
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Екатерина Джугашвили | -- | 2792 | 2023-12-16 14:28:20 | | | |
| 2 | Lindsay Dong | Meta information modification | 2792 | 2023-12-18 06:48:03 | | |
Video Upload Options
Ovarian cancer (OC) has the highest mortality rate among all gynecologic cancers and is characterized by early peritoneal spread. The growth and development of OC are associated with the formation of ascitic fluid, creating a unique tumor microenvironment. Understanding the mechanisms of tumor progression is crucial in identifying new diagnostic biomarkers and developing novel therapeutic strategies. Exosomes, lipid bilayer vesicles measuring 30–150 nm in size, are known to establish a crucial link between malignant cells and their microenvironment. Additionally, the confirmed involvement of exosomes in carcinogenesis enables them to mediate the invasion, migration, metastasis, and angiogenesis of tumor cells. Functionally active non-coding RNAs (such as microRNAs, long non-coding RNAs, circRNAs), proteins, and lipid rafts transported within exosomes can activate numerous signaling pathways and modify gene expression.
1. Introduction
2. Formation and Secretion of Exosomes
- (1)
-
Invagination of the plasmalemma and formation of early endosomes.
- (2)
-
Early endosomes mature into multivesicular bodies (MVBs), which contain intraluminal vesicles (ILVs) filled with various proteins, lipids, and nucleic acids. Notably, the cargo composition of ILVs is specific to the parent cell.
- (3)
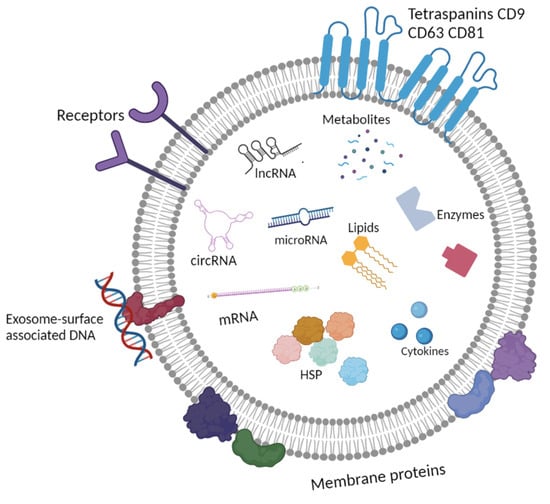
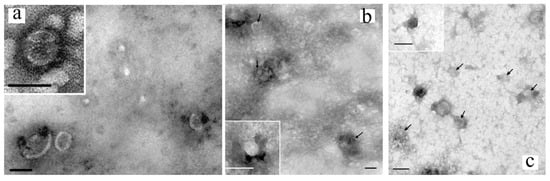
3. Morphology and Content of Exosomes
3.1. Lipidome of Exosomes
3.2. Proteome of Exosomes
| No | Gene Symbol | Uniprot ID | Function |
|---|---|---|---|
| 1 | CD9 | P21926 | Membrane protein. Identified on membranes of oocytes and extracellular exosomes |
| 2 | HSPA8 | P11142 | Chaperone protein |
| 3 | PDCD6IP | Q8WUM4 | Involved in sorting of cargo proteins of the MVBs for incorporation into ILVs |
| 4 | GAPDH | P04406 | Modulates the organization and assembly of the cytoskeleton |
| 5 | ACTB | P60709 | Protein that polymerizes to produce filaments |
| 6 | ANXA2 | P07355 | Calcium-regulated membrane-binding protein |
| 7 | CD63 | P08962 | Cell surface receptor for TIMP1 and plays a role in the activation of cellular signaling cascades AKT, FAK/PTK2 and MAPK |
| 8 | SDCBP | O00560 | Involved in the trafficking of transmembrane proteins, exosome biogenesis, and tumorigenesis |
| 9 | ENO1 | P06733 | Involved in glycolysis, growth control, hypoxia tolerance, and allergic responses |
| 10 | HSP90AA1 | P07900 | Chaperone protein |
| 11 | TSG101 | Q99816 | The component of the ESCRT-I complex mediates the association between the ESCRT-0 and ESCRT-I complex |
| 12 | PKM | P14618 | Catalyzes the final rate-limiting step of glycolysis generating ATP |
| 13 | LDHA | P00338 | Interconverts simultaneously and stereospecifically pyruvate and lactate with concomitant interconversion of NADH and NAD+. |
| 14 | EEF1A1 | P68104 | Translation elongation factor that catalyzes the GTP-dependent binding of aminoacyl-tRNA (aa-tRNA) to the A-site of ribosomes |
| 15 | YWHAZ | P63104 | Adapter protein implicated in the regulation of a large spectrum of signaling pathways |
| 16 | PGK1 | P00558 | Catalyzes one of the two ATP-producing reactions; acts as a polymerase alpha cofactor protein |
| 17 | EEF2 | P13639 | Catalyzes the GTP-dependent ribosomal translocation step during translation elongation |
| 18 | ALDOA | P04075 | Plays a key role in glycolysis and gluconeogenesis; scaffolding protein |
| 19 | HSP90AB1 | P08238 | Chaperone protein |
| 20 | ANXA5 | P08758 | Acts as an indirect inhibitor of the thromboplastin-specific complex |
Proteomic analysis of exosomes obtained from the SKOV3 and HOSEPiC cell cultures revealed 659 universal proteins out of all 1433 identified exosomal proteins [38]. COX2 is one of the most abundant exosomal proteins whose increased expression is associated with hypoxia. Probably, the formation of tumor spheroids and metastasis process are caused by overexpression of this protein [39].
Proteins implicated in carcinogenesis have been identified in the cargo of exosomes obtained from the blood plasma and ascitic fluid of OC patients. Specifically, proteins like ATF2, MTA1, ROCK1/2, and CD147 are involved in tumor angiogenesis, while GNA12, EPHA2, and COIA1 promote migration and metastasis. The Nanog protein plays a role in mediating the proliferation and invasion of tumor cells. Exosomes also contain Hsp90 and Hsc70, MHCI, and MHCII. Additionally, various enzymes have been detected in exosomes, such as phosphate isomerase, peroxiredoxins, aldehyde reductase, fatty acid synthase, and Dicer, which is involved in microRNA maturation, among others [40][41].
3.3. Nucleic Acids Transported by Exosomes
In addition to proteins and lipids, exosomes carry functionally active nucleic acids. It has been shown that exosomes have DNA in their crown, but the proportion of such DNA does not exceed 0.025% of blood plasma DNA in healthy women [30]. It is also known that exosomes contain various types of RNA: mRNA, microRNA, long noncoding RNA, rRNA, tRNA, circRNA, etc. [45]. According to the Exocarta database (www.exocarta.org, accessed on 1 April 2023), exosomes are involved in the transport of more than 2838 microRNAs and 3408 mRNAs. Some researchers also consider lncRNAs and circRNAs as promising diagnostic biomarkers for liquid biopsy due to their ability to influence the carcinogenesis of tumors, including OCs.
4. Conclusions
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249.
- Elias, K.M.; Guo, J.; Bast, R.C., Jr. Early Detection of Ovarian Cancer. Hematol. Oncol. Clin. N. Am. 2018, 32, 903–914.
- Ledermann, J.A.; Raja, F.A.; Fotopoulou, C.; Gonzalez-Martin, A.; Colombo, N.; Sessa, C.; ESMO Guidelines Working Group. Newly diagnosed and relapsed epithelial ovarian carcinoma: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2013, 6, 24–32.
- Nebgen, D.R.; Lu, K.H.; Bast, R.C., Jr. Novel Approaches to Ovarian Cancer Screening. Curr. Oncol. Rep. 2019, 21, 75.
- Dochez, V.; Caillon, H.; Vaucel, E.; Dimet, J.; Winer, N.; Ducarme, G. Biomarkers and algorithms for diagnosis of ovarian cancer: CA125, HE4, RMI and ROMA, a review. J. Ovarian Res. 2019, 12, 28.
- Fischerova, D.; Zikan, M.; Semeradova, I.; Slama, J.; Kocian, R.; Dundr, P.; Nemejcova, K.; Burgetova, A.; Dusek, L.; Cibula, D. Ultrasound in preoperative assessment of pelvic and abdominal spread in patients with ovarian cancer: A prospective study. Ultrasound Obstet. Gynecol. 2017, 49, 263–274.
- Roett, M.A.; Evans, P. Ovarian cancer: An overview. Am. Fam. Physician 2009, 80, 609–616.
- Roze, J.F.; Hoogendam, J.P.; van de Wetering, F.T.; Spijker, R.; Verleye, L.; Vlayen, J.; Veldhuis, W.B.; Scholten, R.J.; Zweemer, R.P. Positron emission tomography (PET) and magnetic resonance imaging (MRI) for assessing tumour resectability in advanced epithelial ovarian/fallopian tube/primary peritoneal cancer. Cochrane Database Syst. Rev. 2018, 10, CD012567.
- Gu, P.; Pan, L.L.; Wu, S.Q.; Sun, L.; Huang, G. CA 125, PET alone, PET–CT, CT and MRI in diagnosing recurrent ovarian carcinoma. Eur. J. Radiol. 2009, 71, 164–174.
- Tenchov, R.; Sasso, J.M.; Wang, X.; Liaw, W.S.; Chen, C.A.; Zhou, Q.A. Exosomes—Nature’s Lipid Nanoparticles, a Rising Star in Drug Delivery and Diagnostics. ACS Nano 2022, 16, 17802–17846.
- Shefer, A.; Yalovaya, A.; Tamkovich, S. Exosomes in Breast Cancer: Involvement in Tumor Dissemination and Prospects for Liquid Biopsy. Int. J. Mol. Sci. 2022, 23, 8845.
- Yunusova, N.; Kolegova, E.; Sereda, E.; Kolomiets, L.; Villert, A.; Patysheva, M.; Rekeda, I.; Grigor’eva, A.; Tarabanovskaya, N.; Kondakova, I.; et al. Plasma Exosomes of Patients with Breast and Ovarian Tumors Contain an Inactive 20S Proteasome. Molecules 2021, 26, 6965.
- Gurung, S.; Perocheau, D.; Touramanidou, L.; Baruteau, J. The exosome journey: From biogenesis to uptake and intracellular signalling. Cell Commun. Signal. 2021, 19, 47.
- Zhang, L.; Yu, D. Exosomes in cancer development, metastasis, and immunity. Biochim. Biophys. Acta Rev. Cancer 2019, 1871, 455–468.
- Huotari, J.; Helenius, A. Endosome maturation. EMBO J. 2011, 30, 3481–3500.
- Beach, A.; Zhang, H.G.; Ratajczak, M.Z.; Kakar, S.S. Exosomes: An overview of biogenesis, composition and role in ovarian cancer. J. Ovarian Res. 2014, 7, 14.
- Colombo, M.; Moita, C.; van Niel, G.; Kowal, J.; Vigneron, J.; Benaroch, P.; Manel, N.; Moita, L.F.; Théry, C.; Raposo, G. Analysis of ESCRT functions in exosome biogenesis, composition and secretion highlights the heterogeneity of extracellular vesicles. J. Cell Sci. 2013, 126, 5553–5565.
- Babst, M. MVB vesicle formation: ESCRT-dependent, ESCRT-independent and everything in between. Curr. Opin. Cell Biol. 2011, 23, 452–457.
- Elsherbini, A.; Bieberich, E. Ceramide and Exosomes: A Novel Target in Cancer Biology and Therapy. Adv. Cancer Res. 2018, 140, 121–154.
- Hurwitz, S.N.; Conlon, M.M.; Rider, M.A.; Brownstein, N.C.; Meckes, D.G., Jr. Nanoparticle analysis sheds budding insights into genetic drivers of extracellular vesicle biogenesis. J. Extracell. Vesicles 2016, 5, 31295.
- Nonaka, T.; Wong, D.T.W. Saliva-Exosomics in Cancer: Molecular Characterization of Cancer-Derived Exosomes in Saliva. Enzymes 2017, 42, 125–151.
- Cao, J.; Zhang, Y.; Mu, J.; Yang, D.; Gu, X.; Zhang, J. Exosomal miR-21-5p contributes to ovarian cancer progression by regulating CDK6. Hum. Cell 2021, 34, 1185–1196.
- Tutanov, O.; Orlova, E.; Proskura, K.; Grigor’eva, A.; Yunusova, N.; Tsentalovich, Y.; Alexandrova, A.; Tamkovich, S. Proteomic Analysis of Blood Exosomes from Healthy Females and Breast Cancer Patients Reveals an Association between Different Exosomal Bioactivity on Non-Tumorigenic Epithelial Cell and Breast Cancer Cell Migration in Vitro. Biomolecules 2020, 10, 495.
- Yunusova, N.V.; Patysheva, M.R.; Molchanov, S.V.; Zambalova, E.A.; Grigor’eva, A.E.; Kolomiets, L.A.; Ochirov, M.O.; Tamkovich, S.N.; Kondakova, I.V. Metalloproteinases at the surface of small extrcellular vesicles in advanced ovarian cancer: Relationships with ascites volume and peritoneal canceromatosis index. Clin. Chim. Acta 2019, 494, 116–122.
- Li, M.; Zeringer, E.; Barta, T.; Schageman, J.; Cheng, A.; Vlassov, A.V. Analysis of the RNA content of the exosomes derived from blood serum and urine and its potential as biomarkers. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014, 369, 20130502.
- Xu, H.; Li, M.; Pan, Z.; Zhang, Z.; Gao, Z.; Zhao, R.; Li, B.; Qi, Y.; Qiu, W.; Guo, Q.; et al. miR-3184-3p enriched in cerebrospinal fluid exosomes contributes to progression of glioma and promotes M2-like macrophage polarization. Cancer Sci. 2022, 113, 2668–2680.
- Kim, K.U.; Kim, W.H.; Jeong, C.H.; Yi, D.Y.; Min, H. More than Nutrition: Therapeutic Potential of Breast Milk-Derived Exosomes in Cancer. Int. J. Mol. Sci. 2020, 21, 7327.
- Tamkovich, S.; Grigor’eva, A.; Eremina, A.; Tupikin, A.; Kabilov, M.; Chernykh, V.; Vlassov, V.; Ryabchikova, E. What information can be obtained from the tears of a patient with primary open angle glaucoma? Clin. Chim. Acta. 2019, 495, 529–537.
- Zhang, W.; Ou, X.; Wu, X. Proteomics profiling of plasma exosomes in epithelial ovarian cancer: A potential role in the coagulation cascade, diagnosis and prognosis. Int. J. Oncol. 2019, 54, 1719–1733.
- Tutanov, O.; Shtam, T.; Grigor’eva, A.; Tupikin, A.; Tsentalovich, Y.; Tamkovich, S. Blood Plasma Exosomes Contain Circulating DNA in Their Crown. Diagnostics 2022, 12, 854.
- Konoshenko, M.; Sagaradze, G.; Orlova, E.; Shtam, T.; Proskura, K.; Kamyshinsky, R.; Yunusova, N.; Alexandrova, A.; Efimenko, A.; Tamkovich, S. Total Blood Exosomes in Breast Cancer: Potential Role in Crucial Steps of Tumorigenesis. Int. J. Mol. Sci. 2020, 21, 7341.
- Quesnel, A.; Broughton, A.; Karagiannis, G.S.; Filippou, P.S. Message in the bottle: Regulation of the tumor microenvironment via exosome-driven proteolysis. Cancer Metastasis Rev. 2022, 41, 789–801.
- Yuana, Y.; Koning, R.I.; Kuil, M.E.; Rensen, P.C.; Koster, A.J.; Bertina, R.M.; Osanto, S. Cryo-electron microscopy of extracellular vesicles in fresh plasma. J. Extracell. Vesicles 2013, 2, 21494.
- Emelyanov, A.; Shtam, T.; Kamyshinsky, R.; Garaeva, L.; Verlov, N.; Miliukhina, I.; Kudrevatykh, A.; Gavrilov, G.; Zabrodskaya, Y.; Pchelina, S.; et al. Cryo-electron microscopy of extracellular vesicles from cerebrospinal fluid. PLoS ONE 2020, 15, e0227949.
- Dzhugashvili, E.I.; Yunusova, N.V.; Yalovaya, A.I.; Grigorieva, A.E.; Sereda, E.E.; Kolomiets, L.A.; Tamkovich, S.N. Comparative assessment of the exosomal tumor-associated microRNA levels in blood plasma and ascitic fluid in ovarian cancer patients. Adv. Mol. Oncol. 2023, 10, 108–116. (In Russia)
- Choi, D.S.; Kim, D.K.; Kim, Y.K.; Gho, Y.S. Proteomics, transcriptomics and lipidomics of exosomes and ectosomes. Proteomics 2013, 13, 1554–1571.
- Liangsupree, T.; Multia, E.; Saarinen, J.; Ruiz-Jimenez, J.; Kemell, M.; Riekkola, M.L. Raman spectroscopy combined with comprehensive gas chromatography for label-free characterization of plasma-derived extracellular vesicle subpopulations. Anal. Biochem. 2022, 647, 114672.
- Cheng, L.; Zhang, K.; Qing, Y.; Li, D.; Cui, M.; Jin, P.; Xu, T. Proteomic and lipidomic analysis of exosomes derived from ovarian cancer cells and ovarian surface epithelial cells. J. Ovarian Res. 2020, 13, 9.
- Ding, Y.; Zhuang, S.; Li, Y.; Yu, X.; Lu, M.; Ding, N. Hypoxia-induced HIF1α dependent COX2 promotes ovarian cancer progress. J. Bioenerg. Biomembr. 2021, 53, 441–448.
- Feng, W.; Dean, D.C.; Hornicek, F.J.; Shi, H.; Duan, Z. Exosomes promote pre-metastatic niche formation in ovarian cancer. Mol. Cancer 2019, 18, 124.
- Tang, M.K.; Wong, A.S. Exosomes: Emerging biomarkers and targets for ovarian cancer. Cancer Lett. 2015, 367, 26–33.
- Zhang, X.; Sheng, Y.; Li, B.; Wang, Q.; Liu, X.; Han, J. Ovarian cancer derived PKR1 positive exosomes promote angiogenesis by promoting migration and tube formation in vitro. Cell Biochem. Funct. 2021, 39, 308–316.
- Shender, V.O.; Pavlyukov, M.S.; Ziganshin, R.H.; Arapidi, G.P.; Kovalchuk, S.I.; Anikanov, N.A.; Altukhov, I.A.; Alexeev, D.G.; Butenko, I.O.; Shavarda, A.L.; et al. Proteome-metabolome profiling of ovarian cancer ascites reveals novel components involved in intercellular communication. Mol. Cell Proteom. 2014, 13, 3558–3571.
- Dorayappan, K.D.P.; Wallbillich, J.J.; Cohn, D.E.; Selvendiran, K. The biological significance and clinical applications of exosomes in ovarian cancer. Gynecol. Oncol. 2016, 142, 199–205.
- Yi, Y.; Wu, M.; Zeng, H.; Hu, W.; Zhao, C.; Xiong, M.; Lv, W.; Deng, P.; Zhang, Q.; Wu, Y. Tumor-Derived Exosomal Non-Coding RNAs: The Emerging Mechanisms and Potential Clinical Applications in Breast Cancer. Front. Oncol. 2021, 11, 738945.




