Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Fernando Baquero | -- | 4028 | 2024-02-23 21:08:53 | | | |
| 2 | Camila Xu | Meta information modification | 4028 | 2024-02-26 01:47:44 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Baquero, F.; Rodríguez-Beltrán, J.; Coque, T.M.; Del Campo, R. Fitness Costs Antibiotic Resistance in the Gut. Encyclopedia. Available online: https://encyclopedia.pub/entry/55402 (accessed on 29 March 2026).
Baquero F, Rodríguez-Beltrán J, Coque TM, Del Campo R. Fitness Costs Antibiotic Resistance in the Gut. Encyclopedia. Available at: https://encyclopedia.pub/entry/55402. Accessed March 29, 2026.
Baquero, Fernando, Jerónimo Rodríguez-Beltrán, Teresa M. Coque, Rosa Del Campo. "Fitness Costs Antibiotic Resistance in the Gut" Encyclopedia, https://encyclopedia.pub/entry/55402 (accessed March 29, 2026).
Baquero, F., Rodríguez-Beltrán, J., Coque, T.M., & Del Campo, R. (2024, February 23). Fitness Costs Antibiotic Resistance in the Gut. In Encyclopedia. https://encyclopedia.pub/entry/55402
Baquero, Fernando, et al. "Fitness Costs Antibiotic Resistance in the Gut." Encyclopedia. Web. 23 February, 2024.
Copy Citation
The acquisition of antibiotic resistance by horizontal gene transfer and by mutational changes in the chromosome or genes located in mobile genetic elements, followed by antibiotic resistance phenotype expression, implies modification of the cellular physiological status, including homeostatic adaptations of the previously susceptible cell.
fitness cost antibiotic resistance
bacterial stress in the gut
antibiotic susceptibility restoration
1. Introduction
The acquisition of antibiotic resistance by horizontal gene transfer and by mutational changes in the chromosome or genes located in mobile genetic elements, followed by antibiotic resistance phenotype expression, implies modification of the cellular physiological status, including homeostatic adaptations of the previously susceptible cell [1]. Thus, in most cases, resistance imposes a type of stress, eventually resulting in a fitness cost, i.e., the growth rate of resistant bacteria might decrease. This cost is absent in susceptible bacteria; thus, hypothetically, the susceptible population is expected to prevail in the medium to long term. Unfortunately, the fitness cost of antibiotic resistance can be reduced after a certain period by the acquisition of compensatory mutations and possibly by phenotypic–epigenetic adaptations.
2. Stress as a Relative Concept: The Case of Intestinal Microbiota
In nature, life is “struggle for life”; i.e., there is no utopia for microorganisms. Even those that are well adapted to their natural niches and that maintain their population in steady-state equilibrium fight against a multiplicity of stresses and do not grow as rapidly as those grown under optimal laboratory conditions (pure cultures, rich media, and controlled physical–chemical variables). Stress might be consequent to changes in the environmental physical–chemical conditions of the occupied niche, defined as a multidimensional environmental space characterized by a variety of conditions, both biotic and abiotic, whose quantitative ranges determine the positive or negative growth rates of the bacterial species [2][3]. Negative growth rates are frequently part of the stress phenotype. The altered-niche hypothesis as a source of stress for the occupant bacterial population can be extended to the whole ecosystem [4][5], including the entire microbiota. The intestine is a flowing open environment (an “invironment”) subject to the host’s circadian rhythms and perhaps influencing the microbiota over a 24 h cycle [6]. It could be true that the most host-adapted populations (the phyla Bacteroidota and Bacillota), with the highest densities in the normal human or most mammals’ microbiota, are generally those that constitute the older population in the co-evolutionary history of intestinal microbial colonization [7]. Thus, they are able to cope with ample ranges of changing conditions and are thereby subject to less stress. Abrupt changes in conditions (i.e., particular biomolecules or chemical conditions) might produce more stress than smooth changes, which frequently require simpler modifications. Most potential human pathogens that are frequently present in the human microbiota, such as the gamma-proteobacteria (Enterobacterales) Escherichia coli, Klebsiella, Serratia, or Enterobacter, constitute a very small proportion of the microbiota (on average less than 1%). Some of these, such as E. coli, are likely of more recent evolutionary acquisition in vertebrates, including mammals [8][9].
However, subpopulations of commensal opportunistic pathogens are frequently involved in urinary tract and bloodstream infections, producing outbreaks both in hospitals and community settings [10]. The proportion of these organisms (also Enterococcus) in the microbiota increases in aged and hospitalized individuals and in people from low-income countries with inadequate sanitation [11][12]. Consequently, the acquisition of antibiotic resistance traits by pathogenic clones of these species is of particular clinical relevance. This change could result in a disadvantage in relation to antibiotic susceptible populations that might replace the resistant ones. This outcome has been confirmed by fecal microbiota transplantation where antibiotic-resistant high-risk clones, were replaced by susceptible clones from the fecal donor [13][14][15]. A schema of the different stresses to which potentially pathogenic bacteria are exposed during their transit in the intestinal tract is presented in Figure 1.
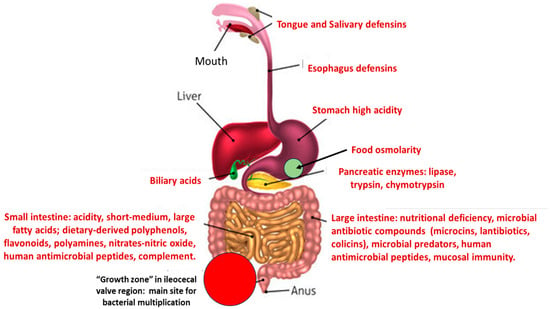
Figure 1. A schema of the main stresses to which potentially pathogenic/antibiotic resistant bacteria are confronted in the human gut, producing a reduction in fitness.
3. The Main Sources of Bacterial Stress in the Intestinal Microbiota
3.1. Acid Stress
One of the first sources of stress faced by bacterial organisms after ingestion is gastric acidity. Under fasting conditions, hydrochloric acid in gastric juice has a highly aggressive pH of 2, whereas Enterobacteriaceae have an optimal pH in the neutral range, between 6.5 and 7.5 (pH 7 for E. coli). Intraluminal acidity is not exclusive to the stomach; the distal duodenum has an acid environment (pH 6), maintained by lactic acid bacteria, which increases until reaching an optimal pH of 7.4 in the terminal ileum, probably where most microbiota growth takes place. Again, acidity increases (probably because of the bacterial production of acids) to a pH of 5.7 in the cecum, ultimately reaching a pH of 6.7 in the rectum. Some pathological conditions might increase intestinal acidity, such as ileocecal resection, chronic pancreatitis, cystic fibrosis, ulcerative colitis, or Crohn’s disease [16]. This increase could be a direct consequence of the altered microbiota, which also occurs with the use of probiotics; in both cases, organic acids are involved. Organic short-chain carboxylic acids, such as fumaric, propionic, acetic, lactic, and butyric acids, are frequently produced by intestinal microorganisms, lowering the pH, which results in stress for a number of bacterial populations [17].
Adaptation to acid stress is an important factor for the transmission of intestinal microbes. In E. coli, resistance to acid stress is guaranteed by the GadE-regulated expression of glutamate and arginine decarboxylases associated with amino acid and Cl−/H+ antiporters [18][19]. Acid stress also forces E. coli to alter the envelope structure and porins in the outer membrane and the cytoplasmic chaperones [20]. Changes in the envelope architecture and in the molecular folding have epistatic consequences altering cellular functions [21]. In fact, there are several overlapping acid survival systems with variable expression and efficacy depending on the growth phase [22][23]. Resistance in E. coli to short-chain organic carboxylic acids also involves changes in the rpoA (influencing folding efficiency and/or chaperone-like activity), rpoC (subunit of RNA polymerase), and rpoS (alternative sigma factor inducing stationary phase) involved in stress response cascades, and probably rho (transcription regulation) and nagA (N-acetyl-d-glucosamine metabolism) [24][25].
3.2. Bile Stress
Bile is stored in the gallbladder and flows into the duodenum by the common bile duct. Taurocholate, glycocholate, and glycochenodeoxycholate are the main bile salts, acting on bacterial membranes of several microorganisms and resulting in potent antimicrobial activity, mostly derived from the highly lipophilic steroid ring. Bile salts contribute to the host’s resistance to upper intestinal bacterial colonization. In the intestine, primary bile acids are susceptible to microbial-mediated oxidation, dihydroxylation, and epimerization, giving rise to the secondary bile acids deoxycholic and lithocholic acid. Bacterial stress derives from cell envelope stress, dissociation of integral membrane proteins, action on membrane lipids, alteration of nutrient uptake, reactive oxygen species-derived nucleic acid damage, and protein misfolding, eventually leading to a bactericidal effect [26][27]. In general, Bacillota, which include the opportunistic pathogen Enterococcus faecalis [28], are more sensitive to the deleterious effects of bile than Enterobacterales, given that the outer membrane’s lipopolysaccharide acts as a protection shield. However, there is also severe bacterial stress in this enteric group, inducing DNA damage, SOS gene stress, and hypermutation [29]. General stress proteins are expressed during E. faecalis bile salt treatment, including molecular chaperones and protectors of DNA-damaging peroxides [27]. Lastly, bilirubin excreted by the bile can have an antibacterial effect on Gram-negative bacteria [30].
Antibiotic-susceptible bacteria respond to the challenge of stress using bile efflux pumps for overexpression of MdtEF-TolC, particularly in acid medium, but at the expense of a fitness cost [31], bile salt hydrolase enzyme, and rewiring the intracellular metabolism and the cell membrane composition [32]. In E. coli, some mutations are bile-hypersensitive such as in AcrAB, EmrAB, and MdtABCD efflux pumps; in OmpF/OmpC outer membrane porin; in HupAB DNA-binding protein (involved in DNA supercoiling); and in genes biosynthesizing the core lipopolysaccharide, showing their effect on bile-resistance. The E. coli SOS gene, dinF, which protects against oxidative stress, also protects from the effect of bile salts [33]. The membrane damage sensors, Cpx and RcsCb, regulate and induce the expression of genes involved in bile stress responses [27].
3.3. Stress by Pancreatic Enzymes
Digestive enzymes such as amylase, lipase, trypsin, and chymotrypsin are released from the pancreatic acini cells (exocrine glands) and flow into the pancreatic duct to reach the duodenum. Lipases might have antibacterial activity, preferentially in Bacillota [34]. Trypsin and chymotrypsin, preferentially in combination, can hydrolyze bacterial outer-membrane proteins in Gram-negative organisms and damage the integrity of surface structures in Bacillota [35]. The extent of these effects in the large intestine is counteracted by trypsin degradation by commensal bacteria [36].
3.4. Stress by Short- and Long-Chain Fatty Acids
Intestinal short-chain fatty acids (2–6 carbons in length) are mostly produced by microorganisms acting on carbohydrates and polyphenols, and these compounds have significant effects in terms of reducing optimal bacterial fitness. Bacillota are mainly butyrate producers, whereas Bacteroidetes excrete acetate and propionate [37][38]. Although these effects are in part due to the reduction in pH (previously treated), they also have a pH-independent antibacterial mode of action. The long-chain fatty acids (12–20 carbons in length) present in the intestinal lumen originate from the host cells, the microbiota, and from dietary sources. The most abundant of these are unsaturated fatty acids, such as oleic and linoleic acids, and saturated fatty acids, such as stearic or palmitic acid. Free fatty acids are bound and further enzymatically released from other compounds, such as glycerol, sugars, or phosphate headgroups, to form lipids [39]. Their lipophilic nature allows them to invade and damage microbial membranes, ultimately leading to a lethal effect, particularly in Bacillota. Gram-negative bacteria such as Enterobacterales are protected in part due to their lipopolysaccharide layer in the outer membrane. However, Enterobacterales might be able to sense extracellular long-chain fatty acids by using a 2-component system that influences gene regulation. Consequently, general metabolism, type 3 secretion systems, or the gene network involved in motility, fimbriae synthesis, and biofilm formation, can be modified, influencing global bacterial fitness [40]. Such effects on gene expression are in part linked to the fact that fatty acids might mimic diffusible signal factors [41].
3.5. Stress by Dietary Compounds
Food–microbiota interaction is one of the cornerstones of intestinal physiology [42]. Among the roles of the microbiota in the assimilation of nutrients by herbivore animals is to degrade complex vegetal molecules (such as cellulose) by symbiotic cellulolytic bacteria, release oligosaccharides, and produce absorbable short-chain fatty acids, ensuring animal nutrition. To a minor degree, intestinal bacteria in humans (such as Enterococcus, frequent in the elderly) contribute to the degradation of complex polysaccharides [43]. However, food–microbiota interactions can result in a challenge for bacterial populations. Some of these causes of stress are examined below.
3.5.1. Stress by Polyphenols
Polyphenols, complex natural molecules containing one or more hydroxylated aromatic rings, are a widely and highly distributed group of diverse natural products (probably over 10,000) found in dietary products such as fruit, vegetables, nuts, seeds, red wine, beer, olive oil, honey, coffee, and tea. Polyphenols, for instance flavonoids and tannins, have been shown to exert antibacterial effects, both in Bacillota species, such as Staphylococcus aureus, and in gamma-Proteobacteria, such as E. coli, Klebsiella pneumoniae, Acinetobacter, and Pseudomonas [44]. In addition, many of them have synergistic activity with antimicrobial agents [45][46]. The antibacterial mode of action of flavonoids appears to involve the perforation and destructuration of the bacterial cytoplasmic membrane, alteration of bacterial transporters, DNA topoisomerase inhibition, and reduction of bacterial energy metabolism by inhibition of nicotinamide adenine dinucleotide hydrogen reductase, all of which are various mechanisms that result in the formation of lethal reactive oxygen species [47][48][49][50]. Bacterial resistance to polyphenols is a poorly explored field of research; however, microorganisms can produce degraded polyphenols, activating glycosidases and esterases, isomerases, and hydrolases, giving rise to simple aromatic metabolites [49]. To which extent these activities are induced by polyphenol stress remains poorly understood.
3.5.2. Stress by Polyamines
Decarboxylation by intestinal microorganisms (mostly anaerobes, such as Bacteroides or Fusobacterium) of aromatic or polycationic amino acids results in polyaminated molecules, biogenic amines, and polyamines. Polyamines include compounds with two amino groups, such as putrescine (1,4-diaminobutane) or cadaverine (1,5-diaminopentane), but also molecules with three or four amino groups, such as spermidine [N-(3-aminopropyl)butane-1,4-diamine] and spermine [N,N′-bis(3-aminopropyl)butane-1,4-diamine], respectively. Bacteria have transport systems allowing uptake of extracellular polyamines, including the polyamine ABC transporter genes, generally organized as four-gene operons, as in the cases of potABCD (spermidine uptake) and potFGHI (putrescine uptake). These compounds have long been known as antibacterials [51], acting on Bacillota species and on those of the family Enterobacteriaceae. They alter bacterial membrane permeability and porin function, they possibly interact with nucleic acids, and these effects are likely highly concentration-dependent. In any case, they have been considered to constitute possible scaffolds for novel antimicrobials or antibiotic enhancers [52][53]. Possible mechanisms of resistance to polyamines involve mutations in these genes or downregulation of operon transcription. However, polyamines might also provide benefits for the bacteria, providing, e.g., resistance to acidity or protection against oxidative stress [54].
3.5.3. Nitric Oxide Stress, Osmolar Stress
Dietary nitrates and nitrites are widespread in food, and they are found naturally in vegetables and fruit or as food additives. They give rise in the gut to reactive nitrogen species and to a human intestinal inflammatory response. Occasionally, bacteria lead to an overproduction of nitric oxide in the gut, with potential antibacterial activity based on lipid peroxidation, nitrosation of membrane proteins, and DNA damage [55][56][57]. The cellular targets of nitric oxide and reactive nitrogen species act as signals, resulting in altered gene expression and synthesis of protective detoxifying enzymes [58]. Osmolarity essentially influences bacteria during their flow or during transient colonization of the small intestine and depends on unabsorbed meal compounds. Osmolality, the concentration of solute particles in a solution, also influences bacterial populations. There is a reduction in bacterial cell volume due to passive water excretion [59]. Bacteria adapt to osmolarity stress by accumulating solutes, such as potassium, glutamate, trehalose, proline, and glycine betaine [60].
3.6. Stress by Nutritional Deficiency
Accessible nutrients for the microbiota in the intestine are always limited, for three main reasons: (1) the host and microbiota compete for nutrients, so that only a small part of dietary food is available for the microbiota; (2) the great density of bacterial cells in the most colonized, anaerobic, and dehydrated part of the intestine, the colon, leads to inter-microbial competition for nutrients; and (3) microbial populations lost daily by defecation need replacement, so doubling time in the gut by a day or more would not be sufficient to maintain a stable population size. It had been proposed that bacterial abundance in the gut fluctuates around the stable carrying capacities of the colonizable gut [61]; thus, many bacterial populations are challenged by conditions close to starvation [62]. It should be noted that nutritional conditions vary along the intestine, being more favorable around the ileocecal valve and proximal colon, probably making it the most effective “growth zone” [63]. Bacterial nutrients from the ileum are dietary but undigested fiber polysaccharides, and secondarily host mucosal glycans and host secretions, as well as microbial exopolysaccharides and capsular material [64]. In the colon, extreme interbacterial competition for nutrients, including nitrogenated compounds and vital metals such as iron or even vitamins, also absorbed by the host, overcomes the presumed higher concentration of these nutrients by host water absorption (also deleterious substances for bacteria concentrate, increasing toxicity) and intermicrobial nutritional cooperation. Microbes in the gut have access to only 1 nitrogen atom for every 10 carbon atoms, whereas free-living organisms (let alone cultures in the lab) have access to 4 nitrogen atoms for every 4 carbon atoms [65]. Bacterial reactions to nitrogen starvation stress in E. coli include global physiological changes (stringent response) mediated by the signal molecule, guanosine tetraphosphate (ppGpp) [66]. In general, nutrient starvation, including inorganic phosphate starvation, produces similar responses, leading to bacteria entering a stationary phase [67]. A poorly explored point is how microbial nutritional starvation influences majority and minority gut populations. The populations with higher densities are probably more resilient to extinction, given that “number is a biological advantage”, as occurs under antibiotic exposure [68]. Many antibiotic-resistant pathogenic bacteria are minorities (less than 1% of the population), and in the absence of antibiotic exposure, resistant populations within a species are also minorities. However, their number generally increases in hospitalized patients.
3.7. Stress Resulting from Microbial Interactions
3.7.1. Stress by Bacterial Antimicrobial Peptides: Microcins, Lantibiotics, Colicins
Microcins are low-molecular-weight antibiotic peptides. They were distinguished in 1976 from colicins, which are higher-molecular-weight antibacterial proteins that are much less stable in the intestinal tract [69]. Microcins are ribosomally synthesized and post-translationally modified peptides (RiPPs), which are mostly produced by Enterobacteriaceae and act on members of this family of microorganisms. They have various mechanisms of action, such as producing pores in the cytoplasmic membrane (MccV, MccE492, and MccL); inhibiting the aspartyl-tRNA involved in protein synthesis (MccC), inhibiting the topoisomerase GyrB, producing double DNA breaks (MccB17); blocking the secondary RNA polymerase channel, impairing transcription and acting on cytochromes inhibiting cellular respiration (MccJ25); or altering the function of the cellular proton channel (MccH47, and possibly MccM and MccI), or the ATP synthase (MccH47). Microcin stress is followed by immunity/resistance mechanisms, including acetyltransferases (MccC), production of immunity proteins (Class IIb microcins), enhanced efflux pumps, and inhibition of DNA gyrase supercoiling activity (MccB17). There is evidence that microcins strongly influence microbial interactions in the gut [70]. The equivalent of microcins in Enterobacteriaceae are lantibiotics in Bacillota, as well as ribosomally produced and modified post-translational peptides [71]. Lantibiotics (lanthionine- and methyllanthionine-containing peptides) can produce holes in the bacterial membrane and eventually interfere with cell wall synthesis [72]. Resistance/protection from lantibiotics is mediated by the production of “immunity proteins”, specialized ABC-transport proteins, modifications in membrane composition, lantibiotic–lytic proteins, spore formation, and immune mimicry [73]. Colicins are much larger and less stable polypeptides in the intestinal environment, and they are produced and active in Enterobacteriaceae. Their mechanism of antibacterial action includes membrane pore formation, degradation of nucleic activity (DNase, 16S rRNase, and tRNase activities), and altering peptidoglycan synthesis. Resistance to colicins involves receptors and translocation mutants (Tol pathway mutants), alteration of outer membrane proteins, including ompF, exbB, and tonB mutations, and enterochelin hyperproduction [74].
3.7.2. Stress by Bacteriophages and Microbial Predators
Bacterial viruses (bacteriophages) have been postulated to be the most abundant microorganisms in the gut. However, most of these phages are prophages, or lysogenic phages that replicate with the host bacterial strain. The “free” (extracellular) phages, which are able to infect new organisms, are comparatively smaller in abundance but can locally increase in number and evolve into a bacteriolytic state when induced (activated) by stressful conditions [75]. The most abundant viral families include Myoviridae, Podoviridae, Siphoviridae, and Microviridae. Most bacterial stress produced by phage invasions derives from envelope (cytoplasmic membrane) stress, fostering a phage-shock-protein (Psp) system, occurring both in Gram-positive and Gram-negative microbes [76]. Classic mechanisms of resistance to phage invasions are alterations in bacterial surface epitopes acting as phage receptors and restriction-modification systems. There is also the production of proteins interfering with the phage infection cycle, and these include variable and evolving CRISPR sequences. There are also phenotypic mechanisms of resistance, which change the metabolic status of the cell and are similar to antibiotic persistence in bacteria [77]. Comparative stress by bacterial predators, such as protozoa, appears to have less importance. However, commensal protozoa, such as Entamoeba or Blastocystis, eating bacteria, might be important for microbiome stability in low-income human populations, particularly in the proximal gut [78]. Bacterial resistance to protozoa is analogous to resistance to phagocytosis and survival in phagolysosomes [79]. The classically described environmental predators, such as Bdellovibrio, can also be abundant in the gut. they penetrate the cell and multiply in the periplasm, killing the prey bacterium; the process is probably too rapid to produce a significant population of stressed bacteria [80].
3.8. Stress by Inflammation and Immunity
Frequently, the relationship between microbiota and the host (particularly in mucus-associated bacteria) can present as a status of “low-grade inflammation”, mostly induced by bacterial exopolysaccharides and cell wall fragments. Innate immune defense is exerted by the secretion of specialized epithelial cells (Paneth cells) of antimicrobial peptides such as α-defensins, which interact and disorganize bacterial membranes, eventually resulting in cell death [81][82]. These cells also produce other antimicrobial peptides, such as CRS4C and the lectin Reg3γ, which disrupt the cell wall [83]. Also, cathelicidins (including indolicidin), produced by intestinal epithelial cells, have significant antibacterial activity [84][85]. If the secretion of these antimicrobial peptides is constitutive, the local invasion by microorganisms could increase their concentration, so that invasive antibiotic-resistant pathogens are facing a higher stress. Mechanisms of bacterial resistance might evolve in Enterobacterales by alteration of the outer membrane lipopolysaccharide. Other molecules of the immune system, such as the complement system, are probably involved in the stress of bacterial cells in contact with the epithelium [86]. Toll-like host receptors recognize microbial-associated molecular patterns, and enterocytes express various complement components. Complement proteins are found among the bacterial-bound proteins detected in intestinal proteomic studies (Concepción Gil, personal communication), and they might kill bacteria directly via large pore-forming complexes [87]. Bacterial resistance to defensins is mediated by expressing proteins such as MprF, which harbors transmembrane domains for lipid lysinilation and defensin repulsion [88][89]. Perhaps as a consequence, MprF plays a crucial role in Staphylococcus aureus virulence and is involved in resistance to daptomycin, which is structurally similar to cationic antimicrobial peptides. Similar effects occur in Enterococcus faecium [90][91].
4. Modulating Intestinal Stress to Select for Antibiotic Susceptibility
The main purpose of this research was to examine the possibility that regulating/modulating or administering physiological molecules of the intestinal tract, which enhance gut stress, might result in fitness costs on microorganisms invading or colonizing the gut (Figure 2).
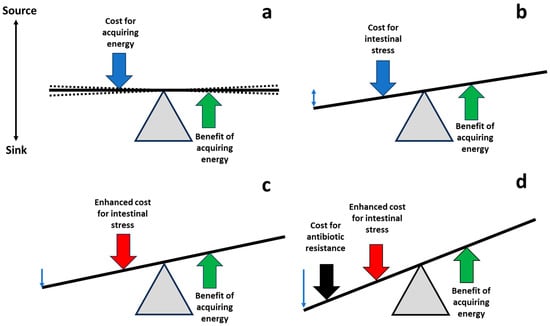
Figure 2. Shifting the balance of costs and benefits to favor antibiotic-susceptible populations. (a) Life is based on equilibrium (with oscillations, broken lines), balancing the costs of acquisition of energy required for fueling biological processes (blue arrow), and the benefits of energy investment, leading to the final goal of bacterial replication (green arrow). (b) During the process of gut invasion and colonization, potentially pathogenic/resistant bacterial populations are exposed to intestinal molecules, reducing their bacterial fitness; compensatory adaptations also contribute to this fitness cost (blue arrow). (c) Managing pharmacological physiological molecules in the gut, it could be possible to increase the cost of these populations in the intestine (red vertical arrow). (d) The increase in intestinal fitness cost might be additive or synergistic with the fitness cost associated with the expression of antibiotic resistance or the cost of harboring carriers (mobile genetic elements) of antibiotic resistance (black arrow). Thus, the antibiotic-susceptible intestinal populations of potential pathogens could have better fitness than the resistant ones, favoring a restoration of susceptibility.
If the expression of resistance mechanisms, including the carriage of mobile resistance elements, in the absence of antibiotics, challenges the bacterial physiology and produces a fitness cost [92][93], it could be of interest to know whether, in the absence of antibiotic exposure, the addition of both types of fitness cost could be untenable for antibiotic- resistant populations but not for susceptible ones. This hypothesis, which suggests that stressful conditions in the gut could exacerbate the fitness costs of resistance, is substantiated by various lines of evidence. Although the precise mechanisms underlying the fitness cost of resistance remain largely unknown, the physiological changes induced by expressing antimicrobial resistance genes overlap with those caused by previously mentioned stressors.
For instance, beta-lactamase expression produces a fitness cost due to the accumulation of enzymes in the periplasmic space, destabilizing the bacterial envelope [21][94][95]. Similarly, bacteriocins, long-chain fatty acids, bile salts, and dietary by-products (among other stressors) exert antimicrobial activity by damaging the cell’s envelope. Therefore, the physiological impact of expressing a β-lactamase and undergoing gut-associated stress will likely synergize, resulting in an unsustainable fitness cost for the antibiotic-resistant bacteria. A different situation emerges if gut stressors enhance antibiotic effectiveness, which is particularly plausible for antibiotics targeting the bacterial envelope (e.g., β-lactams and polymyxins) or those relying on reactive oxygen species for their killing mechanism (e.g., aminoglycosides and quinolones). As mentioned earlier, harsh gut conditions often impact the bacterial envelope and generate oxidative species (such as nitric oxide or bile salts), which could lead to a synergistic effect with specific antibiotics, although further demonstration of this interaction is needed.
Two aspects should be clearly differentiated. First, there is the interaction of intestinal stress with the antibiotic-provoked stress. Some studies have suggested that normal mechanisms of bacterial decontamination in the gut (such as bile production) could increase the antibacterial effect of antimicrobial drugs [96]; furthermore, mechanisms of resistance to gut antibacterial products/conditions could favor cross-resistance with antibiotics [97].
References
- Davies, K.J. Adaptive homeostasis. Mol. Asp. Med. 2016, 49, 1–7.
- Hutchinson, G.E. Concluding remarks. Cold Spring Harb. Symp. 1957, 22, 415–427.
- Baquero, F.; Coque, T.M.; Galán, J.C.; Martinez, J.L. The origin of niches and species in the bacterial world. Front. Microbiol. 2021, 12, 657986.
- Tansley, A.G. The use and abuse of vegetational concepts and terms. In The Future of Nature: Documents of Global Change; Robin, L., Sörlin, S., Warde, P., Eds.; Yale University Press: New Haven, CT, USA, 2013; pp. 220–232.
- Willis, A.J. The ecosystem: An evolving concept viewed historically. Funct. Ecol. 1997, 11, 268–271.
- Wollmuth, E.M.; Angert, E.R. Microbial circadian clocks: Host-microbe interplay in diel cycles. BMC Microbiol. 2023, 23, 1–20.
- Shahab, M.; Shahab, N. Coevolution of the human host and gut microbiome: Metagenomics of microbiota. Cureus 2022, 14, 6.
- Gonzalez-Alba, J.M.; Baquero, F.; Cantón, R.; Galán, J.C. Stratified reconstruction of ancestral Escherichia coli diversification. BMC Genom. 2019, 20, 1–15.
- Gordon, D.M.; Cowling, A. The distribution and genetic structure of Escherichia coli in Australian vertebrates: Host and geographic effects. Microbiology 2003, 149, 3575–3586.
- Rodríguez, I.; Figueiredo, A.S.; Sousa, M.; Aracil-Gisbert, S.; Fernández-de-Bobadilla, M.D.; Lanza, V.F.; Rodríguez, C.; Zamora, J.; Loza, E.; Mingo, P.; et al. A 21-year survey of Escherichia coli from bloodstream infections in a tertiary hospital reveals how community-hospital dynamics of B2 phylogroup clones influence local BSI rates. mSphere 2021, 6, e0086821.
- Tedim, A.P.; Ruiz-Garbajosa, P.; Corander, J.; Rodríguez, C.M.; Cantón, R.; Willems, R.J.; Baquero, F.; Coque, T.M. Population biology of intestinal Enterococcus isolates from hospitalized and non-hospitalized individuals in different age groups. Appl. Environ. Microbiol. 2015, 81, 1820–1831.
- Pehrsson, E.C.; Tsukayama, P.; Patel, S.; Mejía-Bautista, M.; Sosa-Soto, G.; Navarrete, K.M.; Calderon, M.; Cabrera, L.; Hoyos-Arango, W.; Bertoli, M.T.; et al. Interconnected microbiomes and resistomes in low-income human habitats. Nature 2016, 533, 212–221.
- García-Fernández, S.; Morosini, M.I.; Cobo, M.; Foruny, J.R.; López-Sanromán, A.; Cobo, J.; Romero, J.; Cantón, R.; del Campo, R. Gut eradication of VIM-1 producing ST9 Klebsiella oxytoca after fecal microbiota transplantation for diarrhea caused by a Clostridium difficile hypervirulent R027 strain. Diagn. Microbiol. Infect. Dis. 2016, 86, 470–471.
- Nhu, N.T.Q.; Young, V.B. The relationship between the microbiome and antimicrobial resistance. Clin. Infect. Dis. 2023, 77 (Suppl. S6), S479–S486.
- Woodworth, M.H.; Conrad, R.E.; Haldopoulos, M.; Pouch, S.M.; Babiker, A.; Mehta, A.K.; Sitchenko, K.L.; Wang, C.H.; Strudwick, A.; Ingersoll, J.M.; et al. Fecal microbiota transplantation promotes reduction of antimicrobial resistance by strain replacement. Sci. Transl. Med. 2023, 15, eabo2750.
- Fallingborg, J. Intraluminal pH of the human gastrointestinal tract. Dan. Med. Bull. 1999, 46, 183–196.
- Dittoe, D.K.; Ricke, S.C.; Kiess, A.S. Organic acids and potential for modifying the avian gastrointestinal tract and reducing pathogens and disease. Front. Vet. Sci. 2018, 5, 216.
- Foster, J.W. Escherichia coli acid resistance: Tales of an amateur acidophile. Nat. Rev. Microbiol. 2004, 2, 898–907.
- Richard, H.T.; Foster, J.W. Acid resistance in Escherichia coli. Adv. Appl. Microbiol. 2003, 52, 167–186.
- Kanjee, U.; Houry, W.A. Mechanisms of acid resistance in Escherichia coli. Ann. Rev. Microbiol. 2013, 67, 65–81.
- Baquero, F.; Martínez, J.L.; Sánchez, A.; Fernández-de-Bobadilla, M.D.; San-Millán, A.; Rodríguez-Beltrán, J. Bacterial subcellular architecture, structural epistasis, and antibiotic resistance. Biology 2023, 12, 640.
- Castanie-Cornet, M.P.; Penfound, T.A.; Smith, D.; Elliott, J.F.; Foster, J.W. Control of acid resistance in Escherichia coli. J. Bacteriol. 1999, 181, 3525–3535.
- Lin, J.; Lee, I.S.; Frey, J.; Slonczewski, J.L.; Foster, J.W. Comparative analysis of extreme acid survival in Salmonella typhimurium, Shigella flexneri, and Escherichia coli. J. Bacteriol. 1995, 177, 4097–4104.
- Castro, V.S.; Fang, Y.; Yang, X.; Stanford, K. Association of resistance to quaternary ammonium compounds and organic acids with genetic markers and their relationship to Escherichia coli serogroup. Food Microbiol. 2023, 113, 104267.
- Du, B.; Olson, C.A.; Sastry, A.V.; Fang, X.; Phaneuf, P.V.; Chen, K.; Wu, M.; Szubin, R.; Xu, S.; Gao, Y.; et al. Adaptive laboratory evolution of Escherichia coli under acid stress. Microbiology 2020, 166, 141–148.
- Kurdi, P.; Kawanishi, K.; Mizutani, K.; Yokota, A. Mechanism of growth inhibition by free bile acids in lactobacilli and bifidobacteria. J. Bacteriol. 2006, 188, 1979–1986.
- Begley, M.; Gahan, C.G.; Hill, C. The interaction between bacteria and bile. FEMS Microb. Rev. 2005, 29, 625–651.
- Flahaut, S.; Hartke, A.; Giard, J.C.; Auffray, Y. Alkaline stress response in Enterococcus faecalis: Adaptation, cross-protection and changes in protein synthesis. Appl. Environ. Microbiol. 1997, 63, 812–814.
- Sistrunk, J.R.; Nickerson, K.P.; Chanin, R.B.; Rasko, D.A.; Faherty, C.S. Survival of the fittest: How bacterial pathogens utilize bile to enhance infection. Clin. Microbiol. Rev. 2016, 29, 819–836.
- Terzi, H.A.; Kardes, H.; Atasoy, A.R.; Aykan, S.B.; Karakece, E.; Ustundag, G.H. The antibacterial effects of bilirubin on gram-negative bacterial agents of sepsis. Biomed. Res. 2016, 27, 207–209.
- Schaffner, S.H.; Lee, A.V.; Pham, M.T.; Kassaye, B.B.; Li, H.; Tallada, S.; Lis, C.; Lang, M.; Liu, Y.; Ahmed, N.; et al. Extreme acid modulates fitness trade-offs of multidrug efflux pumps MdtEF-TolC and AcrAB-TolC in Escherichia coli K-12. Appl. Environ. Microbiol. 2021, 87, e00724-21.
- Bustos, A.Y.; de Valdez, G.F.; Fadda, S.; Taranto, M.P. New insights into bacterial bile resistance mechanisms: The role of bile salt hydrolase and its impact on human health. Food Res. Intern. 2018, 112, 250–262.
- Rodríguez-Beltrán, J.; Rodríguez-Rojas, A.; Guelfo, J.R.; Couce, A.; Blázquez, J. Oxidative stress and bile salts. PLoS ONE 2012, 7, e34791.
- Isaacs, C.E.; Litov, R.E.; Marie, P.; Thormar, H. Addition of lipases to infant formulas produces antiviral and antibacterial activity. J. Nutr. Biochem. 1992, 3, 304–308.
- Zhang, X.H.; Wang, Z.; Yin, B.; Wu, H.; Tang, S.; Wu, L.L.; Su, Y.N.; Lin, Y.; Liu, X.Q.; Pang, B.; et al. A complex of trypsin and chymotrypsin effectively inhibited growth of pathogenic bacteria inducing cow mastitis and showed synergistic antibacterial activity with antibiotics. Livestock Sci. 2016, 188, 25–36.
- Li, Y.; Watanabe, E.; Kawashima, Y.; Plichta, D.R.; Wang, Z.; Ujike, M.; Ang, Q.Y.; Wu, R.; Furuichi, M.; Takeshita, K.; et al. Identification of trypsin-degrading commensals in the large intestine. Nature 2022, 609, 582–589.
- Den Besten, G.; Van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lip. Res. 2013, 54, 2325–2340.
- Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Cheng, J.; Duncan, A.E.; Kau, A.L.; Griffin, N.W.; Lombard, V.; Henrissat, B.; Bain, J.R.; et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 2013, 341, 1241214.
- Desbois, A.P.; Smith, V.J. Antibacterial free fatty acids: Activities, mechanisms of action and biotechnological potential. Appl. Microbiol. Biotechnol. 2010, 85, 1629–1642.
- Mitchell, M.K.; Ellermann, M. Long chain fatty acids and virulence repression in intestinal bacterial pathogens. Front. Cell. Infect. Microbiol. 2022, 12, 928503.
- Prasun, K.; Jin-Hyung, L.; Haluk, B.; Jintae, L. Fatty acids as antibiofilm and antivirulence agents. Trends Microbiol. 2020, 28, 753–768.
- Berding, K.; Kaulmann, D.; Cryan, J.F. Food-gut microbiota interactions. In Probiotics, Advanced Food and Health Applications; Brandelli, A., Ed.; Academic Press: Cambridge, MA, USA, 2022; pp. 233–256.
- Freitas, A.R.; Tedim, A.P.; Novais, C.; Ruiz-Garbajosa, P.; Werner, G.; Laverde-Gomez, J.A.; Cantón, R.; Peixe, L.; Baquero, F.; Coque, T.M. Global spread of the hyl colonization-virulence gene in megaplasmids of the Enterococcus faecium CC17 polyclonal subcluster. Antimicrob. Agents Chemother. 2010, 54, 2660–2665.
- Coppo, E.; Marchese, A. Antibacterial activity of polyphenols. Curr. Pharm. Biotechnol. 2014, 15, 380–390.
- Manso, T.; Lores, M.; de Miguel, T. Antimicrobial activity of polyphenols and natural polyphenolic extracts on clinical isolates. Antibiotics 2021, 11, 46.
- Ivanov, M.; Novović, K.; Malešević, M.; Dinić, M.; Stojković, D.; Jovčić, B.; Soković, M. Polyphenols as inhibitors of antibiotic resistant bacteria–mechanisms underlying rutin interference with bacterial virulence. Pharmaceuticals 2022, 15, 385.
- Cushnie, T.T.; Lamb, A.J. Recent advances in understanding the antibacterial properties of flavonoids. Int. J. Antimicrob. Agents 2011, 38, 99–107.
- Chambers, C.S.; Viktorová, J.; Řehořová; Biedermann, D.; Turková, L.; Macek, T.; Křen, V.; Valentová, K. Defying multidrug resistance! Modulation of related transporters by flavonoids and flavonolignans. J. Agric. Food. Chem. 2019, 68, 1763–1779.
- Kemperman, R.A.; Bolca, S.; Roger, L.C.; Vaughan, E.E. Novel approaches for analysing gut microbes and dietary polyphenols: Challenges and opportunities. Microbiology 2010, 156, 3224–3231.
- Zhong, W.; Tang, M.; Xie, Y.; Huang, X.; Liu, Y. Tea polyphenols inhibit the activity and toxicity of Staphylococcus aureus by destroying cell membranes and accumulating reactive oxygen species. Foodb. Pathog. Dis. 2023, 20, 294–302.
- Bachrach, U.; Weinstein, A. Effect of aliphatic polyamines on growth and macromolecular syntheses in bacteria. Microbiology 1970, 60, 159–165.
- Blanchet, M.; Borselli, D.; Brunel, J.M. Polyamine derivatives: A revival of an old neglected scaffold to fight resistant Gram-negative bacteria? Future Med. Chem. 2016, 8, 963–973.
- Cadelis, M.M.; Edmeades, L.R.; Chen, D.; Gill, E.S.; Fraser, K.; Rouvier, F.; Bourguet-Kondracki, M.L.; Brunel, J.M.; Copp, B.R. Investigation of naphthyl–polyamine conjugates as antimicrobials and antibiotic enhancers. Antibiotics 2023, 12, 1014.
- Shah, P.; Swiatlo, E. A multifaceted role for polyamines in bacterial pathogens. Mol. Microbiol. 2008, 68, 4–16.
- Porrini, C.; Ramarao, N.; Tran, S.L. Dr. NO and Mr. Toxic—The versatile role of nitric oxide. Biol. Chem. 2020, 401, 547–572.
- Leclerc, M.; Bedu-Ferrari, C.; Etienne-Mesmin, L.; Mariadassou, M.; Lebreuilly, L.; Tran, S.L.; Brazeau, L.; Mayeur, C.; Delmas, J.; Rué, O.; et al. Nitric oxide impacts human gut microbiota diversity and functionalities. Msystems 2021, 6, e00558-21.
- Dykhuizen, R.S.; Frazer, R.; Duncan, C.; Smith, C.C.; Golden, M.; Benjamin, N.; Leifert, C. Antimicrobial effect of acidified nitrite on gut pathogens: Importance of dietary nitrate in host defense. Antimicrob. Agents Chemother. 1996, 40, 1422–1425.
- Poole, R.K. Nitric oxide and nitrosative stress tolerance in bacteria. Biochem. Soc. Transact. 2005, 33, 176–180.
- Tropini, C.; Moss, E.L.; Merrill, B.D.; Ng, K.M.; Higginbottom, S.K.; Casavant, E.P.; Gonzalez, C.G.; Fremin, B.; Bouley, D.M.; Elias, J.E.; et al. Transient osmotic perturbation causes long-term alteration to the gut microbiota. Cell 2018, 173, 1742–1754.e17.
- Csonka, L.N. Physiological and genetic responses of bacteria to osmotic stress. Microbiol. Rev. 1989, 53, 121–147.
- Lim, J.J.; Diener, C.; Wilson, J.; Valenzuela, J.J.; Baliga, N.S.; Gibbons, S.M. Growth phase estimation for abundant bacterial populations sampled longitudinally from human stool metagenomes. Nat. Commun. 2023, 14, 5682.
- Gibson, B.; Wilson, D.J.; Feil, E.; Eyre-Walker, A. The distribution of bacterial doubling times in the wild. Proc. Biol. Sci. 2018, 285, 20180789.
- Arnoldini, M.; Cremer, J.; Hwa, T. Bacterial growth, flow, and mixing shape human gut microbiota density and composition. Gut Microb. 2018, 9, 559–566.
- Glowacki, R.W.P.; Martens, E.C. If you eat it, or secrete it, they will grow: The expanding list of nutrients utilized by human gut bacteria. J. Bacteriol. 2020, 203, e00481-20.
- Reese, A.T.; Pereira, F.C.; Schintlmeister, A.; Berry, D.; Wagner, M.; Hale, L.P.; Wu, A.; Jiang, S.; Durand, H.K.; Zhou, X.; et al. Microbial nitrogen limitation in the mammalian large intestine. Nat. Microbiol. 2018, 3, 1441–1450.
- Brown, D.; Barton, G.; Pan, Z.; Buck, M.; Wigneshweraraj, S. Nitrogen stress response and stringent response are coupled in Escherichia coli. Nat. Commun. 2014, 5, 4115.
- Peterson, C.N.; Mandel, M.J.; Silhavy, T.J. Escherichia coli starvation diets: Essential nutrients weigh in distinctly. J. Bacteriol. 2005, 187, 7549–7553.
- Hansen, E.; Karslake, J.; Woods, R.J.; Read, A.F.; Wood, K.B. Antibiotics can be used to contain drug-resistant bacteria by maintaining sufficiently large sensitive populations. PLoS Biol. 2020, 18, e3000713.
- Asensio, C.; Pérez-Díaz, J.C.; Martínez, M.C.; Baquero, F. A new family of low molecular weight antibiotics from enterobacteria. Biochem. Biophys. Res. Commun. 1976, 69, 7–14.
- Baquero, F.; Lanza, V.F.; Baquero, M.R.; del Campo, R.; Bravo-Vázquez, D.A. Microcins in Enterobacteriaceae: Peptide antimicrobials in the eco-active intestinal chemosphere. Front. Microbiol. 2019, 10, 2261.
- McAuliffe, O.; Ross, R.P.; Hill, C. Lantibiotics: Structure, biosynthesis and mode of action. FEMS Microbiol. Rev. 2001, 25, 285–308.
- Guo, L.; Wambui, J.; Wang, C.; Muchaamba, F.; Fernandez-Cantos, M.V.; Broos, J.; Tasara, T.; Kuipers, O.P.; Stephan, R. Cesin, a short natural variant of nisin, displays potent antimicrobial activity against major pathogens despite lacking two C-terminal macrocycles. Microbiol Spectr. 2023, 11, e0531922.
- Draper, L.A.; Cotter, P.D.; Hill, C.; Ross, R.P. Lantibiotic resistance. Microbiol. Mol. Biol. Rev. 2015, 79, 171–191.
- Feldgarden, M.; Riley, M.A. The phenotypic and fitness effects of colicin resistance in Escherichia coli K-12. Evolution 1999, 53, 1019–1027.
- Sutcliffe, S.G.; Reyes, A.; Maurice, C.F. Bacteriophages playing nice: Lysogenic bacteriophage replication stable in the human gut microbiota. Iscience 2023, 26, 106007.
- Manganelli, R.; Gennaro, M.L. Protecting from envelope stress: Variations on the phage-shock-protein theme. Trends Microbiol. 2017, 25, 205–216.
- De Sordi, L.; Lourenço, M.; Debarbieux, L. The battle within: Interactions of bacteriophages and bacteria in the gastrointestinal tract. Cell Host Microb. 2019, 25, 210–218.
- Chabé, M.; Lokmer, A.; Ségurel, L. Gut protozoa: Friends or foes of the human gut microbiota? Trends Parasitol. 2017, 33, 925–934.
- Sun, S.; Noorian, P.; McDougald, D. Dual role of mechanisms involved in resistance to predation by protozoa and virulence to humans. Front. Microbiol. 2018, 9, 1017.
- Iebba, V.; Santangelo, F.; Totino, V.; Nicoletti, M.; Gagliardi, A.; De Biase, R.V.; Cucchiara, S.; Nencioni, L.; Conte, M.P.; Schippa, S. Higher prevalence and abundance of Bdellovibrio bacteriovorus in the human gut of healthy subjects. PLoS ONE 2013, 8, e61608.
- Reinoso Webb, C.; Koboziev, I.; Furr, K.L.; Grisham, M.B. Protective and pro-inflammatory roles of intestinal bacteria. Pathophysiology 2016, 2, 67–80.
- Adolph, T.E.; Mayr, L.; Grabherr, F.; Tilg, H. Paneth cells and their antimicrobials in intestinal immunity. Curr. Pharmac. Des. 2018, 24, 1121–1129.
- Andersson, M.; Karlsson-Sjöberg, J.; Pütsep, K.A. CRS-peptides: Unique defense peptides of mouse Paneth cells. Mucosal Immunol. 2012, 5, 367–376.
- Batista Araujo, J.; Sastre de Souza, G.; Lorenzon, E.N. Indolicidin revisited: Biological activity, potential applications and perspectives of an antimicrobial peptide not yet fully explored. World J. Microbiol. Biotechnol. 2022, 38, 39.
- Liang, W.; Enée, E.; Andre-Vallee, C.; Falcone, M.; Sun, J.; Diana, J. Intestinal cathelicidin antimicrobial peptide shapes a protective neonatal gut microbiota against pancreatic autoimmunity. Gastroenterology 2022, 162, 1288–1302.
- Sina, C.; Kemper, C.; Derer, S. The intestinal complement system in inflammatory bowel disease: Shaping intestinal barrier function. Semin. Immunol. 2018, 37, 66–73.
- Heesterbeek, D.A.C.; Angelier, M.L.; Harrison, R.A.; Rooijakkers, S.H.M. Complement and bacterial infections: From molecular mechanisms to therapeutic applications. J. Inn. Immun. 2018, 10, 455–464.
- Menendez, A.; Finlay, B.B. Defensins in the immunology of bacterial infections. Curr. Opin. Immunol. 2007, 19, 385–391.
- Ernst, C.M.; Staubitz, P.; Mishra, N.N.; Yang, S.J.; Hornig, G.; Kalbacher, H.; Bayer, A.S.; Kraus, D.; Peschel, A. The bacterial defensin resistance protein MprF consists of separable domains for lipid lysinylation and antimicrobial peptide repulsion. PLoS Path. 2009, 5, e1000660.
- Yang, S.J.; Mishra, N.N.; Rubio, A.; Bayer, A.S. Causal role of single nucleotide polymorphisms within the mprF gene of Staphylococcus aureus in daptomycin resistance. Antimicrob. Agents Chemother. 2013, 57, 5658–5664.
- Diaz, L.; Tran, T.T.; Munita, J.M.; Miller, W.R.; Rincon, S.; Carvajal, L.P.; Arias, C.A. Whole-genome analyses of Enterococcus faecium isolates with diverse daptomycin MICs. Antimicrob. Agents Chemother. 2014, 58, 4527–4534.
- López, C.; Ayala, J.A.; Bonomo, R.A.; González, L.J.; Vila, A.J. Protein determinants of dissemination and host specificity of metallo-β-lactamases. Nat. Commun. 2019, 10, 3617.
- López, C.; Delmonti, J.; Bonomo, R.A.; Vila, A.J. Deciphering the evolution of metallo-β-lactamases: A journey from the test tube to the bacterial periplasm. J. Biol. Chem. 2022, 298, 101665.
- Andersson, D.I.; Hughes, D. Antibiotic resistance and its cost: Is it possible to reverse resistance? Nat. Rev. Microbiol. 2010, 8, 260–271.
- San Millan, A.; MacLean, R.C. Fitness costs of plasmids: A limit to plasmid transmission. Microbiol. Spectr. 2017, 5, 10–1128.
- Le, V.V.H.; Olivera, C.; Spagnuolo, J.; Davies, I.G.; Rakonjac, J. In vitro synergy between sodium deoxycholate and furazolidone against enterobacteria. BMC Microbiol. 2020, 20, 1–11.
- Gipson, K.S.; Nickerson, K.P.; Drenkard, E.; Llanos-Chea, A.; Dogiparthi, S.K.; Lanter, B.B.; Hibbler, R.M.; Yonker, L.M.; Hurley, B.P.; Faherty, C.S. The great ESKAPE: Exploring the crossroads of bile and antibiotic resistance in bacterial pathogens. Infect Immun. 2020, 88, e00865-19.
More
Information
Subjects:
Health Care Sciences & Services
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
579
Revisions:
2 times
(View History)
Update Date:
26 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





