
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Federica Barbagallo | -- | 3877 | 2024-02-19 14:19:45 | | | |
| 2 | Lindsay Dong | + 3 word(s) | 3880 | 2024-02-20 02:17:03 | | |
Video Upload Options
Since its discovery, irisin has attracted much attention for its potential involvement in metabolic and reproductive diseases. It appears to play an important role in different physiological and pathological conditions that can involve women throughout their entire lives. Irisin appears to be an important factor for the hypothalamic–pituitary–gonadal axis activation, and it seems to play a role in the timing of puberty onset. Additionally, serum irisin levels have been proposed as a biomarker for predicting the future development of gestational diabetes (GDM). Its role in polycystic ovary syndrome (PCOS) is still controversial, although an “irisin resistance” mechanism has been hypothesized. Beyond its influence on metabolism, irisin also appears to influence bone health. Irisin levels are inversely correlated with the prevalence of fractures in postmenopausal women. Similar mechanisms have also been postulated in young women with functional hypothalamic amenorrhea (FHA).
1. Introduction
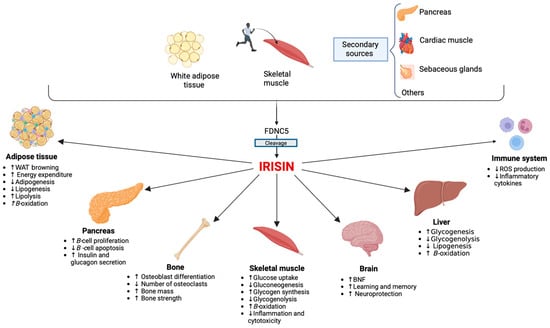
2. Irisin and Sex Differences: Towards Gender Medicine
3. Irisin and Pubertal Development
4. Irisin and Polycystic Ovarian Syndrome (PCOS)
5. Irisin and Functional Hypothalamic Amenorrhea
6. Irisin and Endometriosis
7. Irisin and Gestational Diabetes Mellitus
8. Irisin and Menopause
9. Conclusions
References
- Boström, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Boström, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 2012, 481, 463–468.
- Pukajło, K.; Kolackov, K.; Łaczmański, Ł.; Daroszewski, J. Irisin, a new mediator of energy homeostasis. Adv. Hyg. Exp. Med. 2015, 69, 233–242.
- Perakakis, N.; Triantafyllou, G.A.; Fernández-Real, J.M.; Huh, J.Y.; Park, K.H.; Seufert, J.; Mantzoros, C.S. Physiology and role of irisin in glucose homeostasis. Nat. Rev. Endocrinol. 2017, 13, 324–337.
- Natalicchio, A.; Marrano, N.; Giorgino, F. Irisina: Ruolo nell’omeostasi del glucosio. L’Endocrinologo 2018, 19, 292.
- Norman, D.; Drott, C.J.; Carlsson, P.O.; Espes, D. Irisin—A Pancreatic Islet Hormone. Biomedicines 2022, 10, 258.
- Song, R.; Zhao, X.; Zhang, D.Q.; Wang, R.; Feng, Y. Lower levels of irisin in patients with type 2 diabetes mellitus: A meta-analysis. Diabetes Res. Clin. Pract. 2021, 175, 108788.
- Marrano, N.; Biondi, G.; Borrelli, A.; Cignarelli, A.; Perrini, S.; Laviola, L.; Giorgino, F.; Natalicchio, A. Irisin and Incretin Hormones: Similarities, Differences, and Implications in Type 2 Diabetes and Obesity. Biomolecules 2021, 11, 286.
- Martinez Munoz, I.Y.; Camarillo Romero, E.D.S.; Garduno Garcia, J.J. Irisin a Novel Metabolic Biomarker: Present Knowledge and Future Directions. Int. J. Endocrinol. 2018, 2018, 7816806.
- Korta, P.; Pocheć, E.; Mazur-Biały, A. Irisin as a Multifunctional Protein: Implications for Health and Certain Diseases. Medicina 2019, 55, 485.
- Ma, C.; Ding, H.; Deng, Y.; Liu, H.; Xiong, X.; Yang, Y. Irisin: A New Code Uncover the Relationship of Skeletal Muscle and Cardiovascular Health During Exercise. Front. Physiol. 2021, 2, 620608.
- Yi, P.; Park, J.S.; Melton, D.A. Betatrophin: A hormone that controls pancreatic β cell proliferation. Cell 2013, 53, 747–758.
- Park, M.J.; Kim, D.I.; Choi, J.H.; Heo, Y.R.; Park, S.H. New role of irisin in hepatocytes: The protective effect of hepatic steatosis in vitro. Cell Signal 2015, 27, 1831–1839.
- Wrann, C.D.; White, J.P.; Salogiannnis, J.; Laznik-Bogoslavski, D.; Wu, J.; Ma, D.; Lin, J.D.; Greenberg, M.E.; Spiegelman, B.M. Exercise induces hippocampal BDNF through a PGC-1α/FNDC5 pathway. Cell Metab. 2013, 18, 649–659.
- Colaianni, G.; Cuscito, C.; Mongelli, T.; Oranger, A.; Mori, G.; Brunetti, G.; Colucci, S.; Cinti, S.; Grano, M. Irisin enhances osteoblast differentiation in vitro. Int. J. Endocrinol. 2014, 2014, 902186.
- Mazur-Bialy, A.I.; Pocheć, E.; Zarawski, M. Anti-Inflammatory Properties of Irisin, Mediator of Physical Activity, Are Connected with TLR4/MyD88 Signaling Pathway Activation. Int. J. Mol. Sci. 2017, 18, 701.
- Luo, Y.; Qiao, X.; Xu, L.; Huang, G. Irisin: Circulating levels in serum and its relation to gonadal axis. Endocrine 2022, 75, 663–671.
- Al-Daghri, N.M.; Alkharfy, K.M.; Rahman, S.; Amer, O.E.; Vinodson, B.; Sabico, S.; Piya, M.K.; Harte, A.L.; McTernan, P.G.; Alokail, M.S.; et al. Irisin as a predictor of glucose metabolism in children: Sexually dimorphic effects. Eur. J. Clin. Investig. 2014, 44, 119–124.
- Anastasilakis, A.D.; Polyzos, S.A.; Saridakis, Z.G.; Kynigopoulos, G.; Skouvaklidou, E.C.; Molyvas, D.; Vasiloglou, M.F.; Apostolou, A.; Karagiozoglou-Lampoudi, T.; Siopi, A.; et al. Circulating irisin in healthy, young individuals: Day-night rhythm, effects of food intake and exercise, and associations with gender, physical activity, diet, and body composition. J. Clin. Endocrinol. Metab. 2014, 99, 3247–3255.
- Moreno-Navarrete, J.M.; Ortega, F.; Serrano, M.; Guerra, E.; Pardo, G.; Tinahones, F.; Ricart, W.; Fernández-Real, J.M. Irisin is expressed and produced by human muscle and adipose tissue in association with obesity and insulin resistance. J. Clin. Endocrinol. Metab. 2013, 98, E769–E778.
- Wahab, F.; Khan, I.U.; Polo, I.R.; Zubair, H.; Drummer, C.; Shahab, M.; Behr, R. Irisin in the primate hypothalamus and its effect on GnRH in vitro. J. Endocrinol. 2019, 241, 175–187.
- Wahab, F.; Shahab, M.; Behr, R. Hypothesis: Irisin is a metabolic trigger for the activation of the neurohormonal axis governing puberty onset. Med. Hypotheses 2016, 95, 1–4.
- Ulker, N.; Yardimci, A.; Kaya Tektemur, N.; Bulmus, O.; Ozer Kaya, S.; Gulcu Bulmus, F.; Turk, G.; Ozcan, M.; Canpolat, S. Irisin may have a role in pubertal development and regulation of reproductive function in rats. Reproduction 2020, 160, 281–292.
- Jiang, Q.; Zhang, Q.; Lian, A.; Xu, Y. Irisin stimulates gonadotropins gene expression in tilapia (Oreochromis niloticus) pituitary cells. Anim. Reprod. Sci. 2017, 85, 140–147.
- Poretsky, L.; Islam, J.; Avtanski, D.; Lin, Y.K.; Shen, Y.L.; Hirth, Y.; Lesser, M.; Rosenwaks, Z.; Seto-Young, D. Reproductive effects of irisin: Initial in vitro studies. Reprod. Biol. 2017, 17, 285–288.
- Bastu, E.; Zeybek, U.; Gurel Gurevin, E.; Yüksel Ozgor, B.; Celik, F.; Okumus, N.; Demiral, I.; Dural, O.; Celik, C.; Bulut, H.; et al. Effects of Irisin and Exercise on Metabolic Parameters and Reproductive Hormone Levels in High-Fat Diet-Induced Obese Female Mice. Reprod. Sci. 2018, 25, 281–291.
- Tekin, S.; Beytur, A.; Erden, Y.; Beytur, A.; Cigremis, Y.; Vardi, N.; Turkoz, Y.; Tekedereli, I.; Sandal, S. Effects of intracerebroventricular administration of irisin on the hypothalamus-pituitary-gonadal axis in male rats. J. Cell Physiol. 2019, 234, 8815–8824.
- Klenke, U.; Taylor-Burds, C.; Wray, S. Metabolic influences on reproduction: Adiponectin attenuates GnRH neuronal activity in female mice. Endocrinology 2014, 155, 1851–1863.
- Castellano, J.M.; Tena-Sempere, M. Metabolic control of female puberty: Potential therapeutic targets. Expert. Opin. Ther. Targets 2016, 20, 1181–1193.
- Kutlu, E.; Özgen, I.T.; Bulut, H.; Koçyiǧit, A.; Otçu, H.; Cesur, Y. Serum Irisin Levels in Central Precocious Puberty and Its Variants. J. Clin. Endocrinol. Metab. 2021, 106, E247–E254.
- Reinehr, T.; Elfers, C.; Lass, N.; Roth, C.L. Irisin and its relation to insulin resistance and puberty in obese children: A longitudinal analysis. J. Clin. Endocrinol. Metab. 2015, 100, 2123–2130.
- Azziz, R.; Carmina, E.; Chen, Z.; Dunaif, A.; Laven, J.S.; Legro, R.S.; Lizneva, D.; Natterson-Horowtiz, B.; Teede, H.J.; Yildiz, B.O. Polycystic ovary syndrome. Nat. Rev. Dis. Primers 2016, 2, 16057.
- Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum. Reprod. 2004, 19, 41–47.
- Lim, S.S.; Davies, M.J.; Norman, R.J.; Moran, L.J. Overweight, obesity and central obesity in women with polycystic ovary syndrome: A systematic review and meta-analysis. Hum. Reprod. Update 2012, 18, 618–637.
- Amisi, C.A. Markers of insulin resistance in Polycystic ovary syndrome women: An update. World J. Diabetes 2022, 13, 129–149.
- Pant, P.; Chitme, H.; Sircar, R.; Prasad, R.; Prasad, H.O. Genome-wide association study for single nucleotide polymorphism associated with mural and cumulus granulosa cells of PCOS (polycystic ovary syndrome) and non-PCOS patients. Future J. Pharm. Sci. 2023, 27.
- Gizaw, M.; Anandakumar, P.; Debela, T. A Review on the Role of Irisin in Insulin Resistance and Type 2 Diabetes Mellitus. J. Pharmacopunct. 2017, 20, 235–242.
- Chang, C.L.; Huang, S.Y.; Soong, Y.K.; Cheng, P.J.; Wang, C.J.; Liang, I.T. Circulating irisin and glucose-dependent insulinotropic peptide are associated with the development of polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2014, 99, E2539–E2548.
- Pukajło, K.; Łaczmański, Ł.; Kolackov, K.; Kuliczkowska-Płaksej, J.; Bolanowski, M.; Milewicz, A.; Daroszewski, J. Irisin plasma concentration in PCOS and healthy subjects is related to body fat content and android fat distribution. Gynecol. Endocrinol. 2015, 31, 907–911.
- Abali, R.; Temel Yuksel, I.; Yuksel, M.A.; Bulut, B.; Imamoglu, M.; Emirdar, V.; Unal, F.; Guzel, S.; Celik, C. Implications of circulating irisin and Fabp4 levels in patients with polycystic ovary syndrome. J. Obs. Gynaecol. 2016, 36, 897–901.
- Wang, C.; Zhang, X.Y.; Sun, Y.; Hou, X.G.; Chen, L. Higher circulating irisin levels in patients with polycystic ovary syndrome: A meta-analysis. Gynecol. Endocrinol. 2018, 34, 290–293.
- Cai, X.; Qiu, S.; Li, L.; Zügel, M.; Steinacker, J.M.; Schumann, U. Circulating irisin in patients with polycystic ovary syndrome: A meta-analysis. Reprod. Biomed. Online 2018, 36, 172–180.
- Li, Q.; Jia, S.; Xu, L.; Li, B.; Chen, N. Metformin-induced autophagy and irisin improves INS-1 cell function and survival in high-glucose environment via AMPK/SIRT1/PGC-1α signal pathway. Food Sci. Nutr. 2019, 7, 1695–1703.
- Crujeiras, A.B.; Pardo, M.; Arturo, R.R.; Navas-Carretero, S.; Zulet, M.A.; Martínez, J.A.; Casanueva, F.F. Longitudinal variation of circulating irisin after an energy restriction-induced weight loss and following weight regain in obese men and women. Am J. Hum. Biol. 2014, 26, 198–207.
- Zhang, L.; Fang, X.; Li, L.; Liu, R.; Zhang, C.; Liu, H.; Tan, M.; Yang, G. The association between circulating irisin levels and different phenotypes of polycystic ovary syndrome. J. Endocrinol. Investig. 2018, 41, 1401–1407.
- Li, H.; Xu, X.; Wang, X.; Liao, X.; Li, L.; Yang, G.; Gao, L. Free androgen index and Irisin in polycystic ovary syndrome. J. Endocrinol. Investig. 2016, 39, 549–556.
- Gordon, C.M.; Ackerman, K.E.; Berga, S.L.; Kaplan, J.R.; Mastorakos, G.; Misra, M.; Murad, M.H.; Santoro, N.F.; Warren, M.P. Functional Hypothalamic Amenorrhea: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2017, 102, 1413–1439.
- Practice Committee of the American Society for Reproductive Medicine. Current evaluation of amenorrhea. Fertil. Steril. 2006, 86, S148–S155.
- Indirli, R.; Lanzi, V.; Mantovani, G.; Arosio, M.; Ferrante, E. Bone health in functional hypothalamic amenorrhea: What the endocrinologist needs to know. Front. Endocrinol. 2022, 13, 946695.
- Ma, Y.; Qiao, X.; Zeng, R.; Cheng, R.; Zhang, J.; Luo, Y.; Nie, Y.; Hu, Y.; Yang, Z.; Zhang, J.; et al. Irisin promotes proliferation but inhibits differentiation in osteoclast precursor cells. FASEB J. 2018, 32, 5813–5823.
- Lee, P.; Brychta, R.J.; Collins, M.T.; Linderman, J.; Smith, S.; Herscovitch, P.; Millo, C.; Chen, K.Y.; Celi, F.S. Cold-activated brown adipose tissue is an independent predictor of higher bone mineral density in women. Osteoporos. Int. 2013, 4, 1513–1518.
- Klangjareonchai, T.; Nimitphong, H.; Saetung, S.; Bhirommuang, N.; Samittarucksa, R.; Chanprasertyothin, S.; Sudatip, R.; Ongphiphadhanakul, B. Circulating sclerostin and irisin are related and interact with gender to influence adiposity in adults with prediabetes. Int. J. Endocrinol. 2014, 2014, 261545.
- Gamal, R.M.; Mohamed, M.E.; Hammam, N.; El Fetoh, N.A.; Rashed, A.M.; Furst, D.E. Preliminary study of the association of serum irisin levels with poor sleep quality in rheumatoid arthritis patients. Sleep Med. 2020, 67, 71–76.
- Slate-Romano, J.J.; Yano, N.; Zhao, T.C. Irisin reduces inflammatory signaling pathways in inflammation-mediated metabolic syndrome. Mol. Cell Endocrinol. 2022, 552, 111676.
- Zondervan, K.T.; Becker, C.M.; Koga, K.; Missmer, S.A.; Taylor, R.N.; Viganò, P. Endometriosis. Nat. Rev. Dis. Primers 2018, 4, 9.
- American Diabetes Association. Classification and diagnosis of diabetes: Standards of medical care in diabetes-2021. Diabetes Care 2021, 44, S15–S33.
- Wang, H.; Li, N.; Chivese, T.; Werfalli, M.; Sun, H.; Yuen, L.; Hoegfeldt, C.A.; Elise Powe, C.; Immanuel, J.; Karuranga, S.; et al. IDF Diabetes Atlas: Estimation of Global and Regional Gestational Diabetes Mellitus Prevalence for 2021 by International Association of Diabetes in Pregnancy Study Group’s Criteria. Diabetes Res. Clin. Pract. 2022, 183, 109050.
- Catalano, P.M.; Tyzbir, E.D.; Roman, N.M.; Amini, S.B.; Sims, E.A.H. Longitudinal changes in insulin release and insulin resistance in nonobese pregnant women. Am. J. Obstet. Gynecol. 1991, 165, 1667–1672.
- Vohr, B.R.; Boney, C.M. Gestational diabetes: The forerunner for the development of maternal and childhood obesity and metabolic syndrome? J. Matern.-Fetal Neonatal Med. 2008, 21, 149–157.
- Lain, K.Y.; Catalano, P.M. Factors That Affect Maternal Insulin Resistance and Modify Fetal Growth and Body Composition. Metab. Syndr. Relat. Disord. 2006, 4, 91–100.
- Briana, D.D.; Malamitsi-Puchner, A. Adipocytokines in Normal and Complicated Pregnancies. Reprod. Sci. 2009, 16, 921–937.
- Lowe, W.L.; Karban, J. Genetics, genomics and metabolomics: New insights into maternal metabolism during pregnancy. Diabet. Med. 2014, 31, 254–262.
- Al-Ghazali, M.J.; Ali, H.A.; Al-Rufaie, M.M. Serum irisin levels as a potential marker for diagnosis of gestational diabetes mellitus. Acta Biomed. 2020, 91, 56–63.
- Garcés, M.F.; Peralta, J.J.; Ruiz-Linares, C.E.; Lozano, A.R.; Poveda, N.E.; Torres-Sierra, A.L.; Eslava-Schmalbach, J.H.; Alzate, J.P.; Sánchez, Á.Y.; Sanchez, E.; et al. Irisin Levels During Pregnancy and Changes Associated With the Development of Preeclampsia. J. Clin. Endocrinol. Metab. 2014, 99, 2113–2119.
- Kuzmicki, M.; Telejko, B.; Lipinska, D.; Pliszka, J.; Szamatowicz, M.; Wilk, J.; Zbucka-Kretowska, M.; Laudanski, P.; Kretowski, A.; Gorska, M.; et al. Serum irisin concentration in women with gestational diabetes. Gynecol. Endocrinol. 2014, 30, 636–639.
- Yuksel, M.A.; Oncul, M.; Tuten, A.; Imamoglu, M.; Acikgoz, A.S.; Kucur, M.; Madazli, R. Maternal serum and fetal cord blood irisin levels in gestational diabetes mellitus. Diabetes Res. Clin. Pract. 2014, 104, 171–175.
- Erol, O.; Erkal, N.; Ellidağ, H.Y.; İsenlik, B.S.; Aydın, Ö.; Derbent, A.U.; Yılmaz, N. Irisin as an early marker for predicting gestational diabetes mellitus: A prospective study. J. Matern.-Fetal Neonatal Med. 2016, 29, 3590–3595.
- Ebert, T.; Stepan, H.; Schrey, S.; Kralisch, S.; Hindricks, J.; Hopf, L.; Platz, M.; Lossner, U.; Jessnitzer, B.; Drewlo, S.; et al. Serum levels of irisin in gestational diabetes mellitus during pregnancy and after delivery. Cytokine 2014, 65, 153–158.
- Piya, M.K.; Harte, A.L.; Sivakumar, K.; Tripathi, G.; Voyias, P.D.; James, S.; Sabico, S.; Al-Daghri, N.M.; Saravanan, P.; Barber, T.M.; et al. The identification of irisin in human cerebrospinal fluid: Influence of adiposity, metabolic markers, and gestational diabetes. Am. J. Physiol. Endocrinol. Metab. 2014, 306, 512–518.
- Roca-Rivada, A.; Castelao, C.; Senin, L.L.; Landrove, M.O.; Baltar, J.; Crujeiras, A.B.; Seoane, L.M.; Casanueva, F.F.; Pardo, M. FNDC5/Irisin Is Not Only a Myokine but Also an Adipokine. PLoS ONE 2013, 8, e60563.
- Cai, L.; Wu, W.; Lin, L.; Chen, Y.; Gao, R.; Shi, B.; Ma, B.; Chen, Y.; Jing, J. Association between plasma irisin and glucose metabolism in pregnant women is modified by dietary n-3 polyunsaturated fatty acid intake. J. Diabetes Investig. 2020, 11, 1326–1335.
- Wang, P.; Ma, H.H.; Hou, X.Z.; Song, L.L.; Song, X.L.; Zhang, J.F. Reduced plasma level of irisin in first trimester as a risk factor for the development of gestational diabetes mellitus. Diabetes Res. Clin. Pract. 2018, 142, 130–138.
- Cui, L.; Qiao, T.; Xu, F.; Li, Z.; Chen, T.; Su, H.; Chen, G.; Zhang, L.; Xu, D.; Zhang, X. Circulating irisin levels of prenatal and postnatal patients with gestational diabetes mellitus: A systematic review and meta-analysis. Cytokine 2020, 126, 154924.
- Ersahin, S.S.; Yurci, A. Cord blood and maternal serum preptin and irisin concentrations are regulated independently in GDM. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 1954–1958.
- Yong, E.; Logan, S. Menopausal osteoporosis: Screening, prevention and treatment. Singap. Med. J. 2021, 62, 159–166.
- Zhou, K.; Qiao, X.; Cai, Y.; Li, A.; Shan, D. Lower circulating irisin in middle-aged and older adults with osteoporosis: A systematic review and meta-analysis. Menopause 2019, 26, 1302–1310.
- Akkawi, I.; Zmerly, H. Osteoporosis: Current Concepts. Joints 2018, 6, 122–127.
- Kornel, A.; Den Hartogh, D.J.; Klentrou, P.; Tsiani, E. Role of the myokine irisin on bone homeostasis: Review of the current evidence. Int. J. Mol. Sci. 2021, 22, 9136.
- Qiao, X.; Nie, Y.; Ma, Y.; Chen, Y.; Cheng, R.; Yin, W.; Hu, Y.; Xu, W.; Xu, L. Irisin promotes osteoblast proliferation and differentiation via activating the MAP kinase signaling pathways. Sci. Rep. 2016, 6, 18732.





