Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sebnem Sozcu | -- | 6250 | 2023-12-27 22:40:11 | | | |
| 2 | Rita Xu | Meta information modification | 6250 | 2023-12-28 02:59:39 | | | | |
| 3 | Rita Xu | Meta information modification | 6250 | 2023-12-28 03:15:11 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Sozcu, S.; Venkataraman, M.; Wiener, J.; Tomkova, B.; Militky, J.; Mahmood, A. Incorporation of Cellulose-Based Aerogels into Textile Structures. Encyclopedia. Available online: https://encyclopedia.pub/entry/53197 (accessed on 03 June 2026).
Sozcu S, Venkataraman M, Wiener J, Tomkova B, Militky J, Mahmood A. Incorporation of Cellulose-Based Aerogels into Textile Structures. Encyclopedia. Available at: https://encyclopedia.pub/entry/53197. Accessed June 03, 2026.
Sozcu, Sebnem, Mohanapriya Venkataraman, Jakub Wiener, Blanka Tomkova, Jiri Militky, Aamir Mahmood. "Incorporation of Cellulose-Based Aerogels into Textile Structures" Encyclopedia, https://encyclopedia.pub/entry/53197 (accessed June 03, 2026).
Sozcu, S., Venkataraman, M., Wiener, J., Tomkova, B., Militky, J., & Mahmood, A. (2023, December 27). Incorporation of Cellulose-Based Aerogels into Textile Structures. In Encyclopedia. https://encyclopedia.pub/entry/53197
Sozcu, Sebnem, et al. "Incorporation of Cellulose-Based Aerogels into Textile Structures." Encyclopedia. Web. 27 December, 2023.
Copy Citation
Given their exceptional attributes, aerogels are viewed as a material with immense potential. Being a natural polymer, cellulose offers the advantage of being both replenishable and capable of breaking down naturally. Cellulose-derived aerogels encompass the replenish ability, biocompatible nature, and ability to degrade naturally inherent in cellulose, along with additional benefits like minimal weight, extensive porosity, and expansive specific surface area.
bio-based aerogel
multifunctional properties
thermal insulation
flame retardant
textile applications
1. Introduction
Aerogels are fascinating substances of the twenty-first century due to their unique structure [1]. They possess remarkable properties like high porosity, low density, huge surface area, and superb heat and sound insulation. However, their low mechanical strength and high production costs restrict their usefulness [2].
Aerogels are extremely porous nanostructured materials invented by Kistler in 1931 [2][3]. Aerogels may be created using either supercritical drying or freeze-drying processes. The material’s microporous structure stays intact throughout drying in both circumstances. Kistler’s first aerogel, prepared by supercritical drying, was silica-based. During the creation of silica aerogels, toxic precursors were used, and the aerogels formed were not biodegradable [4]. The costly process of making this type of aerogel, with the restrictions described earlier (non-biodegradable and toxic precursors), confirms the restricted use of this material [4].
Aerogels are also used to develop a wide range of tools, such as in optoelectronics, adsorption catalysis, sound insulation, pharmaceutical materials, and aerospace materials [5][6][7][8][9][10]. However, some drawbacks, together with the huge expenses associated with their fabrication, have severely limited the use of aerogels [11]. Aerogels can be formed using a wide range of materials, including inorganic ones [12], synthetic polymer-based materials [13], and natural polymer-based materials [14][15] such as cellulose [16][17], depending on the starting substance used for their manufacture [2][18]. Aerogels, which are praised for being among the extraordinary materials of the twenty-first century due to their outstanding mechanical properties and extremely low weight, have gained attention as “miracle materials”. These aerogels have drawn a lot of interest from researchers in a variety of sectors due to their remarkable endurance under harsh working conditions [19].
Cellulose aerogels, in particular, have cellulose’s renewability, biocompatibility, and biodegradability, as well as additional benefits like low density values (0.0005–0.35 g·cm−3), enhanced porosity (84.0–99.9%), and a huge specific surface area, representing the materials with the highest potential in the 21st century [11]. Cellulose aerogels possess greater compressive strength (0.0052–16.67 MPa) and superior biodegradability [11]. So, cellulose aerogels are eco-friendly and versatile modern materials with enormous opportunities for application in adsorption and oil/water separation, heat separation, biomedical materials, metal nanoparticle/metal oxide carriers, and carbon aerogel precursors, and they possess high reusability, thereby minimizing economic losses. Moreover, the notable advantage of bio-based aerogels lies in their natural decomposition, avoiding additional environmental harm. Drawing on knowledge from the body of literature already in existence, future studies related to bio-based aerogel absorbents may center on evaluating their stability in harsh conditions. This strategy could lead to improvements in the development of more effective solutions, especially for dealing with oil spills, which would eventually reduce financial losses [20].
Cellulose aerogels, as porous solids, are typically produced through a two-step process: Cellulose or cellulose derivatives are dissolved/dispersed, forming a cellulose sol using the sol–gel method. Subsequently, the cellulose sol is dried by preserving the sol’s three-dimensional porous structure [11].
2. Creating Cellulose Aerogels
Cellulose can be derived from various sources [21][22][23], such as plants or plant-based materials, e.g., rice straw. It can also be derived from the most commonly used sources [24], such as cannabis [25], cotton [26][27], wood [28][29], potato tubers [30], coconut (coir) [31], and bagasse [32]. The extraction of cellulose involves obtaining it from specific plant species, employing various production processes, including pretreatment, post-treatment, and disintegration processes, which determine its performance characteristics, like size, molecular chain length (degree of polymerization (DP)), thermal stability, and degree of crystallinity [33][34]. As a result, the plant source significantly impacts the structure and performance of cellulose aerogels [34][35].
Cellulose is a member of the polysaccharide family, which are the primary building elements for plants. Plants are the first or most basic link in the food chain (which details the feeding interactions of all living organisms) [35][36][37]. Cellulose is a key component of many natural fibers, including cotton and other plants [38][39].
Cellulose exhibits insolubility in water and the majority of common solvents [36], owing to strong intramolecular and intermolecular hydrogen bonding between individual chains [35]. Cellulose is employed in a variety of products despite its poor solubility, including composites, netting, upholstery, coatings, packaging, and paper. To make cellulose more processable and to produce cellulose derivatives, which can be customized for certain industrial purposes, cellulose is chemically modified [40][41].
Aerogel materials can benefit from the mechanical qualities and moisture affinity of cellulose and its derivatives [42][43][44]. To keep their solid network, the cellulose is dissolved in appropriate media, such as NMMO, hydrates of some molten salts, and ionic liquids. Then drying processes such as supercritical drying or freeze-drying [45][46][47][48][49] are used for the preparation of cellulose aerogels. There are several strategies for creating cellulose aerogels that have been published in the literature [46][47][50][51][52]. The manufacturing of cellulose aerogels and their applications are depicted in Figure 1.
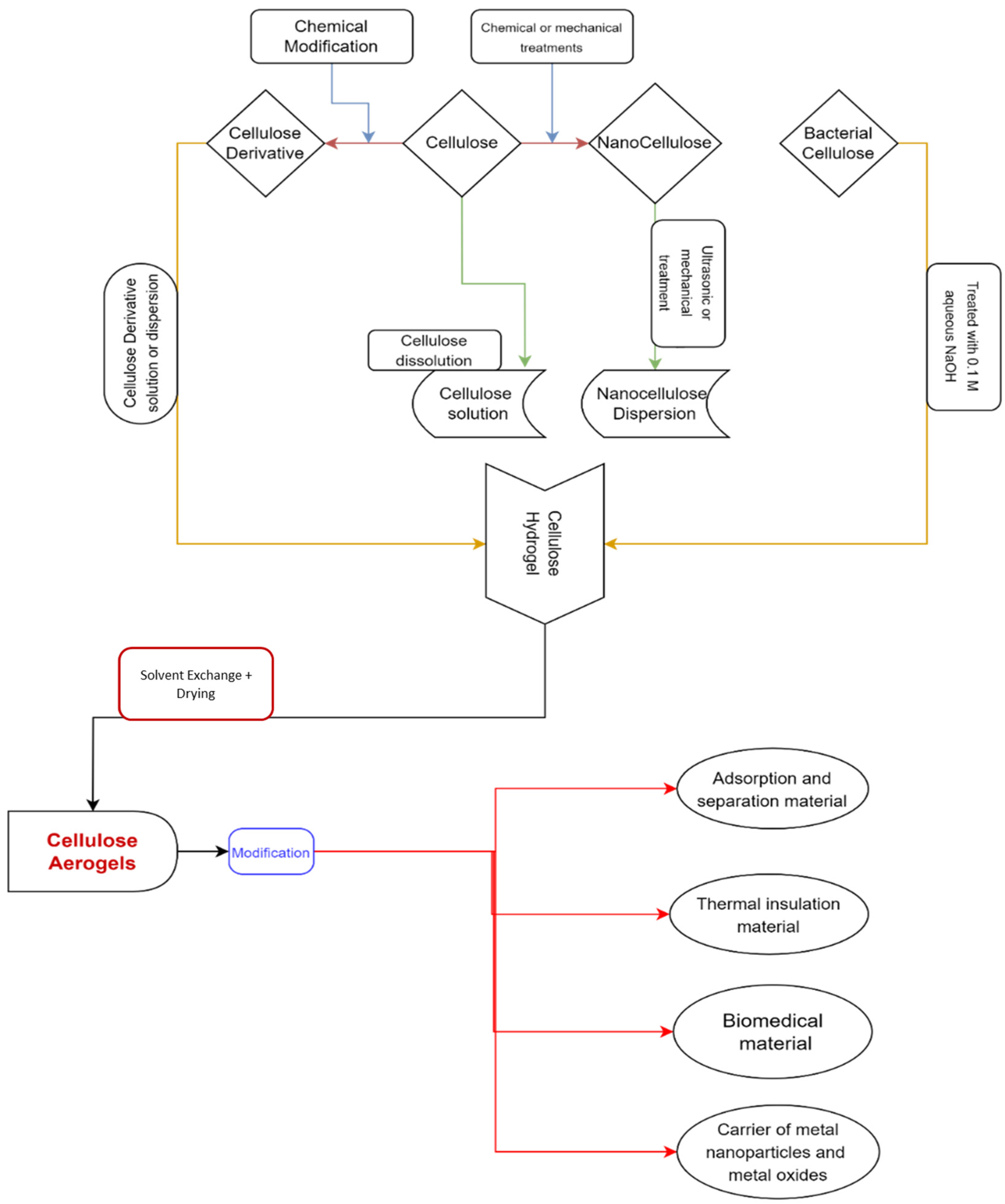
Figure 1. A graphical representation depicting the manufacture and application of cellulose aerogels.
Furthermore, employing cellulose as a precursor in the production of aerogels has the following benefits: (1) The supply of cellulose raw materials is infinite and renewable. (2) Because the cellulose chain contains a lot of hydroxyl groups, no crosslinking agent is needed during the aerogel production process. A stable three-dimensional (3D) network structure can be created by employing hydrogen bond physical crosslinking both within and between molecules, making the aerogel production technique incredibly easy. (3) Chemical cellulose modification is a quick and easy method of enhancing the structural integrity and mechanical strength of cellulose aerogels. The performance and concentration of the cellulose have a significant impact on the manufacturing process and structural characteristics of cellulose aerogels [11]. To create cellulose gels, the cellulose morphology and the structure of the cellulose fibers must be changed. This is done by using a suitable solvent [53][54][55] that is capable of breaking the large hydrogen bonding network that does not degrade along the cellulose polymer chain, or by beginning polymer chain derivation processes [46].
Because of their numerous interior pores and effective heat insulation, cellulose aerogels are among the most capable thermal insulation materials for construction or domestic applications (for example, refrigerator insulation materials) and have the potential to improve their poor properties, such as flame retardancy, huge swelling, and antibacterial properties [56][57].
2.1. Sol–Gel Procedure
As shown in Figure 2, all steps of the production process influence the gel structure, determining its characteristics and, as a result, its utilization [58]. Not only the material type used to prepare the cellulose aerogel, the agent used to dissolve the cellulose material, the type of drying method, etc., but also other techniques, as listed below, are frequently employed in improving the structural attributes and characteristics of cellulose-based gels [59].
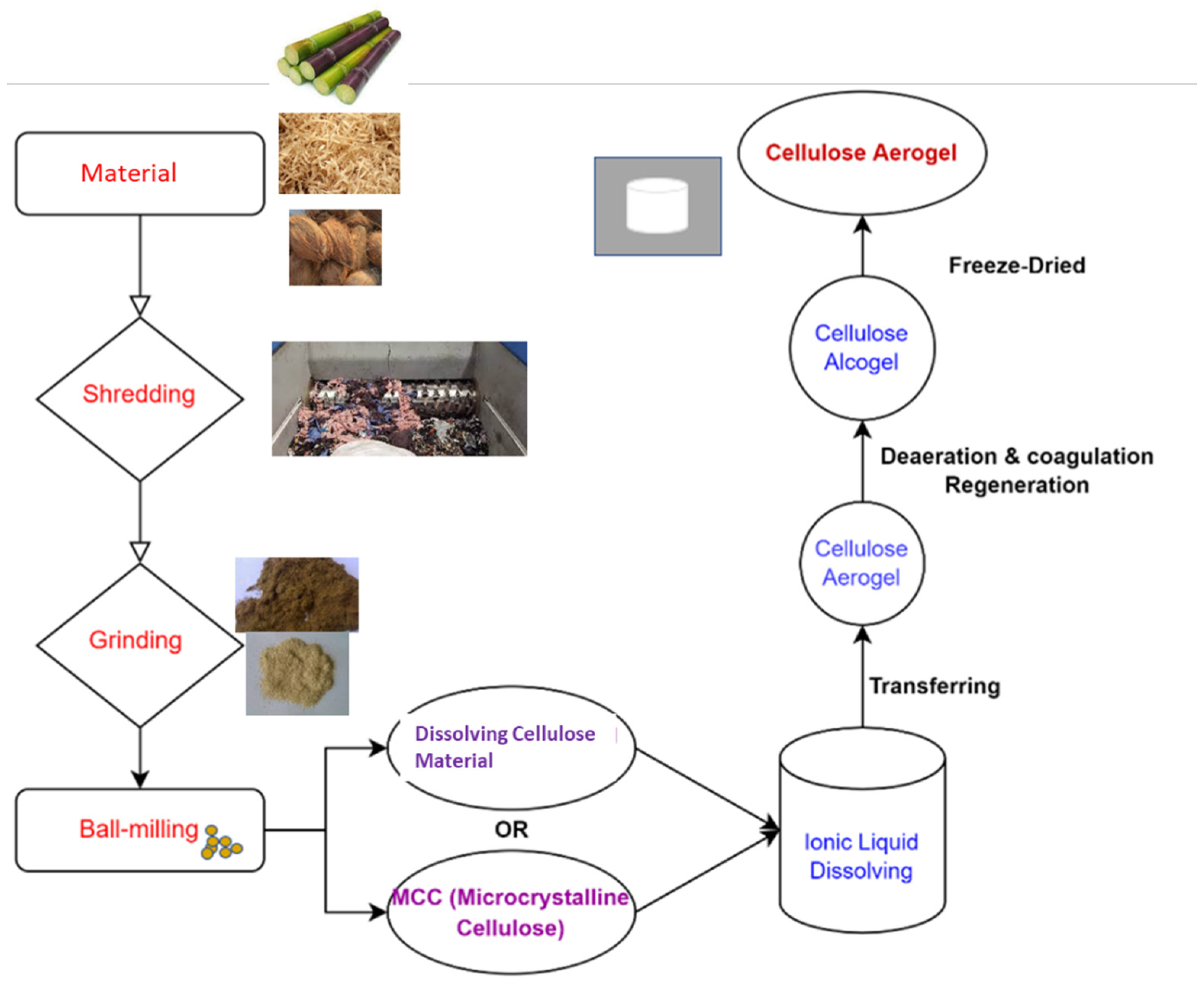
Figure 2. A schematic diagram depicting the cellulose aerogel preparation process.
-
A colloidal suspension is produced by dispersing solid nanoscale particles formed from a reactant in a liquid.
-
Adding an acidic or basic catalyst initiates crosslinking and leads to the linkage and spreading of particles, forming an interlinked network configuration.
-
Gel aging: to strengthen the gel’s backbone and material toughness, it is aged in its mother solution.
-
To avoid gel fractures, the solvent is extracted from the pores of the gel during drying. [60].
The procedure starts with the creation of a colloidal solution, often known as a sol. A solution of reactants and solvents contains solid nanoparticles or initiator materials.
In the course of this procedure, the introduction of a catalyst aids in polymerization, involving hydrolysis and polycondensation chemical reactions. This leads to the formation of crosslinks and branches between polymeric species, giving rise to the creation of a three-dimensional porous network within a wet, gel-like structure before aging as mentioned in the bullet points above for creating aerogels [60][61].
Sol–gel products may be manufactured from a variety of substances, including oxides (such as silicon dioxide and oxide minerals), natural compounds (such as large molecules like plant-derived materials), and carbon-based substances (such as 2D carbon allotropes and carbon nanopipes) [60][62]. In this study, pineapple-fiber (PF) aerogels were successfully created by pretreating PFs with naturally decomposable polyvinyl alcohol (PVA). The PVA solution preparation was combined with PFs and freeze-dried. According to the findings, the PFs have high porosities (~99%), ultralow densities, and microporous formations, as shown by field-emission scanning electron microscopy, Brunauer–Emmett–Teller isotherm, and X-ray diffraction analysis. The exceptionally low thermal conductivity of the PF aerogel demonstrated its applicability for thermal barrier uses. A thermal coat wrapped over a water bottle with a PF aerogel filling can unquestionably keep the water temperature near 0 °C (just above the freezing temperature) for up to 6 h (initial temperature: −3 °C) and above 40 °C for up to 2.5 h (initial temperature: 90 °C). The thermal coat has a potential thermal barrier that is nearly three times that of a product that is currently on the market [63]. In another study, the goal was to develop a thermal coat for army canteens based on a paper waste cellulose aerogel to increase the life of ice slurry for dynamic army troops in exercises or operations. However, because of the minimal stretching capacity and the ease with which the bio-based aerogel structure can be damaged, the bio-based aerogel must be sandwiched between two protective layers to make the thermal coat more durable. The paper waste was combined with deionized water and crosslinked with Kymene chemicals (crosslinkers based on polyamide-epichlorohydrin resin) before being frozen overnight. After freezing, the gel was dried using the lyophilization drying technique at −91 °C to create cellulose aerogels, followed by the crosslinking process in the dryer for 3 h at 120 °C. Following all measurements, the results showed that the heat barrier function of the developed thermal coats was significantly better than that of marketed thermal flasks and similar to that of vacuum flasks for the same duration of 4 h and the same surrounding temperatures [64]. Cellulose aerogels were made from dissolvable cellulose filaments in melts of calcium thiocyanate salt hydrate in this study, followed by regenerating in ethanol and drying under supercritical CO2. It is possible to create uniform-structured bio-based aerogels with minimal bulk mass. The microstructure of bio-based aerogels exhibited a continuous 3D network with a large specific surface ratio coupled with a significantly sponge-like structure (up to 98%). This research enabled the examination of increased cellulose amounts of up to 6 wt%. Bio-based aerogels displayed remarkable physical strength and heat transfer efficiency for textile applications at atmospheric pressure. Moreover, the Young’s modulus of cellulose aerogels showed that it can be reached at 13.5 MPa, while the Poisson ratio was near zero [65]. Yangyang exploited discarded cotton textiles to enhance the anti-flaming capabilities of cellulose aerogels by producing magnesium hydroxide nanoparticles in situ in cellulose gel nanostructures. In addition, three-dimensional nanoporous cellulose gels were produced by disintegrating and coagulating cellulose in an aqueous NaOH/urea solution, and these were employed as patterns for the unclustered production of magnesium hydroxide nanoparticles. According to the findings, the produced mixture–matrix aerogels have extremely porous architectures and exceptional thermal isolation characteristics with minimal heat transfer. In addition, effective flame-retardant and mechanical characteristics were obtained [66].
The sol–gel process is linked to the organic polymer type. Because the molecular composition of organic polymer variants contains a restricted amount of active (e.g., hydroxyl) groups, a connecting agent is often necessary to achieve the required gel structure [11].
The creation of bio-based aerogels from nanoscale crystalline polysaccharides and dissolvable organic polymers from various materials is shown schematically in Figure 2, achieved simply by recovering them as a coagulant from their liquid solution, followed by lyophilization and the resulting regenerated cellulose aerogel [67].
2.2. Drying Methods of Cellulose-Based Aerogels
The most crucial phase in the manufacture of aerogels is drying. The drying process influences the shape of cellulose aerogels. Due to the capillary pressure, traditional drying processes can result in the collapse of the gel pore structure. Supercritical drying (using alcohol, acetone, or CO2) and vacuum freeze-drying are extensively used for cellulose aerogel manufacturing procedures [68][69]. The sublimation of a solid, such as frozen water, from a moist precursor’s pores is identified as freeze-drying. As a result of the formation of ice during the process of water freezing, freeze-drying produces a sheet-like cellulose network with large and linked holes with a width of numerous micrometers [69] Under supercritical (sc) conditions, the absence of a liquid/gas meniscus results in the complete elimination of surface tension between the liquid and gas phases. ScCO2 dried aerogels usually have a cauliflower-like cellulose arrangement, which is an assemblage of tiny shaggy beads.
2.2.1. Drying with Supercritical Carbon Dioxide
Aerogels, resulting from the drying of wet gels while preserving their intrinsic porosity, are commonly fabricated through the sol–gel method, modifying the gel’s molecular structure [70]. Typically, gels exhibit a porous structure strengthened via washing, with pores partially filled by an organic solvent such as ethanol. Drying under specific conditions may lead to collapsed structures, giving rise to xerogels or cryogels. Supercritical drying, employing compressed CO2 above the critical point, helps sustain the porous structure by eliminating capillary forces [71][72][73]. This single-phase process ensures effective extraction. The resulting dried gel, upon exposure to air, experiences CO2 exchange, undergoing transformation into an aerogel [70]. CO2 is a fluid that is normally employed in the drying of cellulose aerogels because of its reasonable critical point (304 K, 7.4 MPa) and the benefits of low cost and great safety.
Supercritical drying is distinguished by the two-way mass transfer of the liquid gel solvent CO2 into and out of the wet gel pores [74]. To begin, the drying is largely caused by high scCO2 dissolution in the liquid gel solvent, which results in an expanded liquid and spilling of the extra liquid volume removed from the gel network. Second, the amount of CO2 increases over time until supercritical conditions are reached for the fluid mixture in the pores, without any intermediary vapor–liquid transitions. Finally, the presence of supercritical fluid mixtures in pores with no liquid phases causes a lack of surface tension, which precludes pore collapse in the gel structure during solvent removal [74].
Water with a high surface tension might destroy a cellulose network’s delicate and extremely porous structure, which is generated during the drying process. The reasons behind this phenomenon include variances in the specific energies during the transitions between solid–liquid and liquid–gas phases, along with the generation of inward forces near the solvent menisci along the capillary walls. As a result, it is necessary to entirely replace the high-surface-tension water [75]. In an NMMO (N-methylmorpholine-N-oxide) solvent system, for example, while manufacturing regenerated cellulose aerogels, the cellulose gel requires re-priming with water, followed by either ethanol and acetone exchange or solely acetone exchange [76][77]. In the case of employing an ionic liquid as the solvent system, the cellulose gel necessitates an initial re-priming step with water, followed by subsequent acetone exchanges conducted repeatedly [49]. Ethanol exchange is a popular treatment for natural cellulose aerogels [78][79]. It has been demonstrated that cellulose solvent residues reduce drying efficacy [47]. Furthermore, the surface tension of various liquids, as well as the shaking involved during the re-priming and solvent exchange procedures, may destroy the cellulose gel structure [47][80]. The solvent exchange process is exceedingly slow, requiring an average of 2–3 days. Finally, supercritical drying using scCO2 can be helpful to reduce damage caused by capillary pressure inside the pores, which can be advantageous, as it promotes the production of aerogel materials with enhanced uniformity in their 3D network.
However, even though liquid CO2 is costless, because a high-pressure tank is required, this method is costly [11]. Nevertheless, the SCD processes offer a notable advantage in that the choice of solvent for the gelation process is highly versatile, allowing for a wide range of options. This approach is applicable across several types of gel materials and is not limited to specific ones. Two fundamental supercritical CO2 drying strategies exist: (A) high-temperature (HT) drying and (B) low-temperature (LT) drying [81].
(A) High-temperature scCO2 drying: After a pre-pressurization stage, the solvent is heated to above its critical point in the high-temperature (HT) process. This method requires heating the wet samples and solvent to supercritical temperatures in a sealed autoclave. For commonly used organic solvents, which have a critical point above 200 °C and a critical pressure ranging from 40 to 80 bar, the desired conditions can be achieved through this method. Subsequently, a slow depressurization is carried out [82][83][84][85][86]. The process and instrumentation involved in the high-temperature (HT) approach are straightforward, as it does not require pumps, and it enables direct surface modification to produce hydrophobic aerogels. However, one disadvantage of utilizing organic solvents is the risk of fire in the case of an unintentional or uncontrolled discharge, and the higher temperature may cause damage to heat-sensitive components. An alternative to the normal HT method is to drop the temperature slightly, maintaining it below the solvent’s critical temperature [87][88].
(B) Low-temperature scCO2 drying: Low-temperature drying uses supercritical carbon dioxide (CO2), since it has a low critical temperature (31 °C), is non-flammable, and is ecologically friendly. To eliminate all solvents, wet gel samples are periodically or continuously flushed with supercritical CO2. Heat exchangers and a liquid CO2 pump are critical equipment components for both periodic and continuous operations. The continuous low-temperature (LT) method may need a greater volume of CO2; nevertheless, particular drying costs can be greatly reduced via careful optimization and scaling to an industrial level [89][90]. Supercritical conditions offer the possibility of functionalizing the aerogel skeleton [91]. Considering the significance of aerogels in both scientific research and industrial applications, extensive studies have been carried out to examine the influence of drying conditions, diffusion, chemical composition, and temperature profiles on the quality of aerogels [70][71][72][73][92][93][94][95].
2.2.2. Direct Vacuum Freeze-Drying and Freezing Facilitated by an Organic Solvent
Cellulose aerogels can be produced using a straightforward and environmentally friendly method known as vacuum freeze-drying. At a temperature below the freezing point of the liquid medium, which is usually water, the gel is initially frozen in this process. Much of the liquid is then eliminated through sublimation, which is an essential step to avoid structural collapse and reduce shrinkage. The implementation of freeze-drying can be done in two different ways: without solvent exchange, by using direct freezing; and by using organic solvent exchange. Direct freeze-drying is a process of low-temperature dehydration, involving freezing, primary drying, and secondary drying stages. Ice crystals, formed during the freezing phase, serve as templates for the structures of porous materials. During primary drying, amorphous ice crystals are sublimated at temperatures below the melting point of ice, by preventing the collapse of the pore structure. Subsequently, in the secondary drying phase, all ice crystals are removed, eliminating the bound water that is adsorbed on the material’s surface [96]. Consequently, the pore structure of porous aerogels, including their pore morphology and distribution, is influenced by the liquid crystallization process and growth behavior, which are controlled by the cooling rate and temperature. Additionally, various factors, such as cellulose content, gel size, shape, and temperature, affect the rate of sublimation, which is typically slow [11].
Another way of freeze-drying is to replace the solvent within the hydrogels with t-BuOH, which involves freezing and sublimation. Altogether, within the parameters mentioned above, freeze-dried aerogels that are made of nanocellulose and its derivatives are commonly encountered with a specific surface area, although the self-agglomeration of nanocellulose may reduce. For this reason, freeze-drying with an organic solvent exchange is another parameter that can affect the porous structure before drying. In particular, tert-butyl alcohol, which can be used instead of methanol or ethanol during solvent exchange, possesses a low interfacial tension and a single hydroxyl group, enabling it to create hydrogen bonds with the surface hydroxyl or carboxyl groups of nanocellulose and its derivatives [96]. Altogether, the presence of multiple butyl groups creates a steric barrier that inhibits the aggregation of nanocellulose. Consequently, when employed in solvent exchange, tert-butyl alcohol has the potential to preserve the gel structure of nanocellulose and its derivatives more effectively than water, thereby preventing the collapse of the cellulose aerogel structure [51][97][98][99].
By employing liquid nitrogen or liquid propane to enhance thermal conductivity, it is possible to quickly cool the cellulose gel. This process effectively reduces cellulose agglomeration and the formation of ice crystals, while simultaneously increasing the porosity of the resulting aerogel. Zhang et al. examined three different chilling rates in their study: liquid nitrogen (−196 °C for 30 min), a freezer with an extremely low temperature (−80 °C for 12 h), and a standard refrigerator (−20 °C for 24 h). Their observations revealed that the use of liquid nitrogen facilitated the rapid formation of ice crystals, thereby effectively mitigating cellulose self-agglomeration and leading to the development of a more homogeneous and seamless surface structure [100]. In order to achieve uniformly structured aerogels, the utilization of both anti-freezing chemicals [101] and spray freeze-drying techniques [102][103] depends on accelerating the freezing rate. However, before the advancement of the solid–liquid interface, comparable freezing rates and localized temperature gradients are observed, which is similar to the scenario of freeze-drying small samples in a freezer while simultaneously cooling the larger sample [11]. The specific surface area and pore size distribution of a given kind of cellulose aerogel are significantly influenced by the drying process employed [45][78]. Because of the creation of ice crystals and the high interfacial tension of water, freeze-drying typically generates fractures in the aerogel material. Other disadvantages of freeze-drying include its lengthy processing time and significant electric energy usage. In contrast, drying with supercritical carbon dioxide (scCO2) offers improved preservation of the cellulose gel structure, resulting in aerogels with minimal shrinkage, smaller pores, and higher specific surface areas [48][79][104][105].
2.2.3. Ambient Drying
Atmospheric pressure drying of (ligno)cellulose aerogels is still in its infancy. The fundamental issue impeding the development of atmospheric drying for aerogels is significant network shrinkage produced through the liquid meniscus and pressure gradient. Under the same regeneration circumstances, vacuum-dried aerogels show significant shrinkage and collapse as compared to supercritical CO2-dried aerogels. According to ESEM images in the study referred to as [96], the capillary force during vacuum-drying degrades the porous structure [96].
The structure of cellulose aerogels may be adjusted and controlled using drying processes. For this purpose, four drying procedures are outlined. ScCO2 drying can result in mesoporous aerogels with large specific surface areas and high porosities. Although t-BuOH drying can achieve comparable results, the porous structure created is less homogeneous than that produced by ScCO2 drying [96].
3. Multifunctional Application of Cellulose-Based Aerogels on Textile Structures
Due to the robust chemical reactivity of cellulose, the wide range of diverse derivatives with various functions, the adaptable construction process, and the multiple methods of modification, bio-based aerogels exhibit multifunctionality. There exist three primary methods for modifying cellulose aerogels [11]:
- -
-
Other components can be added to the cellulose solution/suspension [11]. For example, the reaction of CNF with N-methylol-dimethyphospylpropionamide (MDPA) and further crosslinking by 1,2,3,4-butane tracarboxylic acid (BTCA) yields a flame retardant with good flexibility and self-extinguishment [106].
- -
-
Coating or adding additional substances to the aerogel structure [11], such as the polyacrylonitrile–silica aerogel coating over viscose nonwoven fabric for protection and comfort [107]. Another area of study is the application of molecular layer-by-layer (m-LBL) technology. This technique enables the deposition of ultrathin layers onto a surface through sequential covalent processes. As a consequence, a precise molecular-scale coating is generated, mostly by surface oligomerization, which is not possible with bulk synthesis techniques [108][109][110].
- -
- -
-
Cellulose aerogels are lightweight 3D porous materials. They are currently employed mostly in insulation, flame retardants [66][112][113], and biological applications [4][11]. Additionally, they find applications in carbon aerogel production, as well as the transportation of metal nanoparticles and metal oxides [11].
3.1. Thermal Insulation Materials
Materials are classified according to their thermal conductivity as thermal conductors (λeff ≥ 0.1 W/(mK), insulators (0.1 W/(mK) > λeff > 0.025 W/(mK), and superinsulators (λeff ≤ 0.025 W(mK). It is known that the thermal conductivity of dry air is around 0.025 W/(mK), which is generally slightly dependent on temperature and moisture content.
Due to their thermal conductivity levels spanning from tens to hundreds of W/(mK), metals are good thermal conductors. Expanded polystyrene, extruded polystyrene, glass wool, mineral wool, and wood exhibit thermal conductivities within the range of 0.1 to 0.026 W/(mK), making them effective insulators against heat transfer [114][115]. Silica aerogels, vacuum insulation panels, and vacuum glasses are regarded as superinsulating materials because their thermal conductivity is below 0.025 W/(mK) [116].
The thermal conduction of aerogels can be classified as solid-state, gas-phase, open-pore, or radiation thermal conduction. Once the pore size of a porous material approaches the average free path of the gas (which is approximately 70 nm when vented), the thermal conductivity of the substance decreases. This is attributed to the fact that the pores impede gas flow and restrict convection, thereby hindering heat transfer. The thermal conductivity of mesoporous cellulose aerogels primarily depends on two factors: solid-state thermal conduction and gas-phase thermal conduction. These factors, in turn, are closely associated with the aerogel’s density (determined by the initial cellulose concentration), the pore size distribution, and the surface structures of the aerogel material [11].
Regenerated cellulose aerogels possess a porous structure with a relatively higher fraction of large pores compared to other cellulose aerogels. This increased presence of large holes within the aerogel structure enhances heat conductivity as it facilitates improved gas transport [11].
Antlauf et al. conducted a study in which cellulose fibers (CFs) and cellulose nanofibers (CNFs) were produced from commercially available birch pulp. The production process involved varying pressure and temperature parameters as experimental variables. For temperatures ranging from 80 to 380 °C, their results exhibited very little fluctuation in thermal conductivity with density (ρsample = 1340–1560 kg/m−3). Furthermore, temperature dependency is independent of fiber size, density, and porosity. Figure 3 depicts their studies on thermal conductivity [117].
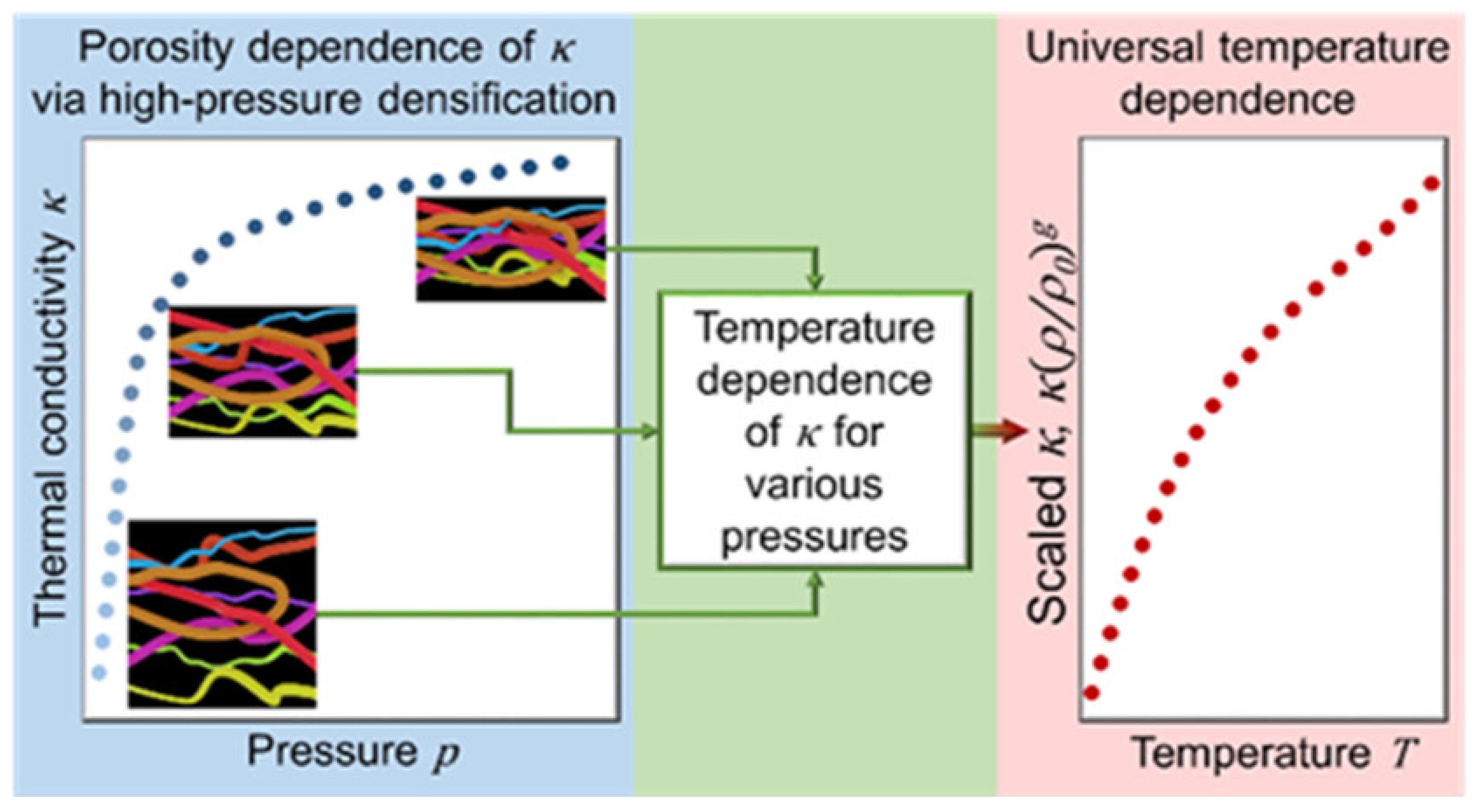
Figure 3. Thermal conductivity against pressure.
3.2. Flame Retardancy
Aerogels with a lightweight composition derived from bio-based materials draw the attention of academics because of their exclusive properties, some examples of which include being environmentally conscious, sustainable, and possessing amazing thermal insulation effectiveness [118][119]. The fire-resistant clothing used for firefighting is a form of specific thermal protection clothing used by firemen during firefighting operations [120][121]. As a result, advanced flame-resistant and thermally insulating materials with exceptional performance are vital in thermal protective garments to safeguard firefighters. Para-aramid polymer is now used mostly in thermal protective gear as a material that provides flame retardancy due to thermal insulation of porous fabrics created from it [122][123]. Using the wet spinning procedure, Liu et al. [122] revealed that a lab-scale nanofibril Kevlar aerogel exhibited strong flame-suppressing properties, characterized by a comparatively slow combustion rate (0.013 cm/s) and the ability to extinguish itself.
It is vital to identify environmentally acceptable thermal insulation materials designed for firefighting apparel [124]. Researchers have recently expressed interest in flame-retardant aerogels made from low-cost biomaterials because of their sustainable nature, eco-friendliness, affordability, lightweight properties, and strong thermal insulation properties [125][126][127]. Due to their high porosity, low thermal conductivity, lightweight structure, and excellent thermal insulation properties, aerogels find extensive utilization in various applications, such as fire resistance and thermal insulation [128][129]. Polymers derived from natural polysaccharides are common renewable biomass resources that are more biodegradable and environmentally friendly compared to fossil-based products [130]. In consequence, several efforts have been undertaken to create aerogels based on polysaccharides that exhibit remarkable low density, porosity, non-toxicity, biodegradability, and bio-sustainability [124]. Among the notable examples are magnesium hydroxide nanoparticles (MH NPs) in waste cotton fabric-based cellulose gel nanostructures [66] obtained by the freeze-drying method, which have demonstrated that the addition of magnesium hydroxide to the gel structure effectively enhances the flame-retardant properties of the aerogel in foam form. According to an experiment conducted by N. Le Thanh, adding NaHCO3 to the material showed a decrease in the combustibility of the material and the burning rate. Figure 4 shows this combustion example accordingly. As the figure and the experiments show, while the pure cellulose aerogel burned quickly (at an average speed of 3.45 mm/s), by increasing the NaHCO3 concentration in the material by between 1–2 and 3%, the combustibility of the material fell and its rate of burning also decreased [131].

Figure 4. Images of a paper cellulose aerogel after a 10 s burn; the following samples were observed: (a) pure cellulose aerogel, (b) cellulose aerogel with 1% NaHCO3, (c) cellulose aerogel with 2% NaHCO3, and (d) cellulose aerogel with 3% NaHCO3.
3.3. Medical Applications
As the most common polymer on the planet, cellulose is mostly obtained from plants and microbiological sources [132]. Nevertheless, due to its unique properties, such as decomposability, compatibility with living systems, and low cytotoxicity, it is one of the most commonly used polymers for manufacturing aerogels [133]. Bio-based aerogels are widely employed in medical treatments such as biological detection, drug release systems, regenerative scaffolds, and anti-infective wound wrap materials [134]. Several studies have previously been published on the sequential evolution of aerogels’ formation and the therapeutic uses of nanofibrillated cellulose aerogels [17]. Nevertheless, little research has been conducted on the utilization of bio-based aerogels for bactericidal administration and wound treatment in textile applications. Many studies have explored strategies for wound healing, including the use of a composite aerogel with collagen and cellulose [135]. Collagen, which is valued for its adhesive, biodegradable, and biocompatible properties, proves suitable for wound dressings. Oxygen permeability and moisture management are crucial for normal cellular function in wound healing [136]. Cellulose-based nanoparticles, particularly nanocellulose polymers, leverage the effective surface area of filamentous biomaterials for cellular absorption. Combined with an antimicrobial substance [137], CNF aerogels emerge as a potential wound dressing solution.
3.4. Water Treatment Containing Textile Dyes
Water pollution, a global concern affecting both water supplies and public health [138], involves contaminants like heavy metals [139], petroleum products, dyes, and various chemical compounds [140]. Ongoing efforts seek optimized methods for removing pollution sources, focusing on sustainable and environmentally friendly materials for water purification [138]. The water treatment method chosen depends on the water composition, quality criteria, and intended usage [141]. Iron removal is crucial for technical purposes to prevent water from becoming unsuitable due to high iron levels and discoloration. Dye release, a minor contributor to water pollution, is visible and undesirable even at low concentrations [140]. Annual dye production, surpassing 700 thousand tons, includes synthetic types posing risks to aquatic organisms and humans [142][143]. In textile manufacturing, dyes fall into categories such as anionic, cationic, or non-ionic [140]. Effluents from the dyeing process exhibit increased levels of color, suspended solids, biochemical oxygen demand, chemical oxygen demand, temperature, metals, and salts [144]. Continuous monitoring and comparison of these parameters with established concentrations are essential in treatment procedures before releasing effluents into water bodies. The assessment of treatment effectiveness also considers additional parameters such as total organic carbon, nitrate–nitrogen, ammonia–nitrogen, and orthophosphate–phosphorus [145]. Capturing dyes in fabrics during dyeing poses a challenge due to their pronounced water solubility, resulting in the generation of considerable wastewater with substantial quantities of these organic compounds [144]. The composition of wastewater in the textile industry exhibits global variations influenced by factors such as the manufacturing process, fabric type, factory equipment, applied chemicals, fabric weight, season, and fashion trends [145].
Various methods, including filtration, oxidation, and microbial approaches, are employed to eliminate dyes from water. However, these methods are associated with high costs, low efficiency, and operational challenges. Despite dyes’ resistance to degradation, certain bacteria, such as Pseudomonas sp. and Sphingomonas sp., have demonstrated effectiveness in decolorizing and mineralizing them [146]. The extensively used adsorption methods, employing traditional adsorbents like clay, bentonite, zeolite, or charcoal [10][11][12][13][147][148][149], are preferred for their cost-effectiveness, high efficiency, and simplicity. Nonetheless, traditional adsorbents face limitations such as a short effective life, regeneration difficulties, and high consumption. Color removal from wastewater involves a range of physical, chemical, and biological treatment methods [150], with adsorption widely acknowledged and employed [151].
Dyes in wastewater pose a significant threat to ecosystems and living organisms due to their persistence, biotoxicity, and bioaccumulation [152][153]. Consequently, there is a growing interest in developing efficient treatment strategies for dye effluents to address multipollutant removal [154]. Among the available technologies, adsorption is considered to be the most competitive, offering easy operation, relatively low costs, and non-toxic byproducts [155][156]. Cellulose-based aerogels are recognized as potential candidates for wastewater treatment due to cellulose’s renewable and biodegradable nature, easily functionalized properties, unique 3D network structure, and high surface area [150][157][158][159]. However, challenges remain, including avoiding toxic crosslinking agents and achieving a balance between high adsorption capacity and selectivity [150][160]. A green and effective strategy is essential to develop cellulose-based adsorption aerogel materials for dye wastewater purification [161]. Produced via the transesterification of cellulose and acetoacetate reagents, cellulose acetoacetate (CAA) is a water-soluble derivative that offers active reaction sites, which enable the creation of functional materials in aqueous environments [162][163][164]. In a noteworthy investigation, Liu and colleagues presented a self-repairing polysaccharide hydrogel formed by combining CAA and chitosan in a solution [165]. This hydrogel, derived from cellulose and employing enamine bonds, exhibited reversible sol–gel transitions that were responsive to pH variations. This highlights the suitability of polymers with amino groups for creating CAA-based gels. Unlike conventional chemical crosslinking agents, the eco-friendly approach of constructing 3D network structures with dynamic enamine bonds can be swiftly achieved at room temperature. The interlocking 3D network structure is also capable of being disassembled for further applications. Additionally, the use of electrostatic attraction, recognized as a simple and effective force, is emphasized for selectively capturing dyes, especially considering that the majority of commercial dyes exist in ion form [166]. In another study, a cationic cellulose aerogel (Q-CNF), derived from cellulose nanofibrils with trimethylammonium chloride groups, was prepared through freeze-drying and aliphatic triisocyanate crosslinking. The rigid porous aerogel demonstrated efficient adsorption of anionic dyes, with capacities of 250, 520, and 600 μmol g−1 (approximately 160, 230, and 560 mg g−1, respectively) for red, blue, and orange dyes, respectively. Electrostatic interactions between CNF surface positive sites and dye sulfonate groups were identified as the main contributors. The adsorption capacity was correlated with the specific surface area and cationic content of the aerogel. Regeneration with KCl in an ethanol–water mixture allowed for multiple adsorption–desorption cycles without significant capacity loss. Q-CNF aerogels exhibit promise as renewable, reusable adsorbents for treating dye-loaded water [167].
3.5. CO2 Capture
Carbon capture and storage (CCS) is a key component in the global effort to reduce carbon dioxide (CO2) emissions by preventing the release of CO2 into the atmosphere. Choosing the right materials is crucial to building dependable and secure infrastructure for CCS technology. Natural cellulose materials are a great option for CCS applications because of their impressive mechanical and physical qualities and eco-friendliness. As CO2 adsorbents and catalyst carriers for CO2 conversion, cellulose-based materials are useful in the field of carbon capture, utilization, and sequestration technologies [168]. Cellulose is a flexible material that may be used as a matrix or filler to produce goods such as films, paper bases, aerogels, and hydrogels with adsorption capabilities. It is known for its renewability and degradability [169][170][171][172]. Creating porous carbon with adsorption properties also requires it. Activation techniques employing physical or chemical activators can be used to modify carbon-based materials. Carbon capture technology appears to benefit from the use of cellulose and its derivatives, such as cellulose nanofibers (CNFs) and cellulose nanocrystals (CNCs), among other sophisticated materials [170][173][174]. Rich and affordable, cellulose aerogels have the potential to revolutionize existing carbon capture techniques, especially when it comes to the manufacturing of commercial nanocellulose [175][176][177]. Notwithstanding their smaller surface area, chemically altered cellulose and nanocellulose aerogels demonstrate adequate CO2 chemisorption, highlighting their potential for use in carbon capture applications [178][179]. By combining cellulose aerogels with large-pore hierarchical porous metal–organic frameworks (HP-MOFs), employing monocarboxylic acid (MA) as a modifier, and growing HP-UIO-66-NH2 on the cellulose aerogels in situ, Yu et al. created hybrid aerogels. The chain length of MA was changed to modify the pore size of the HP-MOFs. The study revealed that the CO2 adsorption capacity followed a trend of initially increasing and then decreasing with the increase in the MOFs’ pore size. Concurrently, the adsorption selectivity for CO2 consistently grew. Notably, among all of the samples, MC-HUN-4, distinguished by a moderate pore size, exhibited the highest CO2 adsorption capacity (1.90 mmol/g at 298 K and 1 bar) and superior adsorption selectivity (13.02 and 2.40 for CO2/N2 and CO2/CH4) [180]. Zhou et al. applied a sol–gel method to create composite aerogels (CSA) by using silica from skimmed cotton and cellulose whiskers. Employing tetraethyl orthosilicates (TEOs) and an alkaline silica solution as precursors, the CSA-TEPA 70% aerogel, with a 70% TEPA (tetraethylenepentamine) loading, demonstrated impressive adsorption, achieving a maximum capacity of 2.25 mmol/g. These adsorbents hold promise for CO2 capture. Additionally, the heightened research interest in metal–organic frameworks (MOFs) is attributed to their high surface area, CO2 affinity, structural diversity, tunable microporosity, and adaptable structure [181]. Using bacterial cellulose (BC) as a substrate, Ma et al. created composite aerogels containing amino-functionalized ZIF-8 (zeolitic imidazolate frameworks) (ZIF-8-NH2). Zinc ions and hydroxyl groups chelated to produce composites with strong interfacial attraction and compatibility when ZIF crystals were evenly encased around cellulose fibers. The resultant aerogel showed a noteworthy CO2 adsorption capability of 1.63 mmol/g. Zinc ions combined with the hydroxyl and oxygen groups in cellulose to create complexes. ZIF-8-NH2 crystals were created as linkers by adding 2-methylimidazole and 2-aminobenzimidazole by wrapping BC chains without the need for binders. By varying the amount of organic linker, the ZIF-8 amino group loading was optimized [182].
Mesoporous cellulose aerogels, derived from old corrugated containers (OCCs) through freeze-drying, exhibited efficient CO2 capture. The aerogel synthesis induced a transition in cellulose crystals from form I to form II while preserving their chemical structures. These aerogels featured excellent thermal stability, comprising highly porous networks with fibrils below 50 nm wide. Their mesopore volumes ranged from 0.73 to 1.53 cm3 g−1, and their specific surface areas varied from 132.72 to 245.19 m2 g−1. Furthermore, the research demonstrated exceptional CO2 adsorption capacities within the range of 1.96–11.78 mmol g−1. In comparison to other sorbents, the CA-2 material (2% weight cellulose) exhibited superior CO2 adsorption capacity at room temperature [183].
References
- Barrios, E.; Fox, D.; Li Sip, Y.Y.; Catarata, R.; Calderon, J.E.; Azim, N.; Afrin, S.; Zhang, Z.; Zhai, L. Nanomaterials in Advanced, High-Performance Aerogel Composites: A Review. Polymers 2019, 11, 726.
- Nita, L.E.; Ghilan, A.; Rusu, A.G.; Neamtu, I.; Chiriac, A.P. New Trends in Bio-Based Aerogels. Pharmaceutics 2020, 12, 449.
- Kistler, S.S. Coherent Expanded Aerogels and Jellies. Nature 1931, 127, 741.
- Revin, V.V.; Nazarova, N.B.; Tsareva, E.E.; Liyaskina, E.V.; Revin, V.D.; Pestov, N.A. Production of Bacterial Cellulose Aerogels with Improved Physico-Mechanical Properties and Antibacterial Effect. Front. Bioeng. Biotechnol. 2020, 8, 1392.
- Maleki, H. Recent Advances in Aerogels for Environmental Remediation Applications: A Review. Chem. Eng. J. 2016, 300, 98–118.
- Hrubesh, L.W. Aerogel Applications. J. Non-Cryst. Solids 1998, 225, 335–342.
- Bheekhun, N.; Abu Talib, A.R.; Hassan, M.R. Aerogels in Aerospace: An Overview. Adv. Mater. Sci. Eng. 2013, 2013, e406065.
- Vareda, J.P.; Valente, A.J.M.; Durães, L. Heavy Metals in Iberian Soils: Removal by Current Adsorbents/Amendments and Prospective for Aerogels. Adv. Colloid Interface Sci. 2016, 237, 28–42.
- Stergar, J.; Maver, U. Review of Aerogel-Based Materials in Biomedical Applications. J. Sol-Gel Sci. Technol. 2016, 77, 738–752.
- Maleki, H.; Durães, L.; Portugal, A. An Overview on Silica Aerogels Synthesis and Different Mechanical Reinforcing Strategies. J. Non-Cryst. Solids 2014, 385, 55–74.
- Long, L.-Y.; Weng, Y.-X.; Wang, Y.-Z. Cellulose Aerogels: Synthesis, Applications, and Prospects. Polymers 2018, 10, 623.
- Jiang, Y.; Chowdhury, S.; Balasubramanian, R. New Insights into the Role of Nitrogen-Bonding Configurations in Enhancing the Photocatalytic Activity of Nitrogen-Doped Graphene Aerogels. J. Colloid Interface Sci. 2019, 534, 574–585.
- Nguyen, B.N.; Meador, M.A.B.; Scheiman, D.; McCorkle, L. Polyimide Aerogels Using Triisocyanate as Cross-Linker. ACS Appl. Mater. Interfaces 2017, 9, 27313–27321.
- Zhu, F. Starch Based Aerogels: Production, Properties and Applications. Trends Food Sci. Technol. 2019, 89, 1–10.
- García-González, C.A.; Uy, J.J.; Alnaief, M.; Smirnova, I. Preparation of Tailor-Made Starch-Based Aerogel Microspheres by the Emulsion-Gelation Method. Carbohydr. Polym. 2012, 88, 1378–1386.
- Li, Z.; Zhong, L.; Zhang, T.; Qiu, F.; Yue, X.; Yang, D. Sustainable, Flexible, and Superhydrophobic Functionalized Cellulose Aerogel for Selective and Versatile Oil/Water Separation. ACS Sustain. Chem. Eng. 2019, 7, 9984–9994.
- Abdul Khalil, H.P.S.; Adnan, A.S.; Yahya, E.B.; Olaiya, N.G.; Safrida, S.; Hossain, M.S.; Balakrishnan, V.; Gopakumar, D.A.; Abdullah, C.K.; Oyekanmi, A.A.; et al. A Review on Plant Cellulose Nanofibre-Based Aerogels for Biomedical Applications. Polymers 2020, 12, 1759.
- Novak, B.M.; Auerbach, D.; Verrier, C. Low-Density, Mutually Interpenetrating Organic-Inorganic Composite Materials via Supercritical Drying Techniques. Chem. Mater. 1994, 6, 282–286.
- Wei, C.; Zhang, Q.; Wang, Z.; Yang, W.; Lu, H.; Huang, Z.; Yang, W.; Zhu, J. Recent Advances in MXene-Based Aerogels: Fabrication, Performance and Application. Adv. Funct. Mater. 2023, 33, 2211889.
- Yang, W.-J.; Yuen, A.C.Y.; Li, A.; Lin, B.; Chen, T.B.Y.; Yang, W.; Lu, H.-D.; Yeoh, G.H. Recent Progress in Bio-Based Aerogel Absorbents for Oil/Water Separation. Cellulose 2019, 26, 6449–6476.
- Fernandes, E.M.; Pires, R.A.; Mano, J.F.; Reis, R.L. Bionanocomposites from Lignocellulosic Resources: Properties, Applications and Future Trends for Their Use in the Biomedical Field. Prog. Polym. Sci. 2013, 38, 1415–1441.
- Moon, R.J.; Martini, A.; Nairn, J.; Simonsen, J.; Youngblood, J. Cellulose Nanomaterials Review: Structure, Properties and Nanocomposites. Chem. Soc. Rev. 2011, 40, 3941–3994.
- Eichhorn, S.J.; Dufresne, A.; Aranguren, M.; Marcovich, N.E.; Capadona, J.R.; Rowan, S.J.; Weder, C.; Thielemans, W.; Roman, M.; Renneckar, S. Current International Research into Cellulose Nanofibres and Nanocomposites. J. Mater. Sci. 2010, 45, 1–33.
- Jianan, C.; Shaoqiong, Y.; Jinyue, R. A Study on the Preparation, Structure, and Properties of Microcrystalline Cellulose. J. Macromol. Sci. Part A Pure Appl. Chem. 2006, 33, 1851–1862.
- Virtanen, T.; Svedström, K.; Andersson, S.; Tervala, L.; Torkkeli, M.; Knaapila, M.; Kotelnikova, N.; Maunu, S.L.; Serimaa, R. A Physico-Chemical Characterisation of New Raw Materials for Microcrystalline Cellulose Manufacturing. Cellulose 2012, 19, 219–235.
- Reddy, N.; Yang, Y. Properties and Potential Applications of Natural Cellulose Fibers from the Bark of Cotton Stalks. Bioresour. Technol. 2009, 100, 3563–3569.
- Mai, T.; Luu, T.; Le, P.; Le, P.; Hoang Nguyen Do, N.; Chau, N.D.Q. Fabrication of Cotton Aerogels and Its Application in Water Treatment. In Proceedings of the 12th Regional Conference on Chemical Engineering (RCChE 2019), Ho Chi Minh City, Vietnam, 15–16 October 2019.
- Wang, X.; Li, H.; Cao, Y.; Tang, Q. Cellulose Extraction from Wood Chip in an Ionic Liquid 1-Allyl-3-Methylimidazolium Chloride (AmimCl). Bioresour. Technol. 2011, 102, 7959–7965.
- Cara, C.; Ruiz, E.; Ballesteros, I.; Negro, M.J.; Castro, E. Enhanced Enzymatic Hydrolysis of Olive Tree Wood by Steam Explosion and Alkaline Peroxide Delignification. Process Biochem. 2006, 41, 423–429.
- Abe, K.; Yano, H. Comparison of the Characteristics of Cellulose Microfibril Aggregates of Wood, Rice Straw and Potato Tuber. Cellulose 2009, 16, 1017–1023.
- Hoang Nguyen Do, N.; Tran, V.; Tran, Q.; Le, K.; Nguyen, P.; Duong, H.; Thai, Q.B.; Le, P. Recycling of Pineapple Leaf and Cotton Waste Fibers into Heat-Insulating and Flexible Cellulose Aerogel Composites. J. Environ. Polym. Degrad. 2021, 29, 1112–1121.
- Sun, J.X.; Sun, X.F.; Zhao, H.; Sun, R.C. Isolation and Characterization of Cellulose from Sugarcane Bagasse. Polym. Degrad. Stab. 2004, 84, 331–339.
- Trache, D.; Hussin, M.H.; Chuin, C.T.H.; Sabar, S.; Fazita, M.N.; Taiwo, O.F.; Hassan, T.M.; Haafiz, M.M. Microcrystalline Cellulose: Isolation, Characterization and Bio-Composites Application—A Review. Int. J. Biol. Macromol. 2016, 93, 789–804.
- Siqueira, G.; Bras, J.; Dufresne, A. Cellulosic Bionanocomposites: A Review of Preparation, Properties and Applications. Polymers 2010, 2, 728–765.
- Hinterstoisser, B.; Salmén, L. Application of Dynamic 2D FTIR to Cellulose. Vib. Spectrosc. 2000, 22, 111–118.
- Bochek, A.M. Effect of Hydrogen Bonding on Cellulose Solubility in Aqueous and Nonaqueous Solvents. Russ. J. Appl. Chem. 2003, 76, 1711–1719.
- Lavanya, D.; Kulkarni, P.; Dixit, M.; Raavi, P.K.; Krishna, L.N.V. Sources of Cellulose and Their Applications—A Review. Int. J. Drug Formul. Res. 2011, 2, 19–38.
- Myasoedova, V.V. Physical Chemistry of Non-Aqueous Solutions of Cellulose and Its Derivatives; Wiley: Chichester, UK, 2000; ISBN 978-0-471-95924-3.
- Orts, W.J.; Sojka, R.E.; Glenn, G.M.; Gross, R.A. Biopolymers from Polysaccharides and Agroproteins, Copyright, Foreword. In Biopolymers from Polysaccharides and Agroproteins; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2001; Volume 786, pp. i–v. ISBN 978-0-8412-3645-5.
- Hon, D.N.-S.; Shiraishi, N. Wood and Cellulosic Chemistry, Second Edition, Revised, and Expanded; CRC Press: New York, NY, USA, 2000; ISBN 978-0-8247-0024-9.
- Surapolchai, W.; Schiraldi, D.A. The Effects of Physical and Chemical Interactions in the Formation of Cellulose Aerogels. Polym. Bull. 2010, 65, 951–960.
- Ahmadi, M.; Madadlou, A.; Saboury, A.A. Whey Protein Aerogel as Blended with Cellulose Crystalline Particles or Loaded with Fish Oil. Food Chem. 2016, 196, 1016–1022.
- Seantier, B.; Bendahou, D.; Bendahou, A.; Grohens, Y.; Kaddami, H. Multi-Scale Cellulose Based New Bio-Aerogel Composites with Thermal Super-Insulating and Tunable Mechanical Properties. Carbohydr. Polym. 2016, 138, 335–348.
- Nguyen, B.N.; Cudjoe, E.; Douglas, A.; Scheiman, D.; McCorkle, L.; Meador, M.A.B.; Rowan, S.J. Polyimide Cellulose Nanocrystal Composite Aerogels. Macromolecules 2016, 49, 1692–1703.
- Liebner, F.; Potthast, A.; Rosenau, T.; Haimer, E.; Wendland, M. Cellulose Aerogels: Highly Porous, Ultra-Lightweight Materials. Holzforschung 2008, 62, 129–135.
- Ratke, L. Monoliths and Fibrous Cellulose Aerogels. In Aerogels Handbook; Aegerter, M.A., Leventis, N., Koebel, M.M., Eds.; Advances in Sol-Gel Derived Materials and Technologies; Springer: New York, NY, USA, 2011; pp. 173–190. ISBN 978-1-4419-7589-8.
- Innerlohinger, J.; Weber, H.K.; Kraft, G. Aerocellulose: Aerogels and Aerogel-like Materials Made from Cellulose. Macromol. Symp. 2006, 244, 126–135.
- Hoepfner, S.; Ratke, L.; Milow, B. Synthesis and Characterisation of Nanofibrillar Cellulose Aerogels. Cellulose 2008, 15, 121–129.
- Sescousse, R.; Gavillon, R.; Budtova, T. Aerocellulose from Cellulose–Ionic Liquid Solutions: Preparation, Properties and Comparison with Cellulose–NaOH and Cellulose–NMMO Routes. Carbohydr. Polym. 2011, 83, 1766–1774.
- Tan, C.; Fung, B.; Newman, J.; Vu, C. Organic Aerogels with Very High Impact Strength. Adv. Mater. 2001, 13, 644–646.
- Jin, H.; Nishiyama, Y.; Wada, M.; Kuga, S. Nanofibrillar Cellulose Aerogels. Colloids Surf. A Physicochem. Eng. Asp. 2004, 240, 63–67.
- Fischer, F.; Rigacci, A.; Pirard, R.; Berthon-Fabry, S.; Achard, P. Cellulose-Based Aerogels. Polymer 2006, 47, 7636–7645.
- Fischer, S. Anorganische Salzhydratschmelzen. Master’s Thesis, Technische Universitaet Bergakademie Freiberg Universitaetsbibliothek “Georgius Agricola”, Freiberg, Germany, 2009.
- Fischer, S.; Leipner, H.; Thümmler, K.; Brendler, E.; Peters, J. Inorganic Molten Salts as Solvents for Cellulose. Cellulose 2003, 10, 227–236.
- Frey, M.W.; Theil, M.H. Calculated Phase Diagrams for Cellulose/Ammonia/Ammonium Thiocyanate Solutions in Comparison to Experimental Results. Cellulose 2004, 11, 53–63.
- Cuce, E.; Cuce, P.M.; Wood, C.J.; Riffat, S.B. Toward Aerogel Based Thermal Superinsulation in Buildings: A Comprehensive Review. Renew. Sustain. Energy Rev. 2014, 34, 273–299.
- Sadineni, S.B.; Madala, S.; Boehm, R.F. Passive Building Energy Savings: A Review of Building Envelope Components. Renew. Sustain. Energy Rev. 2011, 15, 3617–3631.
- Du, A.; Zhou, B.; Zhang, Z.; Shen, J. A Special Material or a New State of Matter: A Review and Reconsideration of the Aerogel. Materials 2013, 6, 941–968.
- Soleimani Dorcheh, A.; Abbasi, M.H. Silica Aerogel; Synthesis, Properties and Characterization. J. Mater. Process. Technol. 2008, 199, 10–26.
- Dervin, S.; Pillai, S. An Introduction to Sol-Gel Processing for Aerogels. In Sol-Gel Materials for Energy, Environment and Electronic Applications; Springer: Cham, Switzerland, 2017; pp. 1–22. ISBN 978-3-319-50142-0.
- Neacşu, I.A.; Nicoară, A.I.; Vasile, O.R.; Vasile, B.Ş. Chapter 9—Inorganic Micro- and Nanostructured Implants for Tissue Engineering. In Nanobiomaterials in Hard Tissue Engineering; Grumezescu, A.M., Ed.; William Andrew Publishing: Norwich, NY, USA, 2016; pp. 271–295. ISBN 978-0-323-42862-0.
- Carraher, C.E. General Topics. Polym. News 2005, 30, 386–388.
- Do, N.H.N.; Luu, T.P.; Thai, Q.B.; Le, D.K.; Chau, N.D.Q.; Nguyen, S.T.; Le, P.K.; Phan-Thien, N.; Duong, H.M. Heat and Sound Insulation Applications of Pineapple Aerogels from Pineapple Waste. Mater. Chem. Phys. 2020, 242, 122267.
- Duong, H.; Xie, Z.; Wei, K.; Nian, N.; Tan, K.; Lim, H.; Li, A.; Chung, K.-S.; Lim, W. Thermal Jacket Design Using Cellulose Aerogels for Heat Insulation Application of Water Bottles. Fluids 2017, 2, 64.
- Karadagli, I.; Milow, B.; Ratke, L.; Schulz, B.; Seide, G.; Gries, T. Synthesis and Characterization of Highly Porous Cellulose Aerogels for Textiles Applications. In Proceedings of the Cellular Materials: CELLMAT 2012, Dresden, Germany, 7–9 November 2012.
- Han, Y.; Zhang, X.; Wu, X.; Lu, C. Flame Retardant, Heat Insulating Cellulose Aerogels from Waste Cotton Fabrics by in Situ Formation of Magnesium Hydroxide Nanoparticles in Cellulose Gel Nanostructures. ACS Sustain. Chem. Eng. 2015, 3, 1853–1859.
- Bao, M.X.; Xu, S.; Wang, X.; Sun, R. Porous Cellulose Aerogels with High Mechanical Performance and Their Absorption Behaviors. Bioresources 2016, 11, 8–20.
- Hüsing, N.; Schubert, U. Aerogels—Airy Materials: Chemistry, Structure, and Properties. Angew. Chem. Int. Ed. 1998, 37, 22–45.
- Buchtová, N.; Budtova, T. Cellulose Aero-, Cryo- and Xerogels: Towards Understanding of Morphology Control. Cellulose 2016, 23, 2585–2595.
- Quiño, J.; Ruehl, M.; Klima, T.; Ruiz, F.; Will, S.; Braeuer, A. Supercritical Drying of Aerogel: In Situ Analysis of Concentration Profiles inside the Gel and Derivation of the Effective Binary Diffusion Coefficient Using Raman Spectroscopy. J. Supercrit. Fluids 2016, 108, 1–12.
- Özbakır, Y.; Erkey, C. Experimental and Theoretical Investigation of Supercritical Drying of Silica Alcogels. J. Supercrit. Fluids 2015, 98, 153–166.
- Sanz-Moral, L.M.; Rueda, M.; Mato, R.; Martín, Á. View Cell Investigation of Silica Aerogels during Supercritical Drying: Analysis of Size Variation and Mass Transfer Mechanisms. J. Supercrit. Fluids 2014, 92, 24–30.
- Griffin, J.S.; Mills, D.H.; Cleary, M.; Nelson, R.; Manno, V.P.; Hodes, M. Continuous Extraction Rate Measurements during Supercritical CO2 Drying of Silica Alcogel. J. Supercrit. Fluids 2014, 94, 38–47.
- García-González, C.A.; Alnaief, M.; Smirnova, I. Polysaccharide-Based Aerogels—Promising Biodegradable Carriers for Drug Delivery Systems. Carbohydr. Polym. 2011, 86, 1425–1438.
- Liebner, F.; Haimer, E.; Wendland, M.; Neouze, M.-A.; Schlufter, K.; Miethe, P.; Heinze, T.; Potthast, A.; Rosenau, T. Aerogels from Unaltered Bacterial Cellulose: Application of scCO2 Drying for the Preparation of Shaped, Ultra-Lightweight Cellulosic Aerogels. Macromol. Biosci. 2010, 10, 349–352.
- Lu, Y.; Qingfeng, S.; Yang, D.; She, X.; Yao, X.; Zhu, G.; Liu, Y.; Zhao, H.; Li, J. Fabrication of Mesoporous Lignocellulose Aerogels from Wood via Cyclic Liquid Nitrogen Freezing–Thawing in Ionic Liquid Solution. J. Mater. Chem. 2012, 22, 13548–13557.
- Pircher, N.; Carbajal, L.; Schimper, C.; Bacher, M.; Rennhofer, H.; Nedelec, J.-M.; Lichtenegger, H.C.; Rosenau, T.; Liebner, F. Impact of Selected Solvent Systems on the Pore and Solid Structure of Cellulose Aerogels. Cellulose 2016, 23, 1949–1966.
- Wang, X.; Zhang, Y.; Jiang, H.; Song, Y.; Zhou, Z.; Zhao, H. Fabrication and Characterization of Nano-Cellulose Aerogels via Supercritical CO2 Drying Technology. Mater. Lett. 2016, 183, 179–182.
- Heath, L.; Thielemans, W. Cellulose Nanowhisker Aerogels. Green Chem. 2010, 12, 1448–1453.
- Schestakow, M.; Karadagli, I.; Ratke, L. Cellulose Aerogels Prepared from an Aqueous Zinc Chloride Salt Hydrate Melt. Carbohydr. Polym. 2016, 137, 642–649.
- Tajiri, K.; Igarashi, K.; Nishio, T. Effects of Supercritical Drying Media on Structure and Properties of Silica Aerogel. J. Non-Cryst. Solids 1995, 186, 83–87.
- Laudise, R.A.; Johnson, D.W. Supercritical Drying of Gels. J. Non-Cryst. Solids 1986, 79, 155–164.
- Kocon, L.; Despetis, F.; Phalippou, J. Ultralow Density Silica Aerogels by Alcohol Supercritical Drying. J. Non-Cryst. Solids 1998, 225, 96–100.
- Stolarski, M.; Walendziewski, J.; Steininger, M.; Pniak, B. Synthesis and Characteristic of Silica Aerogels. Appl. Catal. A Gen. 1999, 177, 139–148.
- Mahadik, D.B.; Lee, Y.K.; Chavan, N.K.; Mahadik, S.A.; Park, H.-H. Monolithic and Shrinkage-Free Hydrophobic Silica Aerogels via New Rapid Supercritical Extraction Process. J. Supercrit. Fluids 2016, 107, 84–91.
- Kong, Y.; Shen, X.-D.; Cui, S. Direct Synthesis of Anatase TiO2 Aerogel Resistant to High Temperature under Supercritical Ethanol. Mater. Lett. 2014, 117, 192–194.
- Kirkbir, F.; Murata, H.; Meyers, D.; Chaudhuri, S.R. Drying of Large Monolithic Aerogels between Atmospheric and Supercritical Pressures. J. Sol-Gel Sci. Technol. 1998, 13, 311–316.
- Kirkbir, F.; Murata, H.; Meyers, D.; Chaudhuri, S.R. Drying of Aerogels in Different Solvents between Atmospheric and Supercritical Pressures. J. Non-Cryst. Solids 1998, 225, 14–18.
- Dowson, M.; Grogan, M.; Birks, T.; Harrison, D.; Craig, S. Streamlined Life Cycle Assessment of Transparent Silica Aerogel Made by Supercritical Drying. Appl. Energy 2012, 97, 396–404.
- van Bommel, M.J.; de Haan, A.B. Drying of Silica Aerogel with Supercritical Carbon Dioxide. J. Non-Cryst. Solids 1995, 186, 78–82.
- Sanz-Moral, L.M.; Rueda, M.; Nieto, A.; Novak, Z.; Knez, Ž.; Martín, Á. Gradual Hydrophobic Surface Functionalization of Dry Silica Aerogels by Reaction with Silane Precursors Dissolved in Supercritical Carbon Dioxide. J. Supercrit. Fluids 2013, 84, 74–79.
- Pajonk, G.M.; Venkateswara Rao, A.; Sawant, B.M.; Parvathy, N.N. Dependence of Monolithicity and Physical Properties of TMOS Silica Aerogels on Gel Aging and Drying conditions. J. Non-Cryst. Solids 1997, 209, 40–50.
- Masmoudi, Y.; Rigacci, A.; Ilbizian, P.; Cauneau, F.; Achard, P. Diffusion During the Supercritical Drying of Silica Gels. Dry. Technol. 2006, 24, 1121–1125.
- García-González, C.A.; Camino-Rey, M.C.; Alnaief, M.; Zetzl, C.; Smirnova, I. Supercritical Drying of Aerogels Using CO2: Effect of Extraction Time on the End Material Textural Properties. J. Supercrit. Fluids 2012, 66, 297–306.
- Wang, Y.-Y.; Gao, Y.-B.; Sun, Y.-H.; Chen, S.-Y. Effect of Preparation Parameters on the Texture of SiO2 Aerogels. Catal. Today 1996, 30, 171–175.
- Lu, Y.; Gao, R.; Xiao, S.; Yin, Y.; Liu, Q.; Li, J. Cellulose Based Aerogels: Processing and Morphology. In Biobased Aerogels: Polysaccharide and Protein-Based Materials; RSC Green Chemistry; The Royal Society of Chemistry: London, UK, 2018; pp. 25–41. ISBN 978-1-78262-765-4.
- Wang, X.; Zhang, Y.; Jiang, H.; Song, Y.; Zhou, Z.; Zhao, H. Tert-Butyl Alcohol Used to Fabricate Nano-Cellulose Aerogels via Freeze-Drying Technology. Mater. Res. Express 2017, 4, 065006.
- Pons, A.; Casas, L.; Estop, E.; Molins, E.; Harris, K.D.M.; Xu, M. A New Route to Aerogels: Monolithic Silica Cryogels. J. Non-Cryst. Solids 2012, 358, 461–469.
- Jiang, F.; Hsieh, Y.-L. Super Water Absorbing and Shape Memory Nanocellulose Aerogels from TEMPO-Oxidized Cellulose Nanofibrils via Cyclic Freezing–Thawing. J. Mater. Chem. A 2013, 2, 350–359.
- Zhang, X.; Yu, Y.; Jiang, Z.; Wang, H. The Effect of Freezing Speed and Hydrogel Concentration on the Microstructure and Compressive Performance of Bamboo-Based Cellulose Aerogel. J. Wood Sci. 2015, 61, 595–601.
- Nakagaito, A.; Kondo, H.; Takagi, H. Cellulose Nanofiber Aerogel Production and Applications. J. Reinf. Plast. Compos. 2013, 32, 1547–1552.
- Jiménez-Saelices, C.; Seantier, B.; Cathala, B.; Grohens, Y. Spray Freeze-Dried Nanofibrillated Cellulose Aerogels with Thermal Superinsulating Properties. Carbohydr. Polym. 2017, 157, 105–113.
- Cai, H.; Sharma, S.; Liu, W.; Mu, W.; Liu, W.; Zhang, X.; Deng, Y. Aerogel Microspheres from Natural Cellulose Nanofibrils and Their Application as Cell Culture Scaffold. Biomacromolecules 2014, 15, 2540–2547.
- Cai, J.; Kimura, S.; Wada, M.; Kuga, S.; Zhang, L. Cellulose Aerogels from Aqueous Alkali Hydroxide–Urea Solution. ChemSusChem 2008, 1, 149–154.
- Beaumont, M.; Kondor, A.; Plappert, S.; Mitterer, C.; Opietnik, M.; Potthast, A.; Rosenau, T. Surface Properties and Porosity of Highly Porous, Nanostructured Cellulose II Particles. Cellulose 2017, 24, 435–440.
- Guo, L.; Chen, Z.; Lyu, S.; Fu, F.; Wang, S. Highly Flexible Cross-Linked Cellulose Nanofibril Sponge-like Aerogels with Improved Mechanical Property and Enhanced Flame Retardancy. Carbohydr. Polym. 2018, 179, 333–340.
- Bhuiyan, M.A.R.; Wang, L.; Shanks, R.A.; Ara, Z.A.; Saha, T. Electrospun Polyacrylonitrile–Silica Aerogel Coating on Viscose Nonwoven Fabric for Versatile Protection and Thermal Comfort. Cellulose 2020, 27, 10501–10517.
- Chan, E.P.; Lee, J.-H.; Chung, J.Y.; Stafford, C.M. An Automated Spin-Assisted Approach for Molecular Layer-by-Layer Assembly of Crosslinked Polymer Thin Films. Rev. Sci. Instrum. 2012, 83, 114102.
- Atoufi, Z.; Reid, M.S.; Larsson, P.A.; Wågberg, L. Surface Tailoring of Cellulose Aerogel-like Structures with Ultrathin Coatings Using Molecular Layer-by-Layer Assembly. Carbohydr. Polym. 2022, 282, 119098.
- Johnson, P.M.; Yoon, J.; Kelly, J.Y.; Howarter, J.A.; Stafford, C.M. Molecular Layer-by-Layer Deposition of Highly Crosslinked Polyamide Films. J. Polym. Sci. Part B Polym. Phys. 2012, 50, 168–173.
- La, P.; Huynh, N.; Bui, K.; Pham, K.; Dao, X.-T.; Tran, T.; Nguyen, T.; Hoang, N.; Mai, P.; Nguyen, H.H. Synthesis and Surface Modification of Cellulose Aerogel from Coconut Peat for Oil Adsorption. Res. Sq. 2021; preprint.
- Kaya, M. Super Absorbent, Light, and Highly Flame Retardant Cellulose-Based Aerogel Crosslinked with Citric Acid. J. Appl. Polym. Sci. 2017, 134, 45315.
- Wicklein, B.; Kocjan, D.; Carosio, F.; Camino, G.; Bergström, L. Tuning the Nanocellulose–Borate Interaction to Achieve Highly Flame Retardant Hybrid Materials. Chem. Mater. 2016, 28, 1985–1989.
- Aegerter, M.A.; Leventis, N.; Koebel, M.M. (Eds.) Aerogels Handbook, 2011th ed.; Advances in Sol-Gel Derived Materials and Technologies; Springer: New York, NY, USA, 2011; ISBN 978-1-4419-7477-8.
- Jelle, B.P. Traditional, State-of-the-Art and Future Thermal Building Insulation Materials and Solutions—Properties, Requirements and Possibilities. Energy Build. 2011, 43, 2549–2563.
- BuyAerogel.com|Airloy® X103 Strong Aerogel Large Panels. Available online: http://www.buyaerogel.com/product/airloy-x103-large-panels/ (accessed on 16 August 2023).
- Antlauf, M.; Boulanger, N.; Berglund, L.; Oksman, K.; Andersson, O. Thermal Conductivity of Cellulose Fibers in Different Size Scales and Densities. Biomacromolecules 2021, 22, 3800–3809.
- Song, M.; Jiang, J.; Qin, H.; Ren, X.; Jiang, F. Flexible and Super Thermal Insulating Cellulose Nanofibril/Emulsion Composite Aerogel with Quasi-Closed Pores. ACS Appl. Mater. Interfaces 2020, 12, 45363–45372.
- Zeng, Z.; Wu, T.; Han, D.; Ren, Q.; Siqueira, G.; Nyström, G. Ultralight, Flexible, and Biomimetic Nanocellulose/Silver Nanowire Aerogels for Electromagnetic Interference Shielding. Acs Nano 2020, 14, 2927–2938.
- Yu, Z.; Suryawanshi, A.; He, H.; Liu, J.; Li, Y.; Lin, X.; Sun, Z. Preparation and Characterisation of Fire-Resistant PNIPAAm/SA/AgNP Thermosensitive Network Hydrogels and Laminated Cotton Fabric Used in Firefighter Protective Clothing. Cellulose 2020, 27, 5391–5406.
- Yu, Z.; Liu, J.; Suryawanshi, A.; He, H.; Wang, Y.; Zhao, Y. Thermal Insulating and Fire-Retarding Behavior of Treated Cotton Fabrics with a Novel High Water-Retaining Hydrogel Used in Thermal Protective Clothing. Cellulose 2021, 28, 2581–2597.
- Liu, Z.; Lyu, J.; Fang, D.; Zhang, X. Nanofibrous Kevlar Aerogel Threads for Thermal Insulation in Harsh Environments. ACS Nano 2019, 13, 5703–5711.
- Kim, S.J.; Kim, H.A. Effect of Fabric Structural Parameters and Weaving Conditions to Warp Tension of Aramid Fabrics for Protective Garments. Text. Res. J. 2018, 88, 987–1001.
- He, H.; Wang, Y.; Yu, Z.; Liu, J.; Zhao, Y.; Ke, Y. Ecofriendly Flame-Retardant Composite Aerogel Derived from Polysaccharide: Preparation, Flammability, Thermal Kinetics, and Mechanism. Carbohydr. Polym. 2021, 269, 118291.
- Cao, M.; Li, S.-L.; Cheng, J.-B.; Zhang, A.-N.; Wang, Y.-Z.; Zhao, H.-B. Fully Bio-Based, Low Fire-Hazard and Superelastic Aerogel without Hazardous Cross-Linkers for Excellent Thermal Insulation and Oil Clean-up Absorption. J. Hazard. Mater. 2021, 403, 123977.
- Wang, B.; Li, P.; Xu, Y.-J.; Jiang, Z.-M.; Dong, C.-H.; Liu, Y.; Zhu, P. Bio-Based, Nontoxic and Flame-Retardant Cotton/Alginate Blended Fibres as Filling Materials: Thermal Degradation Properties, Flammability and Flame-Retardant Mechanism. Compos. Part B Eng. 2020, 194, 108038.
- Zhao, H.-B.; Chen, M.; Chen, H.-B. Thermally Insulating and Flame-Retardant Polyaniline/Pectin Aerogels. ACS Sustain. Chem. Eng. 2017, 5, 7012–7019.
- Chen, J.; Xie, H.; Lai, X.; Li, H.; Gao, J.; Zeng, X. An Ultrasensitive Fire-Warning Chitosan/Montmorillonite/Carbon Nanotube Composite Aerogel with High Fire-Resistance. Chem. Eng. J. 2020, 399, 125729.
- He, C.; Huang, J.; Li, S.; Meng, K.; Zhang, L.; Chen, Z.; Lai, Y. Mechanically Resistant and Sustainable Cellulose-Based Composite Aerogels with Excellent Flame Retardant, Sound-Absorption, and Superantiwetting Ability for Advanced Engineering Materials. ACS Sustain. Chem. Eng. 2018, 6, 927–936.
- He, S.; Liu, C.; Chi, X.; Zhang, Y.; Yu, G.; Wang, H.; Li, B.; Peng, H. Bio-Inspired Lightweight Pulp Foams with Improved Mechanical Property and Flame Retardancy via Borate Cross-Linking. Chem. Eng. J. 2019, 371, 34–42.
- Thanh, N.T.L. Investigation on the Flame-Retardant and Physical Properties of the Modified Cellulosic and Polyurethane Aerogel. Mater. Today Proc. 2022, 66, 2726–2729.
- Gong, J.; Li, J.; Xu, J.; Xiang, Z.; Mo, L. Research on Cellulose Nanocrystals Produced from Cellulose Sources with Various Polymorphs. RSC Adv. 2017, 7, 33486–33493.
- Dong, H.; Xie, Y.; Zeng, G.; Tang, L.; Liang, J.; He, Q.; Zhao, F.; Zeng, Y.; Wu, Y. The Dual Effects of Carboxymethyl Cellulose on the Colloidal Stability and Toxicity of Nanoscale Zero-Valent Iron. Chemosphere 2016, 144, 1682–1689.
- Yahya, E.B.; Alzalouk, M.M.; Alfallous, K.A.; Abogmaza, A.F. Antibacterial Cellulose-Based Aerogels for Wound Healing Application: A Review. Biomed. Res. Ther. 2020, 7, 4032–4040.
- Lu, T.; Li, Q.; Chen, W.; Yu, H. Composite Aerogels Based on Dialdehyde Nanocellulose and Collagen for Potential Applications as Wound Dressing and Tissue Engineering Scaffold. Compos. Sci. Technol. 2014, 94, 132–138.
- Wang, X.; Cheng, F.; Liu, J.; Smått, J.-H.; Gepperth, D.; Lastusaari, M.; Xu, C.; Hupa, L. Biocomposites of Copper-Containing Mesoporous Bioactive Glass and Nanofibrillated Cellulose: Biocompatibility and Angiogenic Promotion in Chronic Wound Healing Application. Acta Biomater. 2016, 46, 286–298.
- Jack, A.A.; Nordli, H.R.; Powell, L.C.; Farnell, D.J.J.; Pukstad, B.; Rye, P.D.; Thomas, D.W.; Chinga-Carrasco, G.; Hill, K.E. Cellulose Nanofibril Formulations Incorporating a Low-Molecular-Weight Alginate Oligosaccharide Modify Bacterial Biofilm Development. Biomacromolecules 2019, 20, 2953–2961.
- Kishor, R.; Purchase, D.; Saratale, G.D.; Saratale, R.G.; Ferreira, L.F.R.; Bilal, M.; Chandra, R.; Bharagava, R.N. Ecotoxicological and Health Concerns of Persistent Coloring Pollutants of Textile Industry Wastewater and Treatment Approaches for Environmental Safety. J. Environ. Chem. Eng. 2021, 9, 105012.
- Lomoko, G.M.N.A.; Paliulis, D.; Valters, K. Removal of Copper (II) Ions from Polluted Water Using Modified Wheat Bran. Environ. Clim. Technol. 2021, 25, 853–864.
- Ardila-Leal, L.D.; Poutou-Piñales, R.A.; Pedroza-Rodríguez, A.M.; Quevedo-Hidalgo, B.E. A Brief History of Colour, the Environmental Impact of Synthetic Dyes and Removal by Using Laccases. Molecules 2021, 26, 3813.
- Altynbayeva, G.; Kadnikova, O.; Aydarhanov, A.; Toretayev, M. Industrial Wastewaters of the Feed Industry: Use of Sodium Ferrate in the Phenol Purification Process. Environ. Clim. Technol. 2021, 25, 829–839.
- Chandanshive, V.; Kadam, S.; Rane, N.; Jeon, B.-H.; Jadhav, J.; Govindwar, S. In Situ Textile Wastewater Treatment in High Rate Transpiration System Furrows Planted with Aquatic Macrophytes and Floating Phytobeds. Chemosphere 2020, 252, 126513.
- Islam, M.T.; Saenz-Arana, R.; Hernandez, C.; Guinto, T.; Ahsan, M.A.; Bragg, D.T.; Wang, H.; Alvarado-Tenorio, B.; Noveron, J.C. Conversion of Waste Tire Rubber into a High-Capacity Adsorbent for the Removal of Methylene Blue, Methyl Orange, and Tetracycline from Water. J. Environ. Chem. Eng. 2018, 6, 3070–3082.
- Wei, F.; Shahid, M.J.; Alnusairi, G.S.H.; Afzal, M.; Khan, A.; El-Esawi, M.A.; Abbas, Z.; Wei, K.; Zaheer, I.E.; Rizwan, M.; et al. Implementation of Floating Treatment Wetlands for Textile Wastewater Management: A Review. Sustainability 2020, 12, 5801.
- Liugė, M.; Paliulis, D. Treatment of Water Containing Dyes Using Cellulose Aerogels. Environ. Clim. Technol. 2023, 27, 314–322.
- Neetha, J.N.; Sandesh, K.; Kumar, K.G.; Chidananda, B.; Ujwal, P. Optimization of Direct Blue-14 Dye Degradation by Bacillus Fermus (Kx898362) an Alkaliphilic Plant Endophyte and Assessment of Degraded Metabolite Toxicity. J. Hazard. Mater. 2019, 364, 742–751.
- Shen, D.; Fan, J.; Zhou, W.; Gao, B.; Yue, Q.; Kang, Q. Adsorption Kinetics and Isotherm of Anionic Dyes onto Organo-Bentonite from Single and Multisolute Systems. J. Hazard. Mater. 2009, 172, 99–107.
- Patel, H. Charcoal as an Adsorbent for Textile Wastewater Treatment. Sep. Sci. Technol. 2018, 53, 2797–2812.
- Javanbakht, V.; Ghoreishi, S.M.; Javanbakht, M. Mathematical Modeling of Batch Adsorption Kinetics of Lead Ions on Modified Natural Zeolite from Aqueous Media. Theor. Found. Chem. Eng. 2019, 53, 1057–1066.
- Lyu, W.; Li, J.; Zheng, L.; Liu, H.; Chen, J.; Zhang, W.; Liao, Y. Fabrication of 3D Compressible Polyaniline/Cellulose Nanofiber Aerogel for Highly Efficient Removal of Organic Pollutants and Its Environmental-Friendly Regeneration by Peroxydisulfate Process. Chem. Eng. J. 2021, 414, 128931.
- Ali Musa, M.; Idrus, S. Physical and Biological Treatment Technologies of Slaughterhouse Wastewater: A Review. Sustainability 2021, 13, 4656.
- Li, W.; Mu, B.; Yang, Y. Feasibility of Industrial-Scale Treatment of Dye Wastewater via Bio-Adsorption Technology. Bioresour. Technol. 2019, 277, 157–170.
- Nguyen, T.A.; Juang, R.-S. Treatment of Waters and Wastewaters Containing Sulfur Dyes: A Review. Chem. Eng. J. 2013, 219, 109–117.
- Rambabu, K.; Bharath, G.; Banat, F.; Show, P.L. Green Synthesis of Zinc Oxide Nanoparticles Using Phoenix Dactylifera Waste as Bioreductant for Effective Dye Degradation and Antibacterial Performance in Wastewater Treatment. J. Hazard. Mater. 2021, 402, 123560.
- Hu, B.-C.; Zhang, H.-R.; Li, S.-C.; Chen, W.-S.; Wu, Z.-Y.; Liang, H.-W.; Yu, H.-P.; Yu, S.-H. Robust Carbonaceous Nanofiber Aerogels from All Biomass Precursors. Adv. Funct. Mater. 2023, 33, 2207532.
- Ilangovan, M.; Guna, V.; Olivera, S.; Ravi, A.; Muralidhara, H.B.; Santosh, M.S.; Reddy, N. Highly Porous Carbon from a Natural Cellulose Fiber as High Efficiency Sorbent for Lead in Waste Water. Bioresour. Technol. 2017, 245, 296–299.
- Jiang, Z.; Ho, S.-H.; Wang, X.; Li, Y.; Wang, C. Application of Biodegradable Cellulose-Based Biomass Materials in Wastewater Treatment. Environ. Pollut. 2021, 290, 118087.
- Li, T.; Chen, C.; Brozena, A.H.; Zhu, J.Y.; Xu, L.; Driemeier, C.; Dai, J.; Rojas, O.J.; Isogai, A.; Wågberg, L.; et al. Developing Fibrillated Cellulose as a Sustainable Technological Material. Nature 2021, 590, 47–56.
- Zhang, J.; Zhang, X.; Tian, Y.; Zhong, T.; Liu, F. Novel and Wet-Resilient Cellulose Nanofiber Cryogels with Tunable Porosity and Improved Mechanical Strength for Methyl Orange Dyes Removal. J. Hazard. Mater. 2021, 416, 125897.
- Liu, J.-L.; Qian, W.-C.; Guo, J.-Z.; Shen, Y.; Li, B. Selective Removal of Anionic and Cationic Dyes by Magnetic Fe3O4-Loaded Amine-Modified Hydrochar. Bioresour. Technol. 2021, 320, 124374.
- Qiu, C.; Li, Y.; Liu, H.; Wang, X.; Hu, S.; Qi, H. A Novel Crosslinking Strategy on Functional Cellulose-Based Aerogel for Effective and Selective Removal of Dye. Chem. Eng. J. 2023, 463, 142404.
- Wu, W.; Yang, Y.; Wang, B.; Rong, L.; Xu, H.; Sui, X.; Zhong, Y.; Zhang, L.; Chen, Z.; Feng, X.; et al. The Effect of the Degree of Substitution on the Solubility of Cellulose Acetoacetates in Water: A Molecular Dynamics Simulation and Density Functional Theory Study. Carbohydr. Res. 2020, 496, 108134.
- Li, L.; Rong, L.; Xu, Z.; Wang, B.; Feng, X.; Mao, Z.; Xu, H.; Yuan, J.; Liu, S.; Sui, X. Cellulosic Sponges with pH Responsive Wettability for Efficient Oil-Water Separation. Carbohydr. Polym. 2020, 237, 116133.
- Peng, F.; Liu, H.; Xiao, D.; Guo, L.; Yue, F.; Würfe, H.; Heinze, T.; Qi, H. Green Fabrication of High Strength, Transparent Cellulose-Based Films with Durable Fluorescence and UV-Blocking Performance. J. Mater. Chem. A 2022, 10, 7811–7817.
- Liu, H.; Sui, X.; Xu, H.; Zhang, L.; Zhong, Y.; Mao, Z. Self-Healing Polysaccharide Hydrogel Based on Dynamic Covalent Enamine Bonds. Macromol. Mater. Eng. 2016, 301, 725–732.
- Li, Z.; Wang, X.; Kuang, W.; Dong, C.; Fan, Y.; Guo, Y.; Qiao, Q.; Zhu, Z.; Liu, Y.; Zhu, Y. Biofiber Waste Derived Zwitterionic and Photocatalytic Dye Adsorbent: Switchable Selectivity, in-Situ Degradation and Multi-Tasking Application. Bioresour. Technol. 2022, 352, 127080.
- Maatar, W.; Boufi, S. Microporous Cationic Nanofibrillar Cellulose Aerogel as Promising Adsorbent of Acid Dyes. Cellulose 2017, 24, 1001–1015.
- Zhang, M.; Xu, T.; Zhao, Q.; Liu, K.; Liang, D.; Si, C. Cellulose-Based Materials for Carbon Capture and Conversion. Carbon Capture Sci. Technol. 2024, 10, 100157.
- Zhang, M.; Wang, Y.; Liu, K.; Liu, Y.; Xu, T.; Du, H.; Si, C. Strong, Conductive, and Freezing-Tolerant Polyacrylamide/PEDOT:PSS/Cellulose Nanofibrils Hydrogels for Wearable Strain Sensors. Carbohydr. Polym. 2023, 305, 120567.
- Ho, N.A.D.; Leo, C.P. A Review on the Emerging Applications of Cellulose, Cellulose Derivatives and Nanocellulose in Carbon Capture. Environ. Res. 2021, 197, 111100.
- Adegoke, K.A.; Oyedotun, K.O.; Ighalo, J.O.; Amaku, J.F.; Olisah, C.; Adeola, A.O.; Iwuozor, K.O.; Akpomie, K.G.; Conradie, J. Cellulose Derivatives and Cellulose-Metal-Organic Frameworks for CO2 adsorption and Separation. J. CO2 Util. 2022, 64, 102163.
- Zheng, Y.; Liu, H.; Yan, L.; Yang, H.; Dai, L.; Si, C. Lignin-Based Encapsulation of Liquid Metal Particles for Flexible and High-Efficiently Recyclable Electronics. Adv. Funct. Mater. 2023, 2310653.
- Liu, H.; Xu, T.; Liang, Q.; Zhao, Q.; Zhao, D.; Si, C. Compressible Cellulose Nanofibrils/Reduced Graphene Oxide Composite Carbon Aerogel for Solid-State Supercapacitor. Adv. Compos. Hybrid Mater. 2022, 5, 1168–1179.
- Xu, T.; Liu, K.; Sheng, N.; Zhang, M.; Liu, W.; Liu, H.; Dai, L.; Zhang, X.; Si, C.; Du, H.; et al. Biopolymer-Based Hydrogel Electrolytes for Advanced Energy Storage/Conversion Devices: Properties, Applications, and Perspectives. Energy Storage Mater. 2022, 48, 244–262.
- Duan, Y.; Yang, H.; Liu, K.; Xu, T.; Chen, J.; Xie, H.; Du, H.; Dai, L.; Si, C. Cellulose Nanofibril Aerogels Reinforcing Polymethyl Methacrylate with High Optical Transparency. Adv. Compos. Hybrid Mater. 2023, 6, 123.
- Liu, W.; Du, H.; Zhang, M.; Liu, K.; Liu, H.; Xie, H.; Zhang, X.; Si, C. Bacterial Cellulose-Based Composite Scaffolds for Biomedical Applications: A Review. ACS Sustain. Chem. Eng. 2020, 8, 7536–7562.
- Liu, W.; Lin, Q.; Chen, S.; Yang, H.; Liu, K.; Pang, B.; Xu, T.; Si, C. Microencapsulated Phase Change Material through Cellulose Nanofibrils Stabilized Pickering Emulsion Templating. Adv. Compos. Hybrid Mater. 2023, 6, 149.
- Xu, T.; Du, H.; Liu, H.; Liu, W.; Zhang, X.; Si, C.; Liu, P.; Zhang, K. Advanced Nanocellulose-Based Composites for Flexible Functional Energy Storage Devices. Adv. Mater. 2021, 33, 2101368.
- Zulaikha, W.; Hassan, M.Z.; Ismail, Z. Recent Development of Natural Fibre for Nanocellulose Extraction and Application. Mater. Today Proc. 2022, 66, 2265–2273.
- Yu, S.; Zhao, X.; Zhang, J.; Liu, S.; Yuan, Z.; Liu, X.; Liu, B.; Yi, X. A Novel Combining Strategy of Cellulose Aerogel and Hierarchically Porous Metal Organic Frameworks (HP-MOFs) to Improve the CO2 Absorption Performance. Cellulose 2022, 29, 6783–6796.
- Zhou, G.; Wang, K.; Liu, R.; Tian, Y.; Kong, B.; Qi, G. Synthesis and CO2 Adsorption Performance of TEPA-Loaded Cellulose Whisker/Silica Composite Aerogel. Colloids Surf. A Physicochem. Eng. Asp. 2021, 631, 127675.
- Ma, H.; Wang, Z.; Zhang, X.-F.; Ding, M.; Yao, J. In Situ Growth of Amino-Functionalized ZIF-8 on Bacterial Cellulose Foams for Enhanced CO2 Adsorption. Carbohydr. Polym. 2021, 270, 118376.
- Miao, Y.; Luo, H.; Pudukudy, M.; Zhi, Y.; Zhao, W.; Shan, S.; Jia, Q.; Ni, Y. CO2 Capture Performance and Characterization of Cellulose Aerogels Synthesized from Old Corrugated Containers. Carbohydr. Polym. 2020, 227, 115380.
More
Information
Subjects:
Materials Science, Textiles
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.9K
Revisions:
3 times
(View History)
Update Date:
28 Dec 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





