Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jihao Li | -- | 1254 | 2024-02-23 04:38:48 | | | |
| 2 | Peter Tang | Meta information modification | 1254 | 2024-02-23 09:06:33 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Fan, K.; Lin, L.; Li, D.; Wang, F.; Li, J. Preparation of Graphene-Based Aerogels Using γ-ray Irradiation Technology. Encyclopedia. Available online: https://encyclopedia.pub/entry/55373 (accessed on 28 March 2026).
Fan K, Lin L, Li D, Wang F, Li J. Preparation of Graphene-Based Aerogels Using γ-ray Irradiation Technology. Encyclopedia. Available at: https://encyclopedia.pub/entry/55373. Accessed March 28, 2026.
Fan, Kai, Lin Lin, Danyi Li, Fangzheng Wang, Jihao Li. "Preparation of Graphene-Based Aerogels Using γ-ray Irradiation Technology" Encyclopedia, https://encyclopedia.pub/entry/55373 (accessed March 28, 2026).
Fan, K., Lin, L., Li, D., Wang, F., & Li, J. (2024, February 23). Preparation of Graphene-Based Aerogels Using γ-ray Irradiation Technology. In Encyclopedia. https://encyclopedia.pub/entry/55373
Fan, Kai, et al. "Preparation of Graphene-Based Aerogels Using γ-ray Irradiation Technology." Encyclopedia. Web. 23 February, 2024.
Copy Citation
Graphene aerogels (GAs) are of significant interest in the scientific community due to their unique attributes, including a three-dimensional porous structure, exceptional specific surface area, and remarkable chemical stability. Researchers have made notable breakthroughs in aerogel preparation, focusing on aspects like porous structures and chemical stability.
graphene aerogels
irradiation
γ-ray
composites
1. Introduction
Graphene aerogel (GA) stands out as an extraordinary three-dimensional porous solid material, distinguished by its ultra-low density, impressive specific surface area, exceptional porosity, and remarkable adsorption capacity. These remarkable properties bestow upon GA a wide spectrum of applications, encompassing energy storage [1][2], catalysis [3][4], sensors [5], environmental protection [6], and beyond. When compared to pristine graphene, GA triumphs over the challenges of agglomeration and the reduction in specific surface area, with issues often stemming from the formidable π-π conjugation and van der Waals forces between graphene sheets during processing. This triumph significantly broadens the horizons for graphene applications, such as energy storage, thermal insulation, and environmental protection. Despite its many potential applications, GA is still expensive to produce and there are no widely available graphene aerogel products on the market today. Therefore, future research and development must focus on reducing production costs, improving efficiency, and developing new application areas.
Traditional synthesis methods for GA encompass hydrothermal reduction, template-based approaches, and sol-gel methods [7]. Among these methods, the hydrothermal reduction method reigns as the most prevalent. It commences with graphene oxide (GO), initiating a process of thermal reduction and self-assembly or chemical cross-linking of GO in aqueous or organic solutions to yield graphene hydrogel (GH) [8]. Subsequently, GA can be meticulously crafted through the freeze-drying or supercritical-CO2-drying of GH, thus realizing its full potential in a myriad of applications [9][10].
Gamma-rays, as a form of ionizing radiation, are characterized by their high energy and are primarily emitted during the decay reactions of radionuclides such as 60Co and 137Cs. When γ-rays interact with various forms of matter, including aqueous solutions, they have the capability to ionize or excite atoms and molecules, resulting in the generation of active particles. This, in turn, initiates a cascade of physical and chemical reactions.
In recent times, the utilization of γ-ray irradiation techniques has gained considerable popularity due to their efficiency, convenience, and ease of use. This technique has found widespread application in the fabrication and modification of inorganic nanomaterials [11][12], as well as the development of polymer functional materials [13][14]. Researchers have harnessed the power of γ-ray irradiation to precisely tailor the properties of these materials for specific applications.
Furthermore, it is worth noting that the versatility of γ-ray irradiation extends to the realm of graphene-based materials. Several studies have demonstrated the successful preparation and enhancement of graphene-based materials using γ-ray irradiation. This innovative approach has shown promise in unlocking new possibilities for graphene-based materials, making them even more versatile and adaptable to a wide range of technological and scientific advancements [15][16][17][18]. However, only a few works have summarized the graphene aerogels prepared using conventional chemical methods and few studies have been reported on the preparation of GAs by γ-ray irradiation.
Gamma-ray irradiation provides a uniform reduction in GO, ensuring consistent and controlled reduction across the entire graphene structure. This results in a more homogeneous and well-defined GA. The use of γ-ray irradiation helps prevent the agglomeration of reduced graphene oxide sheets. This results in a well-dispersed and interconnected graphene network within the aerogel, contributing to improved mechanical and thermal properties. As depicted in Figure 1, the graphene-based aerogels, meticulously crafted through the powerful technique of γ-ray irradiation, exhibit an exceptional degree of reduction and feature a captivating 3D honeycomb-like structure. This structural marvel serves as a testament to the ingenuity of the fabrication process.
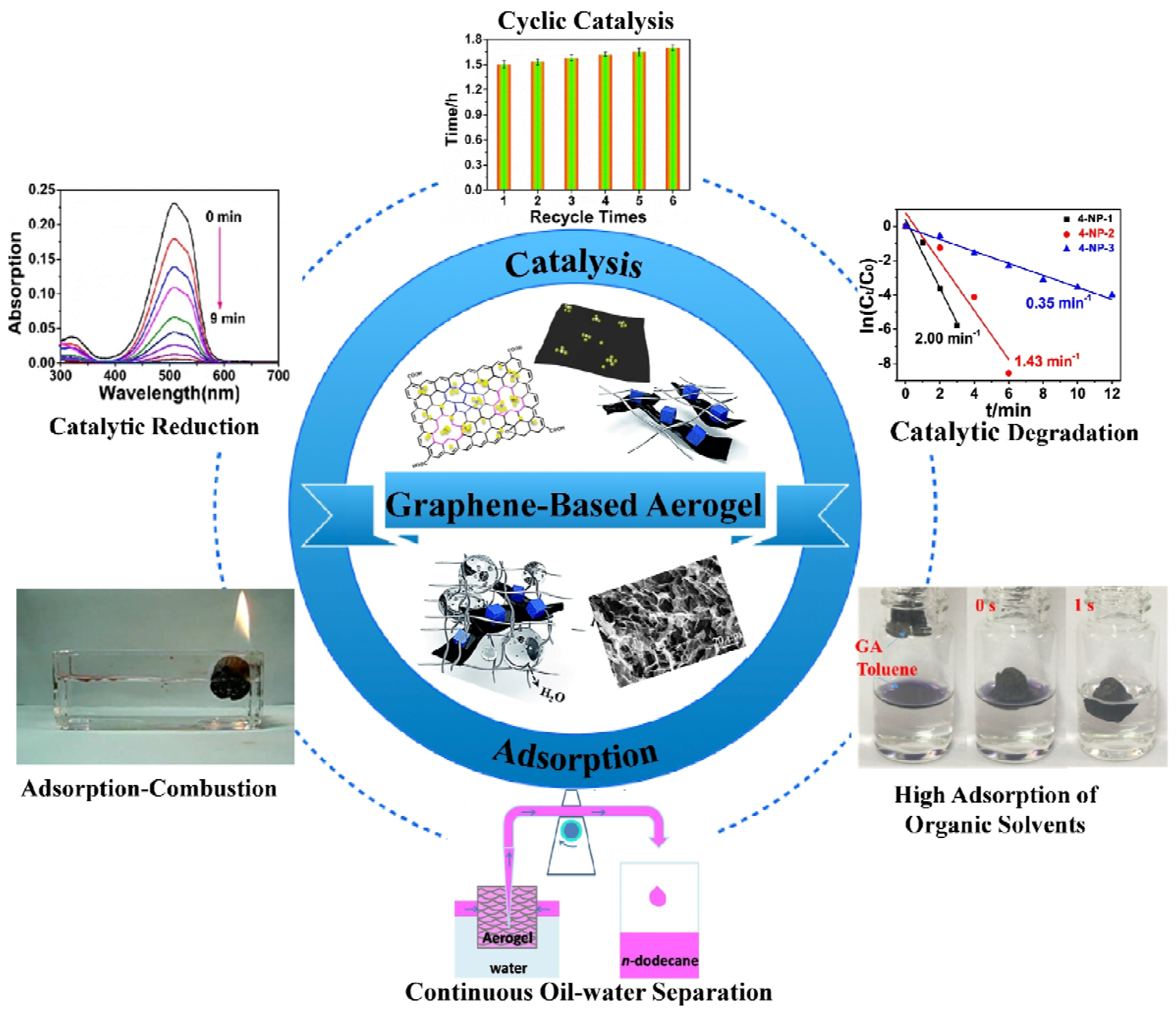
Figure 1. Catalysis and absorption applications of graphene-based aerogels.
These remarkable graphene-based aerogels, hereinafter referred to as GA, offer a plethora of applications driven by their distinctive characteristics. Of particular note are their remarkable roles in catalysis and adsorption. Within the realm of catalysis, GA demonstrates the capability to facilitate the catalytic reduction in various organic substances, thereby contributing to the advancement of green and sustainable chemical processes. Simultaneously, in the realm of adsorption, GA’s unique properties enable its use in the continuous and efficient separation of oil and water, a crucial process in addressing environmental challenges.
2. Radiation Synthesis of GA
Under the influence of γ-ray irradiation, as thoughtfully illustrated in Scheme 1, water undergoes a fascinating transformation, generating a multitude of active products. Among these products are the formation of H2 and H2O2 molecules, the emergence of hydrated electrons (eaq−), and the release of ·OH, H·, HO2· radicals, and H3O+. This diverse array of species plays a pivotal role in the ensuing chemical and physical reactions.
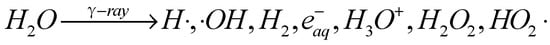
Scheme 1. Radiolysis mechanism of water.
Notably, within this intricate interplay of reactions, there exists a delicate balance between reductive and oxidative species. Hydrated electrons and H· are representative of the reductive species, while ·OH, HO2·, and H2O2 fall within the category of oxidative species. However, it is within this dynamic equilibrium that the presence of radical scavengers in aqueous systems, such as alcohols or amines, becomes significant. When introduced, these scavengers play a remarkable role in eliminating and converting oxidative radicals, particularly the highly reactive ·OH, into reductive radicals. These reductive radicals synergize with the potent eaq− to engage in the reduction of oxidative substances, shaping the complex chemistry of the irradiation process.
This intricate dance of chemical reactions has profound implications for the graphene-based aerogels prepared through the dual techniques of γ-ray irradiation and freeze-drying. These materials are subject to various influencing factors, including pH and the type of radical scavengers, as elegantly depicted in Figure 2. The precise manipulation of these factors allows for the fine-tuning of GA’s properties, rendering it a versatile tool in the realms of catalysis and adsorption.
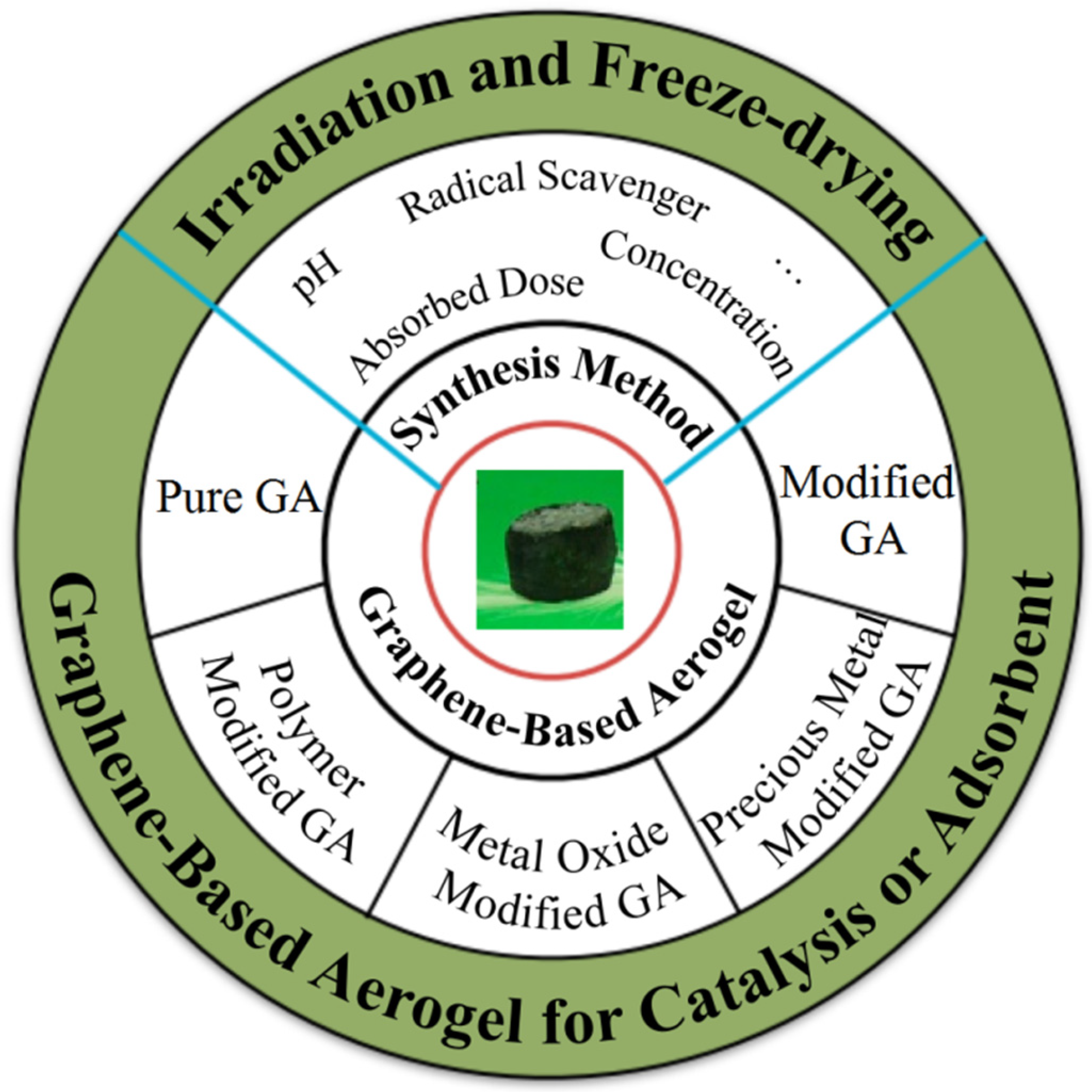
Figure 2. The synthesis method of graphene-based aerogels and their applications.
Furthermore, the versatility of the γ-ray irradiation process extends even further. Through the introduction of different modified species during irradiation, GA can be endowed with enhanced catalytic or adsorption properties, thus expanding its application horizons. This multifaceted approach enables the tailoring of graphene-based aerogels to meet the demands of a diverse range of applications, with the potential for groundbreaking contributions to science and technology.
Radiation-based preparation of GA typically involves utilizing GO as the precursor material. The reduction and self-assembly of GO are induced by γ-ray irradiation to yield a graphene hydrogel. Subsequently, a freeze-drying method is employed to obtain a lightweight GA characterized by a high carbon-to-oxygen (C/O) ratio. It is crucial to note that various factors can influence the reduction and self-assembly process during this approach, including the absorbed dose, solution composition, pH value, and concentration.
To enhance the versatility and application potential of GA, additional techniques can be incorporated. Amines or alcohols are often introduced as free radical scavengers to capture and neutralize species like eaq−, ·OH, and other particles generated through water radiolysis. This not only helps in eliminating oxygen-containing groups but also facilitates the introduction of nitrogen-containing groups or hydroxyl alkylation. Moreover, in a bid to expand the scope of GA applications, metal ions can be introduced into the mixed solution. This has the dual benefit of simultaneously reducing the metal ions and GO while ensuring even dispersion of nanoparticles on the graphene sheets.
The graphene aerogels produced through irradiation methods offer numerous advantages and exhibit substantial potential in applications such as organic solvent absorption, oil spill cleanup, and environmental remediation efforts. However, it is worth mentioning that, despite these promising attributes, there remains a scarcity of research on the radiation-based preparation of graphene aerogels.
References
- Gholipour-Ranjbar, H.; Ganjali, M.R.; Norouzi, P.; Naderi, H.R. Functionalized graphene aerogel with p-phenylenediamine and its composite with porous MnO2: Investigating the effect of functionalizing agent on supercapacitive performance. J. Mater. Sci. Mater. Electron. 2016, 27, 10163–10172.
- Pawar, R.C.; Kang, S.; Khan, H.; Han, H.; Lee, C.S. Study of multi-faceted CoS2 introduced graphene aerogel hybrids via chemical approach for an effective electrocatalytic water splitting. Curr. Appl. Phys. 2021, 32, 78–85.
- Arumugam, M.; Seralathan, K.-K.; Praserthdam, S.; Tahir, M.; Praserthdam, P. Synthesis of novel graphene aerogel encapsulated bismuth oxyiodide composite towards effective removal of methyl orange azo-dye under visible light. Chemosphere 2022, 303, 135121.
- Osman, S.H.; Kamarudin, S.K.; Basri, S.; Karim, N.A. Three-Dimensional Graphene Aerogel Supported on Efficient Anode Electrocatalyst for Methanol Electrooxidation in Acid Media. Catalysts 2023, 13, 879.
- Vrettos, K.; Spyrou, K.; Georgakilas, V. Graphene Aerogel Growth on Functionalized Carbon Fibers. Molecules 2020, 25, 1295.
- Sawut, A.; Wu, T.; Simayi, R.; Wu, T.; Gong, X.; Wang, Z. Reduced graphene oxide-TiO2/sodium alginate/polyacrylamide composite hydrogel for enhanced adsorption-photocatalytic degradation of methylene blue. Colloids Surf. A Physicochem. Eng. Asp. 2023, 678, 132531.
- Wang, Z.; Liu, L.; Zhang, Y.; Huang, Y.; Liu, J.; Zhang, X.; Liu, X.; Teng, H.; Zhang, X.; Zhang, J.; et al. A Review of Graphene-Based Materials/Polymer Composite Aerogels. Polymers 2023, 15, 1888.
- Rathi, K.; Kim, D. Super-compressible and mechanically stable reduced graphene oxide aerogel for wearable functional devices. Sci. Technol. Adv. Mater. 2023, 24, 2214854.
- Kasar, A.K.; Tian, S.; Xiong, G.; Menezes, P.L. Graphene aerogel and its composites: Synthesis, properties and applications. J. Porous Mater. 2022, 29, 1011–1025.
- Zhang, X.; Sui, Z.; Xu, B.; Yue, S.; Luo, Y.; Zhan, W.; Liu, B. Mechanically strong and highly conductive graphene aerogel and its use as electrodes for electrochemical power sources. J. Mater. Chem. 2011, 21, 6494–6497.
- Grushevskaya, H.; Timoshchenko, A.; Lipnevich, I. Topological Defects Created by Gamma Rays in a Carbon Nanotube Bilayer. Nanomaterials 2023, 13, 410.
- Abedini, A.; Daud, A.R.; Hamid, M.A.A.; Othman, N.K. Radiolytic Formation of Fe3O4 Nanoparticles: Influence of Radiation Dose on Structure and Magnetic Properties. PLoS ONE 2014, 9, e90055.
- Borodinov, N.L.; Giarnmarco, J.; Patel, N.; Agarwal, A.; O’Donnell, K.R.; Kucera, C.J.; Jacobsohn, L.G.; Luzinov, I. Stability of Grafted Polymer Nanoscale Films toward Gamma Irradiation. ACS Appl. Mater. Interfaces 2015, 7, 19455–19465.
- Misra, N.; Biswal, J.; Gupta, A.; Sainis, J.K.; Sabharwal, S. Gamma radiation induced synthesis of gold nanoparticles in aqueous polyvinyl pyrrolidone solution and its application for hydrogen peroxide estimation. Radiat. Phys. Chem. 2012, 81, 195–200.
- Anson-Casaos, A.; Puertolas, J.A.; Pascual, F.J.; Hernandez-Ferrer, J.; Castell, P.; Benito, A.M.; Maser, W.K.; Martinez, M.T. The effect of gamma-irradiation on few-layered graphene materials. Appl. Surf. Sci. 2014, 301, 264–272.
- Dumee, L.F.; Feng, C.; He, L.; Allioux, F.-M.; Yi, Z.; Gao, W.; Banos, C.; Davies, J.B.; Kong, L. Tuning the grade of graphene: Gamma ray irradiation of free-standing graphene oxide films in gaseous phase. Appl. Surf. Sci. 2014, 322, 126–135.
- Lin, L.; Wang, W.; Li, D.; Xu, S.; Sun, Y.; Li, L.; Fan, K.; Xing, C.; Zhang, L.; Li, J. Multifunctional graphene/Ag hydrogel with antimicrobial and catalytic properties for efficient solar-driven desalination and wastewater purification. Chem. Eng. J. 2023, 478, 147249.
- Jung, C.-H.; Park, Y.-W.; Hwang, I.-T.; Go, Y.-J.; Na, S.-I.; Shin, K.; Lee, J.-S.; Choi, J.-H. Eco-friendly and simple radiation-based preparation of graphene and its application to organic solar cells. J. Phys. D Appl. Phys. 2014, 47, 015105.
More
Information
Subjects:
Nanoscience & Nanotechnology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
727
Revisions:
2 times
(View History)
Update Date:
23 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





