Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ashoka Sriyani Gamage | -- | 2852 | 2024-02-19 09:56:26 | | | |
| 2 | Sirius Huang | Meta information modification | 2852 | 2024-02-21 02:00:09 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Attanayake, P.; Rupasinghe, D.; Gamage, A.; Madhujith, T.; Merah, O. Annona Seed Oils and Their Chemopreventive Potential. Encyclopedia. Available online: https://encyclopedia.pub/entry/55153 (accessed on 27 May 2026).
Attanayake P, Rupasinghe D, Gamage A, Madhujith T, Merah O. Annona Seed Oils and Their Chemopreventive Potential. Encyclopedia. Available at: https://encyclopedia.pub/entry/55153. Accessed May 27, 2026.
Attanayake, Prabash, Dinesha Rupasinghe, Ashoka Gamage, Terrence Madhujith, Othmane Merah. "Annona Seed Oils and Their Chemopreventive Potential" Encyclopedia, https://encyclopedia.pub/entry/55153 (accessed May 27, 2026).
Attanayake, P., Rupasinghe, D., Gamage, A., Madhujith, T., & Merah, O. (2024, February 19). Annona Seed Oils and Their Chemopreventive Potential. In Encyclopedia. https://encyclopedia.pub/entry/55153
Attanayake, Prabash, et al. "Annona Seed Oils and Their Chemopreventive Potential." Encyclopedia. Web. 19 February, 2024.
Copy Citation
Annona fruit, leaves, seeds, roots, and bark have been conventionally used in many countries for medical treatments as they are considered ideal sources of pharmacologically active compounds, but Annona remains an underutilized fruit in many countries. Annona seed oil contains numerous health-benefiting factors such as vitamins, minerals, bioactive compounds, fatty acids, antioxidants, and phenolic compounds, which are responsible for various biological activities, including antibacterial, antioxidant, and antitumor activities.
Annona muricata
Annona squamosa
Annona reticulata
Annona seed oil
antioxidants
chemopreventive potential
phytochemicals
1. Introduction
Chemoprevention is a method of controlling cancer with which the development of the illness can be completely avoided, slowed down, or reversed by one or more organic or inorganic substances. Annona might offer an indispensable choice besides chemotherapy and radiotherapy, especially for terminally ill patients, as the Annona genus contains secondary metabolites, including phytochemicals, antioxidants, essential oils, FAs, minerals, and vitamins, in nearly every component of Annona plants [1][2]. The structure of bioactive substances affects their ability to treat or prevent cancers. The efficiency of cytotoxic activity is determined by the high number of hydroxyl groups supported by the hydroxyl position flanking the γ-lactone ring and the stereo-chemical configuration of the ring [3][4]. Researchers have recently focused on the anticancer effects of Annona seeds, including ASSO, AMSO, and ARSO, and unique chemical compounds extracted from Annona have shown anticancer activities, which allows for further improvements to the available treatment methods, and this research needs to continue to allow for the development of more efficient but less expensive treatments against cancer [5][6][7].
2. Fatty Acids in Annona Seed Oils
FAs are usually found as components of many complex lipid molecules and can be identified as SFAs or UFAs based on the hydrogen atom bonding to the carbon atoms in the molecule. UFAs can be either MUFAs (mono-unsaturated fatty acids) or PUFAs [8]. In terms of composition, unlike animal oils that are made up mainly of SFAs, ASOs contain varying proportions of SFAs and UFAs tied in their TAG (triacylglyceride) molecules, and the percentage of UFAs is higher than that of SFAs (i.e., FAs esterified to a glycerol moiety). Additionally, this makes ASO an interesting source of two PUFAs; linoleic and linolenic acids, which are termed EFAs (essential fatty acids) because humans must obtain them from their diets. TAG is the principal means of storing FAs in biological systems and is considered cytotoxic [9].
The oil and fat available in Annona seeds consist of different FA compositions and lipid profiles. By utilizing the GC/MS method to examine the methyl esters of the seed oil’s fatty oil, it is possible to ascertain the chemical composition of the oil [10]. In order to be an oil that is nontoxic for humans, the concentration of linoleic acid and oleic acid should not be more than 12% and 1%, respectively, of that of FA methyl esters, according to European guidelines. Despite being toxic, ASO has large levels of UFAs, particularly oleic and linoleic acids. After detoxification, ASO can also be consumed because it contains a lot of UFAs [11]. Consumed oils have a significant impact on human physiology, including lipid metabolism, chronic illness prevention, and general well-being [12][13][14][15][16]. ASO can be widely used to cure cancer because it includes oleic acid, linoleic acid, stearic acid, palmitic acid, and other UFAs [17].
The physio-chemical properties, such as melting point, solid fat index, saponification value, cloud point, flash point, iodine value, color, viscosity, density, specific heat, and the heat of fusion, etc., of oils are largely dependent on the nature of their constituent FA and TAG compositions. Nonetheless, the FA and TAG compositions of different oils from diverse sources mostly determine their qualitative features and applications, and some of them can become source materials for functional lipids [18][19]. In this context, the FA type, the degree of saturation or desaturation, the method of delivery to cancer cells or hosts, and the tumor/cell type must be considered when evaluating the effects of FAs on tumor cell lines [20]. In the literature, the cytotoxic effects of FAs have been individually evaluated in different tumor cell lines with differing results [21][22][23][24][25]. For example, ASSO has been shown to suppress H22 solid tumor development and show selective cytotoxic activity against HepG2 cell lines, which might be attributed to the presence of UFAs and acetogenins [26].
According to the criteria adopted by the American National Cancer Institute and some cytotoxic screening assays, a crude extract oil showing an IC50 value < 30 μg·mL−1 in tumor cell lines is considered promising for anticancer drug development [27]. Since ASO has a high content of UFAs, a well-known antineoplastic activity, and different cytotoxic effects have been reported for some species of Annona, its cytotoxic effects against tumor and non-tumor cell lines should be further studied [28][29].
3. Phytochemicals in Annona Seed Oils
Plant species rich in phytochemicals are believed to reduce disease risk and have therapeutic characteristics, thus having potential medicinal value [11]. Phytochemicals are secondary metabolic compounds, namely acetogenins, alkaloids, phenolic compounds, essential oils, flavonoids, terpenoids, cyclopeptides, carotenoids, amino acids, etc. (Figure 1). Some phytochemicals, such as acetogenins, have shown neurotoxicity in in vitro and in vivo studies. More research is needed to quantify the number, of neurotoxic compounds and to determine the level of human exposure. Metabolic studies are also necessary to determine whether digestive processes decrease or increase the bioactivity and/or neurotoxicity of the active compounds. These studies have been extended to ASOs used in medicinal treatments [30].
Due to their capacity to scavenge potentially damaging free radicals, it has been discovered that the pharmacological effects of ASOs, including anticancer properties, are connected to the availability and content of phytochemicals and are influenced by the structure of these bioactive compounds. Annona has attracted the interest of individuals interested in the utilization of natural compounds that play a crucial role in the food and pharmaceutical industries [3][4]. Due to Annona’s alleged ethno-medical applications against tumors and cancer, extensive anticancer research has been carried out on these plants [31]. Some studies stated that ASO showed high antioxidant activities and phenolic contents and thus may have therapeutic uses [32][33].
The most prevalent bioactive molecules in the Anonaceace family are Anonaceous acetogenins, a special class of long-chain FA derivatives produced via the polyketide pathway. More than 120 acetogenins have been discovered in earlier phytochemical studies on Annona’s leaves, stems, bark, seeds, pulp, and fruit peel [34]. Solamin, annoreticulin-9-one, annomonicin, squamone, and rolliniastatin are the principal five annonaceous acetogenins with cytotoxic properties [6]. The mechanism of acetogenin’s cytotoxic action is the inhibition of mitochondrial complex I [35] and the inhibition of ubiquinone-linked NADH oxidase in the plasma membranes of cancerous cells, causing apoptosis [36]. A broad class of naturally occurring secondary metabolites is called alkaloids, and they are derived from amino acids or the transamination process [37]. Isolated and reported alkaloids include anomurine, anomuricine, isoquinoleic alkaloids, anonaine, and isolaureline [38]. Since the majority of phenolic compounds are water-soluble and the conventional medicinal extract is an aqueous infusion, they are regarded as the most significant phytochemicals [39].
The important bioactive constituents of ARSO are alkaloids, tannins, flavonoids, cardiac glycosides, phenols, terpenoids, steroids, and saponins [40]. The phytochemical concentrations ranged from 0.16 mg/100 g to 16.15 mg/100 g, with phenol found to be significantly prevalent, whilst tannins, alkaloids, and flavonoids were found to be moderately prevalent, and terpenoids and steroids had the lowest prevalence [41][42][43]. In general, flavonoids have the potential to significantly reduce the risk of disease through a variety of physiological processes, including cytotoxic and antioxidant actions [44]. Acetogenins have been identified from ARSOs, and some of these substances have demonstrated strong cytotoxic action against four human cancer cell lines (Table 1). The biological activity of annonacin was investigated after further purification, and it was discovered that this substance killed cells in several cancer cell lines. The biological effects of squamocin were also examined, and this research demonstrated that squamocin is a cytotoxic component for almost all cancer cell lines [45][46][47].
According to preliminary phytochemical analyses, the biological actions of ASSO are primarily brought on by phenolic chemicals such as alkaloids, peptides, amino acids, sterols, tannins, flavonoids, polysaccharides, and tocopherols, which are known to have strong anticancer activities, and they can be used to treat or prevent a variety of illnesses, including cancer [48][49][50][51][52]. ASSO has demonstrated substantial antitumor activity in vitro and in vivo against human hepatoma cells, suggesting the potential for the extract to be developed as a novel anti-liver cancer medication [53][54]. An earlier investigation into the pharmacology of ASSO revealed that it has specific cytotoxic effects on the HepG2 cell line [55].
Table 1. Reported cytotoxic activities of acetogenins present in ASOs.
| Seed Oil | Acetogenins | Reported Cytotoxic Activities |
|---|---|---|
| Annona reticulata seed oil * | bullatacin cis-/trans-isomurisolenin cis-/trans-bullatacinone annoreticulin annoreticulin-9-one cis-/trans-murisolinone squamocin annonacin squamocin |
|
| Annona squamosa seed oil ** | 12, 15-cis-squamostatin-A bullatacin squadiolin A, squadiolin B, squadfosacin B |
|
| Annona muricata Seed oil *** | muricins muricatetrocin A muricatetrocin B longifolicin corossolin corossolone annotacin A and B |
|
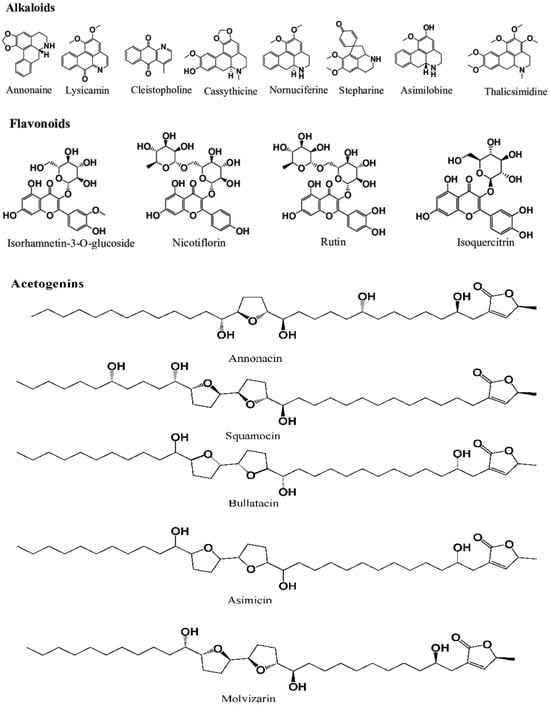
Figure 1. Structures of a few phytochemicals present in ASSO, ARSO, and AMSO [61].
The total acetogenin content in ASSO was 41.00 mg/g, and it exhibited remarkable cytotoxic activities against human tumor cell lines in a study and helped treat cancers of the liver, cervix, pancreas, etc. [53][56]. The study showed that various acetogenin types exhibit various inhibitory effects against various cancer cells (Table 1), and they have been shown to be the major bioactive compounds that possess strong antitumor/anticancer activity [33]. Using high-performance liquid chromatography (HPLC), two significant acetogenins have been identified in ASSO: 12, 15-cis-squamostatin-A (47.98 mg/g) and bullatacin (256.18 mg/g). Other constituents with antitumor activities include annosquacin A, B, and C, annosquatin A and B, squamostanin A, B, and D, squamostolide, and uvarigrandin A. Although it has been established that the fundamental mechanism is the stimulation of apoptosis, ASSO shows selectivity for certain cancer cells. Free radical production is also considered to be a key mechanism for the anticancer action of seed oils [53][58].
Cancer cells are frequently treated with A. muricata, and the entire plant has a promising future as a chemotherapeutic treatment for cancer, according to research. AMSO contains about thirty-seven phenolic compounds, such as acetogenins, alkaloids, phenolic compounds, etc. [62]. In addition to the vitamins and carotenes found in seeds, there have also been shown to be 37 volatile chemicals and 18 essential oils. It improves the capacity of non-cancerous cells in the body. Higher levels of flavonoids are present, which are crucial for reducing cancer cell migration and cellular proliferation, both of which cause a response to reactive oxygen species (ROS) [63]. AMSO-containing acetogenins play a crucial role in preventing cancers (Table 1). Soursop seeds are low in toxins such as tannins, phytates, and cyanides and are high in oil and vitamins [64]. Accordingly, ASOs have huge potential to be used in chemoprevention, which needs to be researched further.
4. Antioxidant Activity of Annona Seed Oils
Natural antioxidants include nitrogen compounds such as alkaloids, amino acids, peptides, and amines and derivatives of chlorophyll, carotenoids, and ascorbic acid, as well as phenolic compounds such as flavonoids, phenolic acids, and tannins [65]. Food antioxidants have a significant impact on maintaining good health and are directly linked to the prevention of degenerative diseases like cancer, cardiovascular disease, and neurological disorders [66]. Free radicals such as peroxide, hydroperoxide, or lipid peroxyl, which are harmful to the body, causing cancer, aging, and cellular damage, are scavenged by antioxidants and prevent the oxidative processes that cause degenerative illnesses [67]. Omega-3 (n-3) UFAs have been linked to cancer prevention [68]. Reactive oxygen species (ROS), also known as oxygen free radicals, play a dual role in biological systems, where both beneficial and detrimental effects are possible [69].
Different antioxidant screening methods were used to examine Annona’s capacity to scavenge free radicals, and the findings showed that ASO has significant antioxidants. Additional research may result in the development of powerful antioxidant agents from ASOs [31][70]. Antioxidant-based chemoprevention has been proposed to have a promising future in the provision of significant fundamental advantages to public health. Many scientists and medical professionals are now considering this as a crucial tactic for preventing, postponing, or even stopping the development of cancer [71]. Although numerous research studies have found a correlation between plant phenolic content and antioxidant capacity, it was noted that data about the antioxidant potential of ASO are rare to find [72]. To develop products, cosmetics, nutraceuticals, and biopharmaceuticals to combat cancer and to add to the database of highly significant medicinal plants, further investigation is needed to identify the bioactive compounds responsible for antioxidant activity [73].
Using a DPPH assay, a study found that ASSO has antioxidant activity, with an IC50 value of 7.88 g/mL [54]. In a different investigation, the antioxidant activity of oral-administered ASSO was evaluated in rats (150–210 g) with alcohol-induced liver damage. Superoxide dismutase, glutathione, and catalase levels were shown to be significantly raised following treatment with the ethanolic seed extract [74]. As a result, ASSO therapy results in the restoration of normal antioxidant enzymes. Through oral administration, ASSO reduced the development of H22 tumor cells in mice by a maximum inhibitory rate of 53.54% [56].
In addition to the six previously reported cytotoxic acetogenins annoreticuin, annoreticuin-9-one, cis-/trans-bullatacinone, bullatacin, cis-/trans-murisolinone, and squamocin, cis-/trans-isomurisolenin was also discovered [72][75]. There have also been reports of spathenelol, muurolene, copaene, and eudesmol, among other terpenes. Cyclohexapeptide glabrin A was recovered from a methanol extract of ARSO, and novel cyclooctapeptides were discovered by analyzing the sequence and three-dimensional structure of cycloreticulins A and B. N-fatty acyl tryptamines were also obtained [75][76]. AMSOs contain a considerable amount of antioxidants, and the antioxidant activity of soursop seed oil was 77.34% (dry basis) [77].
Mother nature has provided us with many naturally occurring medicinal plants, yet we often use them without realizing their therapeutic value [78]. Several medicinal plants have been examined scientifically, and it has been found that their secondary metabolites and bioactive chemicals have the potential to have an anticancer impact. The best example of this is the Annona species. The chosen A. squamosa, A. muricata, and A. reticulata have also grown in favor as a result of more modern research being conducted on the bioactivities and health benefits of various plant parts, such as the seeds, bark, leaves, fruits, etc. Although the seeds of the fruit are usually waste and the fruit itself is edible, literature reviews have reported that ASO primarily consists of fatty acids, phytochemicals, and antioxidants like polyketides, annonaceous acetogenins (neurotoxins), cyclopeptides, carbohydrates, proteins, lipids, oleic acid, and linoleic acid. The phytochemical composition and molecular basis of the bioactivities of Annona seeds have been the subject of a few studies. The demand for innovative treatments is growing as a result of the wide variety of cancer types and their primary causes. A novel cancer medication that can target and prevent apoptosis in cancer cells is the subject of intense research efforts today. Accordingly, it has been noted that plants of the same species collected from various areas have varying levels of secondary metabolites, indicating that the synthesis of a plant’s bioactive components may also vary, impacting how effective it is against cancer cells [79][80].
In conventional medicines, Annona is regarded as an ideal source of pharmacologically active compounds. Several prior investigations on the biological functions of the various Annona spp. components have been conducted [81][82]. Annona seed oil contains phytochemicals, antioxidants, and fatty acids found in ARSO, AMSO, and ASSO, which are employed for the treatment of cancer. In vitro and in vivo studies that show their usefulness in chemoprevention have been explored, as have anticancer agents. Fatty acid-derived lipids serve as the building blocks for cell membrane formation in addition to being crucial for hormone and signal transmission [83]. Due to their rapid proliferation and division, tumor cells require a lot of FAs. FAs make up a large portion of ASO. Tumor cells will undoubtedly absorb more ASO. Additionally, ASO primarily enters cells through passive transport [84].
References
- Pinto, A.C.d.Q.; Corderio, M.C.R.; Andrade, S.R.M.; Ferreira, F.R.; Filguriras, H.A.d.C.; Alves, R.E.; Kinpara, D.I. Annona Species. In International Centre for Underutilised Crops; Williams, J.T., Ed.; University of Southampton: Southampton, UK, 2005; p. 268.
- Lee, S.C.; Chan, J. Evidence for DNA damage as a biological link between diabetes and cancer. Chin. Med. J. 2015, 128, 1543–1548.
- Barbalho, S.; de Goulart, R.; Machado, F.V.F.; da Soares de Souza, M.; Santos Bueno, P.; Guiguer, E.; Araujo, A.; Groppo, M. Annona sp: Plants with Multiple Applications as Alternative Medicine—A Review. Curr. Bioact. Compd. 2012, 8, 277–286.
- Asare, G.A.; Afriyie, D.; Ngala, R.A.; Abutiate, H.; Doku, D.; Mahmood, S.A.; Rahman, H. Antiproliferative activity of aqueous leaf extract of Annona muricata L. on the prostate, BPH-1 cells, and some target genes. Integer. Cancer Ther. 2015, 14, 65–74.
- Taylor, P.; Arsenak, M.; Abad, M.J.; Fernandez, A.; Milano, B.; Gonto, R.; Ruiz, M.C. Screening of Venezuelan medicinal plant extracts for cytostatic and cytotoxic activity against Tumor cell lines. Phytother. Res. 2013, 27, 530–539.
- Chavan, S.S.; Shamkuwar, P.B.; Damale, M.G.; Pawar, D.P. A comprehensive review on Annona reticulata. Int. J. Pharm. Sci. Res. 2014, 5, 45–50.
- Mishra, S.; Ahmad, S.; Kumar, N.; Sharma, B.K. Annona muricata (the cancer killer): A Review. Glob. J. Pharm. Res. 2013, 2, 1613–1618.
- Samaram, S.; Mirhosseini, H.; Tan, C.P.; Ghazali, H.M. Ultrasound-assisted extraction (UAE) and solvent extraction of papaya seed oil: Yield, fatty acid composition, and triacylglycerol profile. Molecules 2013, 18, 12474–12487.
- Lima, L.A.R.S.; Johann, S.; Cisalpino, P.S.; Pimenta, L.P.S.; Boaventura, M.A.D. In vitro antifungal activity of fatty acid methyl esters of the seeds of Annona cornifolia A. St.-Hil. (Annonaceae) against pathogenic fungus Paracoccidioides brasiliensis. J. Braz. Trop. Med. 2011, 44, 777–780.
- Chowdhury, K.; Banu, L.A.; Khan, S.; Latif, A. Studies on the fatty acid composition of edible oils. Bangladesh J. Sci. Ind. Res. 2007, 42, 311–316.
- Akbar, E.; Yaakob, Z.; Kamarudin, S.K.; Smail, M.; Salimon, J. Characteristic and composition of the Jatropha curcas oil seed from Malaysia and its potential as biodiesel feedstock. Eur. J. Sci. Res. 2009, 29, 39–404.
- Thang, T.D.; Kuo, P.C.; Huang, G.J.; Hung, N.H.; Huang, B.S.; Yang, M.L. Chemical Constituents from the Leaves of Annona reticulata and Their Inhibitory Effects on NO Production. Molecules 2013, 18, 4477–4486.
- Valantina, S.R.; Neelamegan, P. Antioxidant potential in vegetable oil—A review paper. Res. J. Chem. Environ. 2012, 16, 87–94.
- Parry, J.; Su, L.; Luther, M.; Zhou, K.; Yurawecz, M.P.; Whittaker, P.; Yu, L. Fatty acid composition and antioxidant properties of cold-pressed marionberry, boysenberry, redraspberry, and blueberry seed oils. J. Agri. Food Chem. 2005, 53, 566–573.
- Mabaleha, M.B.; Mitei, Y.C.; Yeboah, S.O. A comparative study of the properties of selected melon seed oils as potential candidates for development into commercial edible vegetable oils. J. Am. Oil Chem. Soc. 2007, 84, 31–36.
- Anhwange, B.A.; Ajibola, V.; Oniye, S.J. Chemical studies of the seeds Moringa oleifera (LAM) detarium mirocarpum. J. Bio. Sci. 2004, 4, 711–715.
- Nie, Y.L.; Liu, K.X.; Mao, X.Y.; Li, Y.L.; Li, J.; Zhang, M.M. Effect of injection of Brucea javanica oil emulsion plus chemoradiotherapy for lung cancer: A review of clinical evidence. J. Evid.-Based Med. 2012, 5, 216–225.
- Rana, V.S. Fatty Oil and Fatty Acid Composition of Annona squamosa Linn. Seed Kernels. Int. J. Fruit Sci. 2014, 15, 79–84.
- Liyanage, T.; Madhujith, T.; Wijesinghe, K.G.G. Comparative study on major chemical constituents in volatile oil of true cinnamon (Cinnamomum verum) and five wild cinnamon species grown in Sri Lanka. Trop. Agri Res. 2017, 28, 270–280.
- Jiang, W.G.; Bryce, R.P.; Horrobin, D.F. Essential fatty acids: Molecular and cellular basis of their anti-cancer action and clinical implications. Crit. Rev. Oncol. Hematol. 1998, 27, 179–209.
- Meterissian, S.H.; Forse, R.A.; Steele, G.D.; Thomas, P. Effect of membrane free fatty acid alterations on the adhesion of human colorectal carcinoma cells to liver macrophages and extracellular matrix proteins. Cancer Lett. 1995, 89, 145–152.
- Du Toit, P.J.; Van Aswegen, C.H.; Du Plessis, D.J. The effect of essential fatty acids on growth and urokinase-type plasminogen activator production in human prostate DU-145 cells, Prostaglandins Leukot. Essent. Fat. Acids. 1996, 55, 173–177.
- Soto-Guzman, A.; Villegas-Comonfort, S.; Cortes-Reynosa, P.; Perez Salazar, E. Role of arachidonic acid metabolism in Stat5 activation induced by oleic acid in MDA-MB-231 breast cancer cells. Prostaglandins. Leukot. Essent. Fat. Acids. 2013, 88, 243–249.
- Moon, H.S.; Batirel, S.; Mantzoros, C.S. Alpha linolenic acid and oleic acid additively down-regulate malignant potential and positively cross-regulate AMPK/S6 axis in OE19 and OE33 esophageal cancer cells. Metabolism 2014, 63, 1447–1454.
- Wang, Z.; Liu, D.; Zhang, Q.; Wang, J.; Zhan, J.; Xian, X.; Du, Z.; Wang, X.; Hao, A. Palmitic acid affects proliferation and differentiation of neural stem cells in vitro. J. Neurosci. Res. 2014, 92, 574–586.
- Chen, Y.; Chen, Y.; Shi, Y.; Ma, C.; Wang, X.; Li, Y.; Miao, Y.; Chen, J.; Li, X. Antitumor activity of Annona squamosa seed oil. J. Ethnopharmacol. 2016, 193, 362–367.
- Suffness, M.; Pezzuto, J.M. Assays related to cancer drug discovery. In Methods in Plant Biochemistry: Assays for Bioactivity; Hostettmann, K., Ed.; Academic Press: London, UK, 1990; pp. 71–133.
- Fauser, J.K.; Prisciandaro, L.D.; Cummins, A.G.; Howarth, G.S. Fatty acids as potential adjunctive colorectal chemotherapeutic agents. Cancer Biol. Ther. 2011, 11, 724–731.
- Lima, L.A.R.S.; Alves, T.M.A.; Zani, C.L.; Pimenta, L.P.S.; Boaventura, M.A. Antioxidant and citotoxic potential of fatty acid methil esters from the seeds of Annona cornifolia A. ST.-Hil. (Annonaceae). Food Res. Int. 2012, 48, 873–875.
- Thiviya, P.; Gunawardena, N.; Gamage, A.; Madhujith, T.; Merah, O. Apiaceae Family as a Valuable Source of Biocidal Components and their Potential Uses in Agriculture. Horticulturae 2022, 8, 614.
- Adewole, S.; Ojewole, J. Protective effects of Annona muricate Linn. Leaf aqueous extract on serum lipid profiles and oxidative stress in hepatocytes of streptozotocin-treated diabetic rats. Afr. J. Tradit. Complement. Altern. Med. 2009, 6, 30–41.
- Kadarani, D.K.; Setyadjit, S.; Seno, D.S.H.; Sakashih, E. Total phenol & antioxidant from seed & peel of ripe and unripe of Indonesian sugar apple (Annona squamosa L.) extracted with various Pharmacyts IOSR. J. Pharm. 2013, 5, 20–25.
- Eshra, D.H.; Shehata, A.R.; Ahmed, A.N.A.; Saber, J.I. Physicochemical Properties of the Seed Kernels and the Oil of Custard Apple (Annona squamosa L.). Int. J. Food Biotech. 2019, 4, 87–93.
- Sun, S.; Liu, J.; Zhou, N.; Zhu, W.; Dou, Q.P.; Zhou, K. Isolation of three new annonaceous acetogenins from Graviola fruit (Annona muricata) and their anti-proliferation on human prostate cancer cell PC-3. Bioorg. Med. Chem. Let. 2016, 26, 4382–4385.
- Lannuzel, A.; Michel, P.P.; Hôglinger, G.U.; Champy, P.; Jousset, A.; Medja, F.; Ruberg, M. The mitochondrial complex I inhibitor annonacin is toxic to mesencephalic dopaminergic neurons by impairment of energy metabolism. Neuroscience 2003, 121, 287–296.
- Alali, F.Q.; Xiao-Xi, L.; McLaughlin, J.L. Annonaceous acetogenins: Recent progress. J. Nat. Prod. 1999, 62, 504–540.
- Dey, P.; Kundu, A.; Kumar, A.; Gupta, M.; Lee, B.M.; Bhakta, T.; Dash, S.; Kim, H.S. Analysis of alkaloids (indole alkaloids, isoquinoline alkaloids, tropane alkaloids). In Recent Advances in Natural Products Analysis; Elsevier: Amsterdam, The Netherlands, 2020; pp. 505–567.
- Leboeuf, M.; Cavé, A.; Bhaumik, P.; Mukherjee, B.; Mukherjee, R. The phytochemistry of the Annonaceae. Phytochemistry 1980, 21, 2783–2813.
- Coria-Téllez, A.V.; Montalvo-Gónzalez, E.; Yahia, E.M.; Obledo-Vázquez, E.N. Annona muricata: A comprehensive review on its traditional medicinal uses, phytochemicals, pharmacological activities, mechanisms of action and toxicity. Arab. J. Chem. 2016, 11, 662–691.
- Kumar, M.; Changan, S.; Tomar, M.; Prajapati, U.; Saurabh, V.; Hasan, M.; Sasi, M.; Maheshwari, C.; Singh, S.; Dhumal, S.; et al. Custard Apple (Annona squamosa L.) Leaves: Nutritional Composition, Phytochemical Profile, and Health-Promoting Biological Activities. Biomolecules 2021, 11, 614.
- Galeane, M.C.; Martins, C.H.G.; Massuco, J.; Bauab, T.M.; Sacramento, L.V.S. Phytochemical screening of Azadirachta indica A. Juss for antimicrobial activity. Afr. J. Microbiol. Res. 2017, 11, 117–122.
- Ramadass, N.; Subramanian, N.S. Study of phytochemical screening of neem (Azadirachta indica). Int. J. Zool. Stud. 2018, 3, 209–212.
- James, J.; Veettil, A.K.T.; Pratyush, K.; Misra, C.S.; Sagadevan, L.D.M.; Thankamani, V. Phytochemical Investigation and Antibacterial Activity of the Fruits of Alstonia Scholaris. Int. J. Phytopharm. 2012, 3, 74–77.
- Pandey, N.; Barve, D. Phytochemical and Pharmacological Review on Annona squamosa Linn. Int. J. Res. Pharm. Biomed. Sci. 2011, 2, 1404–1412.
- Chang, F.R.; Wu, Y.C.; Duh, C.Y.; Wang, S.K. Studies on the acetogenins of Formosan annonaceous plants. II. Cytotoxic acetogenins from Annona reticulata. J. Nat. Prod. 1993, 56, 1688–1694.
- Yuan, S.S.; Chang, H.L.; Chen, H.W.; Yeh, Y.T.; Kao, Y.H.; Lin, K.H.; Wu, Y.C.; Su, J.H. Annonacin, a mono-tetrahydrofuran acetogenin, arrests cancer cells at the G1 phase and causes cytotoxicity in a Bax- and caspase-3-related pathway. Life Sci. 2003, 72, 2853–2861.
- Yuan, S.S.; Chang, H.L.; Chen, H.W.; Kuo, F.C.; Liaw, C.C.; Su, J.H.; Wu, Y.C. Selective cytotoxicity of squamocin. on T24 bladder cancer cells at the S-phase via Bax-, Bad-, and caspase-3-related pathways. Life Sci. 2006, 78, 869–874.
- Siebra, C.A.; Nardin, J.M.; Florão, A.; Rocha, F.H.; Bastos, D.Z.; Oliveira, B.H.; Weffort-Santos, A.M. Potencial antiinflamatório de Annona glabra, Annonaceae. Rev. Bras. Farmacogn. 2009, 19, 82–88.
- Kintzios, S.E. Terrestrial plant-derived anticancer agents and plant species used in anticancer research. Crit. Rev. Plant Sci. 2006, 25, 79–113.
- Rahman, M.M.; Parvin, S.; Haque, M.E.; Islam, M.E.; Mosaddik, M.A. Antimicrobial and cytotoxic constituents from the seeds of Annona squamosa. Fitoterapia 2005, 76, 484–489.
- Shirwaikar, A.; Rajendran, K.; Kumar, C.D. Oral antidiabetic activity of Annona squamosa leaf alcohol extract in NIDDM rats. Pharm. Biol. 2004, 42, 30–35.
- Madhuri, S.; Pandey, G. Some anticancer medicinal plants of foreign origin. Curr. Sci. 2009, 96, 779–783.
- Chen, Y.; Chen, J.W.; Zhai, J.H.; Wang, Y.; Wang, S.L.; Li, X. Antitumor activity and toxicity relationship of annonaceous acetogenins. Food Chem. Toxicol. 2013, 58, 394–400.
- Rakesh, R.; Mahendra, S. Coumarin lignans from the seeds of Annano Squamosa Linn. J. Chem. 2009, 6, 518–522.
- Richmond, A.; Su, Y.J. Mouse xenograft models vs GEM models for human cancer therapeutics. Dis. Models Mech. 2008, 1, 78–82.
- Dąbrowski, G.; Czaplicki, S.; Konopka, I. Composition and quality of poppy (Papaver somniferum L.) seed oil depending on the extraction method. LWT 2020, 134, 10167.
- Liaw, C.C.; Yang, Y.L.; Chen, M.; Chang, F.R.; Chen, S.L.; Wu, S.H.; Wu, Y.C. Mono-tetrahydrofuran annonaceous acetogenins from Annona squamosa as cytotoxic agents and calcium ion chelators. J. Nat. Prod. 2008, 71, 764–771.
- Yang, H.J.; Zhang, N.; Chen, J.W.; Wang, M.Y. Two new cytotoxic acetogenins from Annona squamosa. J. Asian Nat. Prod. Res. 2009, 11, 250–256.
- Kuete, V.; Dzotam, J.K.; Voukeng, I.K.; Fankam, A.G.; Efferth, T. Cytotoxicity of methanol extracts of Annona muricata, Passiflora edulis, and nine other Cameroonian medicinal plants towards multi-factorial drug-resistant cancer cell lines. SpringerPlus 2016, 5, 1666.
- Chang, F.R.; Wu, Y.C. Novel cytotoxic annonaceous acetogenins from Annona muricate. J. Nat. Prod. 2001, 64, 925–931.
- Kazman, B.S.M.; Al, J.E.; Harnett, A.; Jane, R.H. The Phytochemical Constituents and Pharmacological Activities of Annona atemoya: A Systematic Review. Pharmaceuticals 2020, 13, 269.
- Yang, C.; Gundala, S.R.; Mukkavilli, R.; Vangala, S.; Reid, M.D.; Aneja, R. Synergistic interactions among flavonoids and acetogenins in Graviola (Annona muricata) leaves confer protection against prostate cancer. Carcinogenesis 2015, 36, 656–665.
- Moghadamtousi, S.Z.; Fadaeinasab, M.; Nikzad, S.; Mohan, G.; Ali, H.M.; Kadir, H.A. Annona muricata (Annonaceae): A review of its traditional uses, isolated acetogenins, and biological activities. Int. J. Mol. Sci. 2015, 16, 15625.
- Badrie, N.; Schauss, A. Soursop (Annona muricata L.): Composition, nutritional value, medicinal uses, and toxicology. Bioact. Food Prom. Health 2010, 39, 621–643.
- Hassimotto, N.M.A.; Genovese, M.E.; Lajolo, F.M. Antioxidant capacity of Brazilian fruit, vegetables and commercially-frozen fruit pulps. J. Food Comp. Anal. 2009, 22, 394–396.
- Diplock, A.T.; Miller, N.J.; Rice-Evans, C.A. Evaluation of the total antioxidant activity as a marker of the deterioration of apple juice on storage. J. Agri Food Chem. 1995, 43, 1794–1801.
- Halliwell, B. Free radicals and antioxidants. Nutr. Rev. 1994, 52, 253–265.
- Morris-Stiff, G.J.; Bowrey, D.J.; Oleesky, D.; Davies, M.; Clark, G.W.; Puntis, M.C. The antioxidant profiles of patients with recurrent acute and chronic pancreatitis. Am. J. Gastroenterol. 1999, 94, 2135–2140.
- Bailly, C.; El-Maarouf-Bouteau, H.; Corbineau, F. From intracellular signaling networks to cell death: The dual role of reactive oxygen species in seed physiology. Comptes R. Biol. 2008, 331, 806–814.
- Vikas, B.; Akhil, B.S.; Remani, P.; Sujathan, K. Free Radical Scavenging Properties of Annona squamosa. Asian Pac. J. Cancer Prev. 2017, 18, 2725–2731.
- Shureiqi, I.; Chen, D.; Lotan, R.; Yang, P.; Newman, R.A.; Fischer, S.M.; Lippman, S.M. 15-Lipoxygenase-1 mediates nonsteroidal anti-inflammatory drug-induced apoptosis independently of cyclooxygenase-2 in colon cancer cells. Cancer Res. 2000, 60, 6846–6850.
- Shehata, M.G.; Abu-Serie, M.M.; El-Aziz, A.; Mohammad, N.; El-Sohaimy, S.A. Nutritional, phytochemical, and in vitro anticancer potential of sugar apple (Annona squamosa) fruits. Sci. Rep. 2021, 11, 6224.
- Ma, C.; Chen, Y.; Chen, J.; Li, X.; Chen, Y. A Review on Annona squamosa L.: Phytochemicals and Biological Activities. Am. J. Chin. Med. 2017, 45, 933–964.
- Zahid, M.; Arif, M.; Rahman, M.A.; Singh, K.; Mujahid, M. Solvent extraction and gas chromatography–mass spectrometry analysis of Annona squamosa L. seeds for determination of bioactive, fatty acid/fatty oil composition, and antioxidant activity. J. Diet. Suppl. 2018, 15, 613–623.
- Chang, F.R.; Chen, J.L.; Chiu, H.F.; Wu, M.J.; Wu, Y.C. Acetogenins from seeds of Annona reticulata. Phytochemistry 1998, 47, 1057–1061.
- Meada, U.; Hara, N.; Fujimoto, Y.; Srivastava, A.; Gupta, Y.K.; Sahai, M. ‘N-fatty Acyl Tryptamines from Annona reticulata. Phytochemistry 1993, 34, 1633–1635.
- Silva, A.C.; Jorge, N. Bioactive compounds of the liquid fractions of agro industrial waste. Food Res. Int. 2014, 66, 493–500.
- Pathirana, C.K.; Madhujith, T.; Eeswara, J. Bael (Aegle marmelos L. Corrêa), a Medicinal Tree with Immense Economic Potentials. Adv. Agric. 2020, 2020, 8814018.
- Nugraha, A.S.; Damayanti, Y.D.; Wangchuk, P.; Keller, P.A. Anti-infective and anti-cancer properties of the Annona species: Their ethnomedicinal uses, alkaloid diversity, and pharmacological activities. Molecules 2019, 24, 4419.
- Shital, P.; Rujuta, A.; Sanjay, M. Transbronchial needle aspiration cytology (TBNA) in endobronchial lesions: A valuable technique during bronchoscopy in diagnosing lung cancer and it will decrease repeat bronchoscopy. J. Cancer Res. Clin. Oncol. 2014, 140, 809–815.
- Fridlender, M.; Kapulnik, Y.; Koltai, H. Plant-derived substances with anti-cancer activity: From folklore to practice. Front. Plant Sci. 2015, 6, 799.
- Weerasinghe, M.G.W.K.; Dahanayake, N. A review of Annona species in Sri Lanka. Int. J. Minor. Fruits Med. Aromat. Plants 2022, 8, 1–6.
- Santos, C.R.; Schulze, A. Lipid metabolism in cancer. FEBS J. 2012, 279, 2610–2623.
- Dallavalle, S.; Dobricic, V.; Lazzarato, L.; Gazzano, E.; Machuqueiro, M.; Pajeva, I.; Tsakovska, I.; Zidar, N.; Fruttero, R. Improvement of conventional anti-cancer drugs as new tools against multidrug resistant tumors. Drug Resist. Updates 2020, 50, 100682.
More
Information
Subjects:
Food Science & Technology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
726
Revisions:
2 times
(View History)
Update Date:
21 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





