Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jan Kępczyński | -- | 4088 | 2023-07-04 11:25:53 | | | |
| 2 | Camila Xu | Meta information modification | 4088 | 2023-07-05 03:24:04 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Kępczyński, J.; Kępczyńska, E. Plant-Derived Smoke and Karrikin 1 Activity in Seed. Encyclopedia. Available online: https://encyclopedia.pub/entry/46387 (accessed on 22 May 2026).
Kępczyński J, Kępczyńska E. Plant-Derived Smoke and Karrikin 1 Activity in Seed. Encyclopedia. Available at: https://encyclopedia.pub/entry/46387. Accessed May 22, 2026.
Kępczyński, Jan, Ewa Kępczyńska. "Plant-Derived Smoke and Karrikin 1 Activity in Seed" Encyclopedia, https://encyclopedia.pub/entry/46387 (accessed May 22, 2026).
Kępczyński, J., & Kępczyńska, E. (2023, July 04). Plant-Derived Smoke and Karrikin 1 Activity in Seed. In Encyclopedia. https://encyclopedia.pub/entry/46387
Kępczyński, Jan and Ewa Kępczyńska. "Plant-Derived Smoke and Karrikin 1 Activity in Seed." Encyclopedia. Web. 04 July, 2023.
Copy Citation
Plant-derived smoke and smoke water (SW) can stimulate seed germination in numerous plants from fire-prone and fire-free areas, including cultivated plants and agricultural weeds. Smoke contains thousands of compounds; only several stimulants and inhibitors have been isolated from smoke. Among the six karrikins present in smoke, karrikin 1 (KAR1) seems to be key for the stimulating effect of smoke. The discovery and activity of highly diluted SW and KAR1 at extremely low concentrations (even at ca. 10−9 M) inducing seed germination of a wide array of horticultural and agricultural plants have created tremendous opportunities for the use of these factors in pre-sowing seed treatment through smoke- or KAR1-priming.
smoke-priming
somatic embryogenesis
smoke water
karrikin
KAR1-priming
1. Introduction
After they have been sown, seeds are exposed to various environmental factors influencing seed germination and seedling establishment. These processes seldom take place under optimal conditions. Adverse environmental factors, e.g., extreme temperatures, drought, or high salinity, may slow down germination, prevent uniform germination, and/or cause a low percentage of germination, poor seedling emergence, and irregular plant development, which consequently leads to a reduction in the quality and size of the yield [1][2]. Seed priming technology is an economically successful, viable key strategy to increase crop production under non-stressful and stressful environmental conditions. Conventional seed priming, e.g., hydropriming, osmopriming and matriconditioning, involves seed hydration, allowing metabolism to proceed but preventing radicle protrusion. So far, various priming methods, e.g., hormopriming, gas priming, physical priming, biopriming etc., responsible for the induction of various physio-biochemical traits improving seed vigor, have been developed [2][3] (Figure 1).
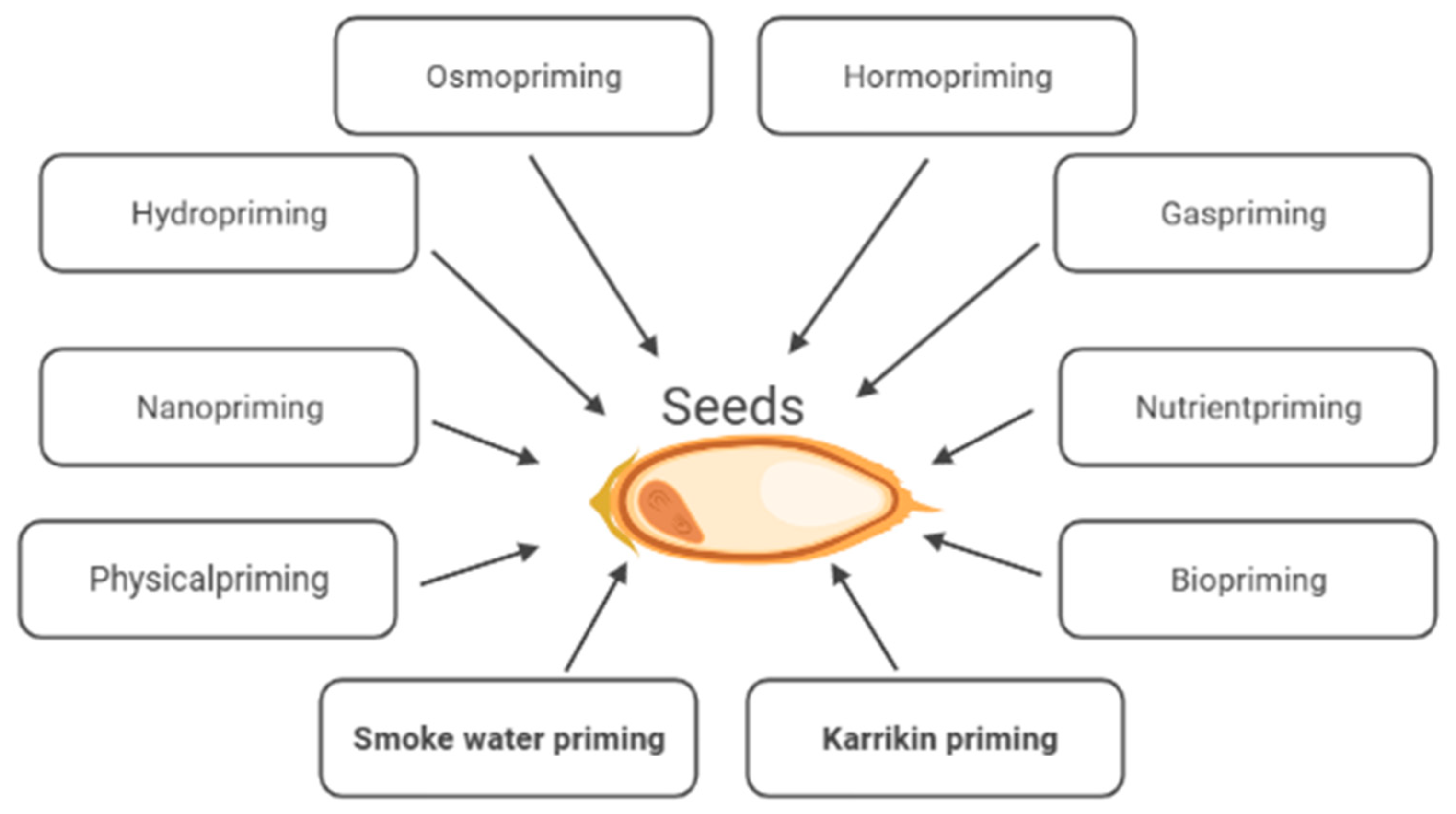
Figure 1. Methods of seed priming.
Since crop cultivation under adverse environmental conditions may reduce yields by up to 50% [4], various cost-effective and easily applicable types of priming should be more widely used, and the search for new pre-sowing techniques should be developed. Demonstrating the ability to stimulate seed germination and seedling growth in many plant crops by plant-derived smoke and smoke-derived KAR1 has generated new opportunities for their use in the pre-sowing treatment of seeds by SW- and KAR1-priming.
2. The Importance of Discovering the Biological Activity of Smoke and Compounds Isolated from It
Having observed that vegetation regenerates after a fire, farmers began to use smoke to treat seeds. In many areas, farmers have used fire and plant-derived smoke to treat seeds to stimulate their germination [5]. Initial data on the effect of smoke on seed germination were published by De Lange and Boucher in 1990 [6]. To those scientists, the discovery of smoke-saturated water (SW) being as effective as smoke alone are owed. This information generated great interest, followed by a large number of publications on the influence of smoke and SW on seed germination in several plant species. The role of smoke in seed germination was also described in reviews [5][7][8][9][10][11][12]. It has been reported that seeds or seedlings of over 1300 plant species respond to smoke or SW [7][12]. Smoke/SW stimulate seed germination in plants derived from areas where fires can occur and crops, vegetables, and weeds [5][9]. Very interesting was the demonstration that SW can be prepared by bubbling, through water, when burning leaves of monocotyledons, dicotyledons and gymnosperms leaves of plants originating not only from fire-prone areas, and also that SW does not lose its activity even after several years of storage [13]. Results of experiments on the beneficial effect of smoke on seed germination and seedling growth were very quickly put into practice by the development of various commercial preparations [14].
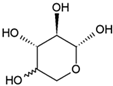
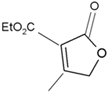
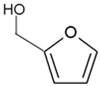
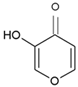
It took many years to search, among thousands of compounds in smoke, for the compound responsible for stimulating smoke-driven germination. The search proved successful only in 2004 when two independent teams isolated butenolide, currently called karrikinolide or karrikin 1 (KAR1), from plant-derived smoke [15] and from burnt cellulose [16] (Figure 2).
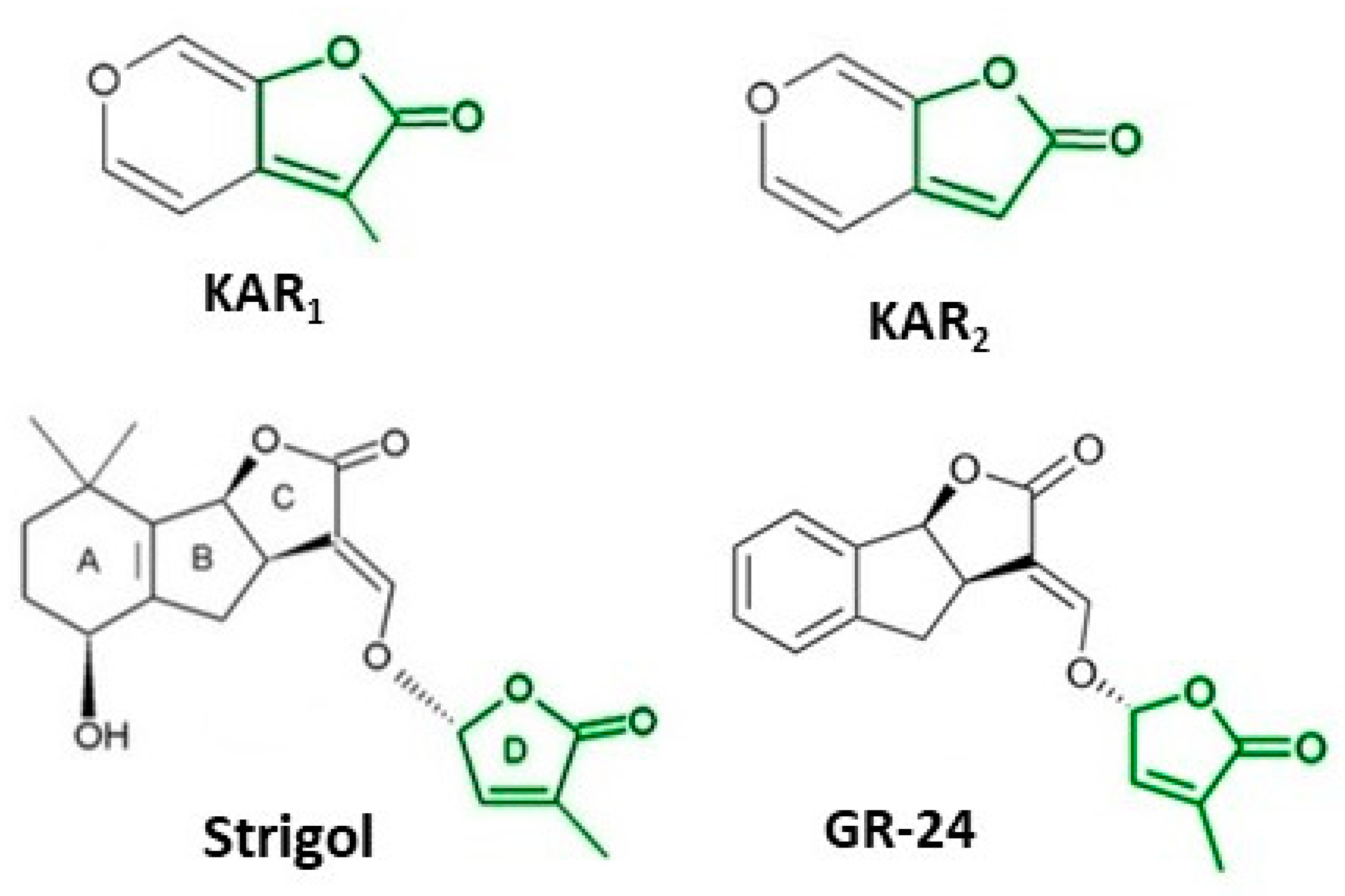
Figure 2. The structure of some bioactive karrikins and strigolactones. KAR1 - 3 methyl-2H-furo[2,3-c]pyran-2-one, most often the most active of the karrikins and found in smoke at the highest concentration; KAR2-karrikin with activity towards Arabidopsis seeds higher than that of KAR1; strigol-natural strigolactone; GR24-synthetic strigolactone. Both karrikins and strigolactones have a butenolide ring. Karrikins and GR24 have similar effects on seed germination, e.g., Arabidopsis and Brassica tournefortii, seedling photomorphogenesis and expression of genes [17][18]. KAR1, in contrast to GR24, could not stimulate the germination of parasitic weed seeds.
Smoke is now known to contain also other karrikins, numbered from KAR1 to KAR6 [8]. KAR1 is usually the most active of all known karrikins; it is present in the highest concentration of smoke, 5.5 to 38 times higher than other karrikins [8]. The KAR1 concentration of 6.7 × 10−7 M was found to correspond to a SW dilution of 1:100 [7]. The compound has been found to stimulate seed germination of plants from fire-prone areas and fire-free regions; the germination of crops and weeds is also stimulated. KAR1 was shown to be very active at extremely low concentrations such as 10−10–10−7 M. Various times of imbibition in a KAR1 solution, e.g., from 1 min for Emmenanthe penduliflora seeds [7] to 6 h for Avena fatua caryopses [19] was sufficient to stimulate germination. KAR1 can be synthesized using different substrates and is commercially available (Table 1).
Table 1. Substrates used for KAR1 synthesis.
Glyceronitrile was reported to appear after fire; in addition to karrikins, ethylene and nitric oxide (NO) are also present in smoke and induce dormancy release in many seeds. They all are recognized as signals in upper soil layers, characteristic of fire and smoke, for dormant seeds to germinate [25] (Table 2).
Table 2. Effects of some compounds present in plant-derived smoke on seed germination. ↑stimulation; ↓ inhibition.
| Compound | Plant Species | References | |
|---|---|---|---|
| Nitrogen dioxide | Emmenanthe penduliflora | ↑ | [26] |
| KAR1 (3-methyl-2H-furo [2, 3-c]pyran-2-one) |
Lactuca sativa | ↑ | [15][16] |
| KAR2, KAR3 | Arabidopsis thaliana | ↑ | [17] |
| KAR4 | Lactuca sativa | ↑ | [27] |
| 3,4,5-trimethylfuran-2(5H)-one (trimethylbutenolide; TMB) | Lactuca sativa | ↓ | [28] |
| Glyceronitrile | Angiozanthos manglesi | ↑ | [29] |
| Hydroquinone | Lactuca sativa | ↑ | [30] |
| 5,5-dimethylfuran-2(5H) -one | Lactuca sativa | ↓ | [31] |
| (5RS) -5-ethylfuran-2(5H) -one | Lactuca sativa | ↓ | [31] |
Seed germination is stimulated by hydroquinone, and seedling growth is improved by catechol, both compounds being detected in smoke as well [30]. Further research has shown that there are seeds which respond to KAR1, but their germination is neither affected nor stimulated by smoke [9]. Moreover, weak—as opposed to strong—SW dilutions inhibit seed germination. Such an effect of SW has been suspected to be related to the presence in the smoke of certain inhibitors antagonistic to karrikins and/or other stimulants. Other experiments revealed the presence of three compounds: 3,4,5-trimethyl furan-2 (5H)-one (TMB), 5,5-dimethylfuran-2(5H)-one and (5RS)-5-ethylfuran-2(5H)-one that inhibited seed germination and exerted an antagonistic effect towards that of KAR1 [28][31]. Thus, the effect of smoke should be considered as a comprehensive biological response to the interacting compounds present in the smoke. Some studies showed TMB to be present in the soil in concentrations higher than those of KAR1 and to be more water-soluble than KAR1 [12]. It has been proposed that TMB accumulated in the soil is diluted and washed away by heavy rainfall, which makes it possible for the stimulatory effect of KAR1 to be expressed. This agrees with a previous suggestion that smoke plays a dual function in regulating seed germination in soil [32].
3. Seed Priming with SW or KAR1 Solution
Since the high effectiveness of SW at high dilutions and KAR1 at low concentrations as stimulants of seed germination and/or seedling development have been demonstrated [8][9][11][33], the agents have become a focus of research on seed priming, a pre-sowing technique. For priming, seeds were imbibed in Petri dishes on filter paper moistened with appropriate solutions of SW or KAR1 or were soaked in solutions of these agents (Table 3).
Table 3. Examples of beneficial effects of SW- or KAR1-priming.
| Plant Species |
SW, Dilution KAR1, M or μg/L−1 |
Methods of Application | Beneficial Effect on: | References |
|---|---|---|---|---|
| Brassica napus L. |
1:1000 10−8 |
imbibition at 25 °C for 12 h, blotting dry |
final percentage, time of germination, at heat stress at 40 °C, CAT activity after 7 d | [34] |
| Brassica napus L. |
1:250 10−7 |
imbibition at 25 °C for 12 h, blotting dry |
growth of seedlings at 25 °C after 7 d | [34] |
| Capsicum annuum L. | 10−7 | imbibition at 25 °C, for 40 h, rinsing, drying at room temp. for 24 h | germination of immature seeds at 25 °C after 10 d, seedling emergence from immature seeds, fresh weight of seedlings from immature and mature seeds at 23 °C after 20 d | [35] |
| Ceratotheca triloba (Bernh.) Hook. | 1:500 10−6 |
imbibition at 25 °C, for 48 h | germination, vigor, seedling growth, at 10/15 °C, 10 °C after 15 d, germination under osmotic stress at 25 °C after 25 d | [36] |
| Coriandrum sativum L. | 10−6 | priming in solution at room temperature for 15 h |
seed germination, seedling growth, chlorophyll a, b, carotenoids and proline contents, membrane stability, leaf osmotic potential, photosynthesis rate, the activity of SOD, POD and CAT at 25 °C after 15 d under cadmium stress | [37] |
| Cucumis melo L. |
10−7 | imbibition at 25 °C for 21 h, rinsing, surface drying |
seedling emergence at 20° and 25 °C, fresh and dry weight of seedling after 24 d |
[38] |
| Daucus carota L. |
51.6 μg/L 1.5 μg/L |
soaking for 12 h, rinsing, drying |
germination in soil, seedling growth at environmental conditions, photosynthesis, ascorbic acid content after 120 d | [39] |
| Eragrostis tef (Zucc.) Trotter | 1:500 10−8 |
imbibition for 48 h at 25 °C | vigor at 20°to 40 °C, seedling length and vigor index, germination at 25–40 °C, at 25 °C under osmotic stress | [40] |
| Lycopersicon esculentum Mill. | 10−7 | soaking at 23 °C for 24 h, drying up to the initial weight |
rate of germination, vigor seedling in water at 10°–35 °C, in salt solutions or PEG solutions at 23 °C after seven days | [41] |
| Oryza sativa L. | 1:1000 1:500 |
primed in solutens for 24 h, air drying at room temperature |
seed germination up to 3 d under salt stress simulated by NaCl at 30 °C, chlorophyll, carotenoids, K+, Ca + contents | [42] |
| Silybum marianum L. Gaertn. Solanum centrale J.M.Black, S.dioicum W.Fitzg, S. orbiculatum Dunal ex Poir |
1:250 1:10 0.67μM |
soaking 1 h at room temperature, drying, soaking for 24 h, rinsing |
speed of germination, vigor, at 25 °C index seedling length after 14 d germination at 26/13 °C, 33/18 °C on water agar, vigor index seedling length after 18 d |
[43] [44] |
| Themeda triandra Forssk. | SW | soaking 1 h at room temperature, drying, and stored at 25 for 3 to 21 days |
germination at 25 °C | [45] |
| Zea mays L. | 1:500 | soaking 6–18 h | seedling growth at 28 °C after 8 d, chlorophyll and carotenoids contents | [46] |
| Zea mays L. | 1:500 10−7 |
soaking 1 h, surface drying | seedling growth in the soil after 30 d in greenhouse conditions | [47] |
Then, the SW- or KAR1-primed seeds were surface dried or dried to air dry level and sown immediately or after storage in Petri dishes or soil. Subsequently, germination, seedling emergence, seedling growth and/or certain indicators of metabolism were determined. SW- or KAR1-priming were applied to seeds of natural plants in the revegetation strategy and to improve seedling emergence and the development of cultivated plants. Early experiments with Themeda triandra, a dominant fire-climax grass, showed seeds imbibed in an SW solution and then dried and stored at 25 °C to germinate better than untreated seeds [45]. The cited authors rightly concluded that such seed pre-treatment could be used to revegetate the plant. Later, other promising results were obtained from research on SW application to prime seeds of cultivated plants. Germination of Solanum centrale, S. dioicum and S. orbiculatum seeds, vigor index and 18-d old seedling length were found to increase when the seeds were imbibed in SW and immediately transferred to Petri dishes with agar [44]. However, it is not known whether the stimulating effect of SW-priming would also persist after seed drying and storage. In another experiment, soaking of Silybum marianum seeds, a plant native to Europe, Asia, and Africa, at present cultivated for the pharmaceutical industry [43] in SW, despite drying, enhanced the germination speed at 25 °C, vigor index and 14-days old seedling length. Soaking Dactylis glomerata seeds in SW solution and drying them before sowing in the field increased germination, seedling emergence under field conditions and biomass after ten weeks of crop cultivation [48]. That beneficial effects can also appear under environmental conditions was an important information. In another experiment, soaking of Zea mays seeds in SW increased the rate of seed germination and fresh weight of shoots and roots in 8-day-old seedlings [46]. Moreover, chlorophyll and carotenoid contents were higher in seedlings obtained from the primed seeds.
Because SW-priming positively affected seed germination, seedling emergence and/or seedling development, experiments were carried out to explore the effects of KAR1-priming or both SW- and KAR1-priming, in one of the experiments, the sowing of Zea mays kernels soaked in an SW or KAR1 solution, followed by surface drying, increased the plant height, root and shoot weight and dry weight after 30 days of greenhouse cultivation [47], indicating the advantages of both SW- and KAR1-priming. However, whether this positive effect persisted until the end of plant development and whether it was reflected in the yield is unknown. Doubtless, the use of primed seeds in commercial practice would be enhanced if positive effects were not reduced due to the post-treatment seed drying and drying and storage. Therefore, it was essential to find out if seeds subjected to KAR1-priming could be stored for some time without losing the beneficial priming effect. In one experiment, the effect of priming on the development of plants after cultivation under environmental conditions was addressed using Daucus carota. SW and KAR1 were applied during the soaking of D. carota seeds, air-dried before sowing, increased germination in soil and seedling growth after 120 days of growth under natural conditions [39]. Moreover, both priming techniques increased the photosynthesis rate and the contents of carotene and ascorbic acid. Capsicum annuum seeds, primed with KAR1 by soaking, showed improved germination of both immature and mature seeds. The technique worked better in immature prime than hydropriming [35]. Likewise, an advantageous effect of KAR1-priming was observed in 20-day-old C. annuum seedlings when the fresh weight of the seedlings grown on a peat moss medium was determined. The stimulatory effect of KAR1 also included an increase in catalase (CAT), ascorbate peroxidase (APX) and superoxide dismutase (SOD) activities in both immature and mature seeds.
So far, various priming technologies have been successfully used to enhance resistance to abiotic stresses, consistently improving overall plant growth [1]. SW and KAR1 have been demonstrated to have a positive effect not only on seed germination and seedling development under optimal conditions but also under stress [11], so it was logical to conduct research aimed at finding out whether SW- and/or KAR1-priming can be used to increase plant tolerance to various abiotic stresses. However, only limited, albeit important, information has been obtained so far through experiments focusing on using both above-priming techniques to improve seed germination, seedling emergence and establishment under various stress conditions. Extreme temperatures, too low or too high, are known to have an adverse impact on seed germination and plant development by disrupting metabolism, damaging structures, and ultimately leading to the death of cells, organ tissues and even the entire organism. Priming Lycopersicon esculentum by soaking the seeds in a KAR1 solution increased the seedling vigor after 7-day incubation at various temperatures, including sub- and supraoptimal [41].
Moreover, KAR1-priming enhanced seedling vigor under salt and osmotic stress—the cultivation of Brassica napus cv. English giant, an annual herbaceous leafy vegetable, can be limited due to the high temperature and water stress. SW-priming and, to a larger extent, KAR1-priming increased seed germination at heat stress (40 °C) [34].
Likewise, SW- and KAR1-priming by imbibing seeds of Eragrostis tef, a major cereal crop in the Horn of Africa countries, were used to test germination and vigor of 7-day-old seedlings at various temperatures, including 40 °C, and at osmotic stress [40]. The treatment improved both germination and seedling vigor. Similarly, SW-priming of Oryza sativa seeds increased seed germination and seedling vigor under salt stress simulated by NaCl solutions [42]. Effects of priming included an increase in the chlorophyll and carotenoid contents. In addition, priming increased the contents of K+ and Ca+, but decreased that of Na+. In Ceratotheca triloba, SW- and KAR1-priming increased seed germination after 25 days under osmotic stress caused by PEG 6000 solutions [36]. Both priming techniques also improved the growth of 25-day-old seedlings under stress. The effect of KAR1-priming on the alleviation of stress caused by cadmium, a pollutant prevalent in arable lands, was studied on seeds of Coriandrum sativum, an important herbaceous plant cultivated in various regions of the world and used in pharmacy, cosmetology, and various cuisines as a spice [37]. Cadmium was found to inhibit seed germination and to reduce the fresh and dry weight of 15-day-old seedlings. The inhibitory effect declined when primed seeds were used. The beneficial effect of priming on cadmium stress tolerance was associated with increased leaf osmotic potential, membrane stability, photosynthesis rate and proline content. Moreover, KAR1-priming resulted in decreased contents of malondialdehyde (MDA) and hydrogen peroxide (H2O2) and electrolyte leakage. In contrast, antioxidant enzymes: superoxide dismutase (SOD), peroxidase (POD) and catalase (CAT) in seedlings were found to enhance their activity. The effect of KAR1-priming of Cucumis melo seeds sown, after surface drying, in peat moss at two depths and kept at 20 and 25 °C in the controlled climatic room for 24 days, on seedling emergence and seedling fresh and dry weight was also examined [38]. The KAR1-priming advantage was particularly evident if the seeds were of poorer quality and were sown too deep at a sub-optimal temperature of 20 °C. KAR1-priming was observed to increase seedling emergence, speed, and final percentage, and seedling fresh weight at 20 °C more effectively than at 25 °C when the seeds were sown deeper.
It is probably still long before SW and/or KAR1 will be commercially applied in priming technology. One can only consider the potential use of SW- and KAR1-priming in increasing seed tolerance to adverse environmental factors. So far, research has been limited to assessing the effects on germination and seedling development, often on Petri dishes. The impact of both types of priming on plant development and yield under environmental conditions in most cases has not been determined. In the experiments, SW- or KAR1-primed seeds were most often used immediately after the treatment or only after surface drying, and it is known that other priming techniques and KAR1-priming diminish the seed priming benefit as a result of drying. Perhaps a solution would be to dry the seeds gradually, similarly to the drying manner recommended for somatic embryos.
Considering the practical application of SW or KAR1 in priming, if the seeds are sensitive to SW, SW-priming seems more valuable because the SW preparation is easy and cost-free. Although KAR1 is expensive due to its effectiveness at very low concentrations, its use is also promising. Knowledge of KAR1-primed seed metabolism is still scant. Application of SW and/or KAR1 in combination with other conditioning methods, e.g., osmopriming or matripriming, to seeds of economically important plants requires further research.
4. SW and KAR1 in Seed Biotechnology
The use of both SW and KAR1 in vitro cultures of various plant materials has been a focus of several studies. As fire and smoke were found to be capable of inducing flowering in some plants, e.g., Cyrtanthus ventricosus and Watsonia borbonica [49][50], experiments were conducted to follow the response of pollen of various plants from fire-prone areas. SW or KAR1 were demonstrated to stimulate pollen germination and tube growth of various plant species; therefore, it was correctly concluded that both agents could increase flower pollination, leading to increased fertilization and seed yield [51][52]. However, no data are showing the effect on the seed yield. It was very interesting and extremely important to discover that, under natural conditions and in vitro, it is possible to generate embryos from somatic cells without fertilization. Somatic embryogenesis, which makes it possible to produce artificial seeds, is of great interest to both the researchers striving to explain the process and the practitioners who could use the process for the mass multiplication of plants. Application of somatic embryogenesis in practice requires optimalisation of the process for the sake of high efficiency, particularly with respect to cotyledonary embryos, the most advanced stage of embryo development. Experiments were carried out to examine the suitability of SW and KAR1 in improving somatic embryogenesis (Table 4).
Table 4. Effect of SW and KAR1 in tissue cultures. + stimulation; − no effect; ND- no data.
| Process | Plant Species | SW | KAR1 | References |
|---|---|---|---|---|
| Somatic embryogenesis Efficiency |
Baloskion tetraphyllum Pelargonium hortorum Pinus wallichiana |
ND + + |
+ ND ND |
[53] [54] [55] |
| Somatic embryo germination | Baloskion tetraphyllum Pinus wallichiana |
ND + |
+ ND |
[53] [55] |
| Conversion to plantlets | Baloskion tetraphyllum Brassica napus Pinus wallichiana |
ND + + |
+ ND ND |
[53] [56] [55] |
| Secondary embryogenesis | Brassica napus | + | ND | [57] |
| Seed germination | Ansellia africana Baloskion tetraphyllum Vanda parviflora Xenikophyton smeeanum |
+ ND + + |
− + ND ND |
[58] [53] [59] [60] |
However, only a few examples of SW or KAR1 are being applied to improve somatic embryogenesis. SW was shown to increase embryogenesis efficiency in Pelargonium hortorum [54] markedly. Important insights were gained, including the observation that SW increased the formation of cotyledonary embryos. When SW was applied to the hypocotyl explant or during the induction phase, the number of cotyledonary embryos increased fivefold. In Pinus wallichiana, SW used in the induction medium increased the number of cotyledonary embryos by about 3.5 [55]. SW was also found to increase the occurrence of secondary embryogenesis in Brassica napus [57]. These data may suggest that karrikin in the smoke was likely responsible for its stimulatory effect. It is important to note that KAR1 accelerated the development of torpedo embryos in Baloskion tetraphyllum [53]. Knowledge of the effect of SW and KAR1 on somatic embryo germination and conversion to plantlets is incomplete. An experiment with P. wallichiana showed SW to stimulate the germination of cotyledonary somatic embryos and to increase the number of surviving seedlings [55]. Likewise, KAR1 improved both somatic embryo germination and plantlet development in B. tetraphyllum [53]. Since SW and KAR1 were initially known as inducers of in vivo seed germination, it is understandable that they were applied to find out if they would be useful in inducing the in vitro seed germination. One of the methods of orchid production involves the sowing of seeds. Therefore, using agents that improve in vitro orchid seed germination is appropriate. SW stimulated the germination of asymbiotic seeds, protocorm differentiation and plant regeneration in Vanda parviflora, an epiphytic orchid [59]. Also, seed germination and plant recovery of the epiphytic orchid Xenikophyton smeeanum and Tulbaghia ludwigiana, a popular garden plant, was increased by SW [60].
Similarly, SW stimulated seed germination and the formation of protocorms in the epiphytic orchid Ansellia africana [58]. However, KAR1 could not induce germination or protocorm formation, which indicates that the SW activity can be associated with compounds other than karrikin(s) or other stimulant(s) present in SW. Seeds of different plant species, e.g., Aloe arborescens, were stimulated to germinate by SW [61]. Therefore, it has been demonstrated that SW could be used in orchid and aloe propagation. An in vitro experiment involving Balsamorhiza deltoidea and B. sagittata, plants from fire-prone areas of America, showed that, like in seeds of some plant species, KAR2—in contrast to—KAR1 was active in vivo as a stimulant [62].
Data on using both SW and KAR1 in seed biotechnology are promising, although too scant. There are no examples of KAR1 interaction with plant hormones in the regulation of somatic embryogenesis. The effects of the compound on the level of phytohormones are unknown, and the mechanism of in vitro treatments that considers the molecular level requires research.
5. Mechanism of Karrikin Signalling
Recently, great progress in elucidating the karrikin signaling mechanism has been observed (Figure 3).
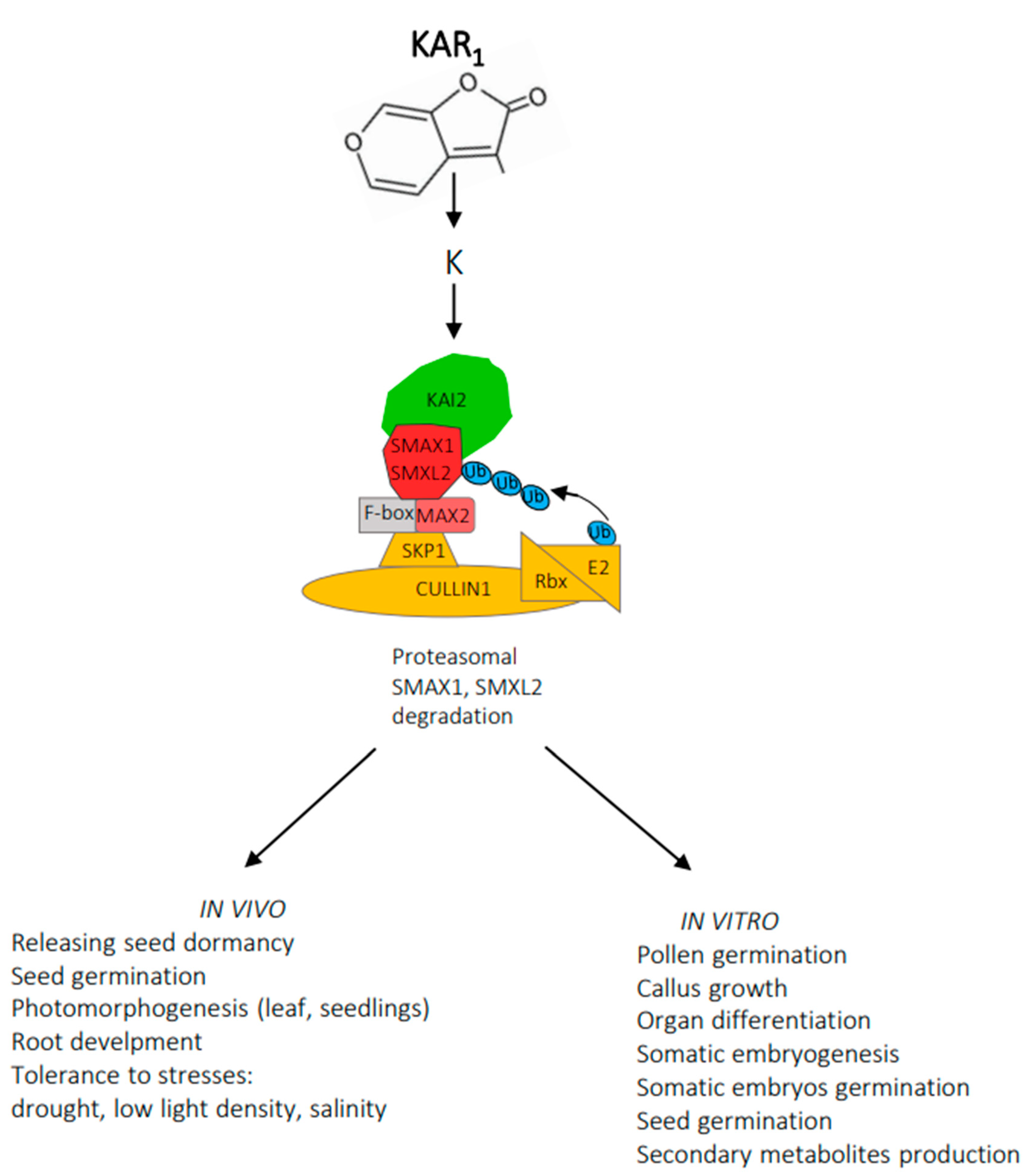
Figure 3. A proposed mechanism of KAR1 signalling. KAI2 (KARRIKIN INSENSITIVE2)-α,β hydrolases performing enzyme and receptor functions; K -a putative karrikin-derived molecule; F-box –recognizes substrate; Skp1 –substrate adaptor; Cullin –regulates complex activity; Rbx –facilitates transfer ubiquitin (Ub); E2 –transfers the ubiquitin to the substrate.
Several studies indicate that KAR1 must be metabolized for an answer to this compound to emerge (63). KAR1 is metabolized to K (a putative karrikin-derived molecule). Activation of KAI2 (KARRIKIN INSENSITIVE2)-α,β hydrolases by K enables interaction with MAX2, an F-box protein part of Skp1-Cullin-F-box (SCF) E3 ubiquitin ligase complex. MAX2 recognizes SMAX1 (SUPPRESSOR OF MAX2), and SMXL2 (SMAX1-LIKE2), proteins that prevent the appearance of a response to KAR1. SMAX1 and SMXL2 proteins undergo polyubiquitination, followed by proteosomal degradation leading to the emergence of a response characteristic of KAR1 [63]. Fascinating is the great similarity in the signalling mechanism of both butenolide molecules, karrikins, unidentified in plants, and plant hormones, strigolactones [63][64]. MAX2 also mediates responses to strigolactones, which activate D14 (DWARF14)- α,β hydrolases structurally similar to KAI2 [18][65]. D14 acts with MAX2 to target SMXL6, SMXL7 and SMXL8 for ubiquitination and degradation. D14 can also target SMAX1 and SMXL2 when an adequate agonist is present. Moreover, there are also similarities in the signaling mechanism of karrikins and plant hormones such as auxins, jasmonates and gibberellins. All response systems involve the Skp1/Cullin/F-box-E3 ubiquitin ligase complex and the ubiquitination of the regulatory protein and its degradation by the 26S proteosome. Studies that involve karrikin and plant hormones pathways will not be referred to here; an extensive review of Blázquez and coworkers provides ample and adequate information [66].
References
- Jisha, K.C.; Vijayakumari, K.; Puthur, J.T. Seed priming for abiotic stress tolerance: An overview. Acta Physiol. Plant 2013, 35, 1381–1396.
- Garcia, D.; Zhao, S.; Arif, S.; Zhao, Y.; Ming, L.C.; Huang, D. Seed Priming technology as a key strategy to increase crop plant production under adverse environmental conditions. J. Agri. Horti. Res. 2022, 5, 27–46.
- Kępczyński, J. Gas-priming as a novel simple method of seed treatment with ethylene, hydrogen cyanide or nitric oxide. Acta Physiol. Plant 2021, 43, 117.
- Farooq, M.; Usman, M.; Nadeem, F.; ur Rehman, H.; Wahid, A.; Basra, S.M.; Siddique, K.H. Seed priming in field crops: Potential benefits, adoption and challenges. Crop Pasture Sci. 2019, 70, 731–771.
- Kulkarni, M.G.; Light, M.E.; Van Staden, J. Plant-derived smoke: Old technology with possibilities for economic applications in agriculture and horticulture. S. Afr. J. Bot. 2011, 77, 972–979.
- De Lange, J.H.; Boucher, C. Auto ecological studies on Audinia capitata (Bruniaceae). I. Plant-derived smoke as a germination cue. S. Afr. J. Bot. 1990, 56, 700–703.
- Dixon, K.W.; Merritt, D.J.; Flematti, G.R.; Ghisalberti, E.L. Karrikinolide–a phytoreactive compound derived from smoke with applications in horticulture, ecological restoration and agriculture. Acta Hort. 2009, 813, 155–170.
- Nelson, D.C.; Flematti, G.R.; Ghisalberti, E.L.; Dixon, K.W.; Smith, S.M. Regulation of seed germination and seedling growth by chemical signals from burning vegetation. Ann. Rev. Plant Biol. 2012, 63, 107–130.
- Kępczyński, J. Induction of agricultural weed seed germination by smoke and smoke-derived karrikin (KAR 1), with a particular reference to Avena fatua L. Acta Physiol. Plant 2018, 40, 87.
- Kępczyński, J. Progress in utilizing plant-derived smoke water and smoke-derived KAR1 in plants tissue culture. Plant Cell Tiss. Organ Cult. 2020, 140, 271–278.
- Banerjee, A.; Tripathi, D.K.; Roychoudhury, A. The Karrikin ‘calisthenics’: Can compounds derived from smoke help in stress tolerance? Physiol. Plant 2019, 165, 290–302.
- Soós, V.; Badics, E.; Incze, N.; Balázs, E. Fire-borne life: A brief review of smoke-induced germination. Nat. Prod. Commun. 2019, 14, 1–5.
- Brown, N.A.C.; Van Staden, J. Smoke as a germination cue: A review. Plant Growth Regul. 1997, 22, 115–124.
- Light, M.E.; van Staden, J. The potential of smoke in seed technology. S. Afr. J. Bot. 2004, 70, 97–101.
- Van Staden, J.; Jager, A.K.; Light, M.E.; Burger, B.V. Isolation of the major germination cue from plant-derived smoke. S. Afr. J. B 2004, 70, 654–659.
- Flematti, G.R.; Ghisalberti, E.L.; Dixon, K.W.; Trengove, R.D. A compound from smoke that promotes seed germination. Science 2004, 305, 977.
- Nelson, D.C.; Riseborough, J.A.; Flematti, G.R.; Stevens, J.; Ghisalberti, E.L.; Dixon, K.W.; Smith, S.M. Karrikins discovered in smoke trigger Arabidopsis seed germination by a mechanism requiring gibberellic acid synthesis and light. Plant Physiol. 2009, 149, 863–873.
- Nelson, D.C.; Scaffidi, A.; Dun, E.A.; Waters, M.T.; Flematti, G.R.; Dixon, K.W.; Beveridge, C.A.; Ghisalberti, E.L.; Smith, S.M. F-box protein MAX2 has dual roles in karrikin and strigolactone signaling in Arabidopsis thaliana. Proc. Nat. Acad. Sci. USA 2011, 108, 8897–8902.
- Kępczyński, J.; Cembrowska, D.; Van Staden, J. Releasing primary dormancy in Avena fatua L. caryopses by smoke-derived butanolide. Plant Growth. Regul. 2010, 62, 85–91.
- Goddard-Borger, E.D.; Ghisalberti, E.L.; Stick, R.V. Synthesis of the Germination Stimulant 3-Methyl-2H-furo pyran-2-one and Analogous Compounds from Carbohydrates. Eur. J. Org. Chem. 2007, 23, 3925–3934.
- Matsuo, K.; Shindo, M. Efficient synthesis of karrikinolide via Cu (II)-catalyzed lactonization. Tetrahedron 2011, 67, 971–975.
- Sun, K.; Chen, Y.; Wagerle, T.; Linnstaedt, D.; Currie, M.; Chmura, P.; Xu, M. Synthesis of butenolides as seed germination stimulants. Tetrahedron Lett. 2008, 49, 2922–2925.
- Nagase, R.; Katayama, M.; Mura, H.; Matsuo, N.; Tanabe, Y. Synthesis of the seed germination stimulant 3-methyl-2H-furo pyran-2-ones utilizing direct and regioselective Ti-crossed aldol addition. Tetrahedron Lett. 2008, 49, 4509–4512.
- Flematti, G.R.; Gavin, R.; Ghisalberti, E.L.; Dixon, K.W.; Trengove, R.D. Synthesis of the seed germination stimulant 3-methyl-2H-furopyran-2-one. Tetrahedron Lett. 2005, 46, 5719–5721.
- Flematti, G.R.; Waters, M.T.; Scaffidi, A.; Merritt, D.J.; Ghisalberti, E.L.; Dixon, K.W.; Smith, S.M. Karrikin and cyanohydrin smoke signals provide clues to new endogenous plant signaling compounds. Mol. Plant. 2013, 6, 29–37.
- Keeley, J.E.; Fotheringham, C.J. Trace gas emissions in smoke-induced germination. Science 1997, 276, 1248–1250.
- Flematti, G.R.; Goddard-Borger, E.D.; Merritt, D.J.; Ghisalberti, E.L.; Dixon, K.W.; Trengove, R.D. Preparation of 2 H-furo pyran-2-one derivatives and evaluation of their germination-promoting activity. J. Agr. Food Chem. 2007, 55, 2189–2194.
- Light, M.E.; Burger, B.V.; Staerk, D.; Kohout, L.; van Staden, J. Butenolides from plant-derived smoke: Natural plant-growth regulators with antagonistic actions on seed germination. J. Nat. Prod. 2010, 73, 267–269.
- Flematti, G.R.; Merritt, D.J.; Piggott, M.J.; Trengove, R.D.; Smith, S.M.; Dixon, K.W.; Ghisalberti, E.L. Burning vegetation produces cyanohydrins that liberate cyanide and stimulate seed germination. Nat. Commun. 2011, 2, 360.
- Kamran, M.; Khan, A.L.; Ali, L.; Hussain, J.; Waqas, M.; Al-Harrasi, A.; Imran, Q.M.; Kim, Y.H.; Kang, S.M.; Yun, B.W.; et al. Hydroquinone: A novel bioactive compopund from plant-derived smoke can cue seed germination of lettuce. Front. Chem. 2017, 5, 30.
- Burger, B.V.; Pošta, M.; Light, M.E.; Kulkarni, M.G.; Viviers, M.Z.; Van Staden, J. More butenolides from plant-derived smoke with germination inhibitory activity against karrikinolide. S. Afr. J. Bot. 2018, 115, 256–263.
- Light, M.E.; Gardner, M.J.; Jager, A.K.; van Staden, J. Dual regulation of seed germination by smoke solutions. Plant Growth Regul. 2002, 37, 135–141.
- Khatoon, A.; Rehman, S.F.; Aslam, M.M.; Jamil, M.; Komatsu, S. Plant-derived smoke affects biochemical mechanism on plant growth and seed germination. Inter. J. Mol. Sci. 2020, 21, 7760.
- Moyo, M.; Amoo, S.O.; Van Staden, J. Seed priming with smoke water and karrikin improves germination and seedling vigor of Brassica napus under varying environmental conditions. Plant Growth Regul. 2022, 97, 315–326.
- Demir, I.; Ozden, E.; Yıldırım, K.C.; Sahin, O.; Van Staden, J. Priming with smoke-derived karrikinolide enhances germination and transplant quality of immature and mature pepper seed lots. S. Afr. J. Bot. 2018, 115, 264–268.
- Masondo, N.A.; Kulkarni, M.G.; Finnie, J.F.; Van Staden, J. Influence of biostimulants-seed-priming on Ceratotheca triloba germination and seedling growth under low temperatures, low osmotic potential and salinity stress. Ecotoxicol. Environ. Saf. 2018, 147, 43–48.
- Sardar, R.; Ahmed, S.; Yasin, N.A. Seed priming with karrikinolide improves growth and physiochemical features of Coriandrum sativum under cadmium stress. Environ. Adv. 2021, 5, 100082.
- Mavi, K.; Light, M.E.; Demir, I.; Van Staden, J.; Yasar, F. Positive effect of smoke-derived butenolide priming on melon seedling emergence and growth. N. Z. J. Crop Hort. Sci. 2010, 38, 147–155.
- Akeel, A.; Khan, M.M.A.; Jaleel, H.; Uddin, M. Smoke-saturated Water and Karrikinolide Modulate Germination, Growth, Photosynthesis and Nutritional Values of Carrot (Daucus carota L.). J. Plant Growth Regul. 2019, 38, 1387–1401.
- Ghebrehiwot, H.M.; Kulkarni, M.G.; Kirkman, K.P.; Van Staden, J. Smoke-water and a smoke-isolated butenolide improve germination and seedling vigour of Eragrostis Tef (Zucc.) Trotter under high temperature and low osmotic potential. J. Agron. Crop Sci. 2008, 194, 270–277.
- Jain, N.; Van Staden, J. The potential of the smoke-derived compound 3-methyl-2H-furopyran-2-one as a priming agent for tomato seeds. Seed Sci. Res. 2007, 17, 175–181.
- Jamil, M.; Kanwal, M.A.; Aslam, M.M.; Khan, S.; Tu, J.; Réhman, S.U. Effect of plant-derived smoke priming on physiological and biochemical characteristics of rice under salt stress condition. Austr. J. Crop Sci. 2014, 8, 159–170.
- Abdollahi, M.R.; Mehrshad, B.; Moosavi, S.S. Effect of method of seed treatment with plant derived smoke solutions on germination and seedling growth of milk thistle (Silybum marianum L.). Seed Sci. Technol. 2011, 39, 225–229.
- Commander, L.E.; Merritt, D.J.; Rokich, D.P.; Flematti, G.R.; Dixon, K.W. Seed germination of Solanum spp. (Solanaceae) for use in rehabilitation and commercial industries. Aust. J. Bot. 2008, 56, 333–341.
- Baxter, B.J.M.; Van Staden, J. Plant-derived smoke: An effective seed pre-treatment. Plant Growth Regul. 1994, 14, 279–282.
- Aslam, M.M.; Jamil, M.; Khatoon, A.; Hendawy, S.E.; Al-Suhaibani, N.A.; Malook, I.; Rehman, S.U. Physiological and biochemical responses of maize (Zea mays L.) to plant derived smoke solution. Pak. J. Bot. 2017, 49, 435–443. Available online: https://w.w.w.researchgate.net/publications/315808314 (accessed on 31 May 2023).
- Van Staden, J.; Sparg, S.G.; Kulkarni, M.G.; Light, M.E. Post-germination effects of the smoke-derived compound 3-methyl-2H-furo pyran-2-one, and its potential as a preconditioning agent. Field Crops Res. 2006, 98, 98–105.
- Abu, Y.; Romo, J.T.; Bai, Y.; Coulman, B. Priming seeds in aqueous smoke solution to improve seed germination and biomass production of perennial forage species. Can. J. Plant Sci. 2016, 96, 551–563.
- Keeley, J.E. Smoke-induced flowering in the fire-lily Cyrtanthus ventricosus. S. Afr. J. Bot. 1993, 59, 638.
- Kulkarni, M.G.; Ascough, G.D.; Van Staden, J. Improved flowering of a South African Watsonia with smoke treatment. S. Afr. J. Bot. 2007, 73, 298.
- Papenfus, H.B.; Kumari, A.; Kulkarni, M.G.; Finnie, J.F.; Van Staden, J. Smoke-water enhances in vitro pollen germination and tube elongation of three species of Amaryllidaceae. S. Afr. J. Bot. 2014, 90, 87–92.
- Kumari, A.; Papenfus, H.B.; Kulkrni, M.G.; Pošta, M.; van Staden, J. Efect of smoke derivatives on in vitro pollen germination and pollen tube elongation of species from diferent plant families. Plant Biol. 2015, 17, 825–830.
- Ma, G.-H.; Bunn, E.; Dixon, K.; Flematti, G. Comparative enhancement of germination and vigor in seed and somatic embryos by the smoke chemical 3-methyl-2H-furo pyran-2-one in Baloskion tetraphyllum (Restionaceae). Vitr. Cell Dev. Biol. 2006, 42, 305–308.
- Senaratna, T.; Dixon, K.; Bunn, E.; Touchell, D. Smoke-saturated water promotes somatic embryogenesis in geranium. Plant Growth Regul. 1999, 28, 95–99.
- Malabadi, R.B.; Nataraja, K. Smoke-saturated water infuences somatic embryogenesis using vegetative shoot apices of mature trees of Pinus wallichiana AB Jacks. J Plant Sci. 2007, 2, 45–53.
- Ghazanfari, P.; Abdollahi, M.R.; Moieni, A.; Moosavi, S.S. Efect of plant-derived smoke extract on in vitro plantlet regeneration from rapeseed (Brassica napus L. cv. Topas) microspore-derived embryos. Int. J. Plant Prod. 2012, 6, 1735–6814.
- Abdollahi, M.R.; Ghazanfari, P.; Corral-Martínez, P.; Moieni, A.; Seguí-Simarro, J.M. Enhancing secondary embryogenesis in Brassica napus by selecting hypocotyl- derived embryos and using plant-derived smoke extract in culture medium. Plant Cell Tissue Organ Cult. 2012, 110, 307–315.
- Papenfus, H.B.; Naidoo, D.; Pošta, M.; Finnie, J.F.; Van Staden, J. The efects of smoke derivatives on in vitro seed germination and development of the leopard orchid Ansellia africana. Plant Biol. 2016, 18, 289–294.
- Malabadi, R.B.; da Silva, J.A.T.; Mulgund, G.S. Smoke-saturated water influences in vitro seed germination of Vanda parviflora Lindl. Seed Sci. Biotechnol. 2008, 2, 65–69.
- Malabadi, R.B.; Vijaykumar, S.; da Silva, J.A.T.; Mulgund, G.S.; Nataraja, K. In vitro seed germination of an epiphytic orchid Xenikophyton smeeanum (Reichb. f.) by using smoke-saturated-water as a natural growth promoter. Int. J. Biol. Technol. 2011, 2, 35–40.
- Espinosa-Leal, C.A.; Garcia-Lara, S. In vitro germination and initial seedling development of krantz aloe by smoke-saturated water and seed imbibition. Hort Technol. 2020, 30, 619–623.
- Monthony, A.S.; Baethke, K.; Erland, L.A.; Murch, S.J. Tools for conservation of Balsamorhiza deltoidea and Balsamorhiza sagittata: Karrikin and thidiazuron-induced growth. Vitr. Cell. Develop. Biol.-Plant. 2020, 56, 398–406.
- Waters, M.T.; Nelson, D.C. Karrikin perception and signalling. New Phytol. 2023, 237, 1525–1541.
- Morffy, N.; Faure, L.; Nelson, D. Smoke and hormone mirrors:action and evolution of Karrikin and strigolactone signaling. Trends Genet. 2016, 3, 176–188.
- Bürger, M.; Mashiguchi, K.; Lee, H.J.; Nakano, M.; Takemoto, K.; Seto, Y.; Yamaguchi, S.; Chory, J. Structural. basis of Karrikin and non-natural strigolactone perception in Physcomitrella patens. Cell Rep. 2019, 26, 855–865.
- Blázquez, M.A.; Nelson, D.C.; Weijers, D. Evolution of plant hormone response pathways. Annu. Rev. Plant. Biol. 2020, 71, 327–353.
More
Information
Subjects:
Plant Sciences
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
05 Jul 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No