
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yves Theoneste Murindangabo | -- | 1860 | 2023-07-04 15:30:44 | | | |
| 2 | Yves Theoneste Murindangabo | + 4 word(s) | 1864 | 2023-07-04 15:33:58 | | |
Video Upload Options
Parameters that determine soil organic matter (SOM) status, soil health, and functions are generally difficult to measure directly. Therefore, they are evaluated by deriving indicators that correlate with soil conditions. Soil condition indicators may be chemical, physical, or biological, and can be either descriptive or quantitative. Descriptive indicators are qualitative and are used in the field, while quantitative indicators are assessed by laboratory analytical procedures. Because total soil organic matter is often not sensitive enough to small and short-term changes due to its complexity levels and background, some studies have recommended using soil organic matter fractions (sub-pools) as more sensitive indicators to detect even small changes over a short period of time. These fractions or sub-pools have been classified by various researchers based on their formation, levels, and ease of decomposition. They include labile, less-stable, and stable fractions. The most labile fraction can decompose in less than a year or two, while the actively decomposing fraction, including partially stabilized organic material from plants and microbial metabolites, may have a turnover of up to 26 years. There is also a chemically stabilized and resistant fraction with a radiocarbon age of up to 2500 years. Quantitative analysis of SOM can be performed using various parameters, including oxidation kinetics, lability, carbon management index, humification degree, humification index, and humification ratio. On the other hand, qualitative evaluation of SOM can involve techniques such as oxidizability, high-performance size-exclusion chromatography, electrospray ionization Fourier transform ion cyclotron resonance mass spectrometry, visual examination, smell, assessment of microorganism content, plant growth, cation exchange capacity, type of organic material, and decomposition. These techniques and parameters provide valuable insights into the characteristics and transformation of SOM, enabling a comprehensive understanding of its dynamics. Evaluating SOM dynamics is of utmost importance as it is a determining factor for soil health, fertility, organic matter stability, and sustainability. Therefore, developing SOM models and other assessment techniques based on soil properties, environmental factors, and management practices can serve as a tool for sustainable management. Long-term or extensive short-term experimental data should be used for modeling to obtain reliable results, especially for quantitative SOM transformation analysis, and changes in the quality and quantity of SOM should be considered when developing sustainable soil management strategies.
1. SOM Fractionation
2. Quantitative Techniques for SOM Measurement
3. Qualitative Techniques for SOM Measurement
4. Lability and Stability of Organic Matter in Soils

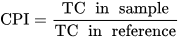
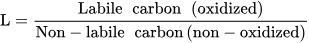
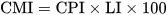
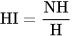

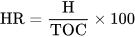
5. Conclusion
The assessment of soil organic matter (SOM) is crucial for understanding soil health, fertility, and sustainability in agricultural systems. Various parameters and indicators have been developed to evaluate SOM status, including quantitative and qualitative techniques. These techniques provide valuable insights into the composition, properties, and functions of SOM.
Quantitative techniques such as dry combustion and wet oxidation methods offer precise measurements of organic carbon content in soil samples. These methods rely on complete combustion or oxidation to determine SOM levels accurately. On the other hand, qualitative techniques involve descriptive evaluation through visual examination, smell tests, and assessments of physical properties like color, texture, and structure. These qualitative indicators can provide valuable information about the quality and characteristics of SOM.
Fractionation of SOM into labile, less-stable, and stable fractions allows for a better understanding of its composition and decomposition rates. Physical, chemical, and biological fractionation methods help in distinguishing different fractions based on their properties and microbial accessibility.
Parameters such as humification index, humification degree, humification ratio, carbon pool index, lability, and carbon management index serve as indicators of SOM stability, humification levels, and carbon cycling. Monitoring changes in these indicators can provide insights into the dynamics of SOM and its response to management practices and environmental factors.
Developing models and assessment techniques based on soil properties, environmental conditions, and management practices can contribute to sustainable soil management. Long-term experimental data and consideration of changes in SOM quality and quantity are crucial for reliable and accurate modeling and analysis of SOM transformation.
References
- Gregorich, E.G.; Beare, M.H.; McKim, U.F.; Skjemstad, J.O. Chemical and Biological Characteristics of Physically Uncomplexed Organic Matter. Soil Sci. Soc. Am. J. 2006, 70, 975–985.
- Golchin, A.; Oades, J.; Skjemstad, J.; Clarke, P. Study of Free and Occluded Particulate Organic Matter in Soils by Solid State 13C Cp/MAS NMR Spectroscopy and Scanning Electron Microscopy. Soil Res. 1994, 32, 285.
- John, B.; Yamashita, T.; Ludwig, B.; Flessa, H. Storage of Organic Carbon in Aggregate and Density Fractions of Silty Soils under Different Types of Land Use. Geoderma 2005, 128, 63–79.
- Scrimgeour, C. Soil Sampling and Methods of Analysis, 2nd ed.; Carter, M.R., Gregorich, E.G., Eds.; CRC Press: Boca Raton, FL, USA, 2008; p. 1224. ISBN 978-0-8593-3586-0.
- Maroušek, J.; Bartoš, P.; Filip, M.; Kolář, L.; Konvalina, P.; Maroušková, A.; Moudrý, J.; Peterka, J.; Šál, J.; Šoch, M.; et al. Advances in the Agrochemical Utilization of Fermentation Residues Reduce the Cost of Purpose-Grown Phytomass for Biogas Production. Energy Sources Part A Recovery Util. Environ. Eff. 2020, 42, 1–11.
- Shirato, Y.; Yokozawa, M. Acid Hydrolysis to Partition Plant Material into Decomposable and Resistant Fractions for Use in the Rothamsted Carbon Model. Soil Biol. Biochem. 2006, 38, 812–816.
- Aranda, V.; Ayora-Cañada, M.J.; Domínguez-Vidal, A.; Martín-García, J.M.; Calero, J.; Delgado, R.; Verdejo, T.; González-Vila, F.J. Effect of Soil Type and Management (Organic vs. Conventional) on Soil Organic Matter Quality in Olive Groves in a Semi-Arid Environment in Sierra Mágina Natural Park (S Spain). Geoderma 2011, 164, 54–63.
- Shibu, M.E.; Leffelaar, P.A.; Van Keulen, H.; Aggarwal, P.K. Quantitative Description of Soil Organic Matter Dynamics—A Review of Approaches with Reference to Rice-Based Cropping Systems. Geoderma 2006, 137, 1–18.
- Bossuyt, H.; Six, J.; Hendrix, P.F. Protection of Soil Carbon by Microaggregates within Earthworm Casts. Soil Biol. Biochem. 2005, 37, 251–258.
- Apesteguia, M.; Plante, A.F.; Virto, I. Methods Assessment for Organic and Inorganic Carbon Quantification in Calcareous Soils of the Mediterranean Region. Geoderma Reg. 2018, 12, 39–48.
- Nelson, D.W.; Sommers, L.E. Total Carbon, Organic Carbon, and Organic Matter. In SSSA Book Series; Soil Science Society of America; American Society of Agronomy: Madison, WI, USA, 2018; pp. 961–1010. ISBN 978-0-89118-866-7.
- Shamrikova, E.V.; Kondratenok, B.M.; Tumanova, E.A.; Vanchikova, E.V.; Lapteva, E.M.; Zonova, T.V.; Lu-Lyan-Min, E.I.; Davydova, A.P.; Libohova, Z.; Suvannang, N. Transferability between Soil Organic Matter Measurement Methods for Database Harmonization. Geoderma 2022, 412, 115547.
- Chang, C.-W.; Laird, D.A.; Mausbach, M.J.; Hurburgh, C.R. Near-Infrared Reflectance Spectroscopy-Principal Components Regression Analyses of Soil Properties. Soil Sci. Soc. Am. J. 2001, 65, 480–490.
- Roberts, C.A.; Workman, J.; Reeves, J.B. (Eds.) Near-Infrared Spectroscopy in Agriculture; Agronomy Monographs; American Society of Agronomy; Crop Science Society of America; Soil Science Society of America: Madison, WI, USA, 2004; ISBN 978-0-89118-236-8.
- Zornoza, R.; Guerrero, C.; Mataix-Solera, J.; Scow, K.M.; Arcenegui, V.; Mataix-Beneyto, J. Near Infrared Spectroscopy for Determination of Various Physical, Chemical and Biochemical Properties in Mediterranean Soils. Soil Biol. Biochem. 2008, 40, 1923–1930.
- Brevik, E.C.; Burgess, L.C. (Eds.) Soils and Human Health; CRC Press: Boca Raton, FL, USA, 2012; ISBN 978-1-4398-4455-7.
- Gregorich, E.G.; Carter, M.R.; Angers, D.A.; Monreal, C.M.; Ellert, B.H. Towards a Minimum Data Set to Assess Soil Organic Matter Quality in Agricultural Soils. Can. J. Soil. Sci. 1994, 74, 367–385.
- Lal, R. Soil Carbon Sequestration to Mitigate Climate Change. Geoderma 2004, 123, 1–22.
- Kopecký, M.; Kolář, L.; Perná, K.; Váchalová, R.; Mráz, P.; Konvalina, P.; Murindangabo, Y.T.; Ghorbani, M.; Menšík, L.; Dumbrovský, M. Fractionation of Soil Organic Matter into Labile and Stable Fractions. Agronomy 2021, 12, 73.
- Váchalová, R.; Borová-Batt, J.; Kolář, L.; Váchal, J. Selectivity of Ion Exchange as a Sign of Soil Quality. Commun. Soil Sci. Plant Anal. 2014, 45, 2673–2679.
- Bongiorno, G. Novel Soil Quality Indicators for the Evaluation of Agricultural Management Practices: A Biological Perspective. Front. Agr. Sci. Eng. 2020, 7, 257.
- Cavalieri-Polizeli, K.M.V.; Marcolino, F.C.; Tormena, C.A.; Keller, T.; de Moraes, A. Soil Structural Quality and Relationships with Root Properties in Single and Integrated Farming Systems. Front. Environ. Sci. 2022, 10, 901302.
- Andriuzzi, W.S. Ecological Functions of Earthworms in Soil; Wageningen University: Wageningen, The Netherlands, 2015; ISBN 978-94-6257-417-5.
- Zhang, G.; Xie, Z. Soil Surface Roughness Decay under Different Topographic Conditions. Soil Tillage Res. 2019, 187, 92–101.
- Steffens, M.; Zeh, L.; Rogge, D.M.; Buddenbaum, H. Quantitative Mapping and Spectroscopic Characterization of Particulate Organic Matter Fractions in Soil Profiles with Imaging VisNIR Spectroscopy. Sci. Rep. 2021, 11, 16725.
- Freitas, V.D.S.; de Babos, D.V.; Guedes, W.N.; Silva, F.P.; de Lima Tozo, M.L.; Martin-Neto, L.; Milori, D.M.B.P.; Villas-Boas, P.R. Assessing Soil Organic Matter Quality with Laser-Induced Fluorescence (LIFS) and Its Correlation to Soil Carbon Stock. In Proceedings of the Latin America Optics and Photonics (LAOP) Conference 2022, Pernambuco, Brazil, 7–11 August 2022; Optica Publishing Group: Recife, Brazil, 2022; p. W3B.5.
- Gao, J.; Liu, L.; Shi, Z.; Lv, J. Characteristics of Fluorescent Dissolved Organic Matter in Paddy Soil Amended with Crop Residues After Column (0–40 Cm) Leaching. Front. Environ. Sci. 2022, 10, 766795.
- Leinweber, P.; Schulten, H.-R. Dynamics of Soil Organic Matter Studied by Pyrolysis—Field Ionization Mass Spectrometry. J. Anal. Appl. Pyrolysis 1993, 25, 123–136.
- Simpson, A.J.; Simpson, M.J. Nuclear Magnetic Resonance Analysis of Natural Organic Matter. In Biophysico-Chemical Processes Involving Natural Nonliving Organic Matter in Environmental Systems; Senesi, N., Xing, B., Huang, P.M., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; pp. 589–650. ISBN 978-0-470-49495-0.
- Brezinski, K.; Gorczyca, B. An Overview of the Uses of High Performance Size Exclusion Chromatography (HPSEC) in the Characterization of Natural Organic Matter (NOM) in Potable Water, and Ion-Exchange Applications. Chemosphere 2019, 217, 122–139.
- Bahureksa, W.; Tfaily, M.M.; Boiteau, R.M.; Young, R.B.; Logan, M.N.; McKenna, A.M.; Borch, T. Soil Organic Matter Characterization by Fourier Transform Ion Cyclotron Resonance Mass Spectrometry (FTICR MS): A Critical Review of Sample Preparation, Analysis, and Data Interpretation. Environ. Sci. Technol. 2021, 55, 9637–9656.
- Kujawinski, E. Electrospray Ionization Fourier Transform Ion Cyclotron Resonance Mass Spectrometry (ESI FT-ICR MS): Characterization of Complex Environmental Mixtures. Environ. Forensics 2002, 3, 207–216.
- Bot, A.; Benites, J. The Importance of Soil Organic Matter: Key to Drought-Resistant Soil and Sustained Food Production; FAO Soils Bulletin; Food and Agriculture Organization of the United Nations: Rome, Italy, 2005; ISBN 978-92-5-105366-9.
- Sainepo, B.M.; Gachene, C.K.; Karuma, A. Assessment of Soil Organic Carbon Fractions and Carbon Management Index under Different Land Use Types in Olesharo Catchment, Narok County, Kenya. Carbon Balance Manag. 2018, 13, 4.
- Blair, G.; Lefroy, R.; Lisle, L. Soil Carbon Fractions Based on Their Degree of Oxidation, and the Development of a Carbon Management Index for Agricultural Systems. Aust. J. Agric. Res. 1995, 46, 1459.
- Bernoux, M.; da Conceição Santana Carvalho, M.; Volkoff, B.; Cerri, C.C. Brazil’s Soil Carbon Stocks. Soil Sci. Soc. Am. J. 2002, 66, 888–896.
- Sisti, C.P.J.; dos Santos, H.P.; Kohhann, R.; Alves, B.J.R.; Urquiaga, S.; Boddey, R.M. Change in Carbon and Nitrogen Stocks in Soil under 13 Years of Conventional or Zero Tillage in Southern Brazil. Soil Tillage Res. 2004, 76, 39–58.
- Plante, A.; Conant, R.T. Soil Organic Matter Dynamics, Climate Change Effects. In Global Environmental Change; Springer: Dordrecht, The Netherlands, 2014; pp. 317–323. ISBN 978-94-007-5783-7.
- Bot, A.; Benites, J. Conservation Agriculture: Case Studies in Latin America and Africa; FAO Soils Bulletin; Food and Agriculture Organization of the United Nations: Rome, Italy, 2001; ISBN 978-92-5-104625-8.





