Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Juan Miguel Gonzalez | -- | 2554 | 2023-06-30 15:34:12 | | | |
| 2 | Rita Xu | Meta information modification | 2554 | 2023-07-03 04:43:13 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Gonzalez, J.M.; Santana, M.M.; Gomez, E.J.; Delgado, J.A. Soil Thermophiles and Their Extracellular Enzymes. Encyclopedia. Available online: https://encyclopedia.pub/entry/46280 (accessed on 09 May 2026).
Gonzalez JM, Santana MM, Gomez EJ, Delgado JA. Soil Thermophiles and Their Extracellular Enzymes. Encyclopedia. Available at: https://encyclopedia.pub/entry/46280. Accessed May 09, 2026.
Gonzalez, Juan M., Margarida M. Santana, Enrique J. Gomez, José A. Delgado. "Soil Thermophiles and Their Extracellular Enzymes" Encyclopedia, https://encyclopedia.pub/entry/46280 (accessed May 09, 2026).
Gonzalez, J.M., Santana, M.M., Gomez, E.J., & Delgado, J.A. (2023, June 30). Soil Thermophiles and Their Extracellular Enzymes. In Encyclopedia. https://encyclopedia.pub/entry/46280
Gonzalez, Juan M., et al. "Soil Thermophiles and Their Extracellular Enzymes." Encyclopedia. Web. 30 June, 2023.
Copy Citation
During this century, a number of reports have described the potential roles of thermophiles in the upper soil layers during high-temperature periods. Soil thermophiles are able to adapt their extracellular enzyme activities to environmental conditions. These enzymes can present optimum activity under high temperatures and reduced water content. Consequently, these microorganisms have been shown to actively process and decompose substances (including pollutants) under extreme conditions (i.e., desiccation and heat) in soils.
thermophiles
soil
extracellular enzyme activity
enzyme persistence
1. Introduction
Soil health and function are strictly linked to microbial activity [1][2][3][4][5]. A large number of processes are carried out mainly or exclusively by microorganisms, and this broad range of activities represents a major asset for soil maintenance and response to multiple variables leading to changing conditions. One of the main factors influencing the functional redundancy of soil processes is microbial diversity [1][5][6][7][8]. Soils are highly heterogeneous, and they present a huge microbial diversity and abundance [9][10]. Current estimates suggest that 1 g of soil contains about 1010 prokaryotic cells and includes about 30,000 different microorganisms [7][9][11]. The duplicity of metabolic capabilities allows soils to preserve functionality, maintaining a stable environment, in spite of drastic changes. Otherwise, a significant decrease in microbial diversity would represent a serious handicap on soils being able to maintain current balances and activities, which would negatively affect soil health and productivity [5][7].
Within the research on the almost unmeasurable high microbial diversity existing in soils, most work has been carried out on their major components, while the low-abundance microorganisms have been poorly considered. This represents a significant limitation because a large number of microbial processes with high relevance to, for instance, the biogeochemical cycling of elements are performed by minority microorganisms. This is, for example, the case for ammonium oxidation, denitrification, metal reduction/oxidation, sulfur oxidation, sulfate reduction, methanogenesis and the decomposition of specific recalcitrant pollutants; these processes are generally carried out by groups represented within the minorities of the natural microbial communities [12].
Within the vast microbial diversity of soils, a permanent component consists in thermophilic bacteria. Although thermophiles are expected to inhabit high-temperature environments, such as hot springs, geothermal areas or compost piles, different reports have confirmed the ubiquitous presence of thermophiles in all examined soils from a wide range of latitudes [13][14][15][16][17][18]. Cold and temperate soils hold thermophiles including distinctive taxa, but the major representatives are Geobacillus-related genera [13][14][15][16][18]. This study will focus on the potential role and consequences of these soil thermophiles within a perspective of global climate warming.
A question has arose about the timing available for thermophiles to grow, assuming these microorganisms are inhabiting the upper soil layers of cold or temperate environments. Previously, an analysis of average number of hot days against latitude [15][18][19] suggested the occurrence of a significant number of hot days (e.g., above 100 hot days/year, around 37° N, in Seville, Spain) [18][19] when soil thermophiles would have an opportunity to grow and show significant activity. At higher latitudes (i.e., above 50° N), the number of hot days is generally low (e.g., around 1–2 hot days/year, around 52° N, at Cambridge, UK) [18][19], but this could be enough to provide time for maintaining thermophile populations and a minimum extracellular enzyme stock in the soil environment. These soil thermophiles survive well through low-temperature periods [20].
Another group of microorganisms that must be mentioned when considering microorganisms thriving under periods of increased temperatures are the thermophilic fungi [21] also present in soils. The role of these fungi in soils during high-temperature events has not been clearly defined yet [21], and additional research is required. Nevertheless, thermophilic fungi can have a role in compost piles, where high temperatures are maintained for much longer time periods than in upper soil layers. Compost piles typically contain a high organic matter load and generate, as a result of microbial growth, a high-temperature environment [22][23][24][25].
Soil organic matter is a major reservoir of C with the potential to greatly influence global climate [3][4]. Most organic carbon is present in the upper soil layers [26]. Rich soils contain a high content of organic matter, represented by a variety of complex compounds. Soil organic matter, besides its carbon content, also includes other elements, some of them critical for plant and microbial growth, such as nitrogen, sulfur and phosphorous, often required as major soil fertilizers [27][28]. Furthermore, complex organics such as humic acids can complex with those elements as well as with metals [29][30]. Within the soil organic matter, large polymers and humic acids need to be broken into smaller compounds or monomers for microorganisms to be able to be taken up and processed as sources of energy and/or biomass [31][32][33]. This breaking down of complex compounds into smaller ones is mediated by extracellular enzymes. In fact, the bottle-neck for soil organic matter mineralization is this step involving the extracellular enzymes [3][19][34]. Microbial extracellular enzyme activity is highly related to organic matter mineralization in soils and has been proposed as a major indicator to evaluate the sink-link issue with the soil–atmosphere C balance [3][4], a critical parameter for modeling climate predictions [5][35][36][37][38].
Extracellular enzyme activity has been proposed as an indicator of soil microbial activity [39][40][41], and it is commonly measured in ecological studies [37][40][41][42][43][44]. Soil thermophiles have been reported as a major source for extracellular enzymes dominating the pool of enzymes in soils [19] because they present higher total activity than the corresponding enzymes from mesophiles (Figure 1) [18][19][45]. Thus, high-temperature events are expected to enhance soil organic matter processing due to the activation of extracellular enzymes from thermophiles. In addition, the current scenario of global warming suggests an expected increase in frequency and duration of high-temperature events in the coming years [4][5][7][46]. As well, high temperature in soil upper layers implies an increased evaporation and therefore a decrease in water content in soils, leading to increased desiccation. Recent reports have also proposed that some soil microorganisms (including some thermophiles) have adapted to dryness by developing extracellular enzymes and metabolisms able to work optimally under dry conditions (at water activity, aw, between 0.3 and 0.8) [43][45]. The levels of desiccation showing maximum extracellular activity by soil thermophilic xerophiles can occur at values below the reported limit for microbial growth (aw 0.605) [47]. Besides extracellular enzyme activity by thermophiles, these cells have been reported to actively decompose recalcitrant pollutants at high temperature [17][48][49][50][51] and under dry conditions [43][45][48], suggesting that cell activity is significant under those extreme conditions in soil upper layers, where these microorganisms can be potential important bioremediation agents [17][48][49][50][51][52][53].
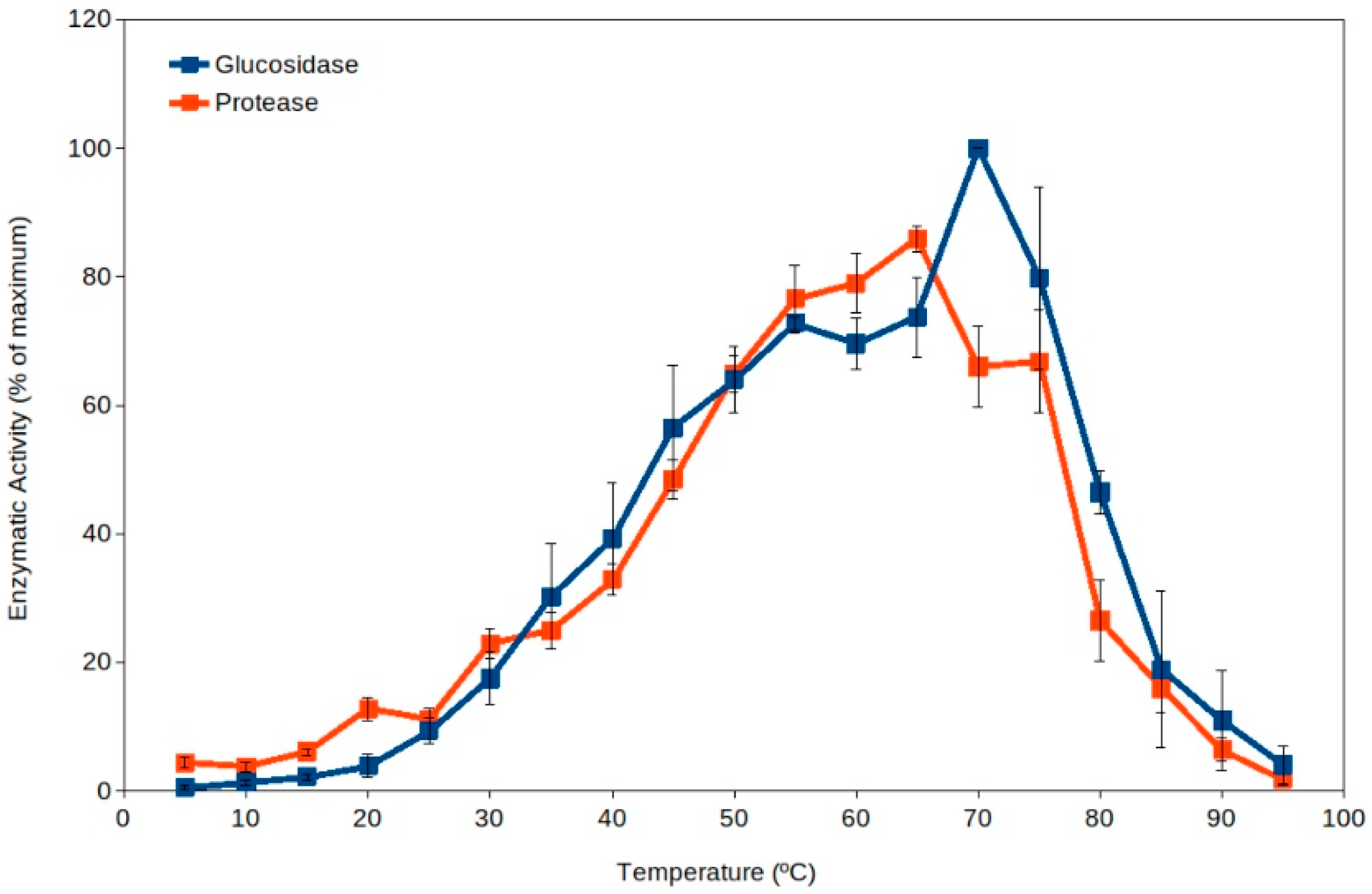
Figure 1. Extracellular enzyme activity in soils over a broad range of temperatures (5 °C to 95 °C). Maximum activity was observed at temperatures in the thermophilic microorganisms range (55 °C–75 °C).
2. Singularity of Soil Thermophiles and Thermophilic Extracellular Enzymes
Soil thermophiles represent a singularity in the microbial communities from cold and temperate soils. However, thermophiles are ubiquitous inhabitants of soils, and their presence as viable cells with an important role and capabilities to survive under those conditions have been reported [14][15]. Thus, thermophiles thriving in temperate environments, depending on periodic/sporadic high-temperature events, can show some growth and produce extracellular enzymes that are required to process organic matter [18][19]. These extracellular enzymes might persist in the environment [54] and become active under heat events. Consequently, these extracellular enzymes actively participate in soil organic matter decomposition [19][43][54].
Mesophilic microorganisms present extracellular enzymes with optimum activity at moderate temperatures (i.e., generally measured around 30 °C or below) [55][56][57], but the extracellular enzymes from thermophiles present optimum activity at temperatures above 50 °C [19][45]. Thus, the activity by extracellular enzymes from thermophiles can be easily detected by carrying out enzyme assays at high temperature (50 °C to 70 °C), and therefore, mesophilic and thermophilic activities can be differentiated. Results discriminating the activity over a range of temperatures (from 5 °C to 95 °C) in a variety of soil samples showed unexpected results [19]. The results clearly indicated that thermophilic activities were always higher than mesophilic ones in all soils tested (Figure 1) [18][19]. This suggests that thermophilic extracellular enzyme activities are dominant in soils. It is important to note that the pioneering work [19] and some subsequent studies [43][45] included samples from soils exposed to hot temperatures and others from cold environments. Additional studies have shown significant roles of thermophiles at higher latitudes [16][17][49][50][51][52], corroborating that soil thermophiles can also show significant environmental activity in relatively cold climate zones.
Because soil thermophiles represent a minority fraction of the total microbial community in cold and temperate soils [13][15][18][43], it is required to look for different potential scenarios to explain that large activity measured in the thermophile temperature range. An easy explanation would be that soil thermophiles show a very high production of extracellular enzymes and/or these enzymes present higher activity than their mesophilic counterparts. Although enzymes from thermophiles have been reported to present higher activity than those from mesophiles [58][59], the difference is not likely to be able to explain the much higher total activity measured in soils due to thermophiles (a minority group) than due to the total mesophilic microbial community, which presents a much higher abundance. According to previous estimates [15], the fraction of thermophiles in soils is generally below 1% of the total community. A potential justification for that large activity at high temperatures in soils could be that soil thermophiles could produce a large amount of extracellular enzymes during hot periods or extreme heat events. Soil thermophiles, such as Geobacillus related taxa, require extracellular enzymes to access complex organic matter in soils and grow, so a high production of enzymes is needed for their development. Nevertheless, the relatively low abundance of thermophiles suggests that the production of thermophilic extracellular enzymes could not be as high as needed—in relationship to the production by mesophiles showing equivalent metabolism—to explain the higher activity in the thermophile temperature range. The level of extracellular enzyme production that could be potentially needed to explain that scenario is likely to be out of reach for the soil thermophilic cells. Otherwise, during hot periods, thermophiles could respond to heat events by growing and producing a moderate amount of enzymes that would persist in the environment over time. This could result in a progressive accumulation of thermophilic enzymes in the environment. These enzymes should be able to persist in the environment at least until the next hot event, and, at that moment, the thermophilic enzymes will show their full activity. For this to be a reasonable explanation, the thermophilic extracellular enzymes should show longer persistence in the soil environment. The potential for accumulation of extracellular enzymes in soils could represent a singular strategy that would allow thermophilic cells to start growing rapidly when the right growth conditions arrive in the ecosystem (e.g., an extreme heat event or hot days during the summer period). A rapid response would take advantage of even the shortest periods at high temperature to recover and grow. During these hot periods, those thermophilic enzymes could decompose soil organic matter, allowing a variety of organisms (both microorganisms and plants) to profit from that release of smaller compounds (and monomers) readily available as substrates for growth and energy [60]. A mechanism facilitating a rapid growth response to high-temperature events (i.e., extracellular enzyme accumulation) represents an interesting adaptive feature for thermophiles to thrive in cold and temperate environments. This type of strategy has not been reported before for microorganisms.
For extracellular enzymes to accumulate in the environment, two aspects need to be fulfilled: a relatively long production of extracellular enzymes and a long persistence in the environment. Generally, enzymes from thermophiles (as well as from other extremophiles) have been reported to present higher durability than those from mesophiles, resulting from a higher stability and resistance to external factors (detergents, denaturants, decomposition, etc.) [58]. Assuming enzymes are produced at a relatively high rate and then show a long persistence in the environment, they could progressively accumulate in soils [54]. In this way, thermophilic extracellular enzymes could generate an active enzymatic pool in soils, readily available to catalyze complex organic matter decomposition as soon as temperatures rise. A recent report [54] has shown that extracellular enzymes from thermophiles are able to persist for a longer time in soils than those from mesophiles (Figure 2). Thermophilic extracellular enzymes maintain their activity in soils even at the highest temperatures and desiccation levels reached in the upper soil layers (Figure 2). Mesophilic enzymes are rapidly denatured during extreme heat events, including summer periods, and their persistence is lower than that for soil thermophiles [54][61]. Thus, the extracellular enzymes from thermophiles are able to persist in the environment, representing a soil asset that could allow the rapid growth of microorganisms, both mesophiles and thermophiles, and so promote soil health and functioning. This unique strategy proposed for soil thermophiles represents a singular mechanism to survive in environments (i.e., cold and temperate soils) considered adverse for thermophiles.
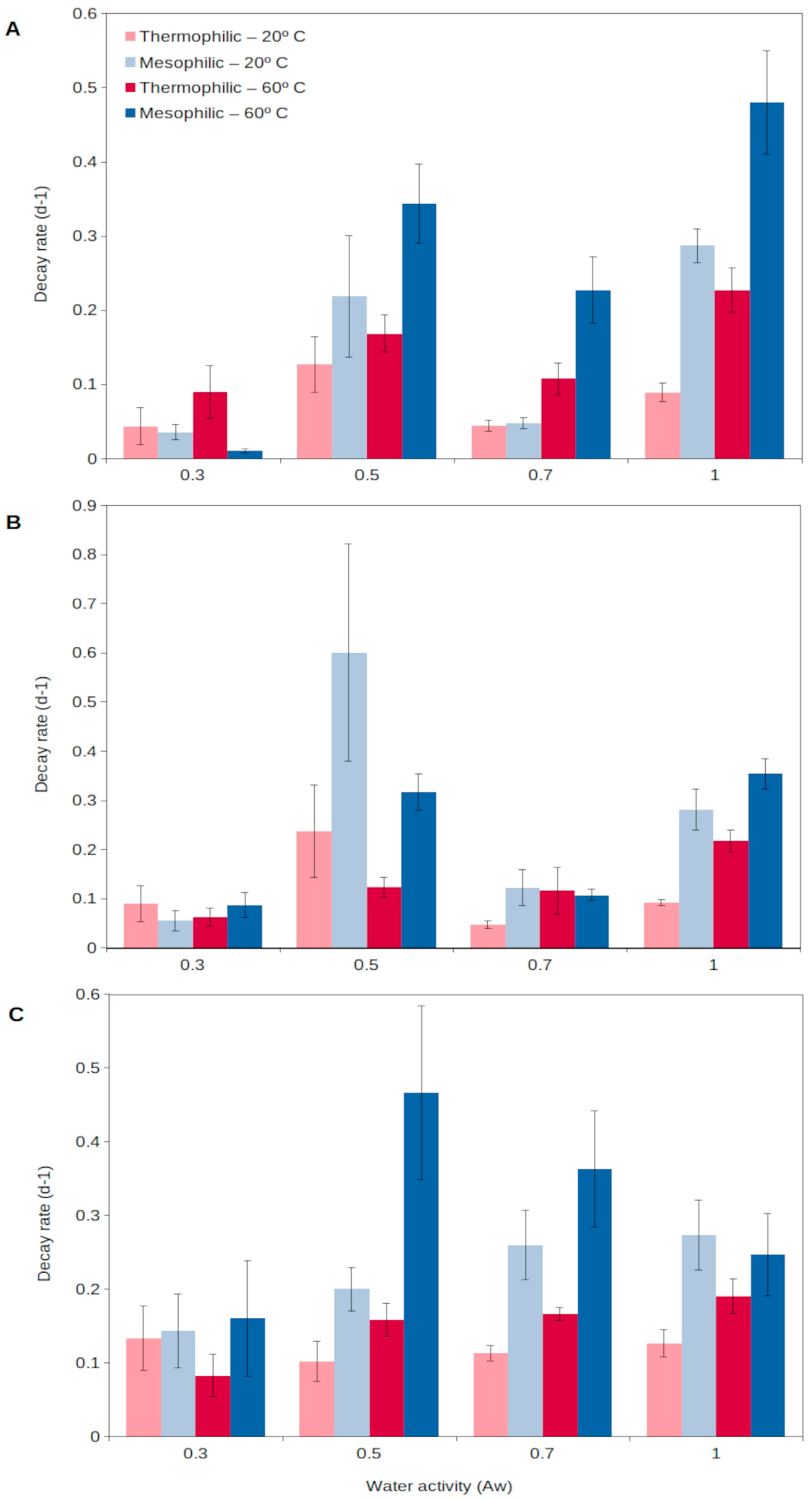
Figure 2. Decay rate of natural extracellular enzymes in soils at different temperatures, 20 °C (light colors) and 60 °C (dark colors), corresponding to the mesophilic (blue) and thermophilic (red) microorganisms, respectively, over a wide spectrum of water contents (water activity, aw) for 3 different enzyme activities: (A), glucanase; (B), phosphatase; (C), protease. High decay rate indicates short persistence, and low decay rate shows long persistence.
High temperature of upper soil layers implies increased evaporation, leading to a reduction in water content and desiccation. It has been generally thought that dried soils present poor or near-null biological activity. Nevertheless, recent work has shown that specific microorganisms present optimum extracellular enzyme activity under dry conditions [43][45][62][63]. This is the case for some soil thermophiles, among other mesophilic bacteria (i.e., Deinococcus). Deinococcus radiodurans, a wide spread soil bacterial species, has been reported to be a model microorganism for resistance to desiccation [64], and some of its extracellular enzymes can present optimum (maximum) activity under dried soil conditions (aw 0.40–0.55) (Figure 3) [45]. Soil thermophiles, those inhabitants of soils frequently exposed to high temperatures and droughts, exhibit an interesting feature: they present optimum extracellular enzyme activities at very low water content (aw 0.3–0.7). Some thermophilic enzymes from sites exposed to hot climate have been shown to reach optimum activities under dry conditions (aw < 0.7), but thermophiles from cooler locations present optima under wet conditions (aw > 0.9). Most mesophilic enzymes in those natural soil samples always presented optimum values in aqueous solutions (aw > 0.9). These results showed that soil thermophiles can adapt to thrive in a variety of environments and have the capacity to adapt their enzyme activities to extremely dry conditions [43][45].
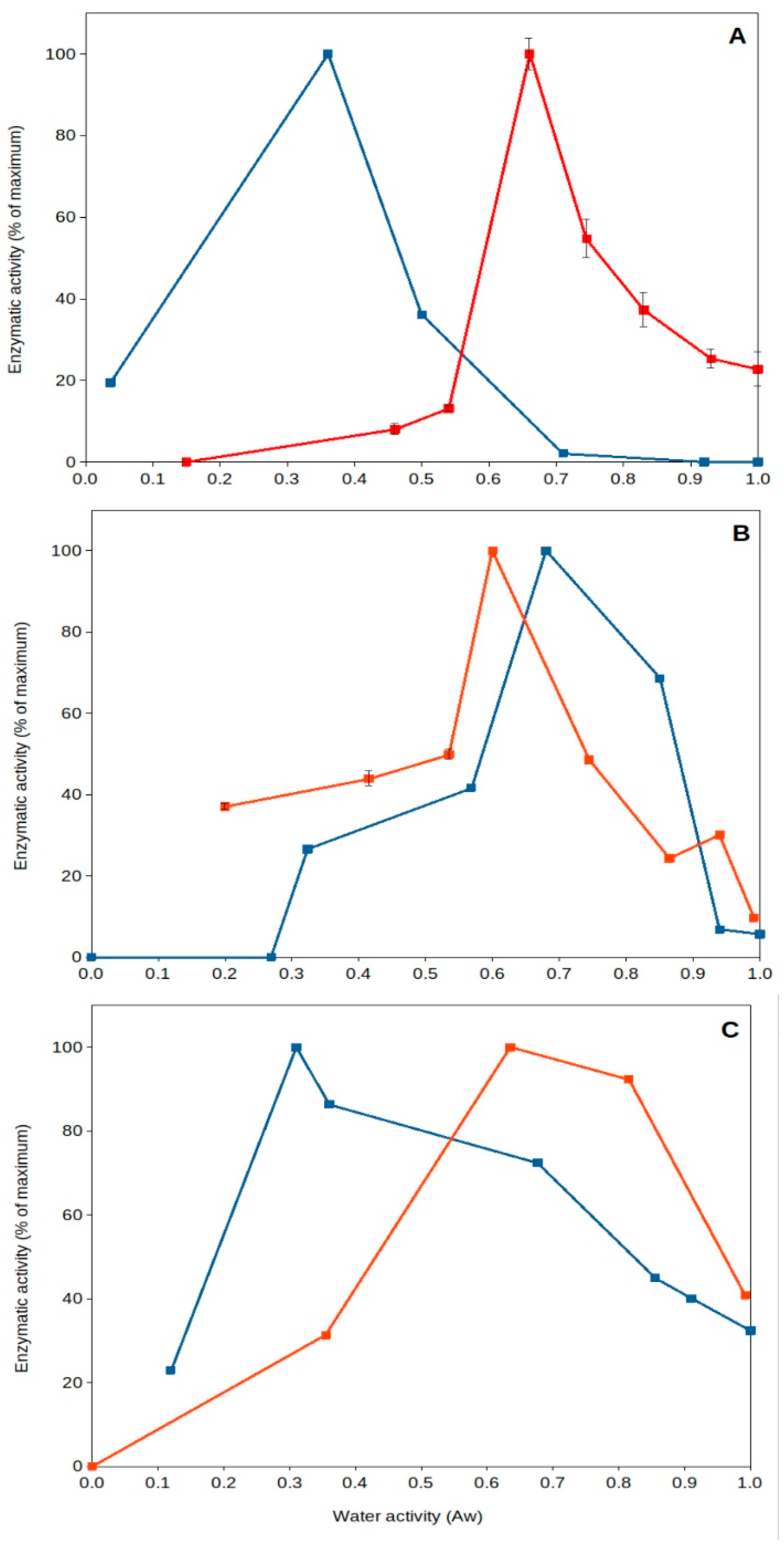
Figure 3. Natural extracellular enzyme activity from Southeastern Spain soils versus water activity. A couple of examples showing optimum extracellular enzyme activity under severe desiccation. Red squares and lines, soil samples collected from Coria del Rio (Seville); blue squares and lines, soil samples collected from Benaocaz (Cadiz). (A), glucanase; (B), phosphatase; (C), protease.
Furthermore, these thermophiles survive and remain viable under these dry conditions (aw 0.5), as shown by a comparative study on pollutant decomposition by Geobacillus (showing maximum pollutant decomposition at aw 0.5) compared to Rhodococcus spp. (showing maximum decomposition at aw > 0.9) [48]. This qualifies soil thermophiles from hot and dry environments as some of the most xerophilic cells reported on Earth. So far, the lowest water activity allowing growth is 0.6 for the fungus Xeromyces bisporus [47], and most microorganisms do not show growth below aw 0.8 [65]. Some soil thermophiles can show optimum extracellular enzymatic activity [43][45] and ability to decompose pollutants [48] at aw around 0.5. Consequently, singular features of soil thermophiles include their potential adaptability to extreme drought and high temperatures, which are valued because of their consequences for the environment [43][45][54], providing evidence of optimal activity under those extremes (Figure 3), as well as a great potential for biotechnological applications in high-temperature and non-aqueous treatments and processes [66][67][68][69].
References
- Whitman, W.B.; Coleman, D.C.; Wiebe, W.J. Prokaryotes: The unseen majority. Proc. Natl. Acad. Sci. USA 1998, 95, 6578–6583.
- Uroz, S.; Calvaruso, C.; Turpault, M.-P.; Frey-Klett, P. Mineral weathering by bacteria: Ecology, actors and mechanisms. Trends Microbiol 2009, 17, 378–387.
- Conant, R.T.; Ryan, M.G.; Ågren, G.I.; Birge, H.E.; Davidson, E.A.; Eliasson, P.E.; Evans, S.E.; Frey, S.D.; Giardina, C.P.; Hopkins, F.; et al. Temperature and soil organic matter decomposition rates—Synthesis of current knowledge and a way forward. Glob. Chang. Biol. 2011, 17, 3392–3404.
- Davidson, E.A.; Janssens, I.A. Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 2006, 440, 165–173.
- Cavicchioli, R.; Ripple, W.J.; Timmis, K.N.; Azam, F.; Bakken, L.R.; Baylis, M.; Behrenfeld, M.J.; Boetius, A.; Boyd, P.W.; Classen, A.T.; et al. Scientist’s warning to humanity: Microorganisms and climate change. Nat. Rev. Microbiol. 2019, 17, 569–586.
- Delgado-Baquerizo, M.; Maestre, F.T.; Gallardo, A.; Bowker, M.A.; Wallenstein, M.D.; Quero, J.L.; Ochoa, V.; Gozalo, B.; García-Gómez, M.; Soliveres, S.; et al. Decoupling of soil nutrient cycles as a function of aridity in global drylands. Nature 2016, 502, 672–676.
- Lladó, S.; López-Mondéjar, R.; Baldrian, P. Forest soil bacteria: Diversity, involvement in ecosystem processes, and response to global change. Microbiol. Mol. Biol. Rev. 2017, 81, e00063-16.
- Maron, P.-A.; Sarr, A.; Kaisermann, A.; Lévêque, J.; Mathieu, O.; Guigue, J.; Karimi, B.; Bernard, L.; Dequiedt, S.; Terrat, S.; et al. High microbial diversity promotes soil ecosystem functioning. Appl. Environ. Microbiol. 2018, 84, e02738-17.
- Curtis, T.P.; Sloan, W.T.; Scannell, J.W. Estimating prokaryotic diversity and its limits. Proc Natl. Acad. Sci. USA 2002, 99, 10494–10499.
- Pedrós-Alió, C. Marine microbial diversity: Can it be determined? Trends Microbiol. 2006, 14, 257–263.
- Roesch, L.F.; Fulthorpe, R.R.; Riva, A.; Casella, G.; Hadwin, A.K.; Kent, A.D.; Daroub, S.H.; Camargo, F.A.; Farmerie, W.G.; Triplett, E.W. Pyrosequencing enumerates and contrasts soil microbial diversity. ISME J. 2007, 4, 283–290.
- Saw, J.H.W. Characterizing the uncultivated microbial minority: Towards understanding the roles of the rare biosphere in microbial communities. mSystems 2021, 6, e00773-21.
- Marchant, R.; Banat, I.M.; Rahman, T.J.; Berzano, M. The frequency and characteristics of highly thermophilic bacteria in cool soil environments. Environ. Microbiol. 2002, 4, 595–602.
- Marchant, R.; Franzetti, A.; Pavlostathis, S.G.; Tas, D.O.; Erdbrugger, I.; Unyayar, A.; Mazmanci, M.A.; Banat, I.M. Thermophilic bacteria in cool temperate soils: Are they metabolically active or continually added by global atmospheric transport? Appl. Microbiol. Biotechnol 2008, 78, 841–852.
- Portillo, M.C.; Santana, M.; Gonzalez, J.M. Presence and potencial role of thermophilic bacteria in temperate terrestrial environments. Naturwissenschaften 2012, 99, 43–53.
- Cockell, C.S.; Cousins, C.; Wilkinson, P.T.; Olsson-Francis, K.; Rozitis, B. Are thermophilic microorganisms active in cold environments? Intl. J. Astrobiol. 2015, 14, 457–463.
- Wong, M.-L.; An, D.; Caffrey, S.; Soh, J.; Dong, X.; Sensen, C.W.; Oldenburg, T.B.P.; Larter, S.R.; Voordouw, G. Roles of thermophiles and fungi in bitumen degradation in mostly cold oil sands outcrops. Appl. Environ. Microbiol. 2015, 81, 6825–6838.
- Santana, M.M.; González, J.M. High temperature microbial activity in upper soil layers. FEMS Microbiol. Lett. 2015, 362, fnv182.
- Gonzalez, J.M.; Portillo, M.C.; Piñeiro-Vidal, M. Latitude-dependent underestimation of microbial extracellular enzyme activity in soils. Int. J. Environ. Sci. Technol. 2015, 12, 2427–2434.
- Milojevic, T.; Cramm, M.A.; Hubert, C.R.J.; Westall, F. “Freezing” thermophiles: From one temperature extreme to another. Microorganisms 2022, 10, 2417.
- Rajasekaran, A.K.; Maheshwari, R. Thermophilic fungi: An assessment of their potential for growth in soil. J. Biosci. 1993, 18, 345–354.
- Zak, J.C.; Howard, G.; Wildman, G. Fungi in stressful environments. In Biodiversity of Fungi. Inventory and Monitoring Methods; Mueller, G.M., Bills, G.F., Foster, M.S., Eds.; Elsevier Academy Press: Burlington, MA, USA, 2004; pp. 305–315.
- Neher, D.A.; Weicht, T.R.; Bates, S.T.; Leff, J.W.; Fierer, N. Changes in bacterial and fungal communities across compost recipes, preparation methods, and composting times. PLoS ONE 2013, 8, e79512.
- Vigneswaran, S.; Kandasamy, J.; Johir, M.A.H. Sustainable operation of composting in solid waste management. Proc. Environ. Sci. 2016, 35, 408–415.
- Diánez, F.; Marín, F.; Santos, M.; Gea, F.J.; Navarro, M.J.; Piñeiro, M.; González, J.M. Genetic analysis and in vitro enzymatic determination of bacterial community in compost teas from different sources. Compost. Sci. Util. 2018, 26, 256–270.
- López-Bellido, R.J.; Lal, R.; Danneberger, T.K.; Street, J.R. Plant growth regulator and nitrogen fertilizer effects on soil organic carbon sequestration in creeping bentgrass fairway turf. Plant Soil 2010, 332, 247–255.
- McGrath, J.M.; Spargo, J.; Penn, C.J. Soil fertility and plant nutrition. In Encyclopedia of Agriculture and Food; van Alfen, N., Ed.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 166–184.
- Ahmad, A.A.; Radowich, T.J.K.; Nguyen, H.V.; Uyeda, J.; Arakaki, A.; Cadby, J.; Paull, R.; Sugano, J.; Teves, G. Use of organic fertilizers to enhance soil fertility, plant growth, and yield in a tropical environment. In Organic Fertilizers; Larramendy, M.L., Soloneski, S., Eds.; IntechOpen: London, UK, 2016.
- Li, Y.; Fang, F.; Wei, J.; Wu, X.; Cui, R.; Li, G.; Zheng, F.; Tan, D. Humic acid fertilizer improved soil properties and soil microbial diversity of continuous cropping peanut: A three-year experiment. Sci. Rep. 2019, 9, 12014.
- Yan, S.; Zhang, N.; Li, J.; Wang, Y.; Liu, Y.; Cao, M.; Yan, Q. Characterization of humic acids from original coal and its oxidization production. Sci. Rep. 2021, 11, 15381.
- Asmar, F.; Eiland, F.; Nielsen, N.E. Effect of extracellular-enzyme activities on solubilization rate of soil organic nitrogen. Biol. Fertil. Soils 1994, 17, 32–38.
- Madigan, M.; Martinko, J.M.; Parker, J. Brock Biology of Microorganisms; Prentice Hall Inc.: Upper Saddle River, NJ, USA, 2003.
- Wallenstein, M.D.; Burns, R.G. Ecology of extracellular enzyme activities and organic matter degradationin soil: A complex community-driven process. In Methods in Soil Enzymology; Dick, R.P., Ed.; Soil Science Society of America Inc.: Madison, WI, USA, 2011; pp. 35–55.
- Cheng, L.; Zhang, N.; Yuan, M.; Xiao, J.; Qin, Y.; Deng, Y.; Tu, Q.; Xue, K.; van Nostrand, J.D.; Wu, L.; et al. Warming enhances old organic carbon decomposition through altering functional microbial communities. ISME J. 2017, 11, 1825–1835.
- Sorouri, B.; Allison, S.D. Microbial extracellular enzyme activity with simulated climate change. Elem. Sci. Anth 2022, 10, 76.
- O’Neill, B.C.; Oppenheimer, M.; Warren, R.; Hallegatte, S.; Kopp, R.E.; Pörtner, H.O.; Scholes, R.; Birkmann, J.; Foden, W.; Licker, R.; et al. IPCC reasons for concern regarding climate change risks. Nat. Clim. Chang. 2017, 7, 28–37.
- Xiao, W.; Chen, X.; Jing, X.; Zhu, B. A meta-analysis of soil extracellular enzyme activities in response to global change. Soil Biol. Biochem. 2018, 123, 21–32.
- Ye, J.S.; Bradford, M.A.; Dacal, M.; Maestre, F.T.; García-Palacios, P. Increasing microbial carbon use efficiency with warming predicts soil heterotrophic respiration globally. Glob. Chang. Biol. 2019, 25, 3354–3364.
- Burns, R.G.; DeForest, J.L.; Marxsen, J.; Sinsabaugh, R.L.; Stromberger, M.E.; Wallenstein, M.D.; Weintraub, M.N.; Zoppini, A. Soil enzymes in a changing environment: Current knowledge and future directions. Soil Biol. Biochem. 2013, 58, 216–234.
- Chen, J.; Sinsabaugh, R.L. Linking microbial functional gene abundance and soil extracellular enzyme activity: Implications for soil carbon dynamics. Glob. Chang. Biol. 2021, 27, 1322–1325.
- Ndabankulu, K.; Egbewale, S.O.; Tcvuura, Z.; Magadlela, A. Soil microbes and associated extracellular enzymes largely impact nutrient bioavailability in acidic and nutrient poor grassland ecosystem soils. Sci. Rep. 2022, 12, 12601.
- Zhou, Y.; Biro, A.; Wong, M.Y.; Batterman, S.A.; Staver, A.C. Fire decreases soil enzyme activities and reorganizes microbially mediated nutrient cycles: A meta-analysis. Ecology 2022, 103, e3807.
- Gomez, E.J.; Delgado, J.A.; Gonzalez, J.M. Environmental factors affect the response of microbial extracellular enzyme activity in soils when determined as a function of water availability and temperature. Ecol. Evol. 2020, 10, 10105–10115.
- Jian, S.; Li, J.; Chen, J.; Wang, G.; Mayes, M.A.; Dzantor, K.E.; Hui, D.; Luo, Y. Soil extracellular enzyme activities, soil carbon and nitrogen storage under nitrogen fertilization: A meta-analysis. Soil Biol. Biochem. 2016, 101, 32–43.
- Gomez, E.J.; Delgado, J.A.; Gonzalez, J.M. Influence of water availability and temperature on estimates of microbial extracellular enzyme activity. PeerJ 2021, 9, e10994.
- Smith, P.; Fang, C.M.; Dawson, J.J.C.; Moncrieff, J.B. Impact of global warming on soil organic carbon. Adv. Agron 2008, 97, 1–43.
- Stevenson, A.; Burkhardt, J.; Cockell, C.S.; Cray, J.A.; Dijksterhuis, J.; Fox-Powell, M.; Kee, T.P.; Kminek, G.; McGenity, T.J.; Timmis, K.N.; et al. Multiplication of microbes below 0.690 water activity: Implications for terrestrial and extraterrestrial life. Environ. Microbiol. 2015, 17, 257–277.
- Moxley, E.; Puerta-Fernández, E.; Gómez, E.J.; González, J.M. Influence of abiotic factors temperature and water content on bacterial 2-chlorophenol biodegradation in soils. Front. Environ. Sci. 2019, 7, 41.
- Awasthi, M.K.; Selvam, A.; Chan, M.T.; Wong, J.W.C. Bio-degradation of oily food waste employing thermophilic bacterial strains. Biores. Technol 2018, 248, 141–147.
- Elumalai, P.; Parthipan, P.; Narenkumar, J.; Anandakumar, B.; Madhavan, J.; Oh, B.-T.; Rajasekar, A. Role of thermophilic bacteria (Bacillus and Geobacillus) on crude oil degradation and biocorrosion in oil reservoir environment. Biotech 2019, 9, 79.
- Marchant, R.; Sharkey, F.H.; Banat, I.M.; Rahman, T.J.; Perfumo, A. The degradation of n-hexadecane in soil by thermophilic geobacilli. FEMS Microbiol. Ecol. 2006, 56, 44–54.
- DePoy, A.N.; King, G.M. Distribution and diversity of anaerobic thermophiles and putative anaerobic nickel-dependent carbon monoxide-oxidizing thermophiles in mesothermal soils and sediments. Front. Microbiol. 2023, 13, 1096186.
- Hutchins, D.A.; Jansson, J.K.; Remains, J.V.; Rich, V.I.; Singh, B.K.; Trivedi, P. Climate change microbiology—Problems and perspectives. Nat. Rev. Microbiol. 2019, 17, 391–396.
- Gomez, E.J.; Delgado, J.A.; Gonzalez, J.M. Persistence of microbial extracellular enzymes in soils under different temperatures and water availabilities. Ecol. Evol. 2020, 10, 10167–10176.
- Townsend, A.; Vitousek, P.M.; Holland, E.A. Tropical soils could dominate the short-term carbon cycle feedbacks to increased global temperatures. Clim. Chang. 1992, 22, 293–303.
- Chróst, R.J. Significance of bacterial ectoenzymes in aquatic environments. Hydrobiologia 1992, 244, 61–70.
- Bai, X.; Dippold, M.A.; An, S.; Wang, B.; Zhang, H.; Loeppmann, S. Extracellular enzyme activity and stoichiometry: The effect of soil microbial element limitation during leaf litter decomposition. Ecol. Indic. 2021, 121, 107200.
- Vieille, C.; Zeikus, G.J. Hyperthermophilic enzymes: Sources, uses, and molecular mechanisms for thermostability. Microbiol. Mol. Biol. Rev. 2001, 65, 1–43.
- Li, G.; Maria-Solano, M.A.; Romero-Riva, A.; Osuna, S.; Reetz, M.T. Inducing high activity of a thermophilic enzyme at ambient temperatures by directed evolution. Chem. Comm. 2017, 53, 9454.
- Santana, M.M.; Carvalho, L.; Melo, J.; Araújo, M.E.; Cruz, C. Unveiling the hidden interaction between thermophiles and plant crops: Wheat and soil thermophilic bacteria. J. Plant Interact. 2020, 15, 127–138.
- Renella, G.; Szukics, U.; Landi, L.; Nannipieri, P. Quantitative assessment of hydrolase production and persistence in soil. Biol. Fertil. Soils 2007, 44, 321–329.
- Wallenstein, M.D.; Weintraub, M.N. Emerging tools for measuring and modeling the in situ activity of soil extracellular enzymes. Soil Biol. Biochem 2008, 40, 2098–2106.
- Vries, F.T.; Griffiths, R.I.; Bailey, M.; Craig, H.; Girlandan, M.; Gweon, H.S.; Hallin, S.; Kaisermann, A.; Keith, A.M.; Kretzschmar, M.; et al. Soil bacterial networks are less stable under drought than fungal networks. Nat. Comm. 2018, 9, 3033.
- Slade, D.; Radman, M. Oxidative stress resistance in Deinococcus radiodurans. Microbiol Mol. Biol. Rev 2011, 75, 133–191.
- Grant, W.D. Life at low water activity. Phil. Trans Royal Soc. London Ser. B Biol. Sci 2004, 359, 1249–1267.
- Hudson, E.P.; Eppler, R.K.; Clark, D.S. Biocatalysis in semi-aqueous and nearly anhydrous conditions. Curr. Opinion Biotechnol. 2005, 16, 637–643.
- Dubey, S.K.; Tripathi, A.K.; Upadhyay, S.N. Exploration of soil bacterial communities for their potential as bioresource. Bioresour. Technol. 2006, 97, 2217–2224.
- Mehta, R.; Singhal, P.; Singh, H.; Damle, D.; Sharma, A.K. Insight into thermophiles and their wide-spectrum applications. Biotech 2016, 6, 81.
- Lyer, P.V.; Ananthanarayan, L. Enzyme stability and stabilization—Aqueous and non-aqueous environment. Process Biochem. 2008, 43, 1019–1032.
More
Information
Subjects:
Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
625
Revisions:
2 times
(View History)
Update Date:
03 Jul 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





