Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Karani S Vimal | -- | 1146 | 2023-07-13 11:45:15 | | | |
| 2 | Dean Liu | -7 word(s) | 1139 | 2023-07-14 03:38:12 | | | | |
| 3 | Dean Liu | -1 word(s) | 1138 | 2023-07-31 08:23:35 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Sekar, P.; Ventura, E.F.; Dhanapal, A.C.T.A.; Cheah, E.S.G.; Loganathan, A.; Quen, P.L.; Appukutty, M.; Taslim, N.A.; Hardinsyah, H.; Md Noh, M.F.; et al. Gene–Diet Interactions on Metabolic Disease-Related Outcomes. Encyclopedia. Available online: https://encyclopedia.pub/entry/46752 (accessed on 19 May 2026).
Sekar P, Ventura EF, Dhanapal ACTA, Cheah ESG, Loganathan A, Quen PL, et al. Gene–Diet Interactions on Metabolic Disease-Related Outcomes. Encyclopedia. Available at: https://encyclopedia.pub/entry/46752. Accessed May 19, 2026.
Sekar, Padmini, Eduard Flores Ventura, Anto Cordelia T. A. Dhanapal, Eddy Seong Guan Cheah, Annaletchumy Loganathan, Phoon Lee Quen, Mahenderan Appukutty, Nurpudji Astuti Taslim, Hardinsyah Hardinsyah, Mohd Fairulnizal Md Noh, et al. "Gene–Diet Interactions on Metabolic Disease-Related Outcomes" Encyclopedia, https://encyclopedia.pub/entry/46752 (accessed May 19, 2026).
Sekar, P., Ventura, E.F., Dhanapal, A.C.T.A., Cheah, E.S.G., Loganathan, A., Quen, P.L., Appukutty, M., Taslim, N.A., Hardinsyah, H., Md Noh, M.F., Lovegrove, J.A., Givens, I., & Vimaleswaran, K.S. (2023, July 13). Gene–Diet Interactions on Metabolic Disease-Related Outcomes. In Encyclopedia. https://encyclopedia.pub/entry/46752
Sekar, Padmini, et al. "Gene–Diet Interactions on Metabolic Disease-Related Outcomes." Encyclopedia. Web. 13 July, 2023.
Copy Citation
Diabetes and obesity are chronic diseases that are a burden to low- and middle-income countries.
systematic review
nutrigenetics
Southeast Asia
genetics
gene–diet interaction
1. Introduction
Metabolic diseases such as obesity and diabetes are now considered epidemics rapidly spreading across developed and developing countries affecting both sexes, age, ethnicities, and socioeconomic groups [1]. This, in turn, has shown to compromise the quality of life that leads to potentially life-threatening conditions such as cancers, cardiovascular diseases, musculoskeletal disorders, and hypertension [2]. According to the 2021 reports from the World Health Organisation (WHO), worldwide obesity has tripled since 1975, with over 650 million obese adults and more than 340 million children and adolescents who are either overweight or obese [3]. By 2022, diabetes reports from WHO indicate that more than 422 million people are diabetic with rising prevalence in low-middle income group countries (LMIC) compared to developed countries [4]. Projected trends also show that diabetes and obesity are rapidly growing and will affect nearly two-thirds of the Southeast (SE) Asian population by the end of 2030, placing a burden on rural and low socioeconomic groups [5][6]. Recent reports from the Association of Southeast Asian Nations (ASEAN) show that the tripling rate of undernutrition has not improved and that obesity and diabetes are now a double burden for these countries [7][8].
Understanding gene–nutrient interactions provide insights regarding nutritional, genetic, and biochemical determinants to better understand complex interactions between environmental factors (including diet) and genes relevant to metabolic health and dis-eases [9][10]. Several studies have also reported the importance of physical activity and nutrient intake which potentially interact with genetic predispositions of a disease that promote the progression and pathogenesis of metabolic diseases [11]. Many studies have also reported the influence of certain gene–diet interactions on metabolic disease-related traits and emphasized the importance of a healthy lifestyle that may modify the outcome of the disease or its related parameters [10][11][12][13][14][15]. A better comprehension of the relationship between genes and diet is key to making correlations between nutrition and wellness, thereby allowing for specific nutritional suggestions that are tailored to individuals or genetic subgroups. This strategy presents an appropriate public health approach [9].
The increasing prevalence of diabetes and obesity in SE Asia can be understood by the nutrition transition phenomenon, environment multiplier theory, and the thrifty gene hypothesis [16][17]. These theories provide an understanding of the dietary shift from traditional high-carbohydrate, low-fat diets towards high-energy diets (high saturated fat, sugars, and salt), and the role of inherited genetic predispositions in over-nutrition-related diseases. Dietary factors can affect the outcome of a disease and there are ethnic-specific genetic variations that influence the mechanism of these nutrient interactions [2]. Furthermore, lifestyle/dietary factors could influence genetic predispositions of metabolic disorders, especially obesity, and diabetes [18][19][20][21], making nutrigenetics research a necessity in ethnically diverse populations such as SE Asia. Nutrition science along with a better understanding of nutrigenetics in different ethnic groups is important to improvise personal and societal health [2][20]. This would ultimately add to the efforts of implementing precision nutrition specific to the populations [22][23].
2. Nutrigenetics Studies in Southeast Asia
Using PubMed and Google Scholar search engines, researchers found 19,031 articles matching the search strings. After the full-text screening, researchers included a total of 20 nutrigenetic studies related to obesity- and diabetes-related parameters carried out in SE Asia. Out of this, 16 studies examined obesity-related outcomes, 13 examined diabetes-related outcomes, and 9 studies observed both obesity, and diabetes-related outcomes. Figure 1 shows the selection of the 20 studies included. From the included studies, only three ASEAN countries (Malaysia, Indonesia, and Singapore) conducted studies to understand gene–diet interactions on metabolic disease-related traits.
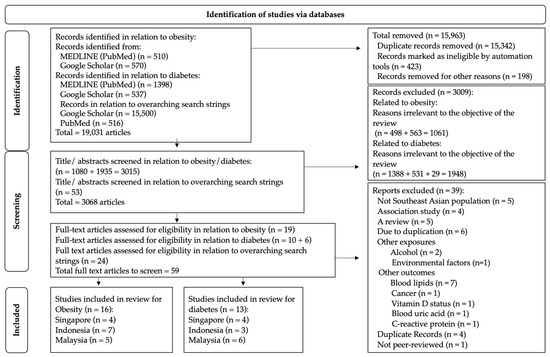
Figure 1. PRISMA flowchart showing the selection of articles based on inclusion and exclusion criteria.
3. Gene–Diet Interactions on Obesity-Related Outcomes in the Southeast Asian Population
Gene–diet interactions on obesity-related traits were observed in five Malaysian, seven Indonesian, and four Singaporean studies (Figure 2, Table 1).
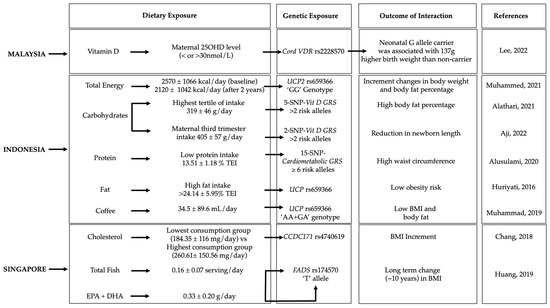
Figure 2. Gene–diet interactions on obesity-related traits. The figure shows the significant gene–diet interactions (p < 0.05) on obesity traits and the dietary factors that have influenced the risk of obesity in individuals carrying specific genetic variations in the Malaysian [24], Indonesian [12][21][25][26][27][28] and Singaporean [29][30] populations. GRS, genetic risk score; BMI, body mass index; EPA + DHA, eicosapentaenoic acid + docosahexaenoic; TEI, total energy intake; plus-minus symbol (±) indicates Standard deviation.
4. Gene–Diet Interactions on Diabetes-Related Outcomes in the Southeast Asian Population
Gene–diet interactions on diabetes-related traits were observed in six Malaysian, three Indonesian, and four Singaporean studies (Figure 3, Table 2).
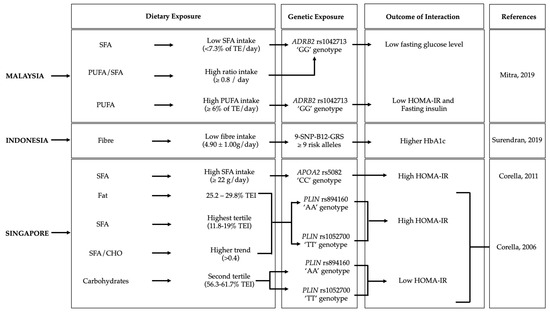
Figure 3. Gene–diet interactions on diabetes-related traits. The figure shows the significant gene–diet interactions (p < 0.05) on diabetes traits and the dietary factors that influenced the risk of diabetes in individuals carrying specific genetic variations in the Malysian [31], Indonesian [32] and Singaporean [33][34] populations. GRS, genetic risk score; BMI, body mass index; SFA, saturated fatty acids; PUFA, polyunsaturated fatty acids; HOMA-IR, homeostatic model assessment—insulin resistance; TE, total energy.
5. Precision Nutrition Approach for the Southeast Asian Population
The understanding of genetic diversity between individuals and among different ethnic groups should be established before designing dietary and nutritional requirements because different individuals respond differently to lifestyle interventions. Human genome sequencing plays a pivotal role in understanding genetic variations among different ethnic groups and has paved the way for the concept of personalized nutrition to frame effective lifestyle intervention strategies [35]. Developments in omics technology provide a better understanding of the whole genome of individuals as well as different ethnic groups along with the transcriptome, proteome, metabolome, and metagenome [36]. Integration of Artificial Intelligence along with gene nutrient analysis, especially in populations such as SE Asia will be useful to develop public health strategies and personalized nutrition plans for cardiometabolic diseases such as obesity and diabetes.
A high-throughput genetic screening has been developed to understand the role of SNPs in cardiometabolic diseases. However, molecular and pathophysiological mechanisms to understand gene–nutrient interactions and its influence on cardiometabolic diseases remain unexplored. In LMIC such as in SE Asia, nutrigenetics is still in its infancy and requires an evidence-based approach before framing precision nutrition strategies for the population. Further, such studies on larger populations and ethnic groups as well as on different levels of nutrition transition, are crucial for the development of accurate and population-specific precision nutrition strategies effective to combat chronic, yet preventable diseases such as obesity and diabetes [2]. While the Western countries have shifted their approach towards nutrigenetics, developing countries like SE Asia still favor traditional methods for evaluating, categorizing, and managing obesity and diabetes. Costly gene testing, lack of knowledge, and experts in this field are the primary impediments of nutrigenetics implementation, particularly in LMIC. Even though this field is expanding globally, there are not many researchers in this discipline in SE Asia. Moving forward, the nutrigenetics approach should be considered for government health programs, particularly those aimed at noncommunicable diseases (NCDs). Currently, lifestyle diseases are a major burden to all countries, and long-term investments in accelerating nutrigenetics research and generating scientific evidence may provide a solution to obesity and its comorbidities through precision nutrition.
References
- Di Cesare, M.; Sorić, M.; Bovet, P.; Miranda, J.J.; Bhutta, Z.; Stevens, G.A.; Laxmaiah, A.; Kengne, A.P.; Bentham, J. The epidemiological burden of obesity in childhood: A worldwide epidemic requiring urgent action. BMC Med. 2019, 17, 212.
- Vimaleswaran, K.S. GeNuIne (gene-nutrient interactions) Collaboration: Towards implementing multi-ethnic population-based nutrigenetic studies of vitamin B(12) and D deficiencies and metabolic diseases. Proc. Nutr. Soc. 2021, 80, 1–11.
- World Health Organisation. Obesity. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 3 November 2022).
- World Health Organisation. Diabetes. Available online: https://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 3 November 2022).
- World Health Organisation. Obesity and Overweight in South East Asia. Available online: https://www.who.int/southeastasia/health-topics/obesity (accessed on 3 November 2022).
- Biswas, T.; Townsend, N.; Magalhaes, R.J.S.; Islam, M.S.; Hasan, M.M.; Mamun, A. Current Progress and Future Directions in the Double Burden of Malnutrition among Women in South and Southeast Asian Countries. Curr. Dev. Nutr. 2019, 3, nzz026.
- Association of Southeast Asian Nations: ASEAN Food and Nutrition Security Report 2021, Volume 1. Available online: https://asean.org/book/asean-food-and-nutrition-security-report-2021-volume-1-the-asean-secretariat-jakarta/ (accessed on 4 November 2022).
- ASEAN Food and Nutrition Security Report 2021 Volume 2—Food and Nutrition Security Country Profiles. Available online: https://asean.org/book/asean-food-and-nutrition-security-report-2021-volume-2-food-and-nutrition-security-country-profiles/ (accessed on 8 November 2022).
- Phillips, C.M. Nutrigenetics and metabolic disease: Current status and implications for personalised nutrition. Nutrients 2013, 5, 32–57.
- Vimaleswaran, K.S.; Franks, P.W.; Barroso, I.; Brage, S.; Ekelund, U.; Wareham, N.J.; Loos, R.J. Habitual energy expenditure modifies the association between NOS3 gene polymorphisms and blood pressure. Am. J. Hypertens. 2008, 21, 297–302.
- Vimaleswaran, K.S.; Li, S.; Zhao, J.H.; Luan, J.; Bingham, S.A.; Khaw, K.T.; Ekelund, U.; Wareham, N.J.; Loos, R.J. Physical activity attenuates the body mass index-increasing influence of genetic variation in the FTO gene. Am. J. Clin. Nutr. 2009, 90, 425–428.
- Alathari, B.E.; Aji, A.S.; Ariyasra, U.; Sari, S.R.; Tasrif, N.; Yani, F.F.; Sudji, I.R.; Lovegrove, J.A.; Lipoeto, N.I.; Vimaleswaran, K.S. Interaction between Vitamin D-Related Genetic Risk Score and Carbohydrate Intake on Body Fat Composition: A Study in Southeast Asian Minangkabau Women. Nutrients 2021, 13, 326.
- Bodhini, D.; Gaal, S.; Shatwan, I.; Ramya, K.; Ellahi, B.; Surendran, S.; Sudha, V.; Anjana, M.R.; Mohan, V.; Lovegrove, J.A.; et al. Interaction between TCF7L2 polymorphism and dietary fat intake on high density lipoprotein cholesterol. PLoS ONE 2017, 12, e0188382.
- Vimaleswaran, K.S.; Bodhini, D.; Lakshmipriya, N.; Ramya, K.; Anjana, R.M.; Sudha, V.; Lovegrove, J.A.; Kinra, S.; Mohan, V.; Radha, V. Interaction between FTO gene variants and lifestyle factors on metabolic traits in an Asian Indian population. Nutr. Metab. 2016, 13, 39.
- Vimaleswaran, K.S.; Cavadino, A.; Hyppönen, E. APOA5 genotype influences the association between 25-hydroxyvitamin D and high density lipoprotein cholesterol. Atherosclerosis 2013, 228, 188–192.
- Bishwajit, G. Nutrition transition in South Asia: The emergence of non-communicable chronic diseases. F1000Res 2015, 4, 8.
- Williams, R.; Periasamy, M. Genetic and Environmental Factors Contributing to Visceral Adiposity in Asian Populations. Endocrinol Metab 2020, 35, 681–695.
- Wuni, R.; Adela Nathania, E.; Ayyappa, A.K.; Lakshmipriya, N.; Ramya, K.; Gayathri, R.; Geetha, G.; Anjana, R.M.; Kuhnle, G.G.C.; Radha, V.; et al. Impact of Lipid Genetic Risk Score and Saturated Fatty Acid Intake on Central Obesity in an Asian Indian Population. Nutrients 2022, 14, 2713.
- Wuni, R.; Kuhnle, G.G.C.; Wynn-Jones, A.A.; Vimaleswaran, K.S. A Nutrigenetic Update on CETP Gene-Diet Interactions on Lipid-Related Outcomes. Curr. Atheroscler. Rep. 2022, 24, 119–132.
- Vimaleswaran, K.S. A nutrigenetics approach to study the impact of genetic and lifestyle factors on cardiometabolic traits in various ethnic groups: Findings from the GeNuIne Collaboration. Proc. Nutr. Soc. 2020, 79, 194–204.
- Alsulami, S.; Aji, A.S.; Ariyasra, U.; Sari, S.R.; Tasrif, N.; Yani, F.F.; Lovegrove, J.A.; Sudji, I.R.; Lipoeto, N.I.; Vimaleswaran, K.S. Interaction between the genetic risk score and dietary protein intake on cardiometabolic traits in Southeast Asian. Genes Nutr. 2020, 15, 19.
- Dhanapal, A.; Wuni, R.; Ventura, E.F.; Chiet, T.K.; Cheah, E.S.G.; Loganathan, A.; Quen, P.L.; Appukutty, M.; Noh, M.F.M.; Givens, I.; et al. Implementation of Nutrigenetics and Nutrigenomics Research and Training Activities for Developing Precision Nutrition Strategies in Malaysia. Nutrients 2022, 14, 5108.
- Thiruvenkataswamy, C.S.; Appukutty, M.; Vimaleswaran, K.S. Role of precision nutrition in improving military performance. Pers. Med. 2022, 19, 167–170.
- Lee, S.S.; Ling, K.H.; Tusimin, M.; Subramaniam, R.; Rahim, K.F.; Loh, S.P. Interplay between Maternal and Neonatal Vitamin D Deficiency and Vitamin-D-Related Gene Polymorphism with Neonatal Birth Anthropometry. Nutrients 2022, 14, 564.
- Muhammad, H.F.L.; Sulistyoningrum, D.C.; Huriyati, E.; Lee, Y.Y.; Muda, W. The interaction between energy intake, physical activity and UCP2 -866G/A gene variation on weight gain and changes in adiposity: An Indonesian Nutrigenetic Cohort (INDOGENIC). Br. J. Nutr. 2021, 125, 611–617.
- Huriyati, E.; Luglio, H.F.; Ratrikaningtyas, P.D.; Tsani, A.F.; Sadewa, A.H.; Juffrie, M. Dyslipidemia, insulin resistance and dietary fat intake in obese and normal weight adolescents: The role of uncoupling protein 2 -866G/A gene polymorphism. TInt. J. Mol. Epidemiol. Genet. 2016, 7, 67–73.
- Muhammad, H.F.L.; Sulistyoningrum, D.C.; Huriyati, E.; Lee, Y.Y.; Manan Wan Muda, W.A. The Interaction between Coffee: Caffeine Consumption, UCP2 Gene Variation, and Adiposity in Adults-A Cross-Sectional Study. J. Nutr. Metab. 2019, 2019, 9606054.
- Aji, A.S.; Lipoeto, N.I.; Yusrawati, Y.; Malik, S.G.; Kusmayanti, N.A.; Susanto, I.; Nurunniyah, S.; Alfiana, R.D.; Wahyuningsih, W.; Majidah, N.M.; et al. Impact of maternal dietary carbohydrate intake and vitamin D-related genetic risk score on birth length: The Vitamin D Pregnant Mother (VDPM) cohort study. BMC Pregnancy Childbirth 2022, 22, 690.
- Chang, X.; Dorajoo, R.; Sun, Y.; Han, Y.; Wang, L.; Khor, C.C.; Sim, X.; Tai, E.S.; Liu, J.; Yuan, J.M.; et al. Gene-diet interaction effects on BMI levels in the Singapore Chinese population. Nutr. J. 2018, 17, 31.
- Huang, T.; Wang, T.; Heianza, Y.; Wiggs, J.; Sun, D.; Choi, H.K.; Chai, J.F.; Sim, X.; Khor, C.C.; Friedlander, Y.; et al. Fish and marine fatty acids intakes, the FADS genotypes and long-term weight gain: A prospective cohort study. BMJ Open 2019, 9, e022877.
- Mitra, S.R.; Tan, P.Y.; Amini, F. Association of ADRB2 rs1042713 with Obesity and Obesity-Related Phenotypes and Its Interaction with Dietary Fat in Modulating Glycaemic Indices in Malaysian Adults. J. Nutr. Metab. 2019, 2019, 8718795.
- Surendran, S.; Aji, A.S.; Ariyasra, U.; Sari, S.R.; Malik, S.G.; Tasrif, N.; Yani, F.F.; Lovegrove, J.A.; Sudji, I.R.; Lipoeto, N.I.; et al. A nutrigenetic approach for investigating the relationship between vitamin B12 status and metabolic traits in Indonesian women. J. Diabetes Metab. Disord. 2019, 18, 389–399.
- Corella, D.; Tai, E.S.; Sorlí, J.V.; Chew, S.K.; Coltell, O.; Sotos-Prieto, M.; García-Rios, A.; Estruch, R.; Ordovas, J.M. Association between the APOA2 promoter polymorphism and body weight in Mediterranean and Asian populations: Replication of a gene-saturated fat interaction. Int. J. Obes. 2011, 35, 666–675.
- Corella, D.; Qi, L.; Tai, E.S.; Deurenberg-Yap, M.; Tan, C.E.; Chew, S.K.; Ordovas, J.M. Perilipin gene variation determines higher susceptibility to insulin resistance in Asian women when consuming a high-saturated fat, low-carbohydrate diet. Diabetes Care 2006, 29, 1313–1319.
- Ferguson, L.R.; De Caterina, R.; Görman, U.; Allayee, H.; Kohlmeier, M.; Prasad, C.; Choi, M.S.; Curi, R.; de Luis, D.A.; Gil, Á.; et al. Guide and Position of the International Society of Nutrigenetics/Nutrigenomics on Personalised Nutrition: Part 1Fields of Precision Nutrition. J. Nutr. Nutr. 2016, 9, 12–27.
- Maria Catherine, B. Otero LFB: Role of Nutrigenomics in Modern-Day Healthcare and Drug Discovery; Elsevier: Amsterdam, The Netherlands, 2023.
More
Information
Subjects:
Genetics & Heredity
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
738
Revisions:
3 times
(View History)
Update Date:
31 Jul 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





