Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | HELOISE BOURIEN | -- | 2577 | 2023-09-05 11:04:03 | | | |
| 2 | Fanny Huang | Meta information modification | 2577 | 2023-09-06 08:55:01 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Bourien, H.; Pircher, C.C.; Guiu, B.; Lamarca, A.; Valle, J.W.; Niger, M.; Edeline, J. Locoregional Treatment in Intrahepatic Cholangiocarcinoma. Encyclopedia. Available online: https://encyclopedia.pub/entry/48813 (accessed on 07 February 2026).
Bourien H, Pircher CC, Guiu B, Lamarca A, Valle JW, Niger M, et al. Locoregional Treatment in Intrahepatic Cholangiocarcinoma. Encyclopedia. Available at: https://encyclopedia.pub/entry/48813. Accessed February 07, 2026.
Bourien, Héloïse, Chiara Carlotta Pircher, Boris Guiu, Angela Lamarca, Juan W Valle, Monica Niger, Julien Edeline. "Locoregional Treatment in Intrahepatic Cholangiocarcinoma" Encyclopedia, https://encyclopedia.pub/entry/48813 (accessed February 07, 2026).
Bourien, H., Pircher, C.C., Guiu, B., Lamarca, A., Valle, J.W., Niger, M., & Edeline, J. (2023, September 05). Locoregional Treatment in Intrahepatic Cholangiocarcinoma. In Encyclopedia. https://encyclopedia.pub/entry/48813
Bourien, Héloïse, et al. "Locoregional Treatment in Intrahepatic Cholangiocarcinoma." Encyclopedia. Web. 05 September, 2023.
Copy Citation
For unresectable intrahepatic cholangiocarcinoma (iCC), different locoregional treatments (LRT) could be proposed to patients, including radiofrequency ablation (RFA) and microwave ablation (MWA), external beam radiotherapy (EBRT) or transarterial treatments, depending on patient and tumor characteristics and local expertise.
cholangiocarcinoma
locoregional treatment
1. Introduction
Biliary tract cancers (BTC) are heterogenous entities comprising intrahepatic cholangiocarcinoma (iCC), perihilar cholangiocarcinoma, distal cholangiocarcinoma, gallbladder cancers and sometimes ampullary cancer. Most patients are diagnosed at an advanced (locally advanced or metastatic) stage. Currently, for these patients, a combination of gemcitabine and cisplatin is recommended in the first-line setting, based on the results of the Advanced Biliary Tract Cancer (ABC)-02 and BT22 trials [1][2]. Recently, durvalumab showed a significant, albeit modest, improvement in overall survival (OS) and was recently granted approval by the FDA [3].
For liver-only iCC, surgeons have to evaluate whether a complete resection, R0, could be performed as surgery is the only potential curative treatment for these patients. Nevertheless, outcomes remain poor, the median OS after curative-intent surgery is about 30 months, and only about a third of patients experience long-term, relapse-free survival (RFS) [4][5][6].
For unresectable liver-only iCC, or for patients not suitable for surgery, radiofrequency ablation (RFA), microwave ablation (MWA) and other locoregional treatments (LRT) like external beam radiotherapy (EBRT) or various transarterial treatments, including transarterial chemoembolization (TACE), radioembolization (also known as selective internal radiation therapy, or SIRT) or hepatic arterial infusion chemotherapy (HAIC), have also been investigated for unresectable non-metastatic iCC.
2. Current Data Regarding Locoregional Treatments of Intrahepatic Cholangiocarcinoma
2.1. Radiofrequency Ablation (RFA) and Microwave Ablation (MWA)
Many studies have reported results of RFA and MWA in iCC. There is no randomized trial evaluating ablative therapy; only prospective cohorts or retrospective studies were published. Studies are heterogenous, which leads to difficulties in cross-trial comparison.
Overall, these studies suggest adequate local control following ablation of iCC. In a recent review, the reported response rate was 93.9% [7].
One of the most important characteristics for using RFA or MWA is the tumor size. In retrospective studies, the tumor size ranged from 0.7 to 10 cm, but the better control of tumors was observed for tumors < 5 cm. For Brandi et al. [8], a tumor size less than 2 cm was an independent factor for improved local tumor progression-free survival. For the cohort of Díaz-González Á [9], the median time to recurrence was significantly lower for a tumor smaller than 2 cm. This correlates well with data from hepatocellular carcinoma (HCC), where RFA is considered equivalent to surgery for tumors up to 2 cm and acceptable for tumors up to 3 cm. Moreover, the ALBI grade and number were independent prognostic factors for ablative therapies in terms of OS [10][11][12]. There is no impact of age on progression-free survival (PFS) or OS after RFA and MWA [13], suggesting that they could be proposed for older patients.
The main reported complications include ascites or pleural effusion, liver abscess, portal vein thrombosis, jaundice and hepatic failure; however, the rate of severe complications is low overall. For Xu et al. [10], the rate of complications of MWA was 5.3%, half that seen with surgery (13.8%). The overall procedure-associated major complication rates range from 2.8% to 5.5% [10][12][13][14]. Kim et al. [15] reported only one complication (liver abscess) out of 13 patients, for a lesion of 7 cm in diameter treated by RFA.
To conclude, RFA and MWA are effective and safe treatments for iCC. They could be proposed as curative-intent treatments in patients deemed unfit for major surgery who have a limited number of lesions (up to 3), lesions under 3 cm, and who have good liver function according to the ALBI score, with or without the presence of cirrhosis (Figure 1A).
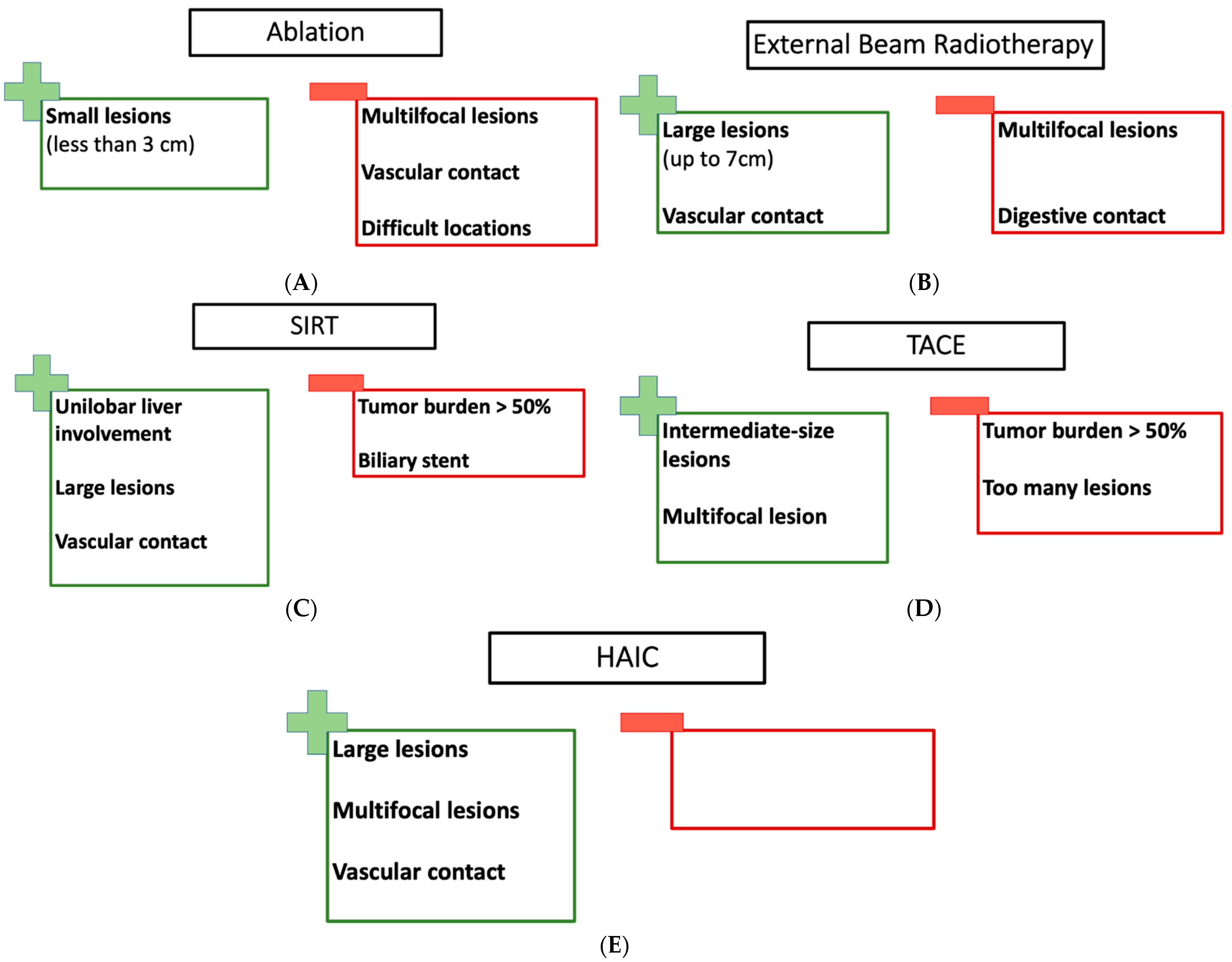
Figure 1. (A) The pros and cons of RFA and MWA; (B) the pros and cons of EBRT; (C) the pros and cons of SIRT; (D) the pros and cons of TACE; (E) the pros and cons of HAIC.
2.2. External Beam Radiotherapy (EBRT)
Several teams evaluated radiotherapy for the management of iCC. Radiotherapy could be proposed in curative or in palliative intent. There is a high heterogeneity concerning dose, schedules or techniques (some used proton therapy, while others used photon therapy or SBRT). Depending on individual studies, the gross tumor volume (GTV) was not the same for liver tumor or regional disease. Moreover, some groups proposed chemotherapy concomitantly [16][17] or sequentially [17][18] with different drugs (5-fluorouracil, gemcitabine, cisplatin, and doxorubicin [17]; erlotinib [19]; or S-1 [20]) administered via hepatic arterial infusion or systemically. Before receiving radiotherapy, patients could have received other treatment, such as surgery, chemotherapy, transarterial treatment, RFA and MWA, or transplantation [21]. One other difficulty in interpreting the data in the literature is the fact that, in some series, the patients included suffered from HCC or iCC, without distinguishing the two entities for toxicity or clinical outcome analyses.
Overall, the literature shows large variations in local control rates and PFS rates, which is probably related to different selections of the population, tumor characteristics, and the technique applied. In the review by Edeline et al. [7], the mean 2-year local control is 69.1% (95% CI: 48.1–90.2).
In the different series of radiation therapy for iCC treatment, grade 3 and higher adverse events were observed in around 10% of patients [16][22][23][24] but can be observed in up to 20% [19]; however, in some series, no grade 3 toxicities were observed [20]. The more frequent grade 3 or higher radiation-related toxicities experienced by patients were cytopenia, gastro-intestinal and hepatic toxicity. Liver failure may occur due to radiation-induced liver disease (RILD) and could be fatal. Depending on the series, RILD was observed in 0 to 7% of patients, but it is rarely fatal. Nevertheless, in a phase I/II study, 2 out of 26 patients (7%) died from liver dysfunction—one probably due to RILD [19]. The likelihood of liver dysfunction and RILD correlates with the volume of spared liver. The addition of chemotherapy to radiotherapy does not seem to increase grade 3 toxicities [17]. The dose of radiation delivered to the hepatobiliary tree seems to be a predictive factor of complication. Shen et al. [25] did not observe toxicities greater than grade 1 for patients with tumors smaller than 5 cm.
To conclude, external beam radiation therapy seems to be a possible treatment for iCC treatment, achieving a satisfactory local control rate for patients with good liver function (Child A or B) and with a tumor size under 5 or 7 cm. EBRT could be proposed for tumors with vascular contact or tumors technically not accessible or too large for RFA or MWA. On one hand, a tumor that received a dose was one of the most important predictive factors of local control; on the other hand, the liver volume spared was correlated with liver dysfunction. Patients need to be carefully selected to obtain the best control rate with the lowest RILD rate. To avoid liver dysfunction and have the best control rate, a tumor size under 7 cm seem to be more suitable [24]. Radiation therapy could be discussed in patients not suitable for surgery, in patients for which RFA or MWA are technically not possible, or in palliative intent (Figure 1B).
2.3. Intra-Arterial Treatment (IAT)
Different intra-arterial treatments could be proposed for iCC, including SIRT, TACE and HAIC [7].
2.3.1. Yttrium-90 Microsphere Selective Internal Radiation Therapy
For cholangiocarcinoma, SIRT indications are not well defined and depend on individual treating centers and their practice. Several groups have reported their retrospective experience of SIRT for the management of patients with cholangiocarcinoma. As with radiotherapy, SIRT may be performed alone or in combination with chemotherapy. In some retrospective series, SIRT has been used as consolidation in patients with no disease progression after first-line chemotherapy and without extrahepatic disease [26]. Only one clinical trial evaluated SIRT with concomitant chemotherapy (n = 41) [27]. Contrary to radiotherapy, chemotherapy used concomitantly with SIRT seems to be standardized and defined by the doublet gemcitabine and cisplatin standard of care regimen [27]. SIRT may be used for downstaging, aiming to render patients suitable for surgery with curative intent or for patients who are in a palliative setting.
There is a large variation in reported efficacy results, probably due to the heterogeneity of the population included. Results in the first-line setting seem promising in the single-arm MISPHEC clinical trial [27] evaluating the activity of gemcitabine-cisplatin with SIRT for patients affected by unresectable iCC with no or limited extrahepatic disease. This strategy permitted a high disease control rate of 98% (95% CI, 80–99%) at 3 months. Of the 41 patients initially considered unresectable, 9 patients (22%) could be downstaged to surgical intervention. Median PFS was 14 months (95% CI, 8–17 months), and median OS was 22 months (95% CI, 14–52 months).
Outside this clinical trial, the majority of patients receiving SIRT were pre-treated (up to 70%), with surgery, TACE, chemotherapy or external beam radiation [28][29]. In most series, patients had extrahepatic metastatic disease (from 1/4 to 1/3 of patients) [29][30][31][32][33][34][35][36].
The aim of SIRT is to deliver a sufficient dose to the tumor in order to induce a local response while sparing healthy liver tissue. A personalized dosimetric approach is important and necessary to select the best candidates for SIRT [37]. Bilobar disease does not permit the delivery of the optimal dose to the tumor in preserving a sufficient hepatic reserve.
A high percentage of liver involvement is frequently associated with poor outcomes [31][38]; therefore, patients with a tumor burden of >50% should not be treated with SIRT. Indeed, the reported median overall survival for patients with a tumor burden > 50% ranged from 1 to 6 months [31][39]. Other prognostic factors were associated with poor outcomes, like previous biliary stenting, a primary location different from iCC, an Eastern Cooperative Oncology Group performance status (ECOG PS) ≥ 1 and progressive tumors after the 1st line of chemotherapy [39].
The most frequent complications occurred in the first 30 days and included gastritis, gastrointestinal ulcerations, radiation cholecystitis and radioembolisation-induced liver disease (REILD). Grade 3 or 4 adverse-related events occurred in less than 10% in most of the series [30][32][33][34][38][40]. This incidence was increased when chemotherapy was given alongside SIRT. In the phase II trial MISPHEC [27], 71% of patients experienced grade 3 or 4 adverse events; hematologic toxicity was prevalent and was probably due to the chemotherapy. Two treatment-related deaths were reported in the 30 days following SIRT for patients with an extrahepatic disease and a PS of 2 [36]. Permanent liver toxicity was associated with the presence of cirrhosis and Child–Pugh score (A6 and B7) [37].
In conclusion, for well-selected patients with unilobar liver involvement, good liver function and a good performance status (PS 0 or 1), SIRT provides promising clinical outcomes. Its preferred indication is in the pre-surgical treatment setting in order to enable surgery in patients with initially unresectable cholangiocarcinoma (Figure 1C). Combination chemotherapy (gemcitabine-cisplatin) with SIRT led to a conversion rate (to resection) of 20% in a well-selected population [27][39].
2.3.2. Transarterial Chemoembolization (TACE)
Several groups have proposed chemoembolization as an LRT for patients with unresectable cholangiocarcinoma. Most of the studies reported to date were retrospective, and the number of patients in each of the studies was small. There was no standardization regarding the chemotherapy drugs used, so data were heterogeneous. In some cases, systemic chemotherapy was also administered concomitantly [41][42][43][44].
Researchers found only one randomized clinical trial (n = 48) that evaluated TACE alongside systemic chemotherapy versus chemotherapy alone [44], with an improvement of PFS of 20 months in the combination arm. The phase 2 trial by Martin et al. [44] reported an improved median overall survival (mOS) for patients in favor of GEMCIS + TACE versus TACE alone (mOS: (33.7 (95% CI 13.5–54.5) months versus 12.6 (95% CI 8.7–33.4) months, p = 0.048)). In a carefully selected population, it could enhance median overall survival (mOS) compared to supportive care [45].
Patients with Child–Pugh B liver function, a PS ≥ 1, a hypovascular tumor, a tumor size larger than 5 cm or a multifocal tumor have the poorest clinical outcomes [46][47][48].
TACE procedures were generally well tolerated with predominant gastro-intestinal (nausea, vomiting and abdominal pain), general (fewer) and hepatic (elevation of transaminases) toxicities and grade 1–2 adverse effects (AE) [49]. In most cases, grade 3–4 AEs were in around 10% of patients [41][48] but could reach 1/4 of patients [45]; principally, GI toxicities or hematological toxicities were seen, but renal failure was as well. If chemotherapy was also given, the rate could reach 1/3 [44], but toxicities were predominantly due to intra-venous chemotherapy. Grade 4 or 5 cardiac toxicities were rare but reported in different series [50][51]. In a series of 18 patients, two adverse fatal events were reported: one due to myocardial infarction and one due to sepsis from biliary abscesses [50]. One hepatorenal death was due to the procedure [42].
In conclusion, for multifocal lesions with a tumor burden < 75 or 50% and in patients with PS ≤ 2 and good liver function (Child-Pugh A5–6 or B7), chemoembolization seems to permit good control of local tumor growth (Figure 1D).
2.3.3. Hepatic Arterial Infusion Chemotherapy (HAIC)
HAIC has been less studied than TACE or SIRT. One of the reasons is probably the necessity for a catheter implementation to access the hepatic artery, sometimes with a placement of a pump, which requires specialist expertise and more challenging logistics than other locoregional options. Some of the series identified were prospective clinical trials, but most studies still had a limited number of patients. In some series or clinical trials, HAIC was given with concomitant systemic chemotherapy. In contrast to some of the earlier techniques, large tumors and multifocal disease can be treated by HAIC.
Two phase I and I/II clinical trials evaluated HAIC prospectively. In the phase I/II trial, 29 patients were treated with hepatic arterial infusion using gemcitabine without systemic treatment. This treatment was well tolerated but not as effective as expected, with a tumor response rate of 7% [52]. In the phase II study, 38 patients received concomitantly HAIC floxuridine and systemic gemcitabine and oxaliplatine. They observed an encouraging response rate of 58%. The median OS was 25.0 months (95% CI, 20.6-not reached), and the median PFS was 11.8 months (one-sided 90% CI, 11.1) [53].
Frequent toxicities observed with HAIC were predominantly elevated liver enzymes and gastro-intestinal toxicities (abdominal pain and nausea) or hematological toxicities, but they were also caused by the catheter placement: extravasation, obstruction or damage of the catheter [52][53].
The most frequent grade 4 relative toxicities that occurred in 4–10% of patients [53][54][55] were infection in the pump pocket, artery aneurysms or portal hypertension.
In conclusion, small prospective trials support the activity of HAIC in iCC. The technique may be most appropriate in the setting of large and/or multifocal tumors. Fewer centers are experienced in the technique (Figure 1E).
2.3.4. Comparisons of the Different Intra-Arterial Therapies
For LRT, IAT could be used in a palliative setting or in a neo adjuvant setting in order to downsize initially unresectable tumors to resection.
There are no randomized clinical trials comparing all the LRT modalities to help clinicians in decision making; nevertheless, based on the available retrospective series and clinical trials, researchers have derived some recommendations that may be applicable in daily practice.
Patients with extrahepatic disease were included in many of the studies, but these patients had unfavorable outcomes; therefore, these patients had to be treated with a systemic treatment. Cirrhotic patients could benefit from LRT but only with preserved liver function and a Child-Pugh score of A.
2.4. Existing Guidelines
For unresectable iCC, LRTs are included in the proposed treatment options; however, there are no clear recommendations for choosing between the different modalities, and there are no specified criteria to help clinicians choose the best option [56].
2.4.1. NCCN (National Comprehensive Cancer Network)
For unresectable iCC, NCCN recommendations [57] propose LRTs as an option. Concomitantly to radiotherapy, the network proposed a chemotherapy with fluoropyrimidine. There is no specific chapter for intra-arterial therapy in iCC, but it proposed the same modalities as in HCC.
2.4.2. ESMO (European Society for Medical Oncology)
In the curative setting, the ESMO guidelines only recommend surgery [58]. For unresectable non-metastatic cholangiocarcinoma, IAT could be proposed as an option and in combination with systemic chemotherapy, but there are no details on which technique is preferred or which criteria would be optimal for a patient or tumor.
Radiotherapy is included as an option for localized disease, and for unresectable iCC, SIRT is also an acceptable option.
References
- Valle, J.; Wasan, H.; Palmer, D.H.; Cunningham, D.; Anthoney, A.; Maraveyas, A.; Madhusudan, S.; Iveson, T.; Hughes, S.; Pereira, S.P.; et al. Cisplatin plus Gemcitabine versus Gemcitabine for Biliary Tract Cancer. N. Engl. J. Med. 2010, 362, 1273–1281.
- Okusaka, T.; Nakachi, K.; Fukutomi, A.; Mizuno, N.; Ohkawa, S.; Funakoshi, A.; Nagino, M.; Kondo, S.; Nagaoka, S.; Funai, J.; et al. Gemcitabine alone or in combination with cisplatin in patients with biliary tract cancer: A comparative multicentre study in Japan. Br. J. Cancer 2010, 103, 469–474.
- Oh, D.-Y.; Ruth, H.A.; Qin, S.; Chen, L.-T.; Okusaka, T.; Vogel, A.; Kim, J.W.; Suksombooncharoen, T.; Ah, L.M.; Kitano, M.; et al. Durvalumab plus Gemcitabine and Cisplatin in Advanced Biliary Tract Cancer. NEJM Evid. 2022, 1, EVIDoa2200015.
- Groot Koerkamp, B.; Fong, Y. Outcomes in biliary malignancy. J. Surg. Oncol. 2014, 110, 585–591.
- Cillo, U.; Fondevila, C.; Donadon, M.; Gringeri, E.; Mocchegiani, F.; Schlitt, H.J.; Ijzermans, J.N.M.; Vivarelli, M.; Zieniewicz, K.; Olde Damink, S.W.M.; et al. Surgery for cholangiocarcinoma. Liver Int. 2019, 39 (Suppl. S1), 143–155.
- Bridgewater, J.; Fletcher, P.; Palmer, D.H.; Malik, H.Z.; Prasad, R.; Mirza, D.; Anthony, A.; Corrie, P.; Falk, S.; Finch-Jones, M.; et al. Long-Term Outcomes and Exploratory Analyses of the Randomized Phase III BILCAP Study. J. Clin. Oncol. 2022, 40, 2048–2057.
- Edeline, J.; Lamarca, A.; McNamara, M.G.; Jacobs, T.; Hubner, R.A.; Palmer, D.; Groot Koerkamp, B.; Johnson, P.; Guiu, B.; Valle, J.W. Locoregional therapies in patients with intrahepatic cholangiocarcinoma: A systematic review and pooled analysis. Cancer Treat. Rev. 2021, 99, 102258.
- Brandi, G.; Rizzo, A.; Dall’Olio, F.G.; Felicani, C.; Ercolani, G.; Cescon, M.; Frega, G.; Tavolari, S.; Palloni, A.; De Lorenzo, S.; et al. Percutaneous radiofrequency ablation in intrahepatic cholangiocarcinoma: A retrospective single-center experience. Int. J. Hyperth. 2020, 37, 479–485.
- Díaz-González, Á.; Vilana, R.; Bianchi, L.; García-Criado, Á.; Rimola, J.; Rodríguez de Lope, C.; Ferrer, J.; Ayuso, C.; Da Fonseca, L.G.; Reig, M.; et al. Thermal Ablation for Intrahepatic Cholangiocarcinoma in Cirrhosis: Safety and Efficacy in Non-Surgical Patients. J. Vasc. Interv. Radiol. 2020, 31, 710–719.
- Xu, C.; Li, L.; Xu, W.; Du, C.; Yang, L.; Tong, J.; Yi, Y. Ultrasound-guided percutaneous microwave ablation versus surgical resection for recurrent intrahepatic cholangiocarcinoma: Intermediate-term results. Int. J. Hyperth. 2019, 36, 351–358.
- Ni, J.-Y.; An, C.; Zhang, T.-Q.; Huang, Z.-M.; Jiang, X.-Y.; Huang, J.-H. Predictive value of the albumin-bilirubin grade on long-term outcomes of CT-guided percutaneous microwave ablation in intrahepatic cholangiocarcinoma. Int. J. Hyperth. 2019, 36, 328–336.
- Zhang, S.-J.; Hu, P.; Wang, N.; Shen, Q.; Sun, A.-X.; Kuang, M.; Qian, G.-J. Thermal ablation versus repeated hepatic resection for recurrent intrahepatic cholangiocarcinoma. Ann. Surg. Oncol. 2013, 20, 3596–3602.
- Zhang, K.; Yu, J.; Yu, X.; Han, Z.; Cheng, Z.; Liu, F.; Liang, P. Clinical and survival outcomes of percutaneous microwave ablation for intrahepatic cholangiocarcinoma. Int. J. Hyperth. 2018, 34, 292–297.
- Xu, H.-X.; Wang, Y.; Lu, M.-D.; Liu, L.-N. Percutaneous ultrasound-guided thermal ablation for intrahepatic cholangiocarcinoma. Br. J. Radiol. 2012, 85, 1078–1084.
- Kim, J.H.; Won, H.J.; Shin, Y.M.; Kim, K.-A.; Kim, P.N. Radiofrequency ablation for the treatment of primary intrahepatic cholangiocarcinoma. AJR Am. J. Roentgenol. 2011, 196, W205–W209.
- Shimizu, S.; Okumura, T.; Oshiro, Y.; Fukumitsu, N.; Fukuda, K.; Ishige, K.; Hasegawa, N.; Numajiri, H.; Murofushi, K.; Ohnishi, K.; et al. Clinical outcomes of previously untreated patients with unresectable intrahepatic cholangiocarcinoma following proton beam therapy. Radiat. Oncol. 2019, 14, 241.
- Cho, Y.; Kim, T.H.; Seong, J. Improved oncologic outcome with chemoradiotherapy followed by surgery in unresectable intrahepatic cholangiocarcinoma. Strahlenther. Onkol. 2017, 193, 620–629.
- Kozak, M.M.; Toesca, D.A.S.; von Eyben, R.; Pollom, E.L.; Chang, D.T. Stereotactic Body Radiation Therapy for Cholangiocarcinoma: Optimizing Locoregional Control With Elective Nodal Irradiation. Adv. Radiat. Oncol. 2020, 5, 77–84.
- Weiner, A.A.; Olsen, J.; Ma, D.; Dyk, P.; DeWees, T.; Myerson, R.J.; Parikh, P. Stereotactic body radiotherapy for primary hepatic malignancies—Report of a phase I/II institutional study. Radiother. Oncol. 2016, 121, 79–85.
- Ohkawa, A.; Mizumoto, M.; Ishikawa, H.; Abei, M.; Fukuda, K.; Hashimoto, T.; Sakae, T.; Tsuboi, K.; Okumura, T.; Sakurai, H. Proton beam therapy for unresectable intrahepatic cholangiocarcinoma. J. Gastroenterol. Hepatol. 2015, 30, 957–963.
- Hong, T.S.; Wo, J.Y.; Yeap, B.Y.; Ben-Josef, E.; McDonnell, E.I.; Blaszkowsky, L.S.; Kwak, E.L.; Allen, J.N.; Clark, J.W.; Goyal, L.; et al. Multi-Institutional Phase II Study of High-Dose Hypofractionated Proton Beam Therapy in Patients With Localized, Unresectable Hepatocellular Carcinoma and Intrahepatic Cholangiocarcinoma. J. Clin. Oncol. 2016, 34, 460–468.
- Smart, A.C.; Goyal, L.; Horick, N.; Petkovska, N.; Zhu, A.X.; Ferrone, C.R.; Tanabe, K.K.; Allen, J.N.; Drapek, L.C.; Qadan, M.; et al. Hypofractionated Radiation Therapy for Unresectable/Locally Recurrent Intrahepatic Cholangiocarcinoma. Ann. Surg. Oncol. 2020, 27, 1122–1129.
- Jung, D.H.; Kim, M.-S.; Cho, C.K.; Yoo, H.J.; Jang, W.I.; Seo, Y.S.; Paik, E.K.; Kim, K.B.; Han, C.J.; Kim, S.B. Outcomes of stereotactic body radiotherapy for unresectable primary or recurrent cholangiocarcinoma. Radiat. Oncol. J. 2014, 32, 163–169.
- Tse, R.V.; Hawkins, M.; Lockwood, G.; Kim, J.J.; Cummings, B.; Knox, J.; Sherman, M.; Dawson, L.A. Phase I study of individualized stereotactic body radiotherapy for hepatocellular carcinoma and intrahepatic cholangiocarcinoma. J. Clin. Oncol. 2008, 26, 657–664.
- Shen, Z.-T.; Zhou, H.; Li, A.-M.; Li, B.; Shen, J.-S.; Zhu, X.-X. Clinical outcomes and prognostic factors of stereotactic body radiation therapy for intrahepatic cholangiocarcinoma. Oncotarget 2017, 8, 93541–93550.
- Ibrahim, S.M.; Mulcahy, M.F.; Lewandowski, R.J.; Sato, K.T.; Ryu, R.K.; Masterson, E.J.; Newman, S.B.; Benson, A.; Omary, R.A.; Salem, R. Treatment of unresectable cholangiocarcinoma using yttrium-90 microspheres: Results from a pilot study. Cancer 2008, 113, 2119–2128.
- Edeline, J.; Touchefeu, Y.; Guiu, B.; Farge, O.; Tougeron, D.; Baumgaertner, I.; Ayav, A.; Campillo-Gimenez, B.; Beuzit, L.; Pracht, M.; et al. Radioembolization Plus Chemotherapy for First-line Treatment of Locally Advanced Intrahepatic Cholangiocarcinoma: A Phase 2 Clinical Trial. JAMA Oncol. 2020, 6, 51–59.
- Azar, A.; Devcic, Z.; Paz-Fumagalli, R.; Vidal, L.L.C.; McKinney, J.M.; Frey, G.; Lewis, A.R.; Ritchie, C.; Starr, J.S.; Mody, K.; et al. Albumin-bilirubin grade as a prognostic indicator for patients with non-hepatocellular primary and metastatic liver malignancy undergoing Yttrium-90 radioembolization using resin microspheres. J. Gastrointest. Oncol. 2020, 11, 715–723.
- Swinburne, N.C.; Biederman, D.M.; Besa, C.; Tabori, N.E.; Fischman, A.M.; Patel, R.S.; Nowakowski, F.S.; Gunasekaran, G.; Schwartz, M.E.; Lookstein, R.A.; et al. Radioembolization for Unresectable Intrahepatic Cholangiocarcinoma: Review of Safety, Response Evaluation Criteria in Solid Tumors 1.1 Imaging Response and Survival. Cancer Biother. Radiopharm. 2017, 32, 161–168.
- Helmberger, T.; Golfieri, R.; Pech, M.; Pfammatter, T.; Arnold, D.; Cianni, R.; Maleux, G.; Munneke, G.; Pellerin, O.; Peynircioglu, B.; et al. Clinical Application of Trans-Arterial Radioembolization in Hepatic Malignancies in Europe: First Results from the Prospective Multicentre Observational Study CIRSE Registry for SIR-Spheres Therapy (CIRT). Cardiovasc. Interv. Radiol. 2021, 44, 21–35.
- Bargellini, I.; Mosconi, C.; Pizzi, G.; Lorenzoni, G.; Vivaldi, C.; Cappelli, A.; Vallati, G.E.; Boni, G.; Cappelli, F.; Paladini, A.; et al. Yttrium-90 Radioembolization in Unresectable Intrahepatic Cholangiocarcinoma: Results of a Multicenter Retrospective Study. Cardiovasc. Interv. Radiol. 2020, 43, 1305–1314.
- Buettner, S.; Braat, A.J.A.T.; Margonis, G.A.; Brown, D.B.; Taylor, K.B.; Borgmann, A.J.; Kappadath, S.C.; Mahvash, A.; IJzermans, J.N.M.; Weiss, M.J.; et al. Yttrium-90 Radioembolization in Intrahepatic Cholangiocarcinoma: A Multicenter Retrospective Analysis. J. Vasc. Interv. Radiol. 2020, 31, 1035–1043.e2.
- White, J.; Carolan-Rees, G.; Dale, M.; Patrick, H.E.; See, T.C.; Bell, J.K.; Manas, D.M.; Crellin, A.; Slevin, N.J.; Sharma, R.A. Yttrium-90 Transarterial Radioembolization for Chemotherapy-Refractory Intrahepatic Cholangiocarcinoma: A Prospective, Observational Study. J. Vasc. Interv. Radiol. 2019, 30, 1185–1192.
- Shaker, T.M.; Chung, C.; Varma, M.K.; Doherty, M.G.; Wolf, A.M.; Chung, M.H.; Assifi, M.M. Is there a role for Ytrrium-90 in the treatment of unresectable and metastatic intrahepatic cholangiocarcinoma? Am. J. Surg. 2018, 215, 467–470.
- Soydal, C.; Kucuk, O.N.; Bilgic, S.; Ibis, E. Radioembolization with (90)Y resin microspheres for intrahepatic cholangiocellular carcinoma: Prognostic factors. Ann. Nucl. Med. 2016, 30, 29–34.
- Saxena, A.; Bester, L.; Chua, T.C.; Chu, F.C.; Morris, D.L. Yttrium-90 radiotherapy for unresectable intrahepatic cholangiocarcinoma: A preliminary assessment of this novel treatment option. Ann. Surg. Oncol. 2010, 17, 484–491.
- Bourien, H.; Palard, X.; Rolland, Y.; Le Du, F.; Beuzit, L.; Uguen, T.; Le Sourd, S.; Pracht, M.; Manceau, V.; Lièvre, A.; et al. Yttrium-90 glass microspheres radioembolization (RE) for biliary tract cancer: A large single-center experience. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 669–676.
- Reimer, P.; Virarkar, M.K.; Binnenhei, M.; Justinger, M.; Schön, M.R.; Tatsch, K. Prognostic Factors in Overall Survival of Patients with Unresectable Intrahepatic Cholangiocarcinoma Treated by Means of Yttrium-90 Radioembolization: Results in Therapy-Naïve Patients. Cardiovasc. Interv. Radiol. 2018, 41, 744–752.
- Mouli, S.; Memon, K.; Baker, T.; Benson, A.B.; Mulcahy, M.F.; Gupta, R.; Ryu, R.K.; Salem, R.; Lewandowski, R.J. Yttrium-90 radioembolization for intrahepatic cholangiocarcinoma: Safety, response, and survival analysis. J. Vasc. Interv. Radiol. 2013, 24, 1227–1234.
- Gangi, A.; Shah, J.; Hatfield, N.; Smith, J.; Sweeney, J.; Choi, J.; El-Haddad, G.; Biebel, B.; Parikh, N.; Arslan, B.; et al. Intrahepatic Cholangiocarcinoma Treated with Transarterial Yttrium-90 Glass Microsphere Radioembolization: Results of a Single Institution Retrospective Study. J. Vasc. Interv. Radiol. 2018, 29, 1101–1108.
- Akinwande, O.; Shah, V.; Mills, A.; Noda, C.; Weiner, E.; Foltz, G.; Saad, N. Chemoembolization versus radioembolization for the treatment of unresectable intrahepatic cholangiocarcinoma in a single institution image-based efficacy and comparative toxicity. Hepat. Oncol. 2017, 4, 75–81.
- Schiffman, S.C.; Metzger, T.; Dubel, G.; Andrasina, T.; Kralj, I.; Tatum, C.; McMasters, K.M.; Scoggins, C.R.; Martin, R.C.G. Precision hepatic arterial irinotecan therapy in the treatment of unresectable intrahepatic cholangiocellular carcinoma: Optimal tolerance and prolonged overall survival. Ann. Surg. Oncol. 2011, 18, 431–438.
- Wright, G.P.; Perkins, S.; Jones, H.; Zureikat, A.H.; Marsh, J.W.; Holtzman, M.P.; Zeh, H.J.; Bartlett, D.L.; Pingpank, J.F. Surgical Resection Does Not Improve Survival in Multifocal Intrahepatic Cholangiocarcinoma: A Comparison of Surgical Resection with Intra-Arterial Therapies. Ann. Surg. Oncol. 2018, 25, 83–90.
- Martin, R.C.G.; Simo, K.A.; Hansen, P.; Rocha, F.; Philips, P.; McMasters, K.M.; Tatum, C.M.; Kelly, L.R.; Driscoll, M.; Sharma, V.R.; et al. Drug-Eluting Bead, Irinotecan Therapy of Unresectable Intrahepatic Cholangiocarcinoma (DELTIC) with Concomitant Systemic Gemcitabine and Cisplatin. Ann. Surg. Oncol. 2022, 29, 5462–5473.
- Park, S.-Y.; Kim, J.H.; Yoon, H.-J.; Lee, I.-S.; Yoon, H.-K.; Kim, K.-P. Transarterial chemoembolization versus supportive therapy in the palliative treatment of unresectable intrahepatic cholangiocarcinoma. Clin. Radiol. 2011, 66, 322–328.
- Ge, Y.; Jeong, S.; Luo, G.-J.; Ren, Y.-B.; Zhang, B.-H.; Zhang, Y.-J.; Shen, F.; Cheng, Q.-B.; Sui, C.-J.; Wang, H.-Y.; et al. Transarterial chemoembolization versus percutaneous microwave coagulation therapy for recurrent unresectable intrahepatic cholangiocarcinoma: Development of a prognostic nomogram. Hepatobiliary Pancreat. Dis. Int. 2020, 19, 138–146.
- Vogl, T.J.; Naguib, N.N.N.; Nour-Eldin, N.-E.A.; Bechstein, W.O.; Zeuzem, S.; Trojan, J.; Gruber-Rouh, T. Transarterial chemoembolization in the treatment of patients with unresectable cholangiocarcinoma: Results and prognostic factors governing treatment success. Int. J. Cancer 2012, 131, 733–740.
- Hyder, O.; Marsh, J.W.; Salem, R.; Petre, E.N.; Kalva, S.; Liapi, E.; Cosgrove, D.; Neal, D.; Kamel, I.; Zhu, A.X.; et al. Intra-arterial therapy for advanced intrahepatic cholangiocarcinoma: A multi-institutional analysis. Ann. Surg. Oncol. 2013, 20, 3779–3786.
- Zhou, T.-Y.; Zhou, G.-H.; Zhang, Y.-L.; Nie, C.-H.; Zhu, T.-Y.; Wang, H.-L.; Chen, S.-Q.; Wang, B.-Q.; Yu, Z.-N.; Wu, L.-M.; et al. Drug-eluting beads transarterial chemoembolization with CalliSpheres microspheres for treatment of unresectable intrahepatic cholangiocarcinoma. J. Cancer 2020, 11, 4534–4541.
- Goerg, F.; Zimmermann, M.; Bruners, P.; Neumann, U.; Luedde, T.; Kuhl, C. Chemoembolization with Degradable Starch Microspheres for Treatment of Patients with Primary or Recurrent Unresectable, Locally Advanced Intrahepatic Cholangiocarcinoma: A Pilot Study. Cardiovasc. Interv. Radiol. 2019, 42, 1709–1717.
- Gusani, N.J.; Balaa, F.K.; Steel, J.L.; Geller, D.A.; Marsh, J.W.; Zajko, A.B.; Carr, B.I.; Gamblin, T.C. Treatment of unresectable cholangiocarcinoma with gemcitabine-based transcatheter arterial chemoembolization (TACE): A single-institution experience. J. Gastrointest. Surg. 2008, 12, 129–137.
- Inaba, Y.; Arai, Y.; Yamaura, H.; Sato, Y.; Najima, M.; Aramaki, T.; Sone, M.; Kumada, T.; Tanigawa, N.; Anai, H.; et al. Phase I/II study of hepatic arterial infusion chemotherapy with gemcitabine in patients with unresectable intrahepatic cholangiocarcinoma (JIVROSG-0301). Am. J. Clin. Oncol. 2011, 34, 58–62.
- Cercek, A.; Boerner, T.; Tan, B.R.; Chou, J.F.; Gönen, M.; Boucher, T.M.; Hauser, H.F.; Do, R.K.G.; Lowery, M.A.; Harding, J.J.; et al. Assessment of Hepatic Arterial Infusion of Floxuridine in Combination With Systemic Gemcitabine and Oxaliplatin in Patients With Unresectable Intrahepatic Cholangiocarcinoma: A Phase 2 Clinical Trial. JAMA Oncol. 2020, 6, 60–67.
- Higaki, T.; Aramaki, O.; Moriguchi, M.; Nakayama, H.; Midorikawa, Y.; Takayama, T. Arterial infusion of cisplatin plus S-1 against unresectable intrahepatic cholangiocarcinoma. Biosci. Trends 2018, 12, 73–78.
- Kasai, K.; Kooka, Y.; Suzuki, Y.; Suzuki, A.; Oikawa, T.; Ushio, A.; Kasai, Y.; Sawara, K.; Miyamoto, Y.; Oikawa, K.; et al. Efficacy of hepatic arterial infusion chemotherapy using 5-fluorouracil and systemic pegylated interferon α-2b for advanced intrahepatic cholangiocarcinoma. Ann. Surg. Oncol. 2014, 21, 3638–3645.
- Banales, J.M.; Cardinale, V.; Carpino, G.; Marzioni, M.; Andersen, J.B.; Invernizzi, P.; Lind, G.E.; Folseraas, T.; Forbes, S.J.; Fouassier, L.; et al. Expert consensus document: Cholangiocarcinoma: Current knowledge and future perspectives consensus statement from the European Network for the Study of Cholangiocarcinoma (ENS-CCA). Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 261–280.
- National Comprehensive Cancer Network® (NCCN®). NCCN Guidelines for Patients Gallbladder and Bile Duct Cancers; National Comprehensive Cancer Network® (NCCN®): Stanford, CA, USA, 2021; p. 102.
- Vogel, A.; Bridgewater, J.; Edeline, J.; Kelley, R.K.; Klümpen, H.J.; Malka, D.; Primrose, J.N.; Rimassa, L.; Stenzinger, A.; Valle, J.W.; et al. Biliary tract cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 2022, 34, 127–140.
More
Information
Subjects:
Oncology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
417
Revisions:
2 times
(View History)
Update Date:
06 Sep 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




