolism in NAFLD obese adolescents: Effects of long-term interdisciplinary therapy. Endocrine 2012, 42, 146–156.
- Estes, C.; Razavi, H.; Loomba, R.; Younossi, Z.; Sanyal, A.J. Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease. Hepatology 2018, 67, 123–133.
- Estes, C.; Anstee, Q.M.; Arias-Loste, M.T.; Bantel, H.; Bellentani, S.; Caballeria, J.; Colombo, M.; Craxi, A.; Crespo, J.; Day, C.P.; et al. Modeling NAFLD disease burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016–2030. J. Hepatol. 2018, 69, 896–904.
- Hagström, H.; Stål, P.; Hultcrantz, R.; Hemmingsson, T.; Andreasson, A. Overweight in late adolescence predicts development of severe liver disease later in life: A 39 years follow-up study. J. Hepatol. 2016, 65, 363–368.
- Piscaglia, F.; Svegliati-Baroni, G.; Barchetti, A.; Pecorelli, A.; Marinelli, S.; Tiribelli, C.; Bellentani, S.; Group, H.-N.I.S. Clinical patterns of hepatocellular carcinoma in nonalcoholic fatty liver disease: A multicenter prospective study. Hepatology 2016, 63, 827–838.
- Eslam, M.; Alkhouri, N.; Vajro, P.; Baumann, U.; Weiss, R.; Socha, P.; Marcus, C.; Lee, W.S.; Kelly, D.; Porta, G.; et al. Defining paediatric metabolic (dysfunction)-associated fatty liver disease: An international expert consensus statement. Lancet Gastroenterol. Hepatol. 2021, 6, 864–873.
- Liu, J.; Mu, C.; Li, K.; Luo, H.; Liu, Y.; Li, Z. Estimating Global Prevalence of Metabolic Dysfunction-Associated Fatty Liver Disease in Overweight or Obese Children and Adolescents: Systematic Review and Meta-Analysis. Int. J. Public Health 2021, 66, 1604371.
- Han, S.K.; Baik, S.K.; Kim, M.Y. Non-alcoholic fatty liver disease: Definition and subtypes. Clin. Mol. Hepatol. 2023, 29 (Suppl. 1), S5–S16.
- Rader, D.J. Effect of insulin resistance, dyslipidemia, and intra-abdominal adiposity on the development of cardiovascular disease and diabetes mellitus. Am. J. Med. 2007, 120 (Suppl. 1), S12–S18.
- Bennett, N.R.; Ferguson, T.S.; Bennett, F.I.; Tulloch-Reid, M.K.; Younger-Coleman, N.O.; Jackson, M.D.; Samms-Vaughan, M.E.; Wilks, R.J. High-Sensitivity C-Reactive Protein is Related to Central Obesity and the Number of Metabolic Syndrome Components in Jamaican Young Adults. Front. Cardiovasc. Med. 2014, 1, 12.
- Cercato, C.; Fonseca, F.A. Cardiovascular risk and obesity. Diabetol. Metab. Syndr. 2019, 11, 74.
- Hedvall Kallerman, P.; Hagman, E.; Edstedt Bonamy, A.K.; Zemack, H.; Marcus, C.; Norman, M.; Westerståhl, M. Obese children without comorbidities have impaired microvascular endothelial function. Acta Paediatr. 2014, 103, 411–417.
- Garibay-Nieto, N.; Hernández-Morán, B.A.; Villanueva-Ortega, E.; Garcés-Hernández, M.J.; Pedraza-Escudero, K.; Arroyo-Valerio, A.; Pedraza-Helvert, C.; Herrera-Rosas, A.; Laresgoiti-Servitje, E.; León-Hernández, M.; et al. Comparison of Carotid Intima-Media Thickness in Children and Adults With and Without Obesity: A Hysteresis Model. Endocr. Pract. 2022, 28, 315–320.
- Robertson, J.; Schaufelberger, M.; Lindgren, M.; Adiels, M.; Schiöler, L.; Torén, K.; McMurray, J.; Sattar, N.; Åberg, M.; Rosengren, A. Higher Body Mass Index in Adolescence Predicts Cardiomyopathy Risk in Midlife. Circulation 2019, 140, 117–125.
- Alpert, M.A.; Omran, J.; Mehra, A.; Ardhanari, S. Impact of obesity and weight loss on cardiac performance and morphology in adults. Prog. Cardiovasc. Dis. 2014, 56, 391–400.
- Jobira, B.; Frank, D.N.; Pyle, L.; Silveira, L.J.; Kelsey, M.M.; Garcia-Reyes, Y.; Robertson, C.E.; Ir, D.; Nadeau, K.J.; Cree-Green, M. Obese Adolescents With PCOS Have Altered Biodiversity and Relative Abundance in Gastrointestinal Microbiota. J. Clin. Endocrinol. Metab. 2020, 105, e2134–e2144.
- Xu, X.R.; Han, M.M.; Yang, Y.Z.; Wang, X.; Hou, D.Y.; Meng, X.C.; Wang, H.; Zhao, W.S.; Zhang, L.; Xu, L. Fifteen-year mortality and prognostic factors in patients with dilated cardiomyopathy: Persistent standardized application of drug therapy and strengthened management may bring about encouraging change in an aging society. J. Geriatr. Cardiol. 2022, 19, 335–342.
- Lee, J.; Kim, J.H. Endocrine comorbidities of pediatric obesity. Clin. Exp. Pediatr. 2021, 64, 619–627.
- Brix, N.; Ernst, A.; Lauridsen, L.L.B.; Parner, E.T.; Arah, O.A.; Olsen, J.; Henriksen, T.B.; Ramlau-Hansena, C.H. Childhood overweight and obesity and timing of puberty in boys and girls: Cohort and sibling-matched analyses. Int. J. Epidemiol. 2020, 49, 834–844.
- Mazur, A.; Zachurzok, A.; Baran, J.; Dereń, K.; Łuszczki, E.; Weres, A.; Wyszyńska, J.; Dylczyk, J.; Szczudlik, E.; Drożdż, D.; et al. Childhood Obesity: Position Statement of Polish Society of Pediatrics, Polish Society for Pediatric Obesity, Polish Society of Pediatric Endocrinology and Diabetes, the College of Family Physicians in Poland and Polish Association for Study on Obesity. Nutrients 2022, 14, 3806.
- Ghergherehchi, R.; Hazhir, N. Thyroid hormonal status among children with obesity. Ther. Adv. Endocrinol. Metab. 2015, 6, 51–55.
- Calcaterra, V.; Magenes, V.C.; Hruby, C.; Siccardo, F.; Mari, A.; Cordaro, E.; Fabiano, V.; Zuccotti, G. Links between Childhood Obesity, High-Fat Diet, and Central Precocious Puberty. Children 2023, 10, 241.
- Liu, Y.; Yu, T.; Li, X.; Pan, D.; Lai, X.; Chen, Y.; Wang, X.; Yu, X.; Fu, S.; Huang, S.; et al. Prevalence of precocious puberty among Chinese children: A school population-based study. Endocrine 2021, 72, 573–581.
- Liu, G.; Guo, J.; Zhang, X.; Lu, Y.; Miao, J.; Xue, H. Obesity is a risk factor for central precocious puberty: A case-control study. BMC Pediatr. 2021, 21, 509.
- Chen, C.; Zhang, Y.; Sun, W.; Chen, Y.; Jiang, Y.; Song, Y.; Lin, Q.; Zhu, L.; Zhu, Q.; Wang, X.; et al. Investigating the relationship between precocious puberty and obesity: A cross-sectional study in Shanghai, China. BMJ Open 2017, 7, e014004.
- Reinehr, T.; Bosse, C.; Lass, N.; Rothermel, J.; Knop, C.; Roth, C.L. Effect of Weight Loss on Puberty Onset in Overweight Children. J. Pediatr. 2017, 184, 143–150.e141.
- Farello, G.; Altieri, C.; Cutini, M.; Pozzobon, G.; Verrotti, A. Review of the Literature on Current Changes in the Timing of Pubertal Development and the Incomplete Forms of Early Puberty. Front. Pediatr. 2019, 7, 147.
- Sultan, C.; Gaspari, L.; Maimoun, L.; Kalfa, N.; Paris, F. Disorders of puberty. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 48, 62–89.
- Su, H.; Su, Z.; Pan, L.; Wang, L.; Xu, Z.; Peng, G.; Li, X. Factors affecting bone maturation in Chinese girls aged 4-8 years with isolated premature thelarche. BMC Pediatr. 2020, 20, 356.
- Kaplowitz, P.B. For Premature Thelarche and Premature Adrenarche, the Case for Waiting before Testing. Horm. Res. Paediatr. 2020, 93, 573–576.
- Banerjee, S.; Bajpai, A. Precocious Puberty. Indian J. Pediatr. 2023, 90, 582–589.
- Itriyeva, K. The effects of obesity on the menstrual cycle. Curr. Probl. Pediatr. Adolesc. Health Care 2022, 52, 101241.
- Elizondo-Montemayor, L.; Hernández-Escobar, C.; Lara-Torre, E.; Nieblas, B.; Gómez-Carmona, M. Gynecologic and Obstetric Consequences of Obesity in Adolescent Girls. J. Pediatr. Adolesc. Gynecol. 2017, 30, 156–168.
- Durá-Travé, T.; Gallinas-Victoriano, F. Hyper-androgenemia and obesity in early-pubertal girls. J. Endocrinol. Investig. 2022, 45, 1577–1585.
- Wood, P.L.; Bauman, D. Gynaecological issues affecting the obese adolescent. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 453–465.
- Kim, J.H.; Lim, J.S. Early menarche and its consequence in Korean female: Reducing fructose intake could be one solution. Clin. Exp. Pediatr. 2021, 64, 12–20.
- Kang, M.J. The adiposity rebound in the 21st century children: Meaning for what? Korean J. Pediatr. 2018, 61, 375–380.
- German, A.; Shmoish, M.; Hochberg, Z. Predicting pubertal development by infantile and childhood height, BMI, and adiposity rebound. Pediatr. Res. 2015, 78, 445–450.
- Koivuaho, E.; Laru, J.; Ojaniemi, M.; Puukka, K.; Kettunen, J.; Tapanainen, J.S.; Franks, S.; Järvelin, M.R.; Morin-Papunen, L.; Sebert, S.; et al. Age at adiposity rebound in childhood is associated with PCOS diagnosis and obesity in adulthood-longitudinal analysis of BMI data from birth to age 46 in cases of PCOS. Int. J. Obes. 2019, 43, 1370–1379.
- Fonseca, M.J.; Moreira, C.; Santos, A.C. Adiposity rebound and cardiometabolic health in childhood: Results from the Generation XXI birth cohort. Int. J. Epidemiol. 2021, 50, 1260–1271.
- Li, W.; Liu, Q.; Deng, X.; Chen, Y.; Yang, B.; Huang, X.; Østbye, T. Association of prepubertal obesity with pubertal development in Chinese girls and boys: A longitudinal study. Am. J. Hum. Biol. 2018, 30, e23195.
- Lee, J.M.; Wasserman, R.; Kaciroti, N.; Gebremariam, A.; Steffes, J.; Dowshen, S.; Harris, D.; Serwint, J.; Abney, D.; Smitherman, L.; et al. Timing of Puberty in Overweight Versus Obese Boys. Pediatrics 2016, 137, e20150164.
- Papadimitriou, A.; Kanakis, G.; Douros, K.; Papadimitriou, D.T.; Boutsiadis, A.H.; Nicolaidou, P.; Fretzayas, A. Constitutional advancement of growth is associated with early puberty in girls. Horm. Res. Paediatr. 2011, 76, 273–277.
- Papadimitriou, A.; Nicolaidou, P.; Fretzayas, A.; Chrousos, G.P. Clinical review: Constitutional advancement of growth, a.k.a. early growth acceleration, predicts early puberty and childhood obesity. J. Clin. Endocrinol. Metab. 2010, 95, 4535–4541.
- Aris, I.M.; Perng, W.; Dabelea, D.; Ganiban, J.M.; Liu, C.; Marceau, K.; Robertson, O.C.; Hockett, C.W.; Mihalopoulos, N.L.; Kong, X.; et al. Analysis of Early-Life Growth and Age at Pubertal Onset in US Children. JAMA Netw. Open 2022, 5, e2146873.
- Papadimitriou, A.; Marakaki, C.; Papadimitriou, D.T. Growth variations with opposite clinical outcomes and the emerging role of IGF-1. Trends Endocrinol. Metab. 2022, 33, 359–370.
- AsghariHanjani, N.; Vafa, M. The role of IGF-1 in obesity, cardiovascular disease, and cancer. Med. J. Islam Repub. Iran 2019, 33, 56.
- Baier, I.; Pereira, A.; Ferrer, P.; Iñiguez, G.; Mericq, V. Higher Prepubertal IGF-1 Concentrations Associate to Earlier Pubertal Tempo in Both Sexes. Horm. Res. Paediatr. 2023, 96, 404–411.
- Kempf, E.; Vogel, M.; Vogel, T.; Kratzsch, J.; Landgraf, K.; Kühnapfel, A.; Gausche, R.; Gräfe, D.; Sergeyev, E.; Pfäffle, R.; et al. Dynamic alterations in linear growth and endocrine parameters in children with obesity and height reference values. EClinicalMedicine 2021, 37, 100977.
- He, Y.; Tian, J.; Blizzard, L.; Oddy, W.H.; Dwyer, T.; Bazzano, L.A.; Hickey, M.; Harville, E.W.; Venn, A.J. Associations of childhood adiposity with menstrual irregularity and polycystic ovary syndrome in adulthood: The Childhood Determinants of Adult Health Study and the Bogalusa Heart Study. Hum. Reprod. 2020, 35, 1185–1198.
- Ambia, A.M.; Pruszynski, J.E.; Fairchild, E.; McIntire, D.D.; Nelson, D.B. Perinatal outcomes of young adolescent pregnancies in an urban inner city. Am. J. Obstet. Gynecol. MFM 2023, 5, 100843.
- Wilson, N.A.; Peña, A.S. Quality of life in adolescent girls with polycystic ovary syndrome. J. Paediatr. Child Health 2020, 56, 1351–1357.
- Kamboj, M.K.; Bonny, A.E. Polycystic ovary syndrome in adolescence: Diagnostic and therapeutic strategies. Transl. Pediatr. 2017, 6, 248–255.
- Li, L.; Feng, Q.; Ye, M.; He, Y.; Yao, A.; Shi, K. Metabolic effect of obesity on polycystic ovary syndrome in adolescents: A meta-analysis. J. Obstet. Gynaecol. 2017, 37, 1036–1047.
- Licenziati, M.R.; Valerio, G.; Vetrani, I.; De Maria, G.; Liotta, F.; Radetti, G. Altered Thyroid Function and Structure in Children and Adolescents Who Are Overweight and Obese: Reversal After Weight Loss. J. Clin. Endocrinol. Metab. 2019, 104, 2757–2765.
- Rumińska, M.; Witkowska-Sędek, E.; Majcher, A.; Pyrżak, B. Thyroid Function in Obese Children and Adolescents and Its Association with Anthropometric and Metabolic Parameters. Adv. Exp. Med. Biol. 2016, 912, 33–41.
- Krause, A.J.; Cines, B.; Pogrebniak, E.; Sherafat-Kazemzadeh, R.; Demidowich, A.P.; Galescu, O.A.; Brady, S.M.; Reynolds, J.C.; Hubbard, V.S.; Yanovski, J.A. Associations between adiposity and indicators of thyroid status in children and adolescents. Pediatr. Obes. 2016, 11, 551–558.
- Witkowska-Sędek, E.; Kucharska, A.; Rumińska, M.; Pyrżak, B. Thyroid dysfunction in obese and overweight children. Endokrynol. Pol. 2017, 68, 54–60.
- Kaltenbach, T.E.; Graeter, T.; Oeztuerk, S.; Holzner, D.; Kratzer, W.; Wabitsch, M.; Denzer, C. Thyroid dysfunction and hepatic steatosis in overweight children and adolescents. Pediatr. Obes. 2017, 12, 67–74.
- Metwalley, K.A.; Farghaly, H.S. Subclinical hypothyroidism in children: Updates for pediatricians. Ann. Pediatr. Endocrinol. Metab. 2021, 26, 80–85.
- Kumar, S.; Dayal, D.; Attri, S.; Gupta, A.; Bhalla, A. Levothyroxine supplementation for obesity-associated thyroid dysfunction in children: A prospective, randomized, case control study. Pediatr. Endocrinol. Diabetes Metab. 2019, 25, 107–113.
- Szeliga, K.; Antosz, A.; Skrzynska, K.; Kalina-Faska, B.; Gawlik, A. Subclinical hypothyroidism in children and adolescents as mild dysfunction of the thyroid gland: A single-center study. Pediatr. Endocrinol. Diabetes Metab. 2023, 29, 97–103.
- Salerno, M.; Capalbo, D.; Cerbone, M.; De Luca, F. Subclinical hypothyroidism in childhood—Current knowledge and open issues. Nat. Rev. Endocrinol. 2016, 12, 734–746.
- Jin, H.Y. Prevalence of subclinical hypothyroidism in obese children or adolescents and association between thyroid hormone and the components of metabolic syndrome. J. Paediatr. Child Health 2018, 54, 975–980.
- Mushannen, T.; Cortez, P.; Stanford, F.C.; Singhal, V. Obesity and Hypogonadism-A Narrative Review Highlighting the Need for High-Quality Data in Adolescents. Children 2019, 6, 63.
- Rey, R.A. Biomarkers of male hypogonadism in childhood and adolescence. Adv. Lab. Med. 2020, 1, 20200024.
- Hassan, M.M.; Sarry Eldin, A.M.; Musa, N.; El-Wakil, K.H.; Ali, M.; Ahmed, H.H. Insights into the implication of obesity in hypogonadism among adolescent boys. J. Pediatr. Endocrinol. Metab. 2022, 35, 1497–1504.
- Reyes-Angel, J.; Kaviany, P.; Rastogi, D.; Forno, E. Obesity-related asthma in children and adolescents. Lancet Child Adolesc. Health 2022, 6, 713–724.
- Deng, X.; Ma, J.; Yuan, Y.; Zhang, Z.; Niu, W. Association between overweight or obesity and the risk for childhood asthma and wheeze: An updated meta-analysis on 18 articles and 73252 children. Pediatr. Obes. 2019, 14, e12532.
- Azizpour, Y.; Delpisheh, A.; Montazeri, Z.; Sayehmiri, K.; Darabi, B. Effect of childhood BMI on asthma: A systematic review and meta-analysis of case-control studies. BMC Pediatr. 2018, 18, 143.
- Brüske, I.; Flexeder, C.; Heinrich, J. Body mass index and the incidence of asthma in children. Curr. Opin. Allergy Clin. Immunol. 2014, 14, 155–160.
- Pavlidou, E.; Mantzorou, M.; Tolia, M.; Antasouras, G.; Poutsidi, A.; Psara, E.; Poulios, E.; Fasoulas, A.; Vasios, G.K.; Giaginis, C. Childhood overweight and obesity and abnormal birth anthropometric measures are associated with a higher prevalence of childhood asthma in preschool age. J. Asthma. 2023, 60, 1316–1325.
- Di Genova, L.; Penta, L.; Biscarini, A.; Di Cara, G.; Esposito, S. Children with Obesity and Asthma: Which Are the Best Options for Their Management? Nutrients 2018, 10, 1634.
- Fainardi, V.; Passadore, L.; Labate, M.; Pisi, G.; Esposito, S. An Overview of the Obese-Asthma Phenotype in Children. Int. J. Environ. Res. Public Health 2022, 19, 636.
- Diaz, J.; Farzan, S. Clinical implications of the obese-asthma phenotypes. Immunol. Allergy Clin. North Am. 2014, 34, 739–751.
- Sikorska-Szaflik, H.; Połomska, J.; Sozańska, B. The Impact of Dietary Intervention in Obese Children on Asthma Prevention and Control. Nutrients 2022, 14, 4322.
- Mangova, M.; Lipek, T.; Vom Hove, M.; Körner, A.; Kiess, W.; Treudler, R.; Prenzel, F. Obesity-associated asthma in childhood. Allergol. Select. 2020, 4, 76–85.
- Dooley, A.A.; Pillai, D.K. Paediatric obesity-related asthma: Disease burden and effects on pulmonary physiology. Paediatr. Respir. Rev. 2021, 37, 15–17.
- Forno, E.; Han, Y.Y.; Mullen, J.; Celedón, J.C. Overweight, Obesity, and Lung Function in Children and Adults-A Meta-analysis. J. Allergy Clin. Immunol. Pract. 2018, 6, 570–581.e510.
- Forno, E.; Weiner, D.J.; Mullen, J.; Sawicki, G.; Kurland, G.; Han, Y.Y.; Cloutier, M.M.; Canino, G.; Weiss, S.T.; Litonjua, A.A.; et al. Obesity and Airway Dysanapsis in Children with and without Asthma. Am. J. Respir. Crit. Care Med. 2017, 195, 314–323.
- Carr, T.F.; Granell, R.; Stern, D.A.; Guerra, S.; Wright, A.; Halonen, M.; Henderson, J.; Martinez, F.D. High Insulin in Early Childhood Is Associated with Subsequent Asthma Risk Independent of Body Mass Index. J Allergy Clin. Immunol. Pract. 2022, 10, 785–792.e785.
- Forno, E.; Han, Y.Y.; Muzumdar, R.H.; Celedón, J.C. Insulin resistance, metabolic syndrome, and lung function in US adolescents with and without asthma. J. Allergy Clin. Immunol. 2015, 136, 304–311.e308.
- Ko, S.H.; Jeong, J.; Baeg, M.K.; Han, K.D.; Kim, H.S.; Yoon, J.S.; Kim, H.H.; Kim, J.T.; Chun, Y.H. Lipid profiles in adolescents with and without asthma: Korea National Health and nutrition examination survey data. Lipids Health Dis. 2018, 17, 158.
- Lim, J.E.; Kim, H.M.; Kim, J.H.; Baek, H.S.; Han, M.Y. Association between dyslipidemia and asthma in children: A systematic review and multicenter cohort study using a common data model. Clin. Exp. Pediatr. 2023, 66, 357–365.
- Brustad, N.; Bønnelykke, K.; Chawes, B. Dietary prevention strategies for childhood asthma. Pediatr. Allergy Immunol. 2023, 34, e13984.
- Tobias, T.A.M.; Wood, L.G.; Rastogi, D. Carotenoids, fatty acids and disease burden in obese minority adolescents with asthma. Clin. Exp. Allergy 2019, 49, 838–846.
- Lautenbacher, L.A.; Jariwala, S.P.; Markowitz, M.E.; Rastogi, D. Vitamin D and pulmonary function in obese asthmatic children. Pediatr. Pulmonol. 2016, 51, 1276–1283.
- Hu, M.; Zhao, X.; Liu, Y.; Zhou, H.; You, Y.; Xue, Z. Complex interplay of gut microbiota between obesity and asthma in children. Front. Microbiol. 2023, 14, 1264356.
- Dávila-Sotelo, E.; Hinojosa-Amaya, L.; Garza-Davila, C.; Botello-Hernández, E.; Garcia-Espinosa, P. Predictors of sleep pattern disturbances. Results from a third-level university hospital. Pediatr. Endocrinol. Diabetes Metab. 2022, 28, 257–262.
- Savini, S.; Ciorba, A.; Bianchini, C.; Stomeo, F.; Corazzi, V.; Vicini, C.; Pelucchi, S. Assessment of obstructive sleep apnoea (OSA) in children: An update. Acta Otorhinolaryngol. Ital. 2019, 39, 289–297.
- Alsubie, H.S.; BaHammam, A.S. Obstructive Sleep Apnoea: Children are not little Adults. Paediatr. Respir. Rev. 2017, 21, 72–79.
- Bhattacharjee, E.B.; Sun, X.; Malhotra, A.; Tantisira, K.G.; Landeo-Gutierrez, J.S.; Jain, S.; Bhattacharjee, R. Association of body anthropometry and obstructive sleep apnea in children: Variations observed in Hispanic children. Obes. Sci. Pract. 2023, 9, 210–217.
- Saint-Fleur, A.L.; Christophides, A.; Gummalla, P.; Kier, C. Much Ado about Sleep: Current Concepts on Mechanisms and Predisposition to Pediatric Obstructive Sleep Apnea. Children 2021, 8, 1032.
- Xiao, L.; Su, S.; Liang, J.; Jiang, Y.; Shu, Y.; Ding, L. Analysis of the Risk Factors Associated With Obstructive Sleep Apnea Syndrome in Chinese Children. Front. Pediatr. 2022, 10, 900216.
- Andersen, I.G.; Holm, J.C.; Homøe, P. Impact of weight-loss management on children and adolescents with obesity and obstructive sleep apnea. Int. J. Pediatr. Otorhinolaryngol. 2019, 123, 57–62.
- Morąg, B.; Kozubek, P.; Gomułka, K. Obesity and Selected Allergic and Immunological Diseases-Etiopathogenesis, Course and Management. Nutrients 2023, 15, 3813.
- Agón-Banzo, P.J.; Sanmartin, R.; García-Malinis, A.J.; Hernández-Martín, Á.; Puzo, J.; Doste, D.; Pardos, C.; Gilaberte, Y. Body mass index and serum lipid profile: Association with atopic dermatitis in a paediatric population. Australas. J. Dermatol. 2020, 61, e60–e64.
- Zhou, J.; Luo, F.; Han, Y.; Lou, H.; Tang, X.; Zhang, L. Obesity/overweight and risk of allergic rhinitis: A meta-analysis of observational studies. Allergy 2020, 75, 1272–1275.
- Hayashi, K.; Tsujiguchi, H.; Hori, D.; Yamada, Y.; Shimizu, Y.; Nguyen, T.T.T.; Hibino, Y.; Kambayashi, Y.; Hara, A.; Nakamura, H. The association between overweight and prevalence of food allergy in Japanese children: A cross-sectional study. Environ. Health Prev. Med. 2021, 26, 44.
- Koenigsberg, R.; Gupta, S.; Slaven, J.E.; Sarin, T.; Vitalpur, G. Body mass index in relation to symptom presentation on diagnosis of eosinophilic esophagitis in children. Ann. Allergy Asthma. Immunol. 2023, 131, 482–486.
- Gaier, E.D.; Heidary, G. Pediatric Idiopathic Intracranial Hypertension. Semin. Neurol. 2019, 39, 704–710.
- Barmherzig, R.; Szperka, C.L. Pseudotumor Cerebri Syndrome in Children. Curr. Pain Headache Rep. 2019, 23, 58.
- Paley, G.L.; Sheldon, C.A.; Burrows, E.K.; Chilutti, M.R.; Liu, G.T.; McCormack, S.E. Overweight and obesity in pediatric secondary pseudotumor cerebri syndrome. Am. J. Ophthalmol. 2015, 159, 344–352.e341.
- Matthews, Y.Y.; Dean, F.; Lim, M.J.; Mclachlan, K.; Rigby, A.S.; Solanki, G.A.; White, C.P.; Whitehouse, W.P.; Kennedy, C.R. Pseudotumor cerebri syndrome in childhood: Incidence, clinical profile and risk factors in a national prospective population-based cohort study. Arch. Dis. Child. 2017, 102, 715–721.
- Mahajnah, M.; Genizi, J.; Zahalka, H.; Andreus, R.; Zelnik, N. Pseudotumor Cerebri Syndrome: From Childhood to Adulthood Risk Factors and Clinical Presentation. J. Child. Neurol. 2020, 35, 311–316.
- Farello, G.; Ferrara, P.; Antenucci, A.; Basti, C.; Verrotti, A. The link between obesity and migraine in childhood: A systematic review. Ital. J. Pediatr. 2017, 43, 27.
- Tarantino, S.; Papetti, L.; Di Stefano, A.; Messina, V.; Ursitti, F.; Ferilli, M.A.N.; Sforza, G.; Moavero, R.; Vigevano, F.; Gentile, S.; et al. Anxiety, Depression, and Body Weight in Children and Adolescents With Migraine. Front. Psychol. 2020, 11, 530911.
- Walter, S.M.; Dai, Z.; Wang, K. Obesity, Migraine, and Overlapping Comorbidities in a Rural Pediatric Population. J. Neurosci. Rural. Pract. 2021, 12, 524–529.
- Manohar, N.; Hayen, A.; Fahey, P.; Arora, A. Obesity and dental caries in early childhood: A systematic review and meta-analyses. Obes. Rev. 2020, 21, e12960.
- Lock, N.C.; Susin, C.; Damé-Teixeira, N.; Maltz, M.; Alves, L.S. Sex differences in the association between obesity and gingivitis among 12-year-old South Brazilian schoolchildren. J. Periodontal. Res. 2020, 55, 559–566.
- Goodson, J.M. Disease reciprocity between gingivitis and obesity. J. Periodontol. 2020, 91 (Suppl. 1), S26–S34.
- Panagiotou, E.; Agouropoulos, A.; Vadiakas, G.; Pervanidou, P.; Chouliaras, G.; Kanaka-Gantenbein, C. Oral health of overweight and obese children and adolescents: A comparative study with a multivariate analysis of risk indicators. Eur. Arch. Paediatr. Dent. 2021, 22, 861–868.
- Alshihri, A.A.; Rogers, H.J.; Alqahtani, M.A.; Aldossary, M.S. Association between Dental Caries and Obesity in Children and Young People: A Narrative Review. Int. J. Dent. 2019, 2019, 9105759.
- Guaré, R.O.; Perez, M.M.; Novaes, T.F.; Ciamponi, A.L.; Gorjão, R.; Diniz, M.B. Overweight/obese children are associated with lower caries experience than normal-weight children/adolescents. Int. J. Paediatr. Dent. 2019, 29, 756–764.
- García Pérez, A.; Barrera Ortega, C.C.; González-Aragón Pineda, Á.; Villanueva Gutiérrez, T.; Pérez Pérez, N.G.; Calderon Uriostegui, D. An inverse relationship between obesity and dental caries in Mexican schoolchildren: A cross-sectional study. Public Health 2020, 180, 163–167.
- Sapunarova, P.T.; Nihtyanova, T.I.; Petrova, S.G.; Kukleva, M.P. Oral Hygiene Status and Gingivitis in Overweight and Obese Children. Folia. Med. 2019, 61, 594–599.
- Umano, G.R.; Pistone, C.; Tondina, E.; Moiraghi, A.; Lauretta, D.; Miraglia Del Giudice, E.; Brambilla, I. Pediatric Obesity and the Immune System. Front. Pediatr. 2019, 7, 487.
- Räisänen, L.; Lommi, S.; Engberg, E.; Kolho, K.L.; Viljakainen, H. Central obesity in school-aged children increases the likelihood of developing paediatric autoimmune diseases. Pediatr. Obes. 2022, 17, e12857.
- Schreiner, T.G.; Genes, T.M. Obesity and Multiple Sclerosis-A Multifaceted Association. J. Clin. Med. 2021, 10, 2689.
- Huppke, B.; Ellenberger, D.; Hummel, H.; Stark, W.; Röbl, M.; Gärtner, J.; Huppke, P. Association of Obesity With Multiple Sclerosis Risk and Response to First-line Disease Modifying Drugs in Children. JAMA Neurol. 2019, 76, 1157–1165.
- Harroud, A.; Mitchell, R.E.; Richardson, T.G.; Morris, J.A.; Forgetta, V.; Davey Smith, G.; Baranzini, S.E.; Richards, J.B. Childhood obesity and multiple sclerosis: A Mendelian randomization study. Mult. Scler. 2021, 27, 2150–2158.
- Pakpoor, J.; Schmierer, K.; Cuzick, J.; Giovannoni, G.; Dobson, R. Estimated and projected burden of multiple sclerosis attributable to smoking and childhood and adolescent high body-mass index: A comparative risk assessment. Int. J. Epidemiol. 2021, 49, 2051–2057.
- Zakharova, I.; Klimov, L.; Kuryaninova, V.; Nikitina, I.; Malyavskaya, S.; Dolbnya, S.; Kasyanova, A.; Atanesyan, R.; Stoyan, M.; Todieva, A.; et al. Vitamin D Insufficiency in Overweight and Obese Children and Adolescents. Front. Endocrinol. 2019, 10, 103.
- Corsello, A.; Macchi, M.; D’Oria, V.; Pigazzi, C.; Alberti, I.; Treglia, G.; De Cosmi, V.; Mazzocchi, A.; Agostoni, C.; Milani, G.P. Effects of vitamin D supplementation in obese and overweight children and adolescents: A systematic review and meta-analysis. Pharmacol. Res. 2023, 192, 106793.
- Tayde, A.; Mittal, M.; Khadgawat, R.; Sharma, S.; Sreenivas, V.; Rai, A. Response to single oral dose vitamin D in obese vs non-obese vitamin D-deficient children. Eur. J. Pediatr. 2021, 180, 1043–1050.
- Murdaca, G.; Tonacci, A.; Negrini, S.; Greco, M.; Borro, M.; Puppo, F.; Gangemi, S. Emerging role of vitamin D in autoimmune diseases: An update on evidence and therapeutic implications. Autoimmun. Rev. 2019, 18, 102350.
- Mailhot, G.; White, J.H. Vitamin D and Immunity in Infants and Children. Nutrients 2020, 12, 1233.
- Mohan, M.; Cherian, J.J.; Sharma, A. Exploring links between vitamin D deficiency and COVID-19. PLoS Pathog. 2020, 16, e1008874.
- Miettinen, M.E.; Niinistö, S.; Erlund, I.; Cuthbertson, D.; Nucci, A.M.; Honkanen, J.; Vaarala, O.; Hyöty, H.; Krischer, J.P.; Knip, M.; et al. Serum 25-hydroxyvitamin D concentration in childhood and risk of islet autoimmunity and type 1 diabetes: The TRIGR nested case-control ancillary study. Diabetologia 2020, 63, 780–787.
- Mäkinen, M.; Simell, V.; Mykkänen, J.; Ilonen, J.; Veijola, R.; Hyöty, H.; Knip, M.; Simell, O.; Toppari, J.; Hermann, R. An increase in serum 25-hydroxyvitamin D concentrations preceded a plateau in type 1 diabetes incidence in Finnish children. J. Clin. Endocrinol. Metab. 2014, 99, E2353–E2356.
- Mangat, G.; Nair, N.; Barat, O.; Abboud, B.; Pais, P.; Bagga, S.; Raina, R. Obesity-related glomerulopathy in children: Connecting pathophysiology to clinical care. Clin. Kidney J. 2023, 16, 611–618.
- Correia-Costa, L.; Azevedo, A.; Caldas Afonso, A. Childhood Obesity and Impact on the Kidney. Nephron 2019, 143, 8–11.
- Marzuillo, P.; Grandone, A.; Di Sessa, A.; Guarino, S.; Diplomatico, M.; Umano, G.R.; Polito, C.; La Manna, A.; Perrone, L.; Miraglia Del Giudice, E. Anthropometric and Biochemical Determinants of Estimated Glomerular Filtration Rate in a Large Cohort of Obese Children. J. Ren. Nutr. 2018, 28, 359–362.
- Vivante, A.; Golan, E.; Tzur, D.; Leiba, A.; Tirosh, A.; Skorecki, K.; Calderon-Margalit, R. Body mass index in 1.2 million adolescents and risk for end-stage renal disease. Arch. Intern. Med. 2012, 172, 1644–1650.
- Yim, H.E.; Yoo, K.H. Obesity and chronic kidney disease: Prevalence, mechanism, and management. Clin. Exp. Pediatr. 2021, 64, 511–518.
- Radosz, A.; Obuchowicz, A. Lipid metabolism and renal function markers in obese adolescents. Pediatr. Endocrinol. Diabetes Metab. 2023, 29, 91–96.
- Di Sessa, A.; Guarino, S.; Umano, G.R.; Arenella, M.; Alfiero, S.; Quaranta, G.; Miraglia Del Giudice, E.; Marzuillo, P. MAFLD in Obese Children: A Challenging Definition. Children 2021, 8, 247.
- Zdanowicz, K.; Daniluk, J.; Lebensztejn, D.M.; Daniluk, U. The Etiology of Cholelithiasis in Children and Adolescents—A Literature Review. Int. J. Mol. Sci. 2022, 23, 13376.
- Cook, M.B.; Freedman, N.D.; Gamborg, M.; Sørensen, T.I.; Baker, J.L. Childhood body mass index in relation to future risk of oesophageal adenocarcinoma. Br. J. Cancer 2015, 112, 601–607.
- Tambucci, R.; Quitadamo, P.; Ambrosi, M.; De Angelis, P.; Angelino, G.; Stagi, S.; Verrotti, A.; Staiano, A.; Farello, G. Association Between Obesity/Overweight and Functional Gastrointestinal Disorders in Children. J. Pediatr. Gastroenterol. Nutr. 2019, 68, 517–520.
- Krawczyk, M.; Kułaga, Z.; Niewiadomska, O.; Jankowska, I.; Lebensztejn, D.; Więcek, S.; Socha, P. Are children with gallstone disease more overweight? Results of a matched case-control analysis. Clin. Res. Hepatol. Gastroenterol. 2023, 47, 102204.
- Cabrera Chamorro, C.C.; Pabón Arteaga, J.S.; Caicedo Paredes, C.A.; Cabrera Bravo, N.; Villamil Giraldo, C.E.; Chávez Betancourt, G.; Zarama Márquez, R.A.; Rincón Torres, C.A. Cholelithiasis and associated complications in pediatric patients. Cir. Pediatr. 2020, 33, 172–176.
- Zdanowicz, K.; Ryzko, J.; Bobrus-Chociej, A.; Wojtkowska, M.; Lebensztejn, D.M. The role of chemerin in the pathogenesis of cholelithiasis in children and adolescents. J. Paediatr. Child Health 2021, 57, 371–375.
- Ba, H.J.; Xu, L.L.; Qin, Y.Z.; Chen, H.S. Serum Chemerin Levels Correlate With Determinants of Metabolic Syndrome in Obese Children and Adolescents. Clin. Med. Insights Pediatr. 2019, 13, 1179556519853780.
- Donnelly, S. Gallstone Disease and Cholecystectomy in Children and Adolescents With Obesity. J. Pediatr. Surg. Nurs. 2019, 8, 55–57.
- Frybova, B.; Drabek, J.; Lochmannova, J.; Douda, L.; Hlava, S.; Zemkova, D.; Mixa, V.; Kyncl, M.; Zeman, L.; Rygl, M.; et al. Cholelithiasis and choledocholithiasis in children; risk factors for development. PLoS ONE 2018, 13, e0196475.
- Parra-Landazury, N.M.; Cordova-Gallardo, J.; Méndez-Sánchez, N. Obesity and Gallstones. Visc. Med. 2021, 37, 394–402.
- Heida, A.; Koot, B.G.; vd Baan-Slootweg, O.H.; Pels Rijcken, T.H.; Seidell, J.C.; Makkes, S.; Jansen, P.L.; Benninga, M.A. Gallstone disease in severely obese children participating in a lifestyle intervention program: Incidence and risk factors. Int. J. Obes. 2014, 38, 950–953.
- Wang, J.; Xu, C.; Cheng, Q.; Zhao, J.; Wu, S.; Li, W.; Ma, W.; Liu, C.; Jiang, X. RNA Sequencing Revealed Signals of Evolution From Gallbladder Stone to Gallbladder Carcinoma. Front. Oncol. 2020, 10, 823.
- Hatia, R.I.; Eluri, M.; Hawk, E.T.; Shalaby, A.; Karatas, E.; Abdelhakeem, A.; Abdel-Wahab, R.; Chang, P.; Rashid, A.; Jalal, P.K.; et al. Independent of Primary Sclerosing Cholangitis and Cirrhosis, Early Adulthood Obesity Is Associated with Cholangiocarcinoma. Cancer Epidemiol. Biomarkers Prev. 2023, 32, 1338–1347.
- Oze, I.; Ito, H.; Koyanagi, Y.N.; Abe, S.K.; Rahman, M.S.; Islam, M.R.; Saito, E.; Gupta, P.C.; Sawada, N.; Tamakoshi, A.; et al. Obesity is associated with biliary tract cancer mortality and incidence: A pooled analysis of 21 cohort studies in the Asia Cohort Consortium. Int. J. Cancer. 2024, 154, 1174–1190.
- Morales Camacho, W.J.; Molina Díaz, J.M.; Plata Ortiz, S.; Plata Ortiz, J.E.; Morales Camacho, M.A.; Calderón, B.P. Childhood obesity: Aetiology, comorbidities, and treatment. Diabetes Metab. Res. Rev. 2019, 35, e3203.
- Lang, J.E.; Hossain, J.; Holbrook, J.T.; Teague, W.G.; Gold, B.D.; Wise, R.A.; Lima, J.J. Gastro-oesophageal reflux and worse asthma control in obese children: A case of symptom misattribution? Thorax 2016, 71, 238–246.
- Pogodina, A.; Romanitsa, A.; Rychkova, L. Functional Bowel Disorders and Obesity in Children: State of the Problem. Int. J. Biomed. 2020, 10, 316–323.
- Molina-Garcia, P.; Migueles, J.H.; Cadenas-Sanchez, C.; Esteban-Cornejo, I.; Mora-Gonzalez, J.; Rodriguez-Ayllon, M.; Plaza-Florido, A.; Vanrenterghem, J.; Ortega, F.B. A systematic review on biomechanical characteristics of walking in children and adolescents with overweight/obesity: Possible implications for the development of musculoskeletal disorders. Obes. Rev. 2019, 20, 1033–1044.
- Steinberg, N.; Nemet, D.; Pantanowitz, M.; Eliakim, A. Gait Pattern, Impact to the Skeleton and Postural Balance in Overweight and Obese Children: A Review. Sports 2018, 6, 75.
- Merder-Coşkun, D.; Uzuner, A.; Keniş-Coşkun, Ö.; Çelenlioğlu, A.E.; Akman, M.; Karadağ-Saygı, E. Relationship between obesity and musculoskeletal system findings among children and adolescents. Turk. J. Phys. Med. Rehabil. 2017, 63, 207–214.
- Maciałczyk-Paprocka, K.; Stawińska-Witoszyńska, B.; Kotwicki, T.; Sowińska, A.; Krzyżaniak, A.; Walkowiak, J.; Krzywińska-Wiewiorowska, M. Prevalence of incorrect body posture in children and adolescents with overweight and obesity. Eur. J. Pediatr. 2017, 176, 563–572.
- Ferrer, F.S.; Castell, E.C.; Marco, F.C.; Ruiz, M.J.; Rico, J.A.Q.; Roca, A.P.N. Influence of weight status on bone mineral content measured by DXA in children. BMC Pediatr. 2021, 21, 185.
- Kim, S.J.; Ahn, J.; Kim, H.K.; Kim, J.H. Obese children experience more extremity fractures than nonobese children and are significantly more likely to die from traumatic injuries. Acta Paediatr. 2016, 105, 1152–1157.
- Franceschi, R.; Radetti, G.; Soffiati, M.; Maines, E. Forearm Fractures in Overweight-Obese Children and Adolescents: A Matter of Bone Density, Bone Geometry or Body Composition? Calcif. Tissue Int. 2022, 111, 107–115.
- Hirt, P.A.; Castillo, D.E.; Yosipovitch, G.; Keri, J.E. Skin changes in the obese patient. J. Am. Acad. Dermatol. 2019, 81, 1037–1057.
- Gupta, M. Cutaneous manifestations of obesity in children: A prospective study. Indian J. Paediatr. Dermatol. 2017, 18, 28–30.
- Hasse, L.; Jamiolkowski, D.; Reschke, F.; Kapitzke, K.; Weiskorn, J.; Kordonouri, O.; Biester, T.; Ott, H. Pediatric obesity and skin disease: Cutaneous findings and associated quality-of-life impairments in 103 children and adolescents with obesity. Endocr. Connect. 2023, 12, e230235.
- Güven, M.; Anık, A.; Ünüvar, T.; İlgün Gürel, D.; Şendur, N. Cutaneous manifestations of obesity in Turkish children: A comparative study. Pediatr. Dermatol. 2022, 39, 513–519.
- Balgobind, A.; Finelt, N.; Strunk, A.; Garg, A. Association between obesity and hidradenitis suppurativa among children and adolescents: A population-based analysis in the United States. J. Am. Acad. Dermatol. 2020, 82, 502–504.
- Jørgensen, A.R.; Aarestrup, J.; Baker, J.L.; Thomsen, S.F. Association of Birth Weight, Childhood Body Mass Index, and Height With Risk of Hidradenitis Suppurativa. JAMA Dermatol. 2020, 156, 746–753.
- Phan, K.; Lee, G.; Fischer, G. Pediatric psoriasis and association with cardiovascular and metabolic comorbidities: Systematic review and meta-analysis. Pediatr. Dermatol. 2020, 37, 661–669.
- Shreberk-Hassidim, R.; Galili, E.; Hassidim, A.; Ramot, Y.; Merdler, I.; Baum, S.; Zlotogorski, A.; Barzilai, A.; Astman, N. Epidemiology and Comorbidities of Psoriasis among Israeli Adolescents: A Large Cross-Sectional Study. Dermatology 2019, 235, 488–494.
- Darlenski, R.; Mihaylova, V.; Handjieva-Darlenska, T. The Link Between Obesity and the Skin. Front. Nutr. 2022, 9, 855573.
- Sadowska-Przytocka, A.; Gruszczyńska, M.; Ostałowska, A.; Antosik, P.; Czarnecka-Operacz, M.; Adamski, Z.; Łącka, K. Insulin resistance in the course of acne—Literature review. Postepy Dermatol. Alergol. 2022, 39, 231–238.
- Svoboda, S.A.; Shields, B.E. Cutaneous Manifestations of Nutritional Excess: Pathophysiologic Effects of Hyperglycemia and Hyperinsulinemia on the Skin. Cutis 2021, 107, 74–78.
More
Information
Subjects:
Endocrinology & Metabolism
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
609
Revisions:
2 times
(View History)
Update Date:
28 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





