Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Krasimir Iliev Kraev | -- | 2838 | 2023-11-15 19:45:27 | | | |
| 2 | Jessie Wu | Meta information modification | 2838 | 2023-11-16 04:00:36 | | | | |
| 3 | Jessie Wu | -2 word(s) | 2836 | 2023-11-16 04:01:32 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Kraev, K.I.; Geneva-Popova, M.G.; Hristov, B.K.; Uchikov, P.A.; Popova-Belova, S.D.; Kraeva, M.I.; Basheva-Kraeva, Y.M.; Stoyanova, N.S.; Mitkova-Hristova, V.T. Febuxostat’s Multifaceted Therapeutic Application. Encyclopedia. Available online: https://encyclopedia.pub/entry/51630 (accessed on 10 May 2026).
Kraev KI, Geneva-Popova MG, Hristov BK, Uchikov PA, Popova-Belova SD, Kraeva MI, et al. Febuxostat’s Multifaceted Therapeutic Application. Encyclopedia. Available at: https://encyclopedia.pub/entry/51630. Accessed May 10, 2026.
Kraev, Krasimir Iliev, Mariela Gencheva Geneva-Popova, Bozhidar Krasimirov Hristov, Petar Angelov Uchikov, Stanislava Dimitrova Popova-Belova, Maria Ilieva Kraeva, Yordanka Mincheva Basheva-Kraeva, Nina Staneva Stoyanova, Vesela Todorova Mitkova-Hristova. "Febuxostat’s Multifaceted Therapeutic Application" Encyclopedia, https://encyclopedia.pub/entry/51630 (accessed May 10, 2026).
Kraev, K.I., Geneva-Popova, M.G., Hristov, B.K., Uchikov, P.A., Popova-Belova, S.D., Kraeva, M.I., Basheva-Kraeva, Y.M., Stoyanova, N.S., & Mitkova-Hristova, V.T. (2023, November 15). Febuxostat’s Multifaceted Therapeutic Application. In Encyclopedia. https://encyclopedia.pub/entry/51630
Kraev, Krasimir Iliev, et al. "Febuxostat’s Multifaceted Therapeutic Application." Encyclopedia. Web. 15 November, 2023.
Copy Citation
Febuxostat, a xanthine oxidase inhibitor originally introduced for the management of hyperuricemia in patients with gout, has evolved into a multifaceted therapeutic agent with a spectrum of effects extending far beyond its initial scope. The diverse range of impacts exerted by febuxostat on various physiological systems are explored, offering insights into its mechanism of action, efficacy in gout management, cardiovascular implications, renal and hepatic effects, musculoskeletal applications, adverse event profiles, and emerging avenues for future research.
febuxostat
hyperuricemia
inflammation
1. Febuxostat
Febuxostat is a xanthine oxidase inhibitor, a pharmaceutical agent employed in the management of hyperuricemia, particularly in the context of gout, a condition characterized by the deposition of uric acid crystals in joints (Figure 1). By selectively inhibiting the enzyme xanthine oxidase, febuxostat impedes the conversion of xanthine and hypoxanthine to uric acid, thereby reducing the production of uric acid in the body [1][2]. This mechanism of action helps to lower serum uric acid levels, mitigating the risk of acute gout flares and the formation of urate crystals in tissues. Febuxostat is considered a valuable pharmacotherapeutic option for patients with gout, hyperuricemia, or those susceptible to recurrent gout attacks, especially when conventional therapies prove inadequate or intolerable. Its clinical utility extends to individuals with conditions like renal impairment, where alternative medications may be less suitable [3][4].
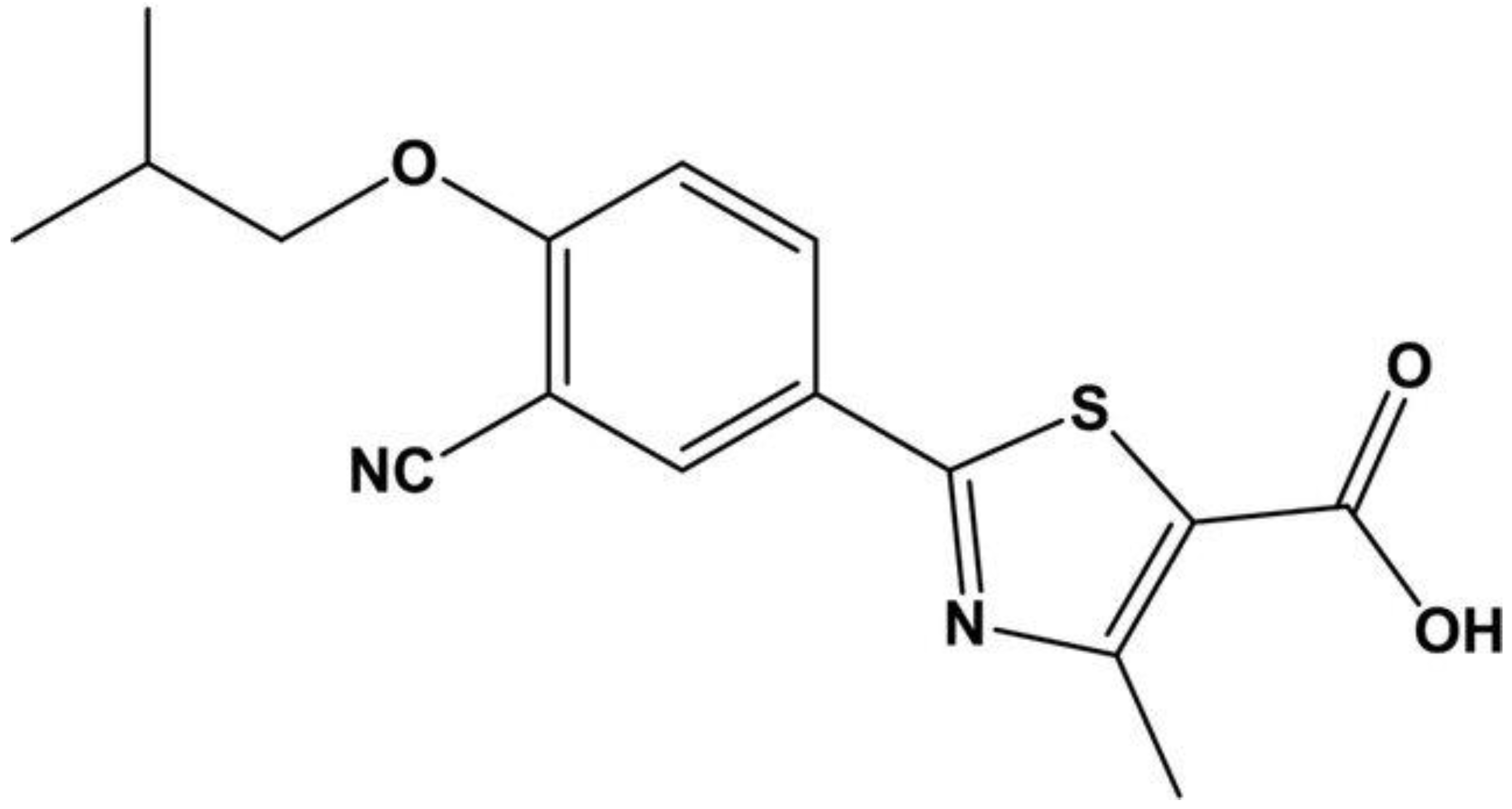
Figure 1. Chemical structure of febuxostat (Kaur, Manpreet et al., “Formulation and in vitro Evaluation of Fast Dissolving Tablets of Febuxostat Using Co-Processed Excipients” [5]).
1.1. Mechanism of Action and Pharmacokinetics
Febuxostat exerts its therapeutic effects through a well-defined mechanism of action and exhibits specific pharmacokinetic properties.
1.1.1. Mechanism of Action
Febuxostat’s primary mechanism of action involves the inhibition of xanthine oxidase, an enzyme crucial in the production of uric acid. By inhibiting xanthine oxidase, febuxostat effectively reduces the conversion of xanthine and hypoxanthine into uric acid. This leads to decreased uric acid levels in the bloodstream, addressing the underlying cause of hyperuricemia, a condition characterized by elevated uric acid levels [4][6].
Febuxostat’s selectivity and potency in inhibiting xanthine oxidase contribute to its effectiveness in reducing uric acid levels. Unlike some older xanthine oxidase inhibitors, such as allopurinol, febuxostat does not rely on dose adjustments based on kidney function, making it a more straightforward option for many patients.
1.1.2. Pharmacokinetics
Febuxostat’s pharmacokinetic properties vary with factors like age, sex, and renal function, necessitating consideration for dosing optimization. Some key pharmacokinetic aspects of febuxostat include:
-
Absorption: febuxostat is well-absorbed after oral administration, with peak plasma concentrations typically reached within 1–1.5 h following ingestion [4].
-
Distribution: The drug has a moderate volume of distribution, indicating its distribution throughout body tissues. It is bound to plasma proteins, primarily albumin [4].
-
Metabolism: Febuxostat undergoes hepatic metabolism, primarily via the cytochrome P450 enzyme system. It is metabolized to inactive metabolites, which are subsequently excreted [6].
-
Excretion: the drug and its metabolites are primarily excreted in the urine, with approximately 49% of the administered dose eliminated as unchanged febuxostat [4].
-
Elimination Half-life: Febuxostat’s elimination half-life varies but is generally in the range of 5–8 h in healthy individuals. This property influences dosing frequency and considerations for patients with renal impairment [6].
-
Dosing Considerations: Dosing adjustments may be necessary based on factors such as age and renal function. Patients with impaired renal function may require lower doses to prevent potential accumulation of the drug [6].
Understanding febuxostat’s pharmacokinetics is essential for healthcare providers to optimize dosing strategies for individual patients. It is particularly crucial to consider factors like age, sex, and renal function when prescribing febuxostat to ensure safe and effective treatment [4][6].
1.2. Efficacy in Gout Management
Febuxostat has proven efficacy in the management of gout, a painful inflammatory arthritis condition characterized by elevated uric acid levels in the blood [7]. By effectively reducing uric acid levels in the body, febuxostat provides relief from gout symptoms and supports long-term gout management [1][2]. Febuxostat’s effectiveness in reducing uric acid levels has been well-documented in clinical trials. It is considered an effective alternative for gout patients who may have contraindications to allopurinol, another common medication for gout [3].
Long-term gout management often involves the use of febuxostat as it supports sustained uric acid control. This is essential to preventing gout flares and the development of tophi, which are urate crystal deposits that can cause joint damage and deformities [3].
However, it is essential to recognize that gout management is not solely about reducing uric acid levels. Lifestyle modifications, dietary changes, and addressing comorbidities also play a crucial role in preventing gout flares and improving overall health.
1.3. Cardiovascular Effects and Safety Profile
Febuxostat has been the subject of an extensive investigation regarding its cardiovascular impact and safety profile [6].
Studies comparing febuxostat to allopurinol, another common medication for gout management, have indicated that febuxostat’s cardiovascular effects are comparable to those of allopurinol. This suggests that febuxostat does not pose a significantly higher cardiovascular risk compared to its counterpart. Furthermore, febuxostat demonstrates a favorable safety profile in various clinical settings. Reports of adverse events associated with febuxostat use are generally low, indicating that it is well-tolerated by many patients. Notably, febuxostat’s safety and tolerability have been confirmed in specific patient populations, including those with comorbidities such as hypertension or diabetes. This suggests that febuxostat may be a suitable choice for gout management in individuals with these common cardiovascular risk factors [8].
In the 2018 article “Cardiovascular Safety of Febuxostat or Allopurinol in Patients with Gout” by White et al., a randomized clinical trial assessed the cardiovascular safety of febuxostat and allopurinol in gout patients with cardiovascular risk factors. The study found that both medications had a similar risk of major cardiovascular events, indicating comparable cardiovascular safety. However, there was a suggestion of a potential increase in all-cause mortality with febuxostat, though this finding was not statistically significant. Gout flare rates did not significantly differ between the two treatment groups. The study’s results emphasize the need for individualized treatment decisions for gout in patients with cardiovascular comorbidities [9].
1.4. Renal Effects and Kidney Function
Febuxostat has garnered interest for its potential impact on renal function and its role in preventing kidney stone formation. Understanding these aspects is crucial for optimizing its clinical utility. Febuxostat exhibits potential nephroprotective effects, particularly in gout patients. It indicates that febuxostat may help prevent renal damage associated with hyperuricemia, which can lead to kidney complications. One significant advantage of febuxostat is its safe and effective use in gout patients with renal impairment.
By preserving renal function and reducing uric acid levels, febuxostat offers potential benefits in preventing kidney stone formation. Hyperuricemia is a known risk factor for kidney stone development, and febuxostat’s role in uric acid reduction may mitigate this risk [10].
A randomized, double-blind, placebo-controlled trial involving 467 patients with stage 3 chronic kidney disease (CKD) and asymptomatic hyperuricemia was conducted by Kimura et al. The goal was to determine if febuxostat, a urate-lowering therapy, could slow CKD progression. The primary outcome was the change in the estimated glomerular filtration rate (eGFR) over 108 weeks. The study found that febuxostat did not significantly impact eGFR decline in the overall group. However, subgroups without proteinuria and lower serum creatinine levels showed potential benefits. Notably, the febuxostat group had a lower incidence of gouty arthritis. In summary, febuxostat did not substantially slow CKD progression in stage 3 CKD patients with asymptomatic hyperuricemia, but some subgroups may benefit [11].
A study by Elsisi aimed to investigate the potential nephroprotective effects of febuxostat, mirtazapine, and their combination in countering gentamicin-induced kidney damage. Nephrotoxicity was induced in study subjects using gentamicin, and various treatments were administered before and during gentamicin exposure. The results demonstrated that both febuxostat and mirtazapine effectively mitigated the biochemical and histopathological changes induced by gentamicin. Moreover, they significantly reduced the renal levels of extracellular signal-regulated protein kinase 1/2 (ERK1/2) and monocyte chemoattractant protein-1 (MCP-1), which are associated with kidney injury. Importantly, the combination of febuxostat and mirtazapine showed a synergistic effect in protecting against gentamicin-induced nephrotoxicity. These findings suggest that nonpurine xanthine oxidase inhibitors like febuxostat, in combination with mirtazapine, may hold promise for future nephroprotective therapies. This novel approach offers potential opportunities for addressing kidney damage caused by nephrotoxic agents like gentamicin [12].
Febuxostat’s impact on kidney function appears to be favorable, but it is essential for healthcare providers to monitor renal parameters during treatment, particularly in patients with pre-existing kidney conditions. Regular kidney function tests can help assess the drug’s safety and effectiveness on an individual basis.
1.5. Liver Function and Hepatic Effects
Febuxostat is generally considered safe and well-tolerated by the liver. In clinical practice, it demonstrates minimal hepatotoxicity, making it a viable option for gout patients with underlying liver conditions. The hepatic effects of febuxostat are generally benign, and the drug does not exhibit significant hepatotoxicity, which is crucial for individuals with liver comorbidities who require uric acid-lowering therapy. Gout patients with liver conditions can benefit from febuxostat’s effectiveness in lowering uric acid levels without compromising liver function. This feature distinguishes it from other gout medications that may pose risks to the liver [6].
2. Emerging Applications for Febuxostat
Recent studies have begun to unveil the broader therapeutic potential of febuxostat beyond its established roles. These emerging applications highlight the versatility of this medication and the promise it holds in various clinical contexts.
2.1. Cardiovascular Health
The cardiovascular implications of febuxostat extend beyond its primary function of managing hyperuricemia and gout. Notably, the medication has demonstrated a favorable cardiovascular safety profile, making it a compelling candidate for further exploration in the field of cardiac care [13].
Numerous studies have ignited interest in the possibility that febuxostat’s multifaceted properties could be harnessed to benefit individuals with various cardiovascular diseases [14]. One of the most intriguing aspects is its potential anti-inflammatory properties. Inflammation plays a pivotal role in the development and progression of conditions like atherosclerosis, heart failure, and other cardiac ailments [15]. Emerging research suggests that febuxostat may possess anti-inflammatory attributes that could offer significant advantages in the management of these cardiovascular disorders.
Atherosclerosis, characterized by the buildup of plaque in the arteries, is a prime example of a condition where inflammation is a key driver [16]. Some studies indicate that febuxostat’s anti-inflammatory properties may help mitigate the inflammatory processes associated with atherosclerosis. By doing so, it could contribute to the stabilization of arterial plaques and the prevention of cardiovascular events such as heart attacks and strokes.
Heart failure, another critical cardiovascular condition, is often accompanied by chronic inflammation [17][18]. The anti-inflammatory potential of febuxostat raises the possibility of managing inflammation in heart failure patients, potentially improving their overall cardiac function and quality of life.
As research in this area advances, future clinical trials are anticipated to provide a more comprehensive understanding of febuxostat’s cardiovascular impact [19]. These trials will play a pivotal role in elucidating whether the medication can be integrated into the existing therapeutic strategies for atherosclerosis, heart failure, and related cardiac conditions [20][21]. The potential for febuxostat to ameliorate inflammation and enhance cardiovascular health represents an exciting frontier in the ongoing pursuit of innovative approaches to cardiac care.
2.2. Neurological Disorders
In recent years, there has been mounting interest in unveiling the potential of febuxostat in the realm of neurological conditions [22]. This interest stems from the medication’s multifaceted properties, including its anti-inflammatory and antioxidative attributes, which have ignited optimism over its neuroprotective capabilities. While the bulk of this research remains in the preclinical phase, the preliminary findings are encouraging and warrant further investigation [23].
Preclinical studies, conducted primarily in animal models, have been instrumental in shedding light on febuxostat’s potential neuroprotective benefits. These studies have ventured into the exploration of its effects on various neurodegenerative diseases, with a particular focus on conditions such as Parkinson’s and Alzheimer’s. The results garnered from these investigations have sparked a surge of interest in advancing to clinical trials to assess the medication’s potential in humans [24].
Parkinson’s disease, a debilitating neurodegenerative disorder, has been a focal point of these preclinical investigations. Animal models of Parkinson’s have offered valuable insights, demonstrating that febuxostat’s anti-inflammatory properties could mitigate the inflammatory processes that contribute to the degeneration of dopaminergic neurons [21]. Additionally, its antioxidative effects may play a crucial role in reducing oxidative stress, a hallmark of Parkinson’s disease [25].
Similarly, research into Alzheimer’s disease has unveiled promising outcomes. The disease is characterized by the accumulation of amyloid-beta plaques and tau tangles in the brain, leading to cognitive decline. Studies in animal models suggest that febuxostat’s anti-inflammatory properties could potentially curtail the neuroinflammation associated with Alzheimer’s. Furthermore, its antioxidative capabilities may help combat the oxidative damage that plays a significant role in disease progression [23][26].
While preclinical studies offer a compelling foundation for further exploration, the translation of these findings to human clinical trials is the next critical step. Clinical trials will be pivotal in determining whether febuxostat can deliver on its neuroprotective potential and provide meaningful benefits to individuals affected by neurological conditions.
2.3. Metabolic Syndrome
Metabolic syndrome, a cluster of conditions including obesity, insulin resistance, and hypertension, represents a significant global health challenge. Some investigations indicate that febuxostat may help mitigate components of metabolic syndrome, possibly through its anti-inflammatory actions and effects on oxidative stress. This area of research holds promise for individuals at risk of metabolic syndrome-related complications [27][28].
A study by Nadwa et al. aimed to compare the effects of febuxostat and allopurinol on metabolic syndrome (MS)-related changes in a rat model. The researchers induced insulin resistance and MS in adult male rats through a high-fructose diet over 8 weeks. They assessed various parameters, including weight, blood pressure, serum biochemistry, and antioxidant enzyme activities. Both febuxostat and allopurinol treatments led to significant improvements, reducing body weight, blood pressure, blood glucose, insulin levels, and lipid profiles, and enhancing kidney function and endothelial integrity compared to untreated rats. Notably, febuxostat was more effective than allopurinol at normalizing fasting glucose, uric acid levels, as well as antioxidant enzyme activities. In conclusion, xanthine oxidase inhibitors, including febuxostat and allopurinol, were found to ameliorate MS-related effects, with febuxostat showing mild superiority in improving various metabolic parameters [11].
2.4. Rheumatoid Arthritis
Rheumatoid arthritis, an autoimmune inflammatory disorder, presents an intriguing arena for febuxostat exploration. Some studies suggest that it may complement existing rheumatoid arthritis treatments by targeting inflammatory pathways. Clinical trials evaluating its role in rheumatoid arthritis management are ongoing, with initial findings suggesting potential efficacy [29][30].
2.5. Cancer Therapy and Oxidative Stress
Emerging evidence suggests that febuxostat’s multifaceted properties extend beyond traditional medical applications, with potential implications in the field of cancer therapy [31]. Notably, febuxostat’s anti-inflammatory and antioxidative properties [32][33] have piqued the interest of researchers in the context of cancer treatment [34]. Early-stage investigations hint at its capacity to enhance the effectiveness of certain cancer treatments while simultaneously mitigating treatment-related inflammation, marking a promising development in the ongoing quest for more effective oncology solutions [35][36].
One study conducted by Fukui et al. [37] delves into the potential impact of febuxostat on oxidative stress in patients. This research encompassed 43 hyperuricemia outpatients, segregated into two groups: one group received febuxostat as a novel treatment, while the other switched from allopurinol to febuxostat. The research evaluated various parameters, including uric acid levels, creatinine levels, estimated glomerular filtration rate, and indicators of oxidative stress, specifically derivatives of reactive oxygen metabolites (d-ROMs) and biological antioxidant potential (BAP). Measurements were taken before and after treatment, offering insights into the medication’s effects.
The results of this research are particularly intriguing. Febuxostat exhibited a significant reduction in uric acid levels, which is essential in the context of hyperuricemia management. Additionally, it yielded a noteworthy reduction in d-ROMs, indicating its potential antioxidative properties. This reduction in oxidative stress markers may have broader implications, especially in conditions where oxidative stress plays a significant role, such as certain types of cancer.
The potential synergy between febuxostat and conventional cancer treatments opens up new avenues in the realm of oncology. By curbing inflammation and addressing oxidative stress [32], febuxostat could improve the overall efficacy of cancer therapies, minimize treatment-related complications, and enhance the quality of life for cancer patients [34]. However, it is crucial to note that this area of research is still in its early stages, and further investigations are warranted to fully understand the scope of febuxostat’s contribution to cancer treatment [34][38].
2.6. Influence of Gender on Uric Acid and Urate-Lowering Therapy
The influence of gender on uric acid and urate-lowering drugs becomes evident through the examination of gout patients with cardiovascular diseases. Cheng et al. reported that the use of febuxostat reveals an increased risk of heart failure (HF) hospitalization, particularly notable among women with gout and heightened cardiovascular risk. This connection highlights the nuanced interplay between sex, gout, and cardiovascular health [39].
Simultaneously, an analysis of clinical trials testing serum uric acid (SUA) lowering drugs reveals a concerning trend of declining enrollment of women over time. This trend varies among different drugs, indicating persistent underrepresentation of women across various classes of SUA-lowering medications. These insights underscore the imperative need to incorporate sex-specific considerations in both real-world settings and clinical trial designs. By doing so, researchers can achieve a more comprehensive understanding of the intricate dynamics surrounding the impact of uric acid and urate-lowering drugs, fostering equitable representation and empowering informed healthcare decisions [40].
References
- Edwards, N.L. Febuxostat: A new treatment for hyperuricaemia in gout. Rheumatology 2009, 48 (Suppl. S2), ii15–ii19.
- Richette, P.; Doherty, M.; Pascual, E.; Barskova, V.; Becce, F.; Castañeda-Sanabria, J.; Coyfish, M.; Guillo, S.; Jansen, T.L.; Janssens, H.; et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann. Rheum. Dis. 2017, 76, 29–42.
- Febuxostat: New drug. Hyperuricaemia: Risk of gout attacks. Prescrire Int. 2009, 18, 63–65.
- Gerriets, V.; Jialal, I. Febuxostat. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023; Available online: https://www.ncbi.nlm.nih.gov/books/NBK544239/ (accessed on 1 October 2023).
- Kaur, M.; Mittal, A.; Gulati, M.; Sharma, D.; Kumar, R. Formulation and in vitro Evaluation of Fast Dissolving Tablets of Febuxostat Using Co-Processed Excipients. Recent Pat. Drug Deliv. Formul. 2020, 14, 48–62.
- Khosravan, R.; Grabowski, B.A.; Wu, J.T.; Joseph-Ridge, N.; Vernillet, L. Pharmacokinetics, pharmacodynamics and safety of febuxostat, a non-purine selective inhibitor of xanthine oxidase, in a dose escalation study in healthy subjects. Clin. Pharmacokinet. 2006, 45, 821–841.
- Ernst, M.E.; Fravel, M.A. Febuxostat: A selective xanthine-oxidase/xanthine-dehydrogenase inhibitor for the management of hyperuricemia in adults with gout. Clin. Ther. 2009, 31, 2503–2518.
- White, W.B.; Saag, K.G.; Becker, M.A.; Borer, J.S.; Gorelick, P.B.; Whelton, A.; Hunt, B.; Castillo, M.; Gunawardhana, L.; CARES Investigators. Cardiovascular Safety of Febuxostat or Allopurinol in Patients with Gout. N. Engl. J. Med. 2018, 378, 1200–1210.
- Kimura, K.; Hosoya, T.; Uchida, S.; Inaba, M.; Makino, H.; Ito, S.; Yamamoto, T.; Tomino, Y.; Ohno, I.; Shibagaki, Y.; et al. Febuxostat Therapy for Patients with Stage 3 CKD and Asymptomatic Hyperuricemia: A Randomized Trial. Am. J. Kidney Dis. 2018, 72, 798–810.
- Abdel-Wahab, B.A.; El-Shoura, E.A.; Habeeb, M.S.; Zafaar, D.K. Febuxostat alleviates Arsenic Trioxide-Induced renal injury in Rats: Insights on the crosstalk between NLRP3/TLR4, Sirt-1/NF-κB/TGF-β signaling Pathways, and miR-23b-3p, miR-181a-5b expression. Biochem. Pharmacol. 2023, 216, 115794.
- Nadwa, E.H.; Morcos, G.N.; Salama, N.M.; Shafik, A.N. Comparing the Effects of Febuxostat and Allopurinol in an Animal Model of Metabolic Syndrome. Pharmacology 2021, 106, 564–572.
- Elsisi, A.E.E.; Sokar, S.S.; Shalaby, M.F.; Abu-Risha, S.E.S. Nephroprotective effects of febuxostat and/or mirtazapine against gentamicin-induced nephrotoxicity through modulation of ERK 1/2, NF-κB and MCP1. Expert Rev. Clin. Pharmacol. 2021, 14, 1039–1050.
- Rashad, A.Y.; Daabees, H.G.; Elagawany, M.; Shahin, M.; Moneim, A.E.A.; Rostom, S.A. Towards the Development of Dual Hypouricemic and Anti-inflammatory Candidates: Design, Synthesis, Stability Studies and Biological Evaluation of Some Mutual Ester Prodrugs of Febuxostat-NSAIDs. Bioorganic Chem. 2023, 135, 106502.
- Feig, D.I.; Kang, D.-H.; Johnson, R.J. Uric acid and cardiovascular risk. N. Engl. J. Med. 2008, 359, 1811–1821.
- Kanbay, M.; Segal, M.; Afsar, B.; Kang, D.H.; Rodriguez-Iturbe, B.; Johnson, R.J. The role of uric acid in the pathogenesis of human cardiovascular disease. Heart 2013, 99, 759–766.
- Bredemeier, M.; Lopes, L.M.; Eisenreich, M.A.; Hickmann, S.; Bongiorno, G.K.; D’Avila, R.; Morsch, A.L.B.; Stein, F.D.S.; Campos, G.G.D. Xanthine oxidase inhibitors for prevention of cardiovascular events: A systematic review and meta-analysis of randomized controlled trials. BMC Cardiovasc. Disord. 2018, 18, 24.
- Grassi, D.; Desideri, G.; Di Giacomantonio, A.V.; Di Giosia, P.; Ferri, C. Hyperuricemia and cardiovascular risk. High Blood Press. Cardiovasc. Prev. 2014, 21, 235–242.
- Borghi, C.; Desideri, G. Urate-Lowering Drugs and Prevention of Cardiovascular Disease: The Emerging Role of Xanthine Oxidase Inhibition. Hypertension 2016, 67, 496–498.
- Borghi, C.; Palazzuoli, A.; Landolfo, M.; Cosentino, E. Hyperuricemia: A novel old disorder—Relationship and potential mechanisms in heart failure. Heart Fail. Rev. 2020, 25, 43–51.
- Ong, S.L.; Vickers, J.J.; Zhang, Y.; McKenzie, K.U.; Walsh, C.E.; Whitworth, J.A. Role of xanthine oxidase in dexamethasone-induced hypertension in rats. Clin. Exp. Pharmacol. Physiol. 2007, 34, 517–519.
- Qu, L.H.; Jiang, H.; Chen, J.H. Effect of uric acid-lowering therapy on blood pressure: Systematic review and meta-analysis. Ann. Med. 2017, 49, 142–156.
- Hong, J.-Y.; Lan, T.-Y.; Tang, G.-J.; Tang, C.-H.; Chen, T.-J.; Lin, H.-Y. Gout and the risk of dementia: A nationwide population-based cohort study. Arthritis Res. Ther. 2015, 17, 139.
- Lu, N.; Dubreuil, M.; Zhang, Y.; Neogi, T.; Rai, S.K.; Ascherio, A.; Hernán, M.A.; Choi, H.K. Gout and the risk of Alzheimer’s disease: A population-based, BMI-matched cohort study. Rheumatol. 2016, 75, 547–551.
- Euser, S.M.; Hofman, A.; Westendorp, R.G.J.; Breteler, M.M.B. Serum uric acid and cognitive function and dementia. Brain 2009, 132, 377–382.
- Singh, J.A.; Cleveland, J.D. Comparative effectiveness of allopurinol versus febuxostat for preventing incident dementia in older adults: A propensity-matched analysis. Arthritis Res. Ther. 2018, 20, 167.
- Mikuls, T.R.; Cheetham, T.C.; Levy, G.D.; Rashid, N.; Kerimian, A.; Low, K.J.; Coburn, B.W.; Redden, D.T.; Saag, K.G.; Foster, P.J.; et al. Adherence and Outcomes with Urate-Lowering Therapy: A Site-Randomized Trial. Am. J. Med. 2019, 132, 354–361.
- Kelley, E.E.; Baust, J.; Bonacci, G.; Golin-Bisello, F.; Devlin, J.E.; Croix, C.M.S.; Watkins, S.C.; Gor, S.; Cantu-Medellin, N.; Weidert, E.R.; et al. Fatty Acid Nitroalkenes Ameliorate Glucose Intolerance and Pulmonary Hypertension in High-Fat Diet-Induced Obesity. Cardiovasc. Res. 2014, 101, 352–363.
- Yisireyili, M.; Hayashi, M.; Wu, H.; Uchida, Y.; Yamamoto, K.; Kikuchi, R.; Hamrah, M.S.; Nakayama, T.; Cheng, X.W.; Matsushita, T.; et al. Xanthine oxidase inhibition by febuxostat attenuates stress-induced hyperuricemia, glucose dysmetabolism, and prothrombotic state in mice. Sci. Rep. 2017, 7, 1266.
- Bou-Salah, L.; Benarous, K.; Linani, A.; Rabhi, F.; Chaib, K.; Chine, I.; Bensaidane, H.; Yousfi, M. Anti-inflammatory drugs as new inhibitors to xanthine oxidase: In vitro and in silico approach. Mol. Cell. Probes 2021, 58, 101733.
- Wang, W.; Pang, J.; Ha, E.H.; Zhou, M.; Li, Z.; Tian, S.; Li, H.; Hu, Q. Development of novel NLRP3-XOD dual inhibitors for the treatment of gout. Bioorganic Med. Chem. Lett. 2020, 30, 126944.
- Ikemura, K.; Hiramatsu, S.-I.; Shinogi, Y.; Nakatani, Y.; Tawara, I.; Iwamoto, T.; Katayama, N.; Okuda, M. Concomitant febuxostat enhances methotrexate-induced hepatotoxicity by inhibiting breast cancer resistance protein. Sci. Rep. 2019, 9, 20359.
- George, J.; Struthers, A.D. Role of urate, xanthine oxidase and the effects of allopurinol in vascular oxidative stress. Vasc. Health Risk Manag. 2009, 5, 265–272.
- Malik, U.Z.; Hundley, N.J.; Romero, G.; Radi, R.; Freeman, B.A.; Tarpey, M.M.; Kelley, E.E. Febuxostat inhibition of endothelial-bound XO: Implications for targeting vascular ROS production. Free. Radic. Biol. Med. 2011, 51, 179–184.
- Lehtisalo, M.; Keskitalo, J.E.; Tornio, A.; Lapatto-Reiniluoto, O.; Deng, F.; Jaatinen, T.; Viinamäki, J.; Neuvonen, M.; Backman, J.T.; Niemi, M. Febuxostat, But Not Allopurinol, Markedly Raises the Plasma Concentrations of the Breast Cancer Resistance Protein Substrate Rosuvastatin. Clin. Transl. Sci. 2020, 13, 1236–1243.
- Krishnamurthy, B.; Rani, N.; Bharti, S.; Golechha, M.; Bhatia, J.; Nag, T.C.; Ray, R.; Arava, S.; Arya, D.S. Febuxostat ameliorates doxorubicin-induced cardiotoxicity in rats. Chem. Interact. 2015, 237, 96–103.
- Zhuge, Z.; Paulo, L.L.; Jahandideh, A.; Brandão, M.C.; Athayde-Filho, P.F.; Lundberg, J.O.; Braga, V.A.; Carlström, M.; Montenegro, M.F. Synthesis and characterization of a novel organic nitrate NDHP: Role of xanthine oxidoreductase-mediated nitric oxide formation. Redox Biol. 2017, 13, 163–169.
- Fukui, T.; Maruyama, M.; Yamauchi, K.; Yoshitaka, S.; Yasuda, T.; Abe, Y. Effects of Febuxostat on Oxidative Stress. Clin. Ther. 2015, 37, 1396–1401.
- Rashad, A.Y.; Daabees, H.G.; Elagawany, M.; Shahin, M.; Moneim, A.E.A.; Rostom, S.A. A New Avenue for Enhanced Treatment of Hyperuricemia and Oxidative Stress: Design, Synthesis and Biological Evaluation of Some Novel Mutual Prodrugs Involving Febuxostat Conjugated with Different Antioxidants. Bioorganic Chem. 2023, 140, 106818.
- Cheng, C.-L.; Yen, C.-T.; Su, C.-C.; Lee, C.-H.; Huang, C.-H.; Yang, Y.-H.K. Sex difference in heart failure risk associated with febuxostat and allopurinol in gout patients. Front. Cardiovasc. Med. 2022, 9, 891606.
- Fogacci, F.; Borghi, C.; Di Micoli, A.; Degli Esposti, D.; Cicero, A.F. Inequalities in enrollment of women and racial minorities in trials testing uric acid lowering drugs. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 3305–3313.
More
Information
Subjects:
Rheumatology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
782
Revisions:
3 times
(View History)
Update Date:
16 Nov 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





