Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Koyeli Girigoswami | -- | 6050 | 2023-07-06 06:58:40 | | | |
| 2 | Rita Xu | Meta information modification | 6050 | 2023-07-06 08:12:05 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Girigoswami, A.; Girigoswami, K. Nanoparticles in Lung Cancer Treatment. Encyclopedia. Available online: https://encyclopedia.pub/entry/46494 (accessed on 06 June 2026).
Girigoswami A, Girigoswami K. Nanoparticles in Lung Cancer Treatment. Encyclopedia. Available at: https://encyclopedia.pub/entry/46494. Accessed June 06, 2026.
Girigoswami, Agnishwar, Koyeli Girigoswami. "Nanoparticles in Lung Cancer Treatment" Encyclopedia, https://encyclopedia.pub/entry/46494 (accessed June 06, 2026).
Girigoswami, A., & Girigoswami, K. (2023, July 06). Nanoparticles in Lung Cancer Treatment. In Encyclopedia. https://encyclopedia.pub/entry/46494
Girigoswami, Agnishwar and Koyeli Girigoswami. "Nanoparticles in Lung Cancer Treatment." Encyclopedia. Web. 06 July, 2023.
Copy Citation
Lung cancer is managed using conventional therapies, including chemotherapy, radiation therapy, or a combination of both. Each of these therapies has its own limitations, such as the indiscriminate killing of normal as well as cancer cells, the solubility of the chemotherapeutic drugs, rapid clearance of the drugs from circulation before reaching the tumor site, the resistance of cancer cells to radiation, and over-sensitization of normal cells to radiation. Other treatment modalities include gene therapy, immunological checkpoint inhibitors, drug repurposing, and in situ cryo-immune engineering (ICIE) strategy. Nanotechnology has come to the rescue to overcome many shortfalls of conventional therapies.
lung cancer
chemotherapy
radiation therapy
nano sensitizers
1. Introduction
Men and women die from lung cancer at a higher rate than any other cancer in the US and around the world. Nearly 84% of all lung cancers belong to non-small-cell lung cancers (NSCLC), while 13% are small-cell lung cancers (SCLC). However, lung cancer screening has expanded, radiation techniques have improved, and treatment advances have changed the prognosis for NSCLC markedly in the past decade. The reported decline in NSCLC mortality is likely a result of these changes. According to a recent study, men’s lung cancer incidence decreased by 3%, and women’s by 1% during the last decade [1]. Over the past decade, increased screening has likely contributed to a rise in stage I NSCLC at diagnosis. As a result of earlier diagnosis of NSCLC and more effective treatment, the prevalence of NSCLC has increased, and five-year survival has improved. Undertreatment may be to blame for the low five-year survival rate among patients aged 65 and older with stage IV NSCLC despite the availability of effective treatments [1][2]. Antihypertensives, anti-hyperlipidemia drugs, anti-inflammatory drugs, anti-diabetics, and antimicrobials are examples of potential candidate drugs that can be repurposed for treating NSCLC [2]. There has been a tremendous leap forward in the field of nanotechnology in recent times, highlighting its applications in the field of targeted drug delivery, imaging, and especially in cancer theranostics [3][4]. Nanobiotechnology-based drug delivery systems are used as drug delivery systems to target tumor cells due to their properties like sustainable biocompatibility, biodistribution, and active targeting. Control of the matter at the nanoscale plays a significant role in these technologies. Nanoparticles are used explicitly due to their properties like confined size, magnetic and optoelectronic properties, and atomic structure. They can also be used for site-specific targeting due to all these properties; nanobiotechnology has advanced in the treatment of cancer [5]. Nanoparticles like liposomes, nanofiber, nanoshells, micelles, dendrimers, carbon nanotubes, and quantum dots are used as nanocarriers that help in carrying the anticancer drug and delivering it to a specific site. These formulations protect the drug from degradation and improve its efficiency and stability. Therefore, they can be used for the targeted therapy of lung cancer [6]. The treatment of cancer with immunotherapy, in which the immune system recognizes and attacks tumors, is a promising approach. The tumor microenvironment (TME) has an immunosuppressive (i.e., immunologically cold) nature, which significantly limits the immune system’s effectiveness. A cryo-immune engineering (ICIE) strategy has been developed for converting a “cold” TME into a “hot” one. Not only in primary tumors with cryosurgery but also in distant tumors without freezing, the ratio of CD8+ cytotoxic T cells to immunosuppressive regulatory T cells increases by more than 100 times after ICIE treatment. The anticancer drug and PD-L1 silencing siRNA are rapidly released into the cytosol following cryosurgery using cold-responsive nanoparticles that target tumors and cause “frostbite” of tumors [7]. Although ICIE therapy has been used recently for breast cancer models in female mice, it can be extrapolated for lung cancer models in the future. A wider holistic approach is necessary to study more about the recent treatment strategies for lung cancer and how they overcome the shortfalls of the conventional treatment modalities.
2. Chemotherapy for Treatment of Lung Cancer
The conventional therapies for the management of lung cancer include chemotherapy and radiation therapy, as well as a combination of both. The different kinds of traditional treatment modalities for lung cancer management are shown in Figure 1.
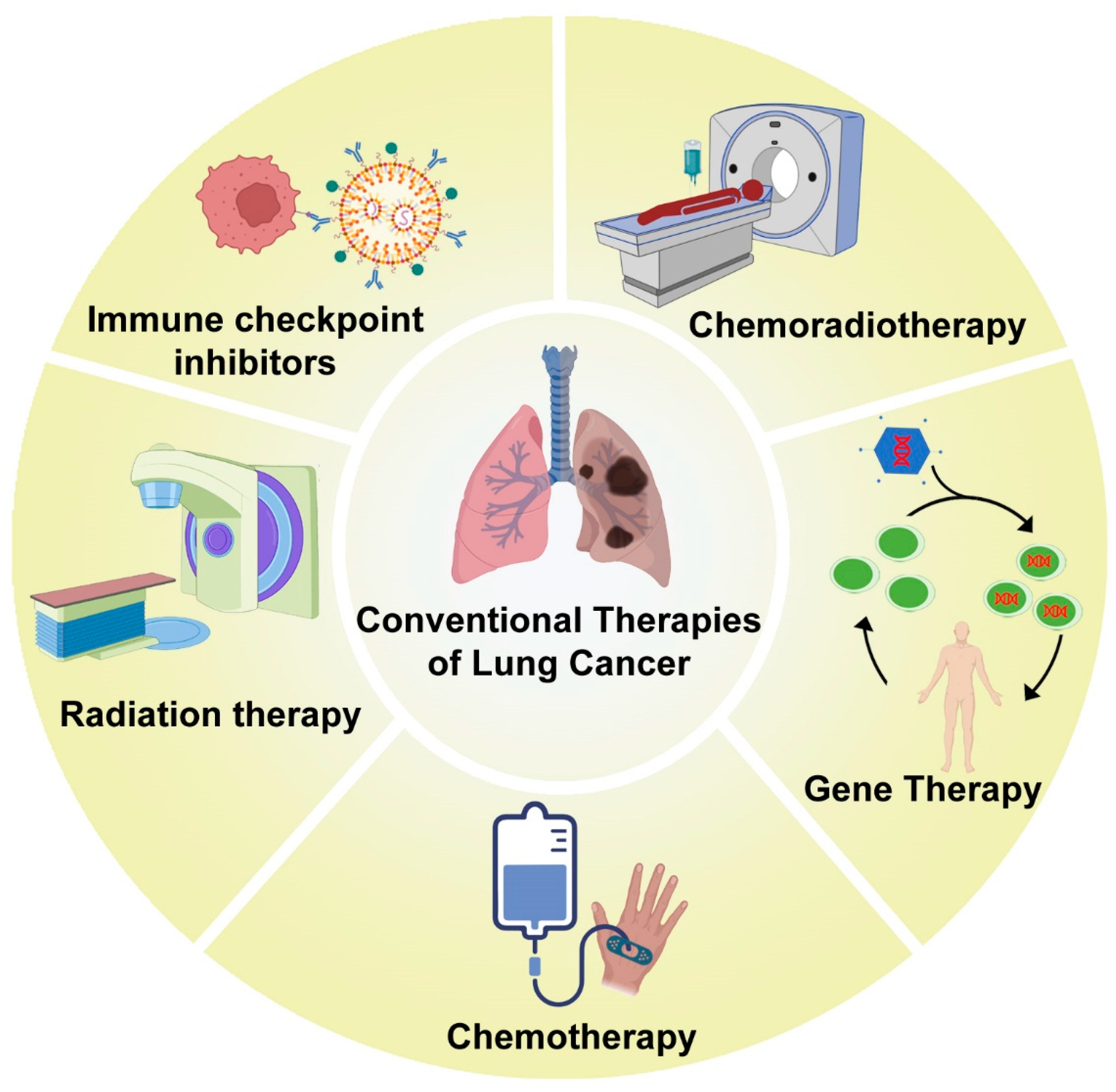
Figure 1. The conventional lung cancer treatment modalities.
Among the chemotherapeutic drugs, the modes of action encompass mitotic inhibitors, alkylating agents, topoisomerase inhibitors, antimetabolites, tubulin-binding agents, etc. [8]. Forde et al. performed an open-label, phase 3 trial by randomly assigning patients with resectable NSCLC stages IB to IIIA. They received (i) nivolumab plus platinum-based chemotherapy or (ii) only platinum-based chemotherapy, followed by surgical resection. By blinded independent review, the primary endpoints were the number of event-free days and the percentage of pathologically complete responses (0% viable tumor detected in resected lymph nodes and lung). As a secondary outcome, overall survival was important, and each patient was assessed for safety. Patients with resectable NSCLC treated with neoadjuvant nivolumab and chemotherapy achieved a higher rate of complete pathological responses than those treated with chemotherapy alone. Neoadjuvant chemotherapy with nivolumab did not increase the incidence of adverse events or impair the feasibility of surgery [9]. A study conducted by Ares et al. examined whether the conjugation of two chemotherapy cycles to nivolumab plus ipilimumab would enhance the clinical benefit further in 1152 patients. A combination of nivolumab plus ipilimumab plus two cycles of chemotherapy significantly improved overall survival compared to chemotherapy alone. Based on these results, this regimen is likely to be an effective first-line therapy for advanced NSCLC patients [10]. According to another study, patients with advanced squamous non-small-cell lung cancer (sq-NSCLC) who received tislelizumab along with chemotherapy had improved PFS. An assessment was performed by an independent review committee (IRC) regarding the PFS. Secondary endpoints were OS, IRC-assessed objective response rate (ORR), investigator-assessed PFS, and IRC-assessed duration of response, along with adverse events (AEs). According to the results of this phase 3 randomized clinical study, patients with advanced sq-NSCLC treated with tislelizumab had significantly longer PFS, higher ORRs, and a more manageable safety/tolerability profile, regardless of the expression of PD-L1 [11].
2.1. Chemoimmunotherapy
At an advanced level, immunotherapy is also combined with chemotherapy to yield a superior result and improve the effective lifespan of the patient. In NSCLC patients with previously treated cancer, atezolizumab (a monoclonal antibody against programmed cell death ligand (PD-L1)) improved overall survival by restoring anticancer immunity. It also demonstrated clinical benefit as a first-line treatment when combined with chemotherapy. For non-squamous NSCLC patients, this study compared the effectiveness and safety of atezolizumab plus chemotherapy with chemotherapy alone. A combined 723 patients from eight countries participated in this study, and it showed that atezolizumab plus chemotherapy improved progression-free survival (PFS) and overall survival (OS) significantly versus chemotherapy when used as first-line of treatment for patients with stage IV non-squamous NSCLC without mutations of EGFR or ALK. Atezolizumab was effective in treating metastatic NSCLC in combination with platinum-based chemotherapy, as supported by this study [12]. Based on the effectiveness of immune checkpoint inhibitors in metastatic NSCLC, researchers planned a trial to assess the efficacy of atezolizumab combined with carboplatin and nab-paclitaxel as neoadjuvant therapy before surgical removal of metastatic NSCLC. Three American hospitals participated in this open-label, single-arm, multicenter, phase 2 trial. It was concluded that nab-paclitaxel and atezolizumab plus carboplatin could be used as the neoadjuvant regimen in resectable NSCLC, showing a high pathological response rate and controllable toxic effects that did not impair the surgical resection in most patients [13].
A checkpoint inhibitor targeting programmed cell death 1 (PD-1) or its ligand (PD-L1) exerted clinical activity in patients with metastatic NSCLC. An open-label, phase 3 randomized clinical trial (MYSTIC) was executed throughout 17 countries at 203 centers for cancer treatment to compare durvalumab, with or without tremelimumab, with chemotherapy as the first-line treatment for metastatic NSCLC. Patients with 25% of tumor cells expressing PD-L1 in the phase 3 MYSTIC trial failed to achieve the primary endpoints showing improved OS after treatment with durvalumab in comparison with chemotherapy, or improved PFS or OS with durvalumab and tremelimumab, both compared to chemotherapy. The combined use of durvalumab plus tremelimumab with a bTMB threshold of 20 mutations per megabase provided an ideal OS benefit [14]. Another study, conducted on 361 and 358 patients, compared nivolumab (NIVO) plus ipilimumab (IPI) plus two cycles of platinum-doublet chemotherapy (chemo) to only chemo in stage IV/recurrent NSCLC in 1 L stage IV. With NSCLC-optimized IPI + NIVO + a limited amount of chemo in comparison to chemotherapy (4 cycles) in 1 L advanced NSCLC, there was a significant OS enhancement. No new safety signals were reported during this study [15]. The same group of scientists reported the first five-year follow-up data from any phase III trial involving immunotherapy as part of a first-line treatment for NSCLC. As part of a randomized controlled trial (RCT), pembrolizumab was compared with platinum-based chemotherapeutic drugs for previously untreated NSCLC diagnosed with at least 50% tumor proportion score in terms of PD-L1 and a lack of sensitizing changes in EGFR or ALK. The patients were assigned randomly (1:1) to receive pembrolizumab (200 mg once every 3 weeks for 35 cycles) or platinum-based chemotherapy. Pembrolizumab could be given to chemotherapy patients with progressive disease, and 305 patients participated in this study. When used for first-line therapy in cases of metastatic NSCLC with at least a 50% PD-L1 tumor proportion score, pembrolizumab provided a durable, clinically meaningful OS benefit over chemotherapy [16].
A study by Provencio et al. examined the antitumor activity of, as well as safety issues for, neoadjuvant chemoimmunotherapy applied to stage IIIA resectable NSCLC. In this study, neoadjuvant nivolumab was added to platinum-based chemotherapy, given to resectable stage IIIA NSCLC patients. Chemoimmunotherapy as a neoadjuvant treatment for locally advanced lung cancer may change perceptions of lung cancer as a potentially lethal disease into a curable one [17].
2.2. Tyrosine Kinase Inhibitor with Chemotherapy
An epidermal growth factor receptor (EGFR)—directed oral inhibitor of tyrosine kinase was the standard first-line treatment in cases of advanced NSCLC. Oral tyrosine kinase inhibitors can be complemented with chemotherapy using pemetrexed and carboplatin. In a phase III randomized trial, Noronha et al. evaluated first-line palliative therapy in advanced NSCLC patients with EGFR-sensitizing mutations and monitored a performance status increment from 0 to 2. According to the results of the study with 350 patients, adding chemotherapy with gefitinib did significantly prolong PFS and OS, but nonetheless increased toxicity [18]. Treatment of advanced NSCLC with mutations in EGFR with a tyrosine kinase inhibitor and cytotoxic chemotherapy was highly effective. A total of 345 patients newly diagnosed with metastatic NSCLC having EGFR mutations were randomly assigned to receive either gefitinib alone or gefitinib along with carboplatin and pemetrexed. According to a hierarchical sequential testing method, progression-free survival (PFS, PFS2) as well as OS were analyzed sequentially. Quality of life, safety, and objective response rate (ORR) were secondary endpoints. Compared to gefitinib alone, the combination of gefitinib with pemetrexed plus carboplatin showed improvement in PFS in untreated advanced NSCLC patients with mutations in EGFR, but further study is needed to determine its OS benefit [19]. Previous studies have investigated whether chemotherapy using single or multiple chemotherapeutic drugs or a combination of immunotherapy with chemotherapy could improve the progression of lung cancer. But it was evident that these treatment modalities had a few limitations, such as toxicity, quality of survival, etc.
3. Lung Cancer Management by Radiation Therapy
The American Society for Radiation Oncology has set clear radiation guidelines for the management of different types and stages of lung cancer [20]. SCLC has recently been challenged in a number of sentinel phase III randomized trials. Both limited-stage (LS) and extensive-stage (ES) SCLC can benefit from thoracic radiotherapy and prophylactic cranial irradiation (PCI). Initially, during the treatment course for LS-SCLC, definitive thoracic RT should be administered once or twice daily. If a patient has positive margins or nodal metastases after surgical resection, adjuvant RT is conditionally recommended. Also, involved field RT used as an advanced treatment modality was recommended strongly for post-chemotherapy volumes. Stereotactic body radiation therapy (SBRT) or conventional fractionation is strongly recommended for patients with stage I or II node-negative cancer, and chemotherapy should be administered before or after SBRT. Patients with stage II or III LS-SCLC who responded to chemoradiation are strongly recommended to undergo PCI; those at high risk of neurocognitive toxicity should be included in the decision. It is strongly recommended to consult a radiation oncologist regarding PCI vs. magnetic resonance surveillance in ES-SCLC. A conditional recommendation is also made for thoracic radiotherapy usage in patients with ES-SCLC following chemotherapy. As in LS-SCLC, RT plays a crucial role in ES-SCLC as well. In SCLC, these guidelines provide guidance on the best clinical practices for local therapy [21].
3.1. Stereotactic Body Radiation Therapy
Radiation therapy has been used for controlling the lung cancerous tumors in 77% of patients [22]. A randomized trial for lung SBRT comparing 34 Gy in one fraction with 48 Gy in four fractions was conducted for presenting the long-term outcome of RTOG 0915/NCCTG N0927. A phase 2 multicenter study involved medically inoperable patients with metastatic peripheral T1 or T2 N0M0 NSCLC. The primary endpoint was one-year toxicity, with secondary endpoints including failure and survival. Neither arm showed a significant increase in late-appearing toxicity. The primary tumor control rates at 5 years were similar between the two arms. In a study with 84 patients, a median time of survival of 4 years suggested similar efficacy for each arm [23]. For patients in which the NSCLC tumor was medically inoperable, SBRT was a standard treatment [24][25]. Thoracic grade 3 or advanced AEs after 30 Gy in one fraction (arm 1) and 60 Gy in three fractions (arm 2) were compared using the Common Terminology Criteria for Adverse Events (CTCAE). It was established that 30 Gy delivered in one fraction corresponded to 60 Gy delivered in three fractions when observed in terms of life expectancy, PFS, toxicity, and OS. A single fraction SBRT was found to be more effective in terms of social functioning and dyspnea measures of QOL [26]. According to a secondary analysis of RTOG 0617, higher radiation doses were related to worse control of tumor and OS in stage III NSCLC. An independent cohort of patients treated at the University of Colorado School of Medicine was studied to determine the influence of the estimated dose of radiation on immune cells (EDRIC). After the definitive treatment of stage III NSCLC, higher radiation doses to the immune system were associated with the progression of tumors and death [27]. In the past, radiation therapy has been observed to produce out-of-field tumor regression (abscopal response), but it has recently gained significant importance with high-precision radiation delivery devices and has been used to treat various cancers, including NSCLC. Radiation therapy-induced abscopal effects in patients with advanced NSCLC were reviewed in a detailed study. When radiation therapy was used in combination with immunotherapy to treat advanced NSCLC or other types of cancers, the results indicated that radiation therapy could induce abscopal effects along with improved potential for boosting these effects. As a result of clinical trials investigating radiation therapy-induced abscopal effects, the use of radiation therapy for advanced NSCLC may be drastically changed, especially when combined with immunotherapy [28].
In peripherally located lung tumors, stereotactic MR-guided adaptive radiation therapy (SMART) has been found to be beneficial for delivering SABR. On an MR Linac or Cobalt-60 system, 23 patients (25 peripheral lung tumors) received SMART in 3–8 fractions. The on-table plan was adapted grounded on the anatomy of that day after each breath-hold MR scan. Under continuous MR guidance, breath-hold gated SABR was delivered using an in-room monitor, resulting in significantly smaller target volumes than if an ITV-based approach had been used. Despite ensuring ablative doses in all fractions with on-table adaptation, dosimetric benefits from daily online plan adaptation were modest in most peripheral lung cancer patients [29].
3.2. Chemoradiation Therapy
Chemoradiation therapy (CRT) was not appropriate for all stage III NSCLC patients. Sequential concurrent CRT had a high local failure rate, and therefore, intensification of treatment was justified. A multicenter feasibility study of intense modulated radiation therapy (IMRT) combining hyperfractionation, acceleration, and dose escalation was conducted. The study included patients with unresectable stage III NSCLC who had a performance status (PS) of 0 to 2 and were not eligible for concurrent chemotherapy. In 14 patients (37.2%), a maximum dose of 79.2 Gy was achieved. Esophagitis grade 3 was confirmed in two patients, but no pneumonitis grade 3 to 4 was reported. In addition to acute radiation pneumonitis, there were bronchopulmonary hemorrhages and acute lung infections of grade 5. In this study, the median survival time was 18.1 months (95% confidence interval [CI], 13.9–30.6), the two-year overall survival rate was 33.6% (95% CI, 17.9–50.1), and the PFS rate was 23.9% (95% CI, 11.3–39.1%) [30]. The rates of toxicity for curative and palliative radiotherapy were assessed and compared using a meta-analysis. Comparatively to individual trials, this provided more accurate quantitative assessments of toxicities. An analysis of randomized trials with >50 patients suffering from unresectable NSCLC who received palliative or curative conventional radiotherapy (RT) with or without chemotherapy was included in this systematic review. Among the data extracted were data on pneumonitis, esophagitis, pulmonary fibrosis, cardiac events, myelopathy, and neutropenia, as well as treatment-related deaths. However, the toxicity rate increased with the intensity of radiation therapy, and there was no significant difference between concurrent and sequential CRT when it came to esophagitis. Clinicians can use this information when making decisions about radiotherapy for NSCLC [31].
Radiation therapy (RT) is effective in treating many malignancies and relieving tumor-related symptoms. RT may, however, produce toxicity because surrounding tissues and organs are exposed to its biophysical effects [32]. Affected organs’ anatomy and physiology play a role in determining the manifestation of toxicity. The dose and volume of radiation applied to normal tissues usually have a direct relationship with the risk of toxicity, which has led to the establishment of guidelines and recommended dose limits for a majority of tissues. In addition to the characteristics of the patient at baseline as well as other treatments that are administered to them, side effects are multifactorial. These limitations of RT make it difficult to implement on a case-to-case basis.
4. Combination of Radiation and Chemotherapy for Lung Cancer Management
In certain conditions of lung cancer, only chemotherapy or only radiation was not sufficient to manage the disease. In such conditions, both chemotherapy as well as radiation were utilized for the tumor progression retardation. The use of definitive concurrent chemoradiotherapy (cCRT) should be considered for patients who have unresectable stage III NSCLC showing good status of performance. A meta-analysis of two large phase 3 randomized studies established the dominance of cCRT over sequential chemoradiotherapy (sCRT). Although cCRT offers greater efficacy, it is associated with more acute toxicity than the sequential treatment. There are currently a number of documented approaches to address this drawback. By using a multidisciplinary team (MDT) approach, the optimal treatment strategy can be determined at the point of diagnosis to minimize risks. Clinical oncologists can also find additional recommendations for defining target volumes and organs at risk in definitive cCRT (and adjuvant radiotherapy) by reviewing the guidelines of the Advisory Committee on Radiation Oncology Practice (ACROP). In addition, radiation oncologists could safely treat the larger tumors of the lung using high radiotherapy doses, resulting in greater accuracy, by utilizing modern advances in radiation therapy treatment planning and delivery. This resulted in reduced radiation dose to healthy tissues surrounding the lung tumor. As a result of these advances in cCRT, comprehensive strategies may be devised to allow the patients to benefit from potentially curative treatment modalities like immunotherapy and minimize risks associated with those treatments [33].
During radiation therapy for lung cancer, it is essential for patients to prevent pulmonary toxicity. Exercise training is not well established for patients with unresectable stage III lung cancer who are candidates for radical treatment. A home-based pulmonary rehabilitation (PR) program was evaluated to determine whether it was feasible to improve respiratory function, exercise capacity, and quality of life through the use of reliable tools. First, 20 patients (interventional group, IG) received PR concurrently with radiation therapy, while another 20 patients were identified as an observational group (OG). The 6 min walking test was performed at baseline (T0), followed by the modified Borg Scale (mBORG), the SF-36 questionnaire (SF-36), and the pulmonary function test (PFT) 8 weeks after the baseline (T2) and 4 weeks after the baseline (T1). After 4 weeks (T1), only the SF-36 questionnaire was administered. Due to the input from the OG, the mBORG scores trended downward; the IG scores, however, showed a slight improvement. There was a decrease in all items of the SF-36 score between T0 and T1 in the OG. There was an increase in the trend from T0 to T2 for all SF-36 items in the IG. There were no clinically significant differences in PFT between the baseline and T2 in either group. Thus, in assessing the effectiveness of PR programs, the 6MWT, mBORG, and SF-36 proved useful. Radio(chemo)therapy resulted in a significant increase in the capacity for functional exercise and a reduction in physiological impairment related to the quality of life [34]. Extensive-stage small cell lung cancer (ES-SCLC) patients have poor survival rates. The combination of cTRT with chemotherapy and upfront immunotherapy along with chemotherapy improved the outcome of patients incrementally but has not been evaluated in a clinical trial yet. After chemotherapy and immunotherapy, cTRT was used to characterize outcomes and toxicities. In two hospitals, researchers identified ES-SCLC patients treated with first-line chemotherapy, immunotherapy, and cTRT. The following outcomes were assessed for patients: PFS, OS, distant progression-free survival (DPFS), local progression-free survival (LPFS), and toxicity. It was observed that the first-line chemoimmunotherapy, which was followed by cTRT, appeared to be safe and produced comparable outcomes as found in published modern clinical trials. In order to determine whether cTRT is beneficial after chemoimmunotherapy, further studies are warranted [35].
SBRT, along with full-dose systemic chemotherapy, was studied in a phase 2 study (ClinicalTrials.gov NCT02568033) for unresectable stage 2 and stage 3 NSCLC. Toxicology and disease-free survival were the primary endpoints. SBRT was administered to all sites of gross disease. There were three fractions of 60 Gy given to peripheral lung tumors, five fractions of 50 Gy for central lung tumors, and five fractions of 40 to 50 Gy for hilar and mediastinal lymph nodes. A total of four cycles of chemotherapy was administered for nonsquamous histology, cisplatin and docetaxel for squamous histology, and cisplatin and paclitaxel for melanoma. In between cycles of chemotherapy, SBRT was given. SBRT was followed by chemotherapy after a seven-day break. Functional assessment of cancer therapy was used to measure the quality of life. SBRT, in combination with full-dose chemotherapy, appeared to be effective and safe for locally advanced NSCLC treatment [36]. The above studies could not conclude whether the combination of radiotherapy and chemotherapy resulted in lower toxicity. There are many limitations of non-specific radiation exposure to benign cells regarding mutation induction, DNA strand breaks, generation of reactive oxygen species, ionization, etc.
5. Other Treatment Strategies for Lung Cancer
Apart from chemotherapy and radiation therapy, there are other treatment modalities for the management of lung cancer. As a neuroendocrine tumor of the lung, SCLC is a potentially aggressive disease that has a metastatic tendency quite early in the course of the disease. The VA staging categorizes the disease as (a) limited stage (LS), which can be confined to one hemithorax and radiated in the same field or (b) extensive stage (ES), which is extended beyond one hemithorax. LS disease is currently treated with concurrent chemoradiation, and ES disease with chemotherapy alone. The current standard treatments will only cure a quarter of patients with LS disease, and most of the patients will eventually succumb to their disease. Despite SCLC’s resistance to conventional therapy and high recurrence rate, a complex genetic landscape provides the basis for effective targeted therapies. The potential roles for several different therapeutic strategies and targeted agents in SCLC have been investigated in recent years. Some of these agents have failed to show a survival advantage in this disease, including BCR-ABL TKIs, mTOR inhibitors, EGFR TKIs, and VEGF inhibitors. In addition, DNA repair inhibitors, antibody-drug conjugates (ADCs), immune therapy with vaccines, cellular development pathway inhibitors, immunomodulators, and immune checkpoint inhibitors are being tested. It is important to note that none of these agents have been approved to be used in SCLC, and most of them are undergoing phase I/II clinical trials, with immune checkpoint inhibitors as the most promising candidates [37].
5.1. Immune Checkpoint Inhibitors in Lung Cancer
In both second- and first-line settings, immunotherapy has demonstrated superior efficacy over chemotherapy alone in treating advanced NSCLC. However, only 20% of patients respond to checkpoint blockade, so novel insights into molecular mechanisms and regimens are needed to improve immunotherapy’s effectiveness. The immune checkpoint inhibitors, combined with chemotherapy, seem to be an effective strategy to prevent tumor cells from evading the immune system through cancer immunoediting. The strategies are: (1) enhance the immune response against tumor cells (immunogenic cell-death), and (2) reduce the immunosuppressive environment around tumors. In combination with chemotherapy, the immune checkpoint inhibitors atezolizumab and pembrolizumab are FDA-approved and recommended already as the first-line treatment for advanced NSCLC. Moreover, as an initial therapeutic approach for metastatic NSCLC, many other chemo-immunotherapeutic regimens have also been evaluated. At the same time, numerous preclinical studies have examined the molecular mechanisms of chemotherapeutic agents used conventionally (antimetabolites, anthracyclines, antimitotic agents, and platinum salts), unraveling effects of drugs and doses/schedules on the immune system that can be exploited for synergistic clinical outcomes [38][39]. The first-line treatment for advanced NSCLC has been changed by immune checkpoint inhibitors (ICIs), which target the PD-1/PD-L1 axis. For patients whose PD-L1 expression is less than 50%, pembrolizumab (a PD-1 inhibitor) is recommended as a monotherapy or combined with chemotherapy. Bevacizumab (an anti-angiogenic antibody) and atezolizumab (PD-L1 inhibitor) can also be used in combination with chemotherapy for first-line treatment of NSCLC regardless of PD-L1 expression. People with high tumor mutational burden (TMB) may also benefit from PD-1/PD-L1 inhibitors in combination with anti-CTLA-4 antibodies for advanced NSCLC compared to platinum-based chemotherapy. For all patients with PD-L1 expression of 1% or more, the FDA has approved the combination of ipilimumab (an anti-CTLA4) and nivolumab (a PD-1 inhibitor). Compared to chemotherapy, immunotherapies alone or in combination with chemotherapy prolong life in people with advanced NSCLC. Chemotherapy may have a higher frequency of side effects than immunotherapy alone. In spite of the widespread use of these antibodies in clinical practice, a few questions remain regarding the best strategy for treatment, the effectiveness of immunotherapy, and the role of different biomarkers in treatment selection, depending on the patient’s clinical characteristics [40][41].
A network meta-analysis (NMA) comparing the efficacy of immune checkpoint inhibitors (ICIs) with or without chemotherapy in metastatic NSCLC patients was conducted based on 12 phase-III studies involving 9236 patients. Combined direct and indirect evidence was analyzed in the NMA, including the results of randomized studies with chemotherapy as the common comparator. Using a frequentist NMA, the hazard ratio (HR) of PFS was estimated. Compared with other treatments studied, chemotherapy combined with pembrolizumab and atezolizumab produced the highest PFS within the overall cohort. Squamous and non-squamous patients both benefited from this superior PFS. Based on non-squamous histology, the pembrolizumab/chemotherapy combination and atezolizumab/bevacizumab/chemotherapy (ABC) provided the best overall survival results in the overall cohort. Again, chemotherapy in combination with atezolizumab or pembrolizumab exhibited significant benefits of PFS, followed by monotherapy using pembrolizumab in patients with high PD-L1 levels. For advanced NSCLC patients, chemotherapy combined with ICIs enhanced treatment efficacy. Compared to chemotherapy alone or any other ICI combination or monotherapy, chemotherapy combined with pembrolizumab or atezolizumab consistently showed higher efficacy in non-squamous cancers [42]. Patients with advanced malignant neoplasms, such as metastatic non-small cell lung cancer (mNSCLC), may benefit from new intermediate endpoints to detect early activity and prioritize new therapies. A study involving more than 150 patients, whose intention-to-treat population was identified, was submitted to the US Food and Drug Administration to examine the milestone rate, an intermediate endpoint for immunotherapy trials. Trial-level milestone ratios were estimated for the overall response rates (ORRs) within 6 months, 9-month PFS rates (PFSs), and 9-month OS rates (OSs). They evaluated the association between milestone ratios and hazard ratios (HRs) using a weighted linear regression model. The Kaplan–Meier survival estimates were compared between the experimental and control arms of trials testing the targeted therapy, immunotherapy, and other therapies. Compared to PFS or 6-month ORR milestones, OS milestones at 12 and 9 months had a moderate association with OS HR. In future trials, however, where immunotherapy may increasingly be the control, new biomarker-enrichment strategies will be deployed, patients with lengthier survival are likely to enroll, and OS HR may not be the optimal time. As a complementary tool or as a secondary outcome in exploratory studies, milestone rates can provide useful information about trial results [43].
5.2. Gene Therapy
In order to treat lung cancer, two novel approaches have been proposed, including gene therapy and immunotherapy. Preclinical data suggest that both treatments may have potential clinical applications. It has been discovered that specific genes are critical to the development of carcinogenesis and that these genes or their products can be targeted for treatment as part of gene therapy programs. As a possible gene therapy strategy, it has been suggested that the adenoviral gene transfer technology (Ad-p53) could be used for the replacement of nonfunctional tumor suppressor genes, like the deleted or mutated p53 genes, with wild-type p53 genes in phase I and phase II lung cancer trials. Direct intratumoral injection and bronchoalveolar lavage have been used in order to achieve transduction of the tumors. By combining Ad-p53 with radiation or perhaps even chemoradiation, these studies have demonstrated a budding role for the radiosensitization of formerly radiation-resistant local tumors [44]. There has been a gradual delineation of the genetic etiology of cancer in the last three decades, but it still has not been completely described. Having a better understanding of the molecular events that take place during the multistep process in bronchogenic carcinogenesis could help researchers to overcome these challenges with greater ease. There has been a great deal of progress made in these three decades when it comes to developing methods for transferring functional genes into mammalian cells. A gene therapy can, for example, prevent the activation of tumor-promoting oncogenes or replace inactivated genes that promote apoptosis or tumor suppression with other tumor-suppressing or apoptosis-promoting genes. It has been discussed by researchers how these molecular changes associated with bronchogenic carcinomas may have therapeutic implications. It has been found that Ras, Erb B-2 (HER-2/neu), Erb B-1 (EGFR), fur, myc, fes, raf, sis, Bcl-2, Bcl-1, and IGF-1 genes may be altered in NSCLC. As far as the therapeutic gene to be transferred is concerned, it falls into one of six categories: RNAi, antisense, or ribozyme sequences against oncogene transcripts; cytokine genes; replacement of tumor suppressor gene; cell surface antigens; suicide genes; as well as multidrug-resistant genes. It is possible to correct the abnormal malignant phenotype by inhibiting the oncogene or by replacing the tumor suppressor gene. Transduced tumor cells would be able to produce toxic metabolites by enzymatically converting otherwise non-toxic substances. It is also possible that the transferred gene will allow cytotoxic drugs to penetrate drug-resistant tumor cells. In order to improve tumor/immune cell interaction and to stimulate the immune response, it would be beneficial if the genes for tumor-specific antigens, MHC molecules, adhesion molecules, co-stimulatory molecules, or cytokine molecules were delivered [45][46].
Apart from immunotherapy and gene therapy, there are a few additional therapeutic approaches developed by researchers. There has been an increase in interest in cancer stem cells (CSCs) in recent years. Essentially, CSCs can self-renew as well as differentiate into different types of cells in order to generate new tumors. Numerous studies report that CSCs mediate tumor recurrence and are resistant to many conventional therapies. A number of markers, such as CD133, CD44, ABCG2, and ALDH1A1, can be used in order to detect CSCs in lung cancer, as well as other characteristics of CSCs, including spheroid formation and colony formation. A potential approach for inhibiting tumor progression would be to target these surface proteins using blocking antibodies and to inhibit ABC transporters and the aldehyde dehydrogenase (ALDH) enzymes utilizing small molecules. There are three signaling cascades that govern the fate of cells during development, the Hh, Notch, and Wnt cascades, and these pathways are involved in the formation of CSCs in a variety of solid tumors. It has also been found that therapeutic approaches can target these signaling pathways in order to inhibit the progression of tumors [47]. A pilot study was conducted to evaluate the efficacy of electrochemical treatment (ECT) as the therapy for 386 patients with NSCLC. In this study, two different ECT methods were employed: firstly, platinum electrodes were introduced transcutaneously inside the tumor under the guidance of X-ray or CT in cases of peripherally located lung cancers. The electrodes were inserted intraoperatively, directly into the tumor for the treatment of central-type lung cancers or for those that could not be operated on during thoracotomy. There were 6–8 V of voltage, 40–100 mA of current, and 100 coulombs of electric charge per cm of tumor diameter. Since the effective area around each electrode was approximately 3 cm in diameter, the number of electrodes was determined by the size of the cancer mass. The clinical results showed that ECT was a simple, safe, and effective therapy with minimal trauma. When lung cancer was conventionally inoperable, unresponsive to chemotherapy or radiotherapy, or unresectable following thoracotomy, ECT offered an alternative treatment option. Further research into ECT is warranted based on the long-term survival rate of treated patients [48]. Despite their potential as cancer therapeutics, microRNAs and siRNAs have been challenging to deliver to most solid tumors. A study showed that a new lung-targeting nanoparticle could deliver miRNA mimics as well as siRNAs to the lung adenocarcinoma cells and tumors in a mouse model of lung cancer whose Kirsten rat sarcoma viral oncogene homolog (Kras) was activated and whose p53 function was lost using genetic engineering. As a result of the therapeutic delivery of miR-34a, a tumor suppressor miRNA that is regulated by p53, miR-34a levels were restored in lung tumors, miR-34a target genes were specifically down-regulated, and tumor growth was slowed. Kras gene expression and MAPK signaling were reduced through siRNA delivery, apoptosis was increased, and tumor growth was inhibited by the delivery of siRNAs targeting Kras. Tumor regression was improved by combining miR-34a with siRNA targeting Kras, compared to either small RNA alone. Further, in this model, chemotherapy plus nanoparticle-based small RNA delivery prolonged survival, compared to chemotherapy alone. As a result of these findings, researchers provided preclinical evidence that small RNA therapies could be used in cancer patients and allowed RNA combination therapy in an autochthonous lung cancer model [49]. Doxorubicin (DOX) and Survivin siRNA have been delivered through a pH-sensitive delivery system to treat metastatic lung cancer. Polyethylenimine-BMPH-DOX (PMD) conjugates were made via a pH-sensitive hydrazine bond (3-aleimidopropionic acid hydrazide, BMPH). B16F10 cells were successfully transfected with DOX and Survivin siRNA and their cytotoxicity was enhanced. There was a preferential accumulation of DOX and siRNA in the lungs of B16F10 tumor-bearing mice after local delivery of PMD/siRNA nanoparticles by pulmonary delivery. In the tumor tissues of the lungs, a considerable amount of DOX and siRNA were observed, whereas limited amounts of DOX and siRNA were observed in the normal tissues of the lungs. Overall, these findings provide a promising strategy for the treatment of metastatic lung cancer by using pulmonary administration as a local delivery method [50]. A schematic representation of the possible killing of the cancer cells by Survivin siRNA is shown in Figure 2.

Figure 2. The delivery of doxorubicin and Survivin siRNA in lung cancer cells using a nanopolymer- based drug delivery system. The drug and siRNA are taken up by the cells via endocytosis. The siRNA makes the RISC complex, thereby degrading the mRNA.
There has been limited success with radiotherapy alone in treating the lung cancer patient population. In a previous study, radiofrequency ablation (RFA) was applied to lung tumors as an image-guided, thermally mediated ablative technique. There has never been a combination therapy that combined both of these treatments before. According to the researchers, a combination of CT-guided radiofrequency ablation along with conventional radiotherapy was used in 24 medically inoperable patients, with a minimum follow-up of 2 years for the survivors. During the course of the treatment, 24 consecutive patients with biopsy-proven, stage I NSCLC who were medically inoperable were treated with CT-guided RFA, followed by 66 Gy of radiotherapy in a dose-dependent manner. RFA was performed using a single or cluster of cool-tip F electrodes, and fluorodeoxyglucose, a radioactive tracer, was used to stage 21 patients before therapy was administered. For patients with medically inoperable stage I NSCLC, RFA followed by conventional radiotherapy may be feasible. Despite the addition of RFA, few procedural complications, and low levels of major toxicities, radiotherapy alone appeared ineffective in controlling local disease and achieving a better survival rate [51].
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA A Cancer J. Clin. 2022, 72, 7–33.
- Doumat, G.; Daher, D.; Zerdan, M.B.; Nasra, N.; Bahmad, H.F.; Recine, M.; Poppiti, R. Drug Repurposing in Non-Small Cell Lung Carcinoma: Old Solutions for New Problems. Curr. Oncol. 2023, 30, 704–719.
- Pallavi, P.; Harini, K.; Crowder, S.; Ghosh, D.; Gowtham, P.; Girigoswami, K.; Girigoswami, A. Rhodamine-Conjugated Anti-Stokes Gold Nanoparticles with Higher ROS Quantum Yield as Theranostic Probe to Arrest Cancer and MDR Bacteria. Appl. Biochem. Biotechnol. 2023, 1–15.
- Gowtham, P.; Girigoswami, K.; Pallavi, P.; Harini, K.; Gurubharath, I.; Girigoswami, A. Alginate-Derivative Encapsulated Carbon Coated Manganese-Ferrite Nanodots for Multimodal Medical Imaging. Pharmaceutics 2022, 14, 2550.
- Soni, A.; Bhandari, M.P.; Tripathi, G.K.; Bundela, P.; Khiriya, P.K.; Khare, P.S.; Kashyap, M.K.; Dey, A.; Vellingiri, B.; Sundaramurthy, S. Nano-biotechnology in tumour and cancerous disease: A perspective review. J. Cell. Mol. Med. 2023, 27, 737–762.
- Jagdale Swati, C.; HableAsawaree, A.; ChabukswarAnuruddha, R. Nanomedicine in lung cancer therapy. Adv. Nov. Formul. Drug Deliv. 2023, 433–448.
- Ou, W.; Stewart, S.; White, A.; Kwizera, E.A.; Xu, J.; Fang, Y.; Shamul, J.G.; Xie, C.; Nurudeen, S.; Tirada, N.P. In-situ cryo-immune engineering of tumor microenvironment with cold-responsive nanotechnology for cancer immunotherapy. Nature Commun. 2023, 14, 392.
- García-Fernández, C.; Fornaguera, C.; Borrós, S. Nanomedicine in non-small cell lung cancer: From conventional treatments to immunotherapy. Cancers 2020, 12, 1609.
- Forde, P.M.; Spicer, J.; Lu, S.; Provencio, M.; Mitsudomi, T.; Awad, M.M.; Felip, E.; Broderick, S.R.; Brahmer, J.R.; Swanson, S.J. Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. N. Engl. J. Med. 2022, 386, 1973–1985.
- Paz-Ares, L.; Ciuleanu, T.-E.; Cobo, M.; Schenker, M.; Zurawski, B.; Menezes, J.; Richardet, E.; Bennouna, J.; Felip, E.; Juan-Vidal, O. First-line nivolumab plus ipilimumab combined with two cycles of chemotherapy in patients with non-small-cell lung cancer (CheckMate 9LA): An international, randomised, open-label, phase 3 trial. Lancet Oncol. 2021, 22, 198–211.
- Wang, J.; Lu, S.; Yu, X.; Hu, Y.; Sun, Y.; Wang, Z.; Zhao, J.; Yu, Y.; Hu, C.; Yang, K. Tislelizumab plus chemotherapy vs chemotherapy alone as first-line treatment for advanced squamous non–small-cell lung cancer: A phase 3 randomized clinical trial. JAMA Oncol. 2021, 7, 709–717.
- West, H.; McCleod, M.; Hussein, M.; Morabito, A.; Rittmeyer, A.; Conter, H.J.; Kopp, H.-G.; Daniel, D.; McCune, S.; Mekhail, T. Atezolizumab in combination with carboplatin plus nab-paclitaxel chemotherapy compared with chemotherapy alone as first-line treatment for metastatic non-squamous non-small-cell lung cancer (IMpower130): A multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2019, 20, 924–937.
- Shu, C.A.; Gainor, J.F.; Awad, M.M.; Chiuzan, C.; Grigg, C.M.; Pabani, A.; Garofano, R.F.; Stoopler, M.B.; Cheng, S.K.; White, A. Neoadjuvant atezolizumab and chemotherapy in patients with resectable non-small-cell lung cancer: An open-label, multicentre, single-arm, phase 2 trial. Lancet Oncol. 2020, 21, 786–795.
- Rizvi, N.A.; Cho, B.C.; Reinmuth, N.; Lee, K.H.; Luft, A.; Ahn, M.-J.; van den Heuvel, M.M.; Cobo, M.; Vicente, D.; Smolin, A. Durvalumab with or without tremelimumab vs standard chemotherapy in first-line treatment of metastatic non–small cell lung cancer: The MYSTIC phase 3 randomized clinical trial. JAMA Oncol. 2020, 6, 661–674.
- Reck, M.; Ciuleanu, T.-E.; Dols, M.C.; Schenker, M.; Zurawski, B.; Menezes, J.; Richardet, E.; Bennouna, J.; Felip, E.; Juan-Vidal, O. Nivolumab (NIVO)+ ipilimumab (IPI)+ 2 cycles of platinum-doublet chemotherapy (chemo) vs 4 cycles chemo as first-line (1L) treatment (tx) for stage IV/recurrent non-small cell lung cancer (NSCLC): CheckMate 9LA. J. Clin. Oncol. 2020, 38 (Suppl. 15), 9501.
- Reck, M.; Rodríguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S. Five-year outcomes with pembrolizumab versus chemotherapy for metastatic non–small-cell lung cancer with PD-L1 tumor proportion score ≥ 50%. J. Clin. Oncol. 2021, 39, 2339.
- Provencio, M.; Nadal, E.; Insa, A.; García-Campelo, M.R.; Casal-Rubio, J.; Dómine, M.; Majem, M.; Rodríguez-Abreu, D.; Martínez-Martí, A.; Carpeño, J.D.C. Neoadjuvant chemotherapy and nivolumab in resectable non-small-cell lung cancer (NADIM): An open-label, multicentre, single-arm, phase 2 trial. Lancet Oncol. 2020, 21, 1413–1422.
- Noronha, V.; Patil, V.M.; Joshi, A.; Menon, N.; Chougule, A.; Mahajan, A.; Janu, A.; Purandare, N.; Kumar, R.; More, S. Gefitinib versus gefitinib plus pemetrexed and carboplatin chemotherapy in EGFR-mutated lung cancer. J. Clin. Oncol. 2020, 38, 124–136.
- Hosomi, Y.; Morita, S.; Sugawara, S.; Kato, T.; Fukuhara, T.; Gemma, A.; Takahashi, K.; Fujita, Y.; Harada, T.; Minato, K. Gefitinib alone versus gefitinib plus chemotherapy for non–small-cell lung cancer with mutated epidermal growth factor receptor: NEJ009 study. J. Clin. Oncol. 2020, 38, 115–123.
- Kris, M.G.; Gaspar, L.E.; Chaft, J.E.; Kennedy, E.B.; Azzoli, C.G.; Ellis, P.M.; Lin, S.H.; Pass, H.I.; Seth, R.; Shepherd, F.A.; et al. Adjuvant Systemic Therapy and Adjuvant Radiation Therapy for Stage I to IIIA Completely Resected Non–Small-Cell Lung Cancers: American Society of Clinical Oncology/Cancer Care Ontario Clinical Practice Guideline Update. J. Clin. Oncol. 2017, 35, 2960–2974.
- Simone II, C.B.; Bogart, J.A.; Cabrera, A.R.; Daly, M.E.; DeNunzio, N.J.; Detterbeck, F.; Faivre-Finn, C.; Gatschet, N.; Gore, E.; Jabbour, S.K. Radiation therapy for small cell lung cancer: An ASTRO clinical practice guideline. Radiat. Oncol. 2020, 10, 158–173.
- Vinod, S.K.; Hau, E. Radiotherapy treatment for lung cancer: Current status and future directions. Respirology 2020, 25, 61–71.
- Videtic, G.M.; Paulus, R.; Singh, A.K.; Chang, J.Y.; Parker, W.; Olivier, K.R.; Timmerman, R.D.; Komaki, R.R.; Urbanic, J.J.; Stephans, K.L. Long-term follow-up on NRG Oncology RTOG 0915 (NCCTG N0927): A randomized phase 2 study comparing 2 stereotactic body radiation therapy schedules for medically inoperable patients with stage I peripheral non-small cell lung cancer. Int. J. Radiat. Oncol. Biol. Phys. 2019, 103, 1077–1084.
- Prezzano, K.M.; Ma, S.J.; Hermann, G.M.; Rivers, C.I.; Gomez-Suescun, J.A.; Singh, A.K. Stereotactic body radiation therapy for non-small cell lung cancer: A review. World J. Clin. Oncol. 2019, 10, 14.
- Cerullo, M.; Lee, H.-J.; Kelsey, C.; Farrow, N.E.; Scales, C.D.; Tong, B.C. Surgical evaluation in patients undergoing radiation therapy for early-stage lung cancer. Ann. Thorac. Surg. 2023, 115, 338–345.
- Singh, A.K.; Gomez-Suescun, J.A.; Stephans, K.L.; Bogart, J.A.; Hermann, G.M.; Tian, L.; Groman, A.; Videtic, G.M. One versus three fractions of stereotactic body radiation therapy for peripheral stage I to II non-small cell lung cancer: A randomized, multi-institution, phase 2 trial. Int. J. Radiat. Oncol. Biol. Phys. 2019, 105, 752–759.
- Ladbury, C.J.; Rusthoven, C.G.; Camidge, D.R.; Kavanagh, B.D.; Nath, S.K. Impact of radiation dose to the host immune system on tumor control and survival for stage III non-small cell lung cancer treated with definitive radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 2019, 105, 346–355.
- D’Andrea, M.A.; Reddy, G.K. Systemic effects of radiation therapy-induced abscopal responses in patients with advanced lung cancer. Oncology 2021, 99, 1–14.
- Finazzi, T.; Palacios, M.A.; Haasbeek, C.J.; Admiraal, M.A.; Spoelstra, F.O.; Bruynzeel, A.M.; Slotman, B.J.; Lagerwaard, F.J.; Senan, S. Stereotactic MR-guided adaptive radiation therapy for peripheral lung tumors. Radiother. Oncol. 2020, 144, 46–52.
- Haslett, K.; Bayman, N.; Franks, K.; Groom, N.; Harden, S.V.; Harris, C.; Hanna, G.; Harrow, S.; Hatton, M.; McCloskey, P. Isotoxic intensity modulated radiation therapy in stage III non-small cell lung cancer: A feasibility study. Int. J. Radiat. Oncol. Biol. Phys. 2021, 109, 1341–1348.
- Or, M.; Liu, B.; Lam, J.; Vinod, S.; Xuan, W.; Yeghiaian-Alvandi, R.; Hau, E. A systematic review and meta-analysis of treatment-related toxicities of curative and palliative radiation therapy in non-small cell lung cancer. Sci. Rep. 2021, 11, 5939.
- Wang, K.; Tepper, J.E. Radiation therapy-associated toxicity: Etiology, management, and prevention. CA A Cancer J. Clin. 2021, 71, 437–454.
- Conibear, J.; Limited, A.U. Rationale for concurrent chemoradiotherapy for patients with stage III non-small-cell lung cancer. Br. J. Cancer 2020, 123, 10–17.
- Borghetti, P.; Branz, J.; Volpi, G.; Pancera, S.; Buraschi, R.; Bianchi, L.N.C.; Bonù, M.L.; Greco, D.; Facheris, G.; Tomasi, C. Home-based pulmonary rehabilitation in patients undergoing (chemo) radiation therapy for unresectable lung cancer: A prospective explorative study. Radiol. Med. 2022, 127, 1322–1332.
- Diamond, B.H.; Verma, N.; Shukla, U.C.; Park, H.S.; Koffer, P.P. Consolidative thoracic radiation therapy after first-line chemotherapy and immunotherapy in extensive-stage small cell lung cancer: A multi-institutional case series. Adv. Radiat. Oncol. 2022, 7, 100883.
- Kubicek, G.J.; Khrizman, P.; Squillante, C.; Callahan, K.; Xu, Q.; Abouzgheib, W.; Boujaoude, Z.; Patel, A.; Hageboutros, A. Stereotactic body radiotherapy and systemic dose chemotherapy for locally advanced lung cancer: Single arm phase 2 study. Am. J. Clin. Oncol. 2022, 45, 129–133.
- Mamdani, H.; Induru, R.; Jalal, S.I. Novel therapies in small cell lung cancer. Transl. Lung Cancer Res. 2015, 4, 533.
- Leonetti, A.; Wever, B.; Mazzaschi, G.; Assaraf, Y.G.; Rolfo, C.; Quaini, F.; Tiseo, M.; Giovannetti, E. Molecular basis and rationale for combining immune checkpoint inhibitors with chemotherapy in non-small cell lung cancer. Drug Resist. Updates 2019, 46, 100644.
- Yang, K.; Li, J.; Bai, C.; Sun, Z.; Zhao, L. Efficacy of immune checkpoint inhibitors in non-small-cell lung cancer patients with different metastatic sites: A systematic review and meta-analysis. Front. Oncol. 2020, 10, 1098.
- Ferrara, R.; Imbimbo, M.; Malouf, R.; Paget-Bailly, S.; Calais, F.; Marchal, C.; Westeel, V. Single or combined immune checkpoint inhibitors compared to first-line platinum-based chemotherapy with or without bevacizumab for people with advanced non-small cell lung cancer. Cochrane Database Syst. Rev. 2020, 12, CD013257.
- Soh, J.; Hamada, A.; Fujino, T.; Mitsudomi, T. Perioperative therapy for non-small cell lung cancer with immune checkpoint inhibitors. Cancers 2021, 13, 4035.
- Dafni, U.; Tsourti, Z.; Vervita, K.; Peters, S. Immune checkpoint inhibitors, alone or in combination with chemotherapy, as first-line treatment for advanced non-small cell lung cancer. A systematic review and network meta-analysis. Lung Cancer 2019, 134, 127–140.
- Blumenthal, G.M.; Zhang, L.; Zhang, H.; Kazandjian, D.; Khozin, S.; Tang, S.; Goldberg, K.; Sridhara, R.; Keegan, P.; Pazdur, R. Milestone analyses of immune checkpoint inhibitors, targeted therapy, and conventional therapy in metastatic non–small cell lung cancer trials: A meta-analysis. JAMA Oncol. 2017, 3, e171029.
- Swisher, S.G.; Roth, J.A.; Carbone, D.P. Genetic and immunologic therapies for lung cancer. Semin. Oncol. 2002, 29, 95–101.
- Toloza, E.M.; Morse, M.A.; Lyerly, H.K. Gene therapy for lung cancer. J. Cell. Biochem. 2006, 99, 1–22.
- Lara-Guerra, H.; Roth, J.A. Gene therapy for lung cancer. Crit. Rev. Oncog. 2016, 21, 115–124.
- Prabavathy, D.; Swarnalatha, Y.; Ramadoss, N. Lung cancer stem cells—Origin, characteristics and therapy. Stem Cell Investig. 2018, 5, 6.
- Xin, Y.l.; Xue, F.z.; Ge, B.s.; Zhao, F.r.; Shi, B.; Zhang, W. Electrochemical treatment of lung cancer. Bioelectromagn. J. Bioelectromagn. Soc. Soc. Phys. Regul. Biol. Med. Eur. Bioelectromagn. Assoc. 1997, 18, 8–13.
- Xue, W.; Dahlman, J.E.; Tammela, T.; Khan, O.F.; Sood, S.; Dave, A.; Cai, W.; Chirino, L.M.; Yang, G.R.; Bronson, R. Small RNA combination therapy for lung cancer. Proc. Natl. Acad. Sci. USA 2014, 111, E3553–E3561.
- Xu, C.; Tian, H.; Wang, P.; Wang, Y.; Chen, X. The suppression of metastatic lung cancer by pulmonary administration of polymer nanoparticles for co-delivery of doxorubicin and Survivin siRNA. Biomater. Sci. 2016, 4, 1646–1654.
- Dupuy, D.E.; DiPetrillo, T.; Gandhi, S.; Ready, N.; Ng, T.; Donat, W.; Mayo-Smith, W.W. Radiofrequency ablation followed by conventional radiotherapy for medically inoperable stage I non-small cell lung cancer. Chest 2006, 129, 738–745.
More
Information
Subjects:
Cell Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
2.3K
Revisions:
2 times
(View History)
Update Date:
06 Jul 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





