Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yeshun Zhang | -- | 4746 | 2023-06-12 12:36:15 | | | |
| 2 | Sirius Huang | Meta information modification | 4746 | 2023-06-13 02:53:50 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Ye, R.; Liu, S.; Zhu, W.; Li, Y.; Huang, L.; Zhang, G.; Zhang, Y. Properties and Biomedical Application of Chitosan-Based Hydrogels. Encyclopedia. Available online: https://encyclopedia.pub/entry/45448 (accessed on 07 June 2026).
Ye R, Liu S, Zhu W, Li Y, Huang L, Zhang G, et al. Properties and Biomedical Application of Chitosan-Based Hydrogels. Encyclopedia. Available at: https://encyclopedia.pub/entry/45448. Accessed June 07, 2026.
Ye, Ruixi, Siyu Liu, Wenkai Zhu, Yurong Li, Long Huang, Guozheng Zhang, Yeshun Zhang. "Properties and Biomedical Application of Chitosan-Based Hydrogels" Encyclopedia, https://encyclopedia.pub/entry/45448 (accessed June 07, 2026).
Ye, R., Liu, S., Zhu, W., Li, Y., Huang, L., Zhang, G., & Zhang, Y. (2023, June 12). Properties and Biomedical Application of Chitosan-Based Hydrogels. In Encyclopedia. https://encyclopedia.pub/entry/45448
Ye, Ruixi, et al. "Properties and Biomedical Application of Chitosan-Based Hydrogels." Encyclopedia. Web. 12 June, 2023.
Copy Citation
The prospective applications of chitosan-based hydrogels (CBHs), a category of biocompatible and biodegradable materials, in biomedical disciplines such as tissue engineering, wound healing, drug delivery, and biosensing have garnered great interest. The synthesis and characterization processes used to create CBHs play a significant role in determining their characteristics and effectiveness.
biomedical application
chitosan
hydrogel
properties
1. Introduction
CBHs are incredible biomaterials with an extensive range of interesting applications. Most CBHs inherit the essential property of benign biocompatibility from chitosan [1]. The marvelous capacity to be characterized, meanwhile, allows the widespread application of CBHs in biomedicine. The characteristics and applications of one hydrogel are generally regarded as being closely connected. Herein, the main properties of CBHs catering to versatile application sectors are methodically introduced. This section provides a thorough overview of the current state-of-the-art and prospects in biomedicine. CBHs could be endowed with versatile properties such as swelling ability, stimuli-responsiveness, antimicrobial activity, enhanced mechanical strength, etc. Consequently, CBHs could be applied in a diversity of biomedical sectors, including tissue engineering, DDSs, wound healing, etc., in accordance with their properties, as shown in Figure 1.
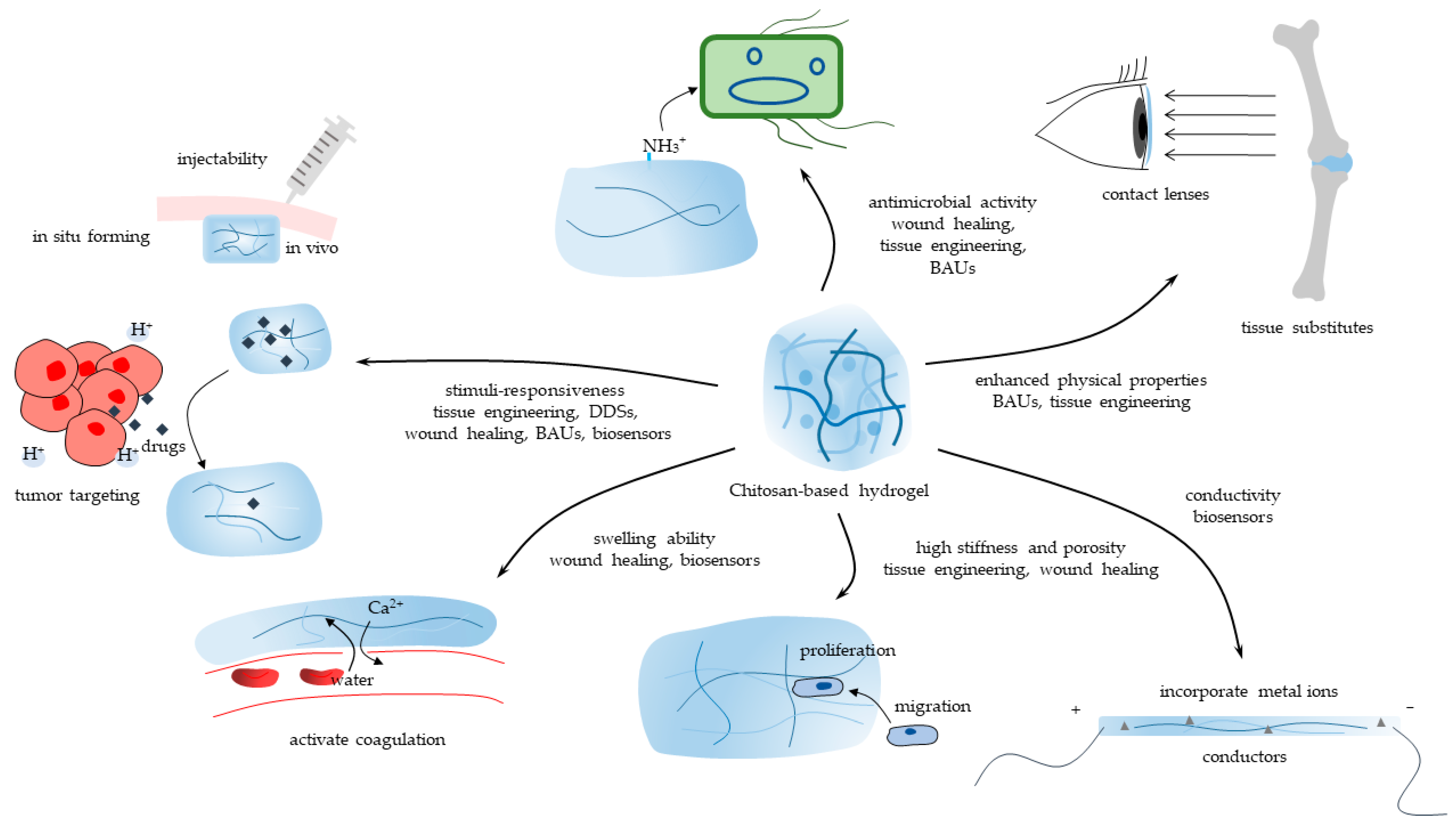
Figure 1. The main properties of CBHs and their biomedical application sectors.
2. Tissue Engineering
Acceleration or augmentation of regeneration procedures is a requisite in tissue engineering due to the insufficient ability of some target tissues to propagate. For instance, cartilage is a form of connective tissue that has a low capability for self-repair due to its avascular and aneural nature [2]. Therefore, one feasible treatment method is to foster regeneration via exotic hydrogels. Hydrogel is a promising material for tissue engineering since its 3D configuration is analogous to the natural extracellular matrix (ECM) of tissues, while the ferous porosity arrangement supports cell adhesion, proliferation, differentiation, and function. In addition, surface modification with other substances can foster the step [3]. Gifted properties facilitate the preparation steps of the scaffold, but the successive activities are tricky, so these are detailed.
2.1. Methods and Techniques to Implant
The scaffold is often fabricated in a laboratory setting before being surgically implanted into the flaw site [4]. This technique incorporates materials or cells into the hydrogel and allows for fine control over the size and shape of the hydrogel. However, it might also impair surrounding tissues or cause an infection or inflammation. 3D bioprinting is an advanced fabrication technique that can produce patient-specific scaffolds with complex geometries and precise control of cell positioning and can achieve large tissue-engineered products, with shear thinning behavior and viscosity being the critical characteristics of the bioink [5][6]. The 3D printing (3DP) technology could overcome limitations in complicated tissue conditions with unique forms [7]. Chitosan is a high MW polymer that produces viscous solutions for 3DP. This technique has the advantages of creating homogenous pore sizes and avoiding deviation compared with conventional CBH construction methods [8]. Xu et al. [9] reported a supramolecular CBH with host-guest connections that had great printability and mechanical strength. Comparatively, in situ tissue engineering avoids the need for pre-culturing cells and tissues in vitro and uses the host’s own cells and biomolecules to regenerate the tissue, improving the integration and functionality of the regenerated tissue while lowering the risk of immunological rejection and infection [10]. In situ-forming CBH, which is a kind of smart hydrogel that is able to commence sol-gel transition in response to exterior inducements, such as pH value and temperature [11], could be injected into the body via minimally invasive surgery (MIS). Chitosan is water-soluble and positively charged under acidic conditions (pH < 6), but would turn uncharged and hydrophobic at physiological pH, forming a compact and physically crosslinked hydrogel [12]. The generation of both parallel and perpendicular crosslinking in CBH network structure arises at higher pH levels (above 6.5) (tissue pH is neutral) as well [13]. The two mechanisms facilitate pH in situ formation. Though mechanical properties would descend unavoidably, concentration and printing temperature are vital parameters affecting the gelation rate and thereby the strength of printed structures in in situ gelation four-dimensional (4D) CBH, which is endowed with thermo-sensitivity [14]. Introduced thermo-responsive constituent parts enable CBHs to respond to temperature via the quantity variance of intramolecular hydrogen bonds, and this has been considered a typical stimulus for smart CBHs [15]. Nevertheless, before application, it is vital to take into account patient variability, hydrogel displacement, lack of stability, etc.
2.2. Microenvironment Adjustment
A non-threatening microenvironment is critical for tissue regeneration. There are two dimensions to improving conditions: one is to eliminate antigens that might trigger an immunoreaction, and the other is to modulate the host's immune response. Bioactive materials, e.g., growth factors, peptides, nucleic acids, and antibiotics, could be loaded by CBHs to facilitate the growth of seed cells. Growth factors can be chemically modified or covalently immobilized via the amino and hydroxyl groups of chitosan, improving their stability and bioactivity in the target tissue matrix [16]. Li et al. [17] reported that CBH could recruit tetrahedral framework nucleic acid (TFNA), which was injected into the articular cavity to enhance cartilage repair. TFNA is a promising DNA nanomaterial for improving the regenerative microenvironment. The antibiotics added to the hydrogel can enhance antimicrobial activity, producing environments that are free of microbes [18]. Inflammation resulting from xenografts is one obstacle to the implantation of CBHs [19]. Therefore, immunomodulation via hydrogel becomes conspicuous. Disordered macrophage activation impedes tissue regeneration. Regulating macrophages from the M1 type to the M2 type is crucial for expediting tissue repair in cartilage [20] and bone regeneration [21] because the M1 type promotes inflammation while the M2 type secretes anti-inflammatory cytokines for pro-tissue repair effects. Various CBHs could regulate this procedure. For instance, chitosan/silk fibroin/cellulose nanoparticle (CS/SF/CNPs) scaffolds facilitate M2 macrophage polarization and influence the osteo-immunomodulatory responses of the cells [21].
2.3. Tissue Regeneration
Cells interact with their microenvironments and are affected by them. Hydrogels must be created with exclusive physical, chemical, and biological qualities in order to account for the unique traits of each type of tissue and foster tissue regeneration.
Regeneration strategies can be divided into two types: encapsulating autologous cells as seed cells in hydrogels and inducing the proliferation of surrounding cells via various substances [2]. The combination of cell-based and proliferative strategies is ideal. In the experiment of Hao et al. [22], CBHs alone were less effective than chondrocyte- and chitosan-coated materials at fully repairing cartilage lesions in vivo, proving the advantages of adding seed cells. Similar to the natural ECM, the matrix stiffness of CBHs is one parameter affecting tissue regeneration by regulating the force exerted on the cells. It has been demonstrated that the hydrogel matrix stiffness affects cell proliferation, with early passage-stage cells being more sensitive [23]. High matrix stiffness typically encourages cell growth, whereas low stiffness causes cell dormancy and stemness [24]. The ECM stiffness varies depending on the tissue type and can influence cell behavior, so modulation is imperative for application. Generally, the parameter can be adjusted by modifying the crosslinking degree; more bonds are beneficial to enhance the deformation resistance, while fewer bonds work in the opposite direction. Chang et al. [25] achieve stiffness tunability by changing the grafting ratio. Dual-crosslinked hydrogels might have higher stiffness than single-crosslinking methods [26]. Moreover, the covalent binding methods unavoidably affect other properties due to the changes in CBH structures, so it turned out to be an optimization problem. Physical contacts are weaker and less stable than dynamic covalent bonds, which are reversible, stimuli-responsive, and adjustable [27], a workable method for controlling the stiffness of hydrogels. However, a few issues with the latter, including achieving homogeneity, incomplete study, and finite reaction type variety, need to be ameliorated [28]. Additionally, the porous structure is available for the proliferation of the tissue because it can not only facilitate the adhesion and proliferation of adjacent cells but also release drugs more efficiently. CBHs could be made permeable using a variety of techniques, divided into physical, chemical, and biological types. The porous framework of CBHs can be generated by 3DP [5], foaming [29], microwave [30], etc. NaHCO3 added to the CBH system, could react with the protonated amino groups in vivo, releasing CO2 to generate micropores [31]. Adding lysozyme encourages the degradation of CBHs to generate pores. By catalyzing the breakdown of CBH, lysozyme can be used to produce porous structures. When mesenchymal stem cells are incorporated into chitosan-lysozyme hydrogels, these spaces advance cell proliferation and migration, which likewise help with osteogenic differentiation [32]. The favorable average diameter of scaffold pores varies depending on the cell type, which has an impact on cell differentiation and gene expression [33], so regulating the average diameter of the hydrogel is vital, with the modifying approaches diverging between techniques. Furthermore, the porosity ratio is another critical parameter to assess the microstructure. In addition, 4D CBH, a type of smart CBH based on the 3DP technique [34], holds the potential to manufacture sophisticated non-sintering scaffolds with improved porosity qualities for hard tissue engineering scaffolds or mimic the ECM of soft ones to foster tissue regeneration [35]. Nonetheless,
2.4. Biodegradation In Vivo
As tissue engineering materials, scaffolds should not induce acute or chronic effects and be biodegradable because the newly formed tissue should be able to replace them simultaneously [36], which prevents the need for surgical removal. Hydrolysis, enzyme-mediated processes, or a combination are utilized [12]. Lysozyme, a critical enzyme catalyzing the degradation of CBH, resides in all mammalian tissues and functions via accidental splitting of β-1,4-glycosidic bonds (depolymerization) and hydrolysis of the N-acetyl linkage (deacetylation) [36]. It has been proven that the degradation of CBH can be tweaked by changing the concentration of lysozyme added to chitosan hydrogels by chemically altering methacrylate groups. Additionally, biodegradable smart CBHs are hydrogels that would break down over time in response to biological or microenvironmental ingredients such as biological substances, pH levels, temperature, etc. Biodegradable smart hydrogels have been utilized for tissue engineering purposes because of their advantages, including controlled degradation kinetics and sensitivity to incentives. Nevertheless, some of the challenges or limitations are their low mechanical strength, inferior stability, multifactorial degradation kinetics, and potential immunogenicity [37]. Notably, CBHs can release loaded drugs, cells, growth factors, etc., following degradation, and the released N-acetyl-β-D-glucosamine induces fibroblast growth [38]. There are several parameters in the synthesis procedure that influence biodegradability, such as the DD and MW of chitosan and the type and concentration of crosslinking agents [39]. Nonetheless, the impact of the concentration of lysozyme in the tissue, which will ascend in some adverse cases, on the biodegradation ratio is still unstudied, which should be considered in the factors affecting it. Last but not least, Reay et al. [40] measured the sizes of the genipin-chitosan hydrogel-degraded particles and reported that the majority of them were as small as 1.7 nm, which is below the renal filtration threshold, meaning that they could be eliminated from the body via urine. The safety of CBH biodegradation remains unclear due to the varied crosslinking methods of CBHs; therefore, further research is crucial.
3. Drug Delivery Systems (DDSs)
CBHs have been manipulated as drug delivery vehicles for various bioactive agents. CBHs could enhance the stability, retention, and bioactivity of the delivered agents and modulate the release kinetics and interactions with the target tissues. Moreover, many medicines with different physicochemical properties could be loaded by the CBH to achieve treatment.
3.1. Drug Release Regulation and Release Kinetics
Stimuli-responsive CBHs are the major component of DDSs made of CBHs, having been well-researched and widely applied, for they are liable to gelatinize and break down under definite stimuli. In addition, their widespread application can be attributed to their injectability, lack of surgical necessity, shape flexibility, etc. They can release drugs, cells, biomass, etc., at one specific occasion, depending on the microenvironment of the tissue, including pH values, temperature, biological factors, etc. [41]. Passively received and steerable stimuli could be used to categorize these signals. The former should be nimble and take into account the hardwired circumstances of the target site, due to the fact that the former is a category of indeterminate parameters, whereas the latter can be regulated because it is generally stable and mostly factitious. As detailed in the tissue engineering part, at an acidic pH, the amino groups of chitosan will be positively charged, and the hydrogel will swell, facilitating the release of cargo. However, most of the groups were depleted by chemical gelation. Silva et al. [42] preserved the original amino acid via a protecting and deprotecting strategy. The physical crosslinking approach is another practical option. It can retain relatively high pH sensitivity and fragility, which are advantageous for the emancipation of medications. It has been demonstrated that sol-gel crosslinking CBH is a pH-responsive matrix that can bulge or dwindle depending on the pH level of the microenvironment [43]. In cancer therapy, the drugs used in DDSs are usually malignant to normal cells as well. The gel formation is rather immobile, favoring adhesion to the targeted tissue by filling it up or wrapping it. Then, different stimuli foster the release of bioactive molecules integrated with chitosan, achieving targeted therapy. The tumor microenvironment (TME) is a multifaceted and continuously evolving object, featuring acidic, hypoxic, excessive metabolite accumulation, and high-depression conditions for immune cells [44][45]. pH-responsive CBHs, especially acid-sensitive ones, would trigger degradation of the hydrogel there [46], thereby releasing the loaded drugs. Another latent reaction, swelling behavior, triggered by acidic stimuli, increases the contact area between the hydrogel and the tumor, facilitating the diffusion of drugs or other agents from the hydrogel [41]. In addition, it can also exert mechanical pressure on the tumor tissue, depressing and hindering tumor growth. The high density of lactic acid in the tissue fluid promotes macrophage transformation into the M2 type, which will foster the escalation of tumors [47]. The immunosuppressive milieu can be reversed in the microenvironment by adding CaCO3, which also lessens the immunosuppressive effect on T cells since the higher pH value encourages macrophage polarization from the M2 type to the M1 type [48]. Additionally, the bionic onion structure is applied to regulate the drug release kinetics [49]. It enlightens that the exquisite structure can be programmed to release alkaline salts or ions in acidic instances before setting free medications, which are not only stimuli-responsive but also can adjust the microenvironment to favor subsequent treatment. Thermosensitive CBHs can be divided into two types. One form of thermosensitive CBH exhibits a lower critical solution temperature (LCST), whereas the other type exhibits an upper critical solution temperature (UCST). When the temperature is raised over a certain degree, the LCST hydrogel contracts and releases contents, whereas the UCST hydrogel expands and absorbs water. LCST could be exploited to control the release of drugs from the hydrogel, for the hydrogel will shrink and release encapsulated drugs within it [50]. By controlling the temperature and the LCST of the CBHs, it is achievable to control the rate and timing of drug release. Comparatively, the delivery mode of in situ-forming hydrogels is the very opposite of the anterior. UCST hydrogel is a fluid that facilitates a mixture of drugs at a lower temperature and becomes a gel after being inoculated into the body. Incidentally, the UCST CBH is widely utilized as a category of injectable hydrogels. The liquid formation facilitates the homogeneous incorporation of drugs or biological molecules, and they can keep their hydrogel formation after injection, achieving relatively sustained drug release. Vaccine antigens could be added when the hydrogel is liquid at room temperature and can be released slowly to trigger the adaptive immune system [51].
3.2. Loaded Medicines
CBHs could perform as DDSs for medications by incorporating or adsorbing them within their network or via covalent bonds. The merits of CBHs as delivery carriers include versatile loading modes, extending the retention of consignments in biological settings, and regulated or controllable release kinetics [52]. Hydrogel is a 3D structure with a high water content, so hydrophilic medications are more likely to load than hydrophobic ones. Considering the increasing needs, there are several strategies to increase the load ratio and retard the release of hydrophobic drugs. It has been demonstrated that boosting the proportion of hydrophobic moieties inside CBH can postpone the release, but the link should be correctly regulated because the relationship is not a linear one [53]. Polymeric micelles, self-assembled configurations of amphiphilic molecules having a hydrophobic core and a hydrophilic shell, can also be exploited to extraordinarily depress diffusion of the hydrophobic substances [54]. A biocompatible, non-ionic microemulsion can be used to encapsulate hydrophobic drugs to enhance the water solubility of the drugs and protect them from degradation [55]. Conspicuously, encapsulating exosomes and micro-vesicles, two types of extracellular vesicles that are small membrane-bound structures released by cells into the extracellular space, are capable of carrying different types of macromolecules, bestowing CBHs with the aptitude to modulate several metabolic pathways in the recipient cells [56][57]. The amalgamation of the two materials is favorable because they have the advantages of being stimuli-responsive, biocompatible, able to steadily release, able to incorporate a variety of chemicals, etc.
4. Wound Healing
The intention of wound healing is to accelerate the natural healing response so that damaged tissues can mend and regenerate. This involves the use of dressings, biomaterials, and drugs that can modulate the hemostasis, inflammation, proliferation, and remodeling phases [58] of wound healing, with CBH functioning mainly in the first three phases. Wound-healing CBHs can be operated on to treat acute wounds, chronic inflammation, ulcers, etc.
4.1. Hemostasis Phase
Hemocompatibility is a prerequisite for wound-dressing materials [59], as these CBHs come into direct contact with gore. To improve the adhesion property of hydrogels on bleeding wound surfaces of internal organs, Chen et al. [60] suggested that creating a local drying setting by expelling the moisture on the wound surface via incorporating a super-hydrophilic substance is one feasible way. Furthermore, the hydrogel could still preserve a humid microenvironment, endorsing wound healing [61], inside the wound by absorbing effusion. Introducing other adhesive components can also help [59]. After being injected into irregular wounds, self-healing CBHs are capable of mending on their own thanks to the design of reversible crosslinking, including Schiff base bonds, the metal coordination bond, host-guest interactions, and electrostatic interactions [62]. Wei et al. [63] developed a particular type of CBH that demonstrated a normal shear-induced gel-to-sol transition as well as the quick self-healing ability and quickly plugged the lesion with the CBH. Additionally, photo-initiated polymerization allows for the tuning of the CBH, which enables it to respond to various wound forms and tissue types. Moreover, another benefit is that the wound dressings can preserve integrity under external forces. After the hydrogels are applied to the location, versatile methods, such as electrostatic interaction [50] and chitosan’s inherent hemostatic property [64], are implemented to concentrate blood cells and platelets to activate coagulation, the initial stage of wound healing. Yu et al. [65] synthesized Janus self-propelled CBH spheres with constructive swelling behaviors, realizing exudate absorption, red blood cell and platelet concentration, and directional movement, moving against blood flow and reaching the bleeding site. Notably, apart from these methods, the native coagulation system in vivo can also be regulated via loaded substances. Ca2+ is widely integrated into CBHs through ionic [59] or calcium salt [66] formation. The ion can induce the conversion of prothrombin to thrombin, a critical step because the latter fosters the formation of fibrin networks [65]. Additionally, tranexamic acid (TXA), a typical antifibrinolytic drug to treat hemophilia and other coagulopathies [67], can be added, avoiding fibrinolysis and preventing the breakdown of blood clots [65]. Song et al. [68] demonstrated that a certain ratio of acetylamino and amino groups in chitosan may also stimulate the coagulation system and encourage platelet and erythrocyte adherence. However, whether there are similar mechanisms in CBHs deserves further research.
4.2. Inflammation Regulation Phase
Bacterial infection is a tricky menace to laceration and escalating inflammation. CBHs that have inherent antibacterial properties and combining antibacterial agents with hydrogels are two viable strategies. The former relies on the intrinsic properties of chitosan (or its derivatives) or is triggered by some substances (e.g., triazole rings [69]). The positively charged amino groups (pH < 6) of chitosan electrostatically interact with negatively charged components on the microbial membrane, thwarting basic roles [70]. Some low-MW chitosan can permeate the membrane and combine the biopolymers, disordering metabolisms [71]. The latter involves the incorporation of external antibacterial agents, such as antibiotics (e.g., lincomycin [72]), metal nanoparticles or ions (e.g., silver nanoparticles (AgNPs) or Mg2+), natural polymers (e.g., antimicrobial peptides [73]), etc., into the hydrogel matrix to eliminate microbes due to its biodegradability. Nonetheless, CBHs that have inherent antibacterial properties may have lower and narrower antibacterial activity and spectrum than those combining antibacterial agents with hydrogels. Therefore, both have merits and demerits, and the choice of the most suitable one depends on the specific application and requirements. Inflammation is a normal response to injury or infection, but excessive or chronic inflammation, more common in the type 2 diabetic patient group, would result in tissue damage. Therefore, on the premise that no microorganisms exist or have any growth activity, anti-inflammation is imperative to decrease the damage. There are versatile methods to downregulate it. For instance, by inhibiting the expression of inducible nitric oxide synthase (iNOS), an enzyme that creates NO in response to inflammation, it was demonstrated that the diminution in NO instigated by CBH had an anti-inflammatory impact, activated by lipopolysaccharide [74]. Nevertheless, there are some concerns that the antibacterial agent would leach out over time, resulting in the loss of antibacterial properties [75], and the anti-inflammation property might work reversely. Yin et al. [76] synthesized hybrid hydrogels, which could elevate the release of both NO and tumor necrosis factor alpha (TNF-α) by stimulating and activating RAW 264.7 macrophages, augmenting their antibacterial biological response, which is proven to advance the healing procedure. In short, the regulation of inflammation should be a comprehensive issue, demanding consideration of the antimicrobial property, adjacent tissue state, patient condition, etc.
4.3. Fostering Proliferation Phase
The third stage is proliferation, where new tissue is formed to rebuild the site, which is detailed in the tissue engineering part. Another way to promote proliferation is to eliminate malignant factors. To a great extent, delayed wound healing results from the depletion of metal ions in the wound microenvironment, so the controlled release sequence of metal ions from CBHs can significantly promote wound healing [77]. Reactive oxygen species (ROS) buildup greater than the antioxidant capacity of cells can obstruct wound healing, especially in cases of chronic and non-healing wounds, whereupon antioxidant treatments are effective in preventing or reducing oxidative stress and improving wound healing [78]. Amino groups of chitosan can be regarded as electron donors to stabilize active oxidizing radicals and further terminate the free radical chain reaction, which endows it as an antioxidant [79], with prolonged N-deacetylation having a stronger antioxidant effect [80]. Phenolic hydroxyl groups [81], magnolol [82], and other oxidants present in the hydrogel matrix can enhance the property.
5. Biocompatible Auxiliary Units (BAUs)
The BAUs are a specific kind of hydrogel with high biocompatibility, which enables them to function as substitutes or auxiliaries for tissues to achieve healing or symptom relief. Generally, BAUs with biodegradability are beneficial for short-term usage, while others are better suited for substitution. Furthermore, the latter, known as tissue substitutes, aim to mimic the properties of original tissues rather than serve as scaffolds for tissue regeneration, which sets them apart from tissue engineering. Biocompatibility and low cytotoxicity are essential to alleviating inflammation [83] and damage to adjacent tissues [84].
Contact lenses made of CBH can be used for eyesight correction or therapy. The former requires premium optical properties, which could be achieved by regulating the orientation of the polymer chains via electrodeposition [85], while the latter is more of a DDS. Jiao et al. [83] produced contact lenses made of one CBH endowed with biodegradability, antioxidant activity, and antimicrobial activity, making them a feasible therapy for bacterial keratitis therapy and corneal repair. In addition, its optical properties are similar to commercial ones.
As tissue substitutes, the gels shall be endowed with equal or enhanced mechanical characteristics. The difficulty of preparing a permanent substitute is another issue, and research on how to preserve the integration of hydrogels is insufficient [36]. The physical crosslinking of CBH preserves the stimuli-responsibility and biodegradability of hydrogels; the latter is disadvantageous in this case. With coordination crosslinking between chitosan and nanoparticles [86] or other polymers, the mechanical properties of hydrogels can be enhanced. Dual-physical crosslinking is able to enhance stability, depending on other precursors [87]. The cytotoxicity of chemical agents and the inability to react to stimuli are obstacles, though they foster stronger covalent crosslinking. Exploiting novel agents with higher biocompatibility and lower cytotoxicity is one feasible way. Combining the two approaches is another potential course of action, which would give CBH some intriguing advantages and make it possible to produce hydrogels with better mechanical and structural stability while preserving biocompatibility and biodegradability [88]. Tough and lubricated BAU is a great artificial cartilage that functions as native tissue [89]. The gel membrane with high toughness, immunomodulatory, and anti-adhesion properties may replace the biological ones such as the dura mater and diaphragm [90]. UCST hydrogels are prone to application due to the in situ formation of these gels, enabling injection in MIS because they are a liquid solution at room temperature and form a gel at body temperature, avoiding surgery [90][91]. Combining this property with pH sensitivity, CBHs could release drugs in a controlled manner [84], achieving the treatment incidentally. Ni et al. [91] synthesized hydrogels, forming a cushion to lift the lesion area, adhere to the wound, and protect the micro-wound from acidic environments.
6. Molecular Detection Biosensors
Biosensors, a type of analytical tool, consist of biological elements with physicochemical detectors that perceive and quantify the presence or concentration of particular compounds in a sample. Herein, molecular detection biosensors made of CBHs have been set into immobilization matrixes and responsive units. The former is quantitative, inexpensive, and uncomplicated to use and detect in vitro. Nonetheless, the sampling process is typically uncomfortable for the patients. The latter makes the most of the properties of the CBHs, including their high stiffness, sensitivity, avoidance of surgery, etc., but it necessitates various instrumentation for configuration and rigorous manufacturing standards for products.
6.1. Immobilization Matrix
The ability of qualitative biosensors to convert or magnify an unquantifiable signal into a quantifiable one is a crucial feature. One strategy is to utilize the rest of the reactive groups of CBH to graft other bioscopes for transformation. The low limit of detection indicates sensitivity. Immobilization ability, linear range, and surface area-to-volume ratio are critical parameters. Most immobilization matrixes are made into membrane or layer formation because they have a higher surface area-to-volume ratio, meaning that more biomolecules or nanomaterials can be attached to the hydrogel surface, which can improve the sensitivity, selectivity, and stability of the biosensor [92]. In order to create hydrogels with improved mechanical stability and enzyme adsorption capacity, additional polymers can be grafted onto chitosan [93]. Another method is to combine nanoparticles with chitosan to create nanocomposites that have improved conductivity and sensitivity [92]. An enzyme, antibody, or nucleic acid, for example, interacts with the target substance to produce a signal that the physicochemical detector can detect and quantify. Inserting nucleic acid could be implemented to find the complement sequence that measures the level of gene expression, making it easier to diagnose at the gene level [94]. The electrochemical reaction enzyme can transform the concentration of the target substrate into detectable electric signals received by the electrode [95][96][97]. While it is important to take precautions before detection because otherwise these molecules are easily deactivated or degraded, the detecting circumstances are typically complex and may have an impact on the stability of bioprobes, CBHs, and the bonding between them. Another concern is that the sensitivity is easily affected by the solution conductivity. Therefore, it is necessary to increase the stability and set the calibration. Additionally, another technique uses variations in optical signals brought on by the blend of a detection target and CBHs [98][99].
6.2. Responsive Units
Ebrahimi et al. [100] reported a method by which the presence and concentration of the enzyme can be indicated by the intensity of the fluorogenic substrate, which is covalently conjugated to chitosan and will be released after degradation. Another strategy is to detect hydrogel conductivity. This method is used in detecting vitality signals, e.g., respiratory movements, heart beating, and motions, by inducing minor deformation, which is enough to change the inherent conductivity of the hydrogel [101][102]. Electrical signals are the common output of this method; they rely on the electrode to sense the current change. Conductivity is critical to the sensitivity of biosensors. Given that the hydrogel itself is not conductive, adding carbon nanotubes [101], salt content [102], metal ions [103], etc., can improve it. In addition to improving wear resistance, oriented 3D networks also give CBH enhanced linearity over a broad strain sensing range [89], increasing accuracy. Moreover, many properties of CBHs facilitate biomedical applications including self-healing and adhesion-elongating duration in vivo [101]. The anti-swelling feature maintains mechanical toughness and strength across a variety of deformations and pressures, guaranteeing the stability and dependability of CBH sensors in a variety of settings and applications [103].
References
- Arif, Z.U.; Khalid, M.Y.; Sheikh, M.F.; Zolfagharian, A.; Bodaghi, M. Biopolymeric sustainable materials and their emerging applications. J. Environ. Chem. Eng. 2022, 10, 108159.
- Wei, W.; Ma, Y.; Yao, X.; Zhou, W.; Wang, X.; Li, C.; Lin, J.; He, Q.; Leptihn, S.; Ouyang, H. Advanced hydrogels for the repair of cartilage defects and regeneration. Bioact. Mater. 2021, 6, 998–1011.
- Palma, P.; Matos, S.; Ramos, J.; Guerra, F.; Figueiredo, M.; Kauser, J. New formulations for space provision and bone regeneration. Biodental Eng. I 2010, 1, 71–76.
- Xu, F.; Dawson, C.; Lamb, M.; Mueller, E.; Stefanek, E.; Akbari, M.; Hoare, T. Hydrogels for tissue engineering: Addressing key design needs toward clinical translation. Front. Bioeng. Biotechnol. 2022, 10, 849831.
- Rajabi, M.; McConnell, M.; Cabral, J.; Ali, M.A. Chitosan hydrogels in 3D printing for biomedical applications. Carbohydr. Polym. 2021, 260, 117768.
- Ahmed, A.; Arya, S.; Gupta, V.; Furukawa, H.; Khosla, A. 4D printing: Fundamentals, materials, applications and challenges. Polymer 2021, 228, 123926.
- Arif, Z.U.; Khalid, M.Y.; Noroozi, R.; Sadeghianmaryan, A.; Jalalvand, M.; Hossain, M. Recent advances in 3D-printed polylactide and polycaprolactone-based biomaterials for tissue engineering applications. Int. J. Biol. Macromol. 2022, 218, 930–968.
- Arif, Z.U.; Khalid, M.Y.; Noroozi, R.; Hossain, M.; Shi, H.H.; Tariq, A.; Ramakrishna, S.; Umer, R. Additive manufacturing of sustainable biomaterials for biomedical applications. Asian J. Pharm. Sci. 2023; 100812, in press.
- Xu, J.; Zhang, M.; Du, W.; Zhao, J.; Ling, G.; Zhang, P. Chitosan-based high-strength supramolecular hydrogels for 3D bioprinting. Int. J. Biol. Macromol. 2022, 219, 545–557.
- Dias, J.R.; Ribeiro, N.; Baptista-Silva, S.; Costa-Pinto, A.R.; Alves, N.; Oliveira, A.L. In situ enabling approaches for tissue regeneration: Current challenges and new developments. Front. Bioeng. Biotechnol. 2020, 8, 85.
- Ding, X.; Wang, Y.; Liu, J.; Zhang, P.; Li, G.; Sun, T.; Xiao, C. Injectable in situ forming double-network hydrogel to enhance transplanted cell viability and retention. Chem. Mater. 2021, 33, 5885–5895.
- Nicodemus, G.D.; Bryant, S.J. Cell encapsulation in biodegradable hydrogels for tissue engineering applications. Tissue Eng. Part B Rev. 2008, 14, 149–165.
- Xu, H.; Matysiak, S. Effect of pH on chitosan hydrogel polymer network structure. Chem. Commun. 2017, 53, 7373–7376.
- Zhou, L.; Ramezani, H.; Sun, M.; Xie, M.; Nie, J.; Lv, S.; Cai, J.; Fu, J.; He, Y. 3D printing of high-strength chitosan hydrogel scaffolds without any organic solvents. Biomater. Sci. 2020, 8, 5020–5028.
- Tang, G.; Tan, Z.; Zeng, W.; Wang, X.; Shi, C.; Liu, Y.; He, H.; Chen, R.; Ye, X. Recent advances of chitosan-based injectable hydrogels for bone and dental tissue regeneration. Front. Bioeng. Biotechnol. 2020, 8, 587658.
- Ashok, N.; Pradeep, A.; Jayakumar, R. Synthesis-structure relationship of chitosan based hydrogels. In Chitosan for Biomaterials III: Structure—Property Relationships; Springer: Berlin/Heidelberg, Germany, 2021; pp. 105–129.
- Li, P.; Fu, L.; Liao, Z.; Peng, Y.; Ning, C.; Gao, C.; Zhang, D.; Sui, X.; Lin, Y.; Liu, S. Chitosan hydrogel/3D-printed poly (ε-caprolactone) hybrid scaffold containing synovial mesenchymal stem cells for cartilage regeneration based on tetrahedral framework nucleic acid recruitment. Biomaterials 2021, 278, 121131.
- Chen, H.; Li, B.; Feng, B.; Wang, H.; Yuan, H.; Xu, Z. Tetracycline hydrochloride loaded citric acid functionalized chitosan hydrogel for wound healing. RSC Adv. 2019, 9, 19523–19530.
- Xu, X.; Zeng, Y.; Chen, Z.; Yu, Y.; Wang, H.; Lu, X.; Zhao, J.; Wang, S. Chitosan-based multifunctional hydrogel for sequential wound inflammation elimination, infection inhibition, and wound healing. Int. J. Biol. Macromol. 2023, 235, 123847.
- Ji, X.; Shao, H.; Li, X.; Ullah, M.W.; Luo, G.; Xu, Z.; Ma, L.; He, X.; Lei, Z.; Li, Q. Injectable immunomodulation-based porous chitosan microspheres/HPCH hydrogel composites as a controlled drug delivery system for osteochondral regeneration. Biomaterials 2022, 285, 121530.
- Patel, D.K.; Dutta, S.D.; Hexiu, J.; Ganguly, K.; Lim, K.-T. 3D-printable chitosan/silk fibroin/cellulose nanoparticle scaffolds for bone regeneration via M2 macrophage polarization. Carbohydr. Polym. 2022, 281, 119077.
- Hao, T.; Wen, N.; Cao, J.-K.; Wang, H.-B.; Lü, S.-H.; Liu, T.; Lin, Q.-X.; Duan, C.-M.; Wang, C.-Y. The support of matrix accumulation and the promotion of sheep articular cartilage defects repair in vivo by chitosan hydrogels. Osteoarthr. Cartil. 2010, 18, 257–265.
- Sumey, J.L.; Harrell, A.M.; Johnston, P.C.; Caliari, S.R. Serial passaging affects stromal cell mechanosensitivity on hyaluronic acid hydrogels. bioRxiv 2003.
- Schrader, J.; Gordon-Walker, T.T.; Aucott, R.L.; van Deemter, M.; Quaas, A.; Walsh, S.; Benten, D.; Forbes, S.J.; Wells, R.G.; Iredale, J.P. Matrix stiffness modulates proliferation, chemotherapeutic response, and dormancy in hepatocellular carcinoma cells. Hepatology 2011, 53, 1192–1205.
- Chang, F.-C.; Tsao, C.-T.; Lin, A.; Zhang, M.; Levengood, S.L.; Zhang, M. PEG-chitosan hydrogel with tunable stiffness for study of drug response of breast cancer cells. Polymers 2016, 8, 112.
- Batool, S.R.; Nazeer, M.A.; Yildiz, E.; Sahin, A.; Kizilel, S. Chitosan-anthracene hydrogels as controlled stiffening networks. Int. J. Biol. Macromol. 2021, 185, 165–175.
- Ye, J.; Fu, S.; Zhou, S.; Li, M.; Li, K.; Sun, W.; Zhai, Y. Advances in hydrogels based on dynamic covalent bonding and prospects for its biomedical application. Eur. Polym. J. 2020, 139, 110024.
- Lu, C.-H.; Yu, C.-H.; Yeh, Y.-C. Engineering nanocomposite hydrogels using dynamic bonds. Acta Biomater. 2021, 130, 66–79.
- Li, Y.; Sun, S.; Gao, P.; Zhang, M.; Fan, C.; Lu, Q.; Li, C.; Chen, C.; Lin, B.; Jiang, Y. A tough chitosan-alginate porous hydrogel prepared by simple foaming method. J. Solid State Chem. 2021, 294, 121797.
- Luo, M.; Wang, X.; Meng, T.; Yang, P.; Zhu, Z.; Min, H.; Chen, M.; Chen, W.; Zhou, X. Rapid one-step preparation of hierarchical porous carbon from chitosan-based hydrogel for high-rate supercapacitors: The effect of gelling agent concentration. Int. J. Biol. Macromol. 2020, 146, 453–461.
- Wasupalli, G.K.; Verma, D. Thermosensitive injectable hydrogel based on chitosan-polygalacturonic acid polyelectrolyte complexes for bone tissue engineering. Carbohydr. Polym. 2022, 294, 119769.
- Aguanell, A.; Del Pozo, M.L.; Pérez-Martín, C.; Pontes, G.; Bastida, A.; Fernández-Mayoralas, A.; García-Junceda, E.; Revuelta, J. Chitosan sulfate-lysozyme hybrid hydrogels as platforms with fine-tuned degradability and sustained inherent antibiotic and antioxidant activities. Carbohydr. Polym. 2022, 291, 119611.
- Han, Y.; Lian, M.; Wu, Q.; Qiao, Z.; Sun, B.; Dai, K. Effect of pore size on cell behavior using melt electrowritten scaffolds. Front. Bioeng. Biotechnol. 2021, 9, 629270.
- Gao, B.; Yang, Q.; Zhao, X.; Jin, G.; Ma, Y.; Xu, F. 4D bioprinting for biomedical applications. Trends Biotechnol. 2016, 34, 746–756.
- Arif, Z.U.; Khalid, M.Y.; Ahmed, W.; Arshad, H. A review on four-dimensional bioprinting in pursuit of advanced tissue engineering applications. Bioprinting 2022, 27, e00203.
- Matica, A.; Menghiu, G.; Ostafe, V. Biodegradability of chitosan based products. New Front. Chem. 2017, 26, 75–86.
- Tan, H.; Marra, K.G. Injectable, biodegradable hydrogels for tissue engineering applications. Materials 2010, 3, 1746–1767.
- Bano, I.; Arshad, M.; Yasin, T.; Ghauri, M.A.; Younus, M. Chitosan: A potential biopolymer for wound management. Int. J. Biol. Macromol. 2017, 102, 380–383.
- Zhang, A.; Liu, Y.; Qin, D.; Sun, M.; Wang, T.; Chen, X. Research status of self-healing hydrogel for wound management: A review. Int. J. Biol. Macromol. 2020, 164, 2108–2123.
- Reay, S.L.; Jackson, E.L.; Ferreira, A.M.; Hilkens, C.M.; Novakovic, K. In vitro evaluation of the biodegradability of chitosan–genipin hydrogels. Mater. Adv. 2022, 3, 7946–7959.
- Kasiński, A.; Zielińska-Pisklak, M.; Oledzka, E.; Sobczak, M. Smart hydrogels–synthetic stimuli-responsive antitumor drug release systems. Int. J. Nanomed. 2020, 15, 4541–4572.
- Da Silva, S.B.; Krolicka, M.; van den Broek, L.A.; Frissen, A.E.; Boeriu, C.G. Water-soluble chitosan derivatives and pH-responsive hydrogels by selective C-6 oxidation mediated by TEMPO-laccase redox system. Carbohydr. Polym. 2018, 186, 299–309.
- Li, C.-P.; Weng, M.-C.; Huang, S.-L. Preparation and characterization of pH sensitive Chitosan/3-Glycidyloxypropyl Trimethoxysilane (GPTMS) hydrogels by sol-gel method. Polymers 2020, 12, 1326.
- Anderson, N.M.; Simon, M.C. The tumor microenvironment. Curr. Biol. 2020, 30, R921–R925.
- Jin, M.-Z.; Jin, W.-L. The updated landscape of tumor microenvironment and drug repurposing. Signal Transduct. Target. Ther. 2020, 5, 166.
- Abdellatif, A.A.; Mohammed, A.M.; Saleem, I.; Alsharidah, M.; Al Rugaie, O.; Ahmed, F.; Osman, S.K. Smart injectable chitosan hydrogels loaded with 5-fluorouracil for the treatment of breast cancer. Pharmaceutics 2022, 14, 661.
- Wang, J.X.; Choi, S.Y.; Niu, X.; Kang, N.; Xue, H.; Killam, J.; Wang, Y. Lactic acid and an acidic tumor microenvironment suppress anticancer immunity. Int. J. Mol. Sci. 2020, 21, 8363.
- Huo, W.; Yang, X.; Wang, B.; Cao, L.; Fang, Z.; Li, Z.; Liu, H.; Liang, X.-J.; Zhang, J.; Jin, Y. Biomineralized hydrogel DC vaccine for cancer immunotherapy: A boosting strategy via improving immunogenicity and reversing immune-inhibitory microenvironment. Biomaterials 2022, 288, 121722.
- Zhang, W.; Jin, X.; Li, H.; Wei, C.-X.; Wu, C.-W. Onion-structure bionic hydrogel capsules based on chitosan for regulating doxorubicin release. Carbohydr. Polym. 2019, 209, 152–160.
- Sponchioni, M.; Palmiero, U.C.; Moscatelli, D. Thermo-responsive polymers: Applications of smart materials in drug delivery and tissue engineering. Mater. Sci. Eng. C 2019, 102, 589–605.
- Bedford, J.G.; Caminschi, I.; Wakim, L.M. Intranasal delivery of a chitosan-hydrogel vaccine generates nasal tissue resident memory CD8+ T cells that are protective against influenza virus infection. Vaccines 2020, 8, 572.
- Tian, B.; Hua, S.; Tian, Y.; Liu, J. Chemical and physical chitosan hydrogels as prospective carriers for drug delivery: A review. J. Mater. Chem. B 2020, 8, 10050–10064.
- Thanou, M.; Verhoef, J.; Junginger, H. Oral drug absorption enhancement by chitosan and its derivatives. Adv. Drug Deliv. Rev. 2001, 52, 117–126.
- Delmar, K.; Bianco-Peled, H. Composite chitosan hydrogels for extended release of hydrophobic drugs. Carbohydr. Polym. 2016, 136, 570–580.
- Ali, M.K.; Moshikur, R.M.; Wakabayashi, R.; Moniruzzaman, M.; Kamiya, N.; Goto, M. Biocompatible ionic liquid surfactant-based microemulsion as a potential carrier for sparingly soluble drugs. ACS Sustain. Chem. Eng. 2020, 8, 6263–6272.
- Guo, L.; Guan, Y.; Liu, P.; Gao, L.; Wang, Z.; Huang, S.; Peng, L.; Zhao, Z. Chitosan hydrogel, as a biological macromolecule-based drug delivery system for exosomes and microvesicles in regenerative medicine: A mini review. Cellulose 2022, 29, 1315–1330.
- Ratajczak, M.Z.; Ratajczak, J. Extracellular microvesicles/exosomes: Discovery, disbelief, acceptance, and the future? Leukemia 2020, 34, 3126–3135.
- Feng, P.; Luo, Y.; Ke, C.; Qiu, H.; Wang, W.; Zhu, Y.; Hou, R.; Xu, L.; Wu, S. Chitosan-based functional materials for skin wound repair: Mechanisms and applications. Front. Bioeng. Biotechnol. 2021, 9, 650598.
- Yang, E.; Hou, W.; Liu, K.; Yang, H.; Wei, W.; Kang, H.; Dai, H. A multifunctional chitosan hydrogel dressing for liver hemostasis and infected wound healing. Carbohydr. Polym. 2022, 291, 119631.
- Chen, Z.; Zhao, J.; Wu, H.; Wang, H.; Lu, X.; Shahbazi, M.-A.; Wang, S. A triple-network carboxymethyl chitosan-based hydrogel for hemostasis of incompressible bleeding on wet wound surfaces. Carbohydr. Polym. 2023, 303, 120434.
- Song, K.; Hao, Y.; Liu, Y.; Cao, R.; Zhang, X.; He, S.; Wen, J.; Zheng, W.; Wang, L.; Zhang, Y. Preparation of pectin-chitosan hydrogels based on bioadhesive-design micelle to prompt bacterial infection wound healing. Carbohydr. Polym. 2023, 300, 120272.
- Zhang, X.; Tan, B.; Wu, Y.; Zhang, M.; Xie, X.; Liao, J. An injectable, self-healing carboxymethylated chitosan hydrogel with mild photothermal stimulation for wound healing. Carbohydr. Polym. 2022, 293, 119722.
- Wei, H.; Yu, S.; Zhang, Y.; Zhang, H.; Ma, Y.; Xu, M.; An, P.; Zhou, Y.; Halila, S.; Wei, Y. Injectable chitosan/xyloglucan composite hydrogel with mechanical adaptivity and endogenous bioactivity for skin repair. Carbohydr. Polym. 2023, 313, 120904.
- Du, X.; Wu, L.; Yan, H.; Jiang, Z.; Li, S.; Li, W.; Bai, Y.; Wang, H.; Cheng, Z.; Kong, D. Microchannelled alkylated chitosan sponge to treat noncompressible hemorrhages and facilitate wound healing. Nat. Commun. 2021, 12, 4733.
- Yu, Q.; Su, B.; Zhao, W.; Zhao, C. Janus self-propelled chitosan-based hydrogel spheres for rapid bleeding control. Adv. Sci. 2023, 10, 2205989.
- Weng, H.; Jia, W.; Li, M.; Chen, Z. New injectable chitosan-hyaluronic acid based hydrogels for hemostasis and wound healing. Carbohydr. Polym. 2022, 294, 119767.
- Tengborn, L.; Blombäck, M.; Berntorp, E. Tranexamic acid—an old drug still going strong and making a revival. Thromb. Res. 2015, 135, 231–242.
- Song, X.; Zhao, Y.; Liu, Y.; Zhang, W.; Yuan, X.; Xu, L.; Zhang, J. Effects of degree of deacetylation on hemostatic performance of partially deacetylated chitin sponges. Carbohydr. Polym. 2021, 273, 118615.
- Tashakkorian, H.; Hasantabar, V.; Mostafazadeh, A.; Golpour, M. Transparent chitosan based nanobiocomposite hydrogel: Synthesis, thermophysical characterization, cell adhesion and viability assay. Int. J. Biol. Macromol. 2020, 144, 715–724.
- Yan, D.; Li, Y.; Liu, Y.; Li, N.; Zhang, X.; Yan, C. Antimicrobial properties of chitosan and chitosan derivatives in the treatment of enteric infections. Molecules 2021, 26, 7136.
- Ke, C.-L.; Deng, F.-S.; Chuang, C.-Y.; Lin, C.-H. Antimicrobial actions and applications of chitosan. Polymers 2021, 13, 904.
- Qing, X.; He, G.; Liu, Z.; Yin, Y.; Cai, W.; Fan, L.; Fardim, P. Preparation and properties of polyvinyl alcohol/N–succinyl chitosan/lincomycin composite antibacterial hydrogels for wound dressing. Carbohydr. Polym. 2021, 261, 117875.
- Huan, Y.; Kong, Q.; Tang, Q.; Wang, Y.; Mou, H.; Ying, R.; Li, C. Antimicrobial peptides/ciprofloxacin-loaded O-carboxymethyl chitosan/self-assembling peptides hydrogel dressing with sustained-release effect for enhanced anti-bacterial infection and wound healing. Carbohydr. Polym. 2022, 280, 119033.
- Hemmingsen, L.M.; Giordani, B.; Pettersen, A.K.; Vitali, B.; Basnet, P.; Škalko-Basnet, N. Liposomes-in-chitosan hydrogel boosts potential of chlorhexidine in biofilm eradication in vitro. Carbohydr. Polym. 2021, 262, 117939.
- Yatvin, J.; Gao, J.; Locklin, J. Durable defense: Robust and varied attachment of non-leaching poly “-onium” bactericidal coatings to reactive and inert surfaces. Chem. Commun. 2014, 50, 9433–9442.
- Yin, M.; Wan, S.; Ren, X.; Chu, C.-C. Development of inherently antibacterial, biodegradable, and biologically active chitosan/pseudo-protein hybrid hydrogels as biofunctional wound dressings. ACS Appl. Mater. Interfaces 2021, 13, 14688–14699.
- Xiao, J.; Zhou, Y.; Ye, M.; An, Y.; Wang, K.; Wu, Q.; Song, L.; Zhang, J.; He, H.; Zhang, Q. Freeze-thawing chitosan/ions hydrogel coated gauzes releasing multiple metal ions on demand for improved infected wound healing. Adv. Healthc. Mater. 2021, 10, 2001591.
- Xu, H.; Zheng, Y.-W.; Liu, Q.; Liu, L.-P.; Luo, F.-L.; Zhou, H.-C.; Isoda, H.; Ohkohchi, N.; Li, Y.-M. Reactive oxygen species in skin repair, regeneration, aging, and inflammation. React. Oxyg. Species Living Cells 2018, 8, 69–88.
- Yuan, Y.; Tan, W.; Zhang, J.; Li, Q.; Guo, Z. Water-soluble amino functionalized chitosan: Preparation, characterization, antioxidant and antibacterial activities. Int. J. Biol. Macromol. 2022, 217, 969–978.
- Yen, M.-T.; Yang, J.-H.; Mau, J.-L. Antioxidant properties of chitosan from crab shells. Carbohydr. Polym. 2008, 74, 840–844.
- Zhong, Y.; Seidi, F.; Wang, Y.; Zheng, L.; Jin, Y.; Xiao, H. Injectable chitosan hydrogels tailored with antibacterial and antioxidant dual functions for regenerative wound healing. Carbohydr. Polym. 2022, 298, 120103.
- Wang, M.; Huang, H.; Huang, C.; Liu, S.; Peng, X. pH-responsive magnolol nanocapsule-embedded magnolol-grafted-chitosan hydrochloride hydrogels for promoting wound healing. Carbohydr. Polym. 2022, 292, 119643.
- Jiao, Z.; Huo, Q.; Lin, X.; Chu, X.; Deng, Z.; Guo, H.; Peng, Y.; Lu, S.; Zhou, X.; Wang, X. Drug-free contact lens based on quaternized chitosan and tannic acid for bacterial keratitis therapy and corneal repair. Carbohydr. Polym. 2022, 286, 119314.
- Wang, S.; Chi, J.; Jiang, Z.; Hu, H.; Yang, C.; Liu, W.; Han, B. A self-healing and injectable hydrogel based on water-soluble chitosan and hyaluronic acid for vitreous substitute. Carbohydr. Polym. 2021, 256, 117519.
- Yang, C.; Wang, M.; Wang, W.; Liu, H.; Deng, H.; Du, Y.; Shi, X. Electrodeposition induced covalent cross-linking of chitosan for electrofabrication of hydrogel contact lenses. Carbohydr. Polym. 2022, 292, 119678.
- Ao, Y.; Zhang, E.; Liu, Y.; Yang, L.; Li, J.; Wang, F. Advanced Hydrogels With Nanoparticle Inclusion for Cartilage Tissue Engineering. Adv. Biomater. Osteochondral Regen. 2023, 16648714, 32.
- Lin, Y.; Wang, S.; Sun, S.; Liang, Y.; Xu, Y.; Hu, H.; Luo, J.; Zhang, H.; Li, G. Highly tough and rapid self-healing dual-physical crosslinking poly (DMAA-co-AM) hydrogel. RSC Adv. 2021, 11, 32988–32995.
- Wang, Y.; Zhang, W.; Gong, X.; Zhao, C.; Liu, Y.; Zhang, C. Construction of carboxymethyl chitosan hydrogel with multiple cross-linking networks for electronic devices at low temperature. ACS Biomater. Sci. Eng. 2022, 9, 508–519.
- Li, K.; Yang, X.; Dong, X.; Cao, H.; Zhuang, S.; Gu, X. Easy regulation of chitosan-based hydrogel microstructure with citric acid as an efficient buffer. Carbohydr. Polym. 2023, 300, 120258.
- Yu, H.C.; Zhang, H.; Ren, K.; Ying, Z.; Zhu, F.; Qian, J.; Ji, J.; Wu, Z.L.; Zheng, Q. Ultrathin κ-carrageenan/chitosan hydrogel films with high toughness and antiadhesion property. ACS Appl. Mater. Interfaces 2018, 10, 9002–9009.
- Ni, P.; Li, R.; Ye, S.; Shan, J.; Yuan, T.; Liang, J.; Fan, Y.; Zhang, X. Lactobionic acid-modified chitosan thermosensitive hydrogels that lift lesions and promote repair in endoscopic submucosal dissection. Carbohydr. Polym. 2021, 263, 118001.
- Jiang, Y.; Wu, J. Recent development in chitosan nanocomposites for surface-based biosensor applications. Electrophoresis 2019, 40, 2084–2097.
- Facin, B.R.; Moret, B.; Baretta, D.; Belfiore, L.A.; Paulino, A.T. Immobilization and controlled release of β-galactosidase from chitosan-grafted hydrogels. Food Chem. 2015, 179, 44–51.
- Mohammadi, S.; Mohammadi, S.; Salimi, A. A 3D hydrogel based on chitosan and carbon dots for sensitive fluorescence detection of microRNA-21 in breast cancer cells. Talanta 2021, 224, 121895.
- Chmayssem, A.; Shalayel, I.; Marinesco, S.; Zebda, A. Investigation of GOx stability in a chitosan matrix: Applications for enzymatic electrodes. Sensors 2023, 23, 465.
- Johnson, D.; Kim, U.; Mobed-Miremadi, M. Nanocomposite films as electrochemical sensors for detection of catalase activity. Front. Mol. Biosci. 2022, 9, 972008.
- Kemp, E.; Palomäki, T.; Ruuth, I.A.; Boeva, Z.A.; Nurminen, T.A.; Vänskä, R.T.; Zschaechner, L.K.; Perez, A.G.; Hakala, T.A.; Wardale, M. Influence of enzyme immobilization and skin-sensor interface on non-invasive glucose determination from interstitial fluid obtained by magnetohydrodynamic extraction. Biosens. Bioelectron. 2022, 206, 114123.
- Pal, A.K.; Goddard, N.J.; Dixon, H.J.; Gupta, R. A self-referenced diffraction-based optical leaky waveguide biosensor using photofunctionalised hydrogels. Biosensors 2020, 10, 134.
- Ye, Y.; Qi, X.; Wang, H.; Zhao, B.; Xu, L.; Zhang, Y.; Wang, X.; Zhou, N. A surface-enhanced Raman scattering aptasensor for Escherichia coli detection based on high-performance 3D substrate and hot spot effect. Anal. Chim. Acta 2022, 1221, 340141.
- Sadat Ebrahimi, M.M.; Schönherr, H. Enzyme-sensing chitosan hydrogels. Langmuir 2014, 30, 7842–7850.
- Ren, Z.; Ke, T.; Ling, Q.; Zhao, L.; Gu, H. Rapid self-healing and self-adhesive chitosan-based hydrogels by host-guest interaction and dynamic covalent bond as flexible sensor. Carbohydr. Polym. 2021, 273, 118533.
- Hu, Y.; Liu, N.; Chen, K.; Liu, M.; Wang, F.; Liu, P.; Zhang, Y.; Zhang, T.; Xiao, X. Resilient and self-healing hyaluronic acid/chitosan hydrogel with ion conductivity, low water loss, and freeze-tolerance for flexible and wearable strain sensor. Front. Bioeng. Biotechnol. 2022, 10, 837750.
- Wang, T.; Ren, X.; Bai, Y.; Liu, L.; Wu, G. Adhesive and tough hydrogels promoted by quaternary chitosan for strain sensor. Carbohydr. Polym. 2021, 254, 117298.
More
Information
Subjects:
Materials Science, Biomaterials
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.8K
Entry Collection:
Biopharmaceuticals Technology
Revisions:
2 times
(View History)
Update Date:
13 Jun 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





