
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Pierfrancesco Morganti | -- | 2579 | 2023-06-06 02:11:53 | | | |
| 2 | Lindsay Dong | + 3 word(s) | 2582 | 2023-06-06 10:18:50 | | | | |
| 3 | Lindsay Dong | Meta information modification | 2582 | 2023-06-06 10:19:17 | | |
Video Upload Options
Beauty and personal care became a significant part of the global economy for two reasons: (1) The elderly growing in the global population and (2) the desire of women and men to appear younger and more attractive. Thus, both young and old people are looking for revolutionary nutritional eco-cosmetics (combined use of cosmeceuticals and nutraceuticals) manufactured by natural active ingredients, using biopolymers as substrates, and made by innovative and sustainable technologies. Consequently, the market of both cosmetics and diet supplements is continually growing together with the request of natural active ingredients, including bio-peptides and biological macromolecules such as chitin and lignin. Therefore, both consumers and industry need to recover innovative active ingredients and carriers (vehicles), naturally derived and supported by advanced methods for controlling their effectiveness and safeness on skin and mucous membrane layers. The use of selected bio-ingredients, such as hyaluronic acid and bio-mimetic peptides, obtained by advanced, innovative and sustainable bio nanotechnologies, will be of interest to develop smart cosmeceutical and nutraceutical formulations. Innovation is considered the key business strategy to drive sustainable economic growth. For trying to reduce waste and produce sustainable, biodegradable and innovative products, the realization of new non-woven tissues, used as carriers for making innovative cosmeceuticals and nutraceuticals was considered. Both carriers and active ingredients have been obtained from food waste to reduce loss and pollution.
1. Introduction
2. Active Ingredients of Bio-Nanotechnology
The market for active ingredients for both cosmetics and beauty diet supplements is increasing year by year [1][2]. Thus, consumers and companies, looking for the best way to address a younger appearance try to find more effective formulations that are able to restore a healthy prematurely aged or altered skin [11][12]. As a result, it is essential to focus more studies on recovering active ingredients that can be used to make innovative cosmeceuticals and nutraceuticals, which are effective and safe for both skin health and the environment [13][14][15]. To this end, many scientific research studies have been oriented to the use of nano biotechnology-derived ingredients. With bio-nanotechnology, it seems possible to develop innovative compounds, new delivery systems and tools, which are able to control the product effectiveness and safeness at the level of the cell biology through new smart techniques, including the molecular biology, recombinant DNA, and gene and microbiome regulations [13][14][15]. New active micro-nano ingredients were, therefore, recovered and applied to different biological systems, including skin and mucous membranes, for overcoming the various biological barriers by the use, for example, of dermal and transdermal delivery systems [16][17][18][19]. Some active ingredients, such as chitin nanofibrils(CN) and nanolignin (NL), may be diffused by nanocarriers, such as liposomes, niosomes, innovative nanovesicles, matrices, nanocomposites and nanoparticles applied to biological systems, for example by electromagnetic fields [16][17][18][19].
However, what does bio-nanotechnology mean? It “is the branch of nanotechnology with biological and biochemical applications”, including the use of biomolecules as part of nanotechnological devices, supported by mathematic, physic, engineering, computer science and information technology [20]. This new branch of science, therefore, is currently developing environmentally-friendly processes that produce zero waste and low consumption of water and energy, which are requirements for obtaining a cleaner and healthier planet. Thus, in the sector of nutritional eco-cosmetics (used topically or by oral route), bio-nanotechnology is playing an important role, ameliorating performance and bioavailability of the active ingredients, which, for example, encapsulated or adsorbed on the surface of nanoparticles, might enhance their penetration through skin or mucous membranes [21].
3. Natural Ingredients Used in the Medical, Cosmetic and Diet Supplement Field
| Bioingredients | Source | References |
|---|---|---|
| Flavonoids, polyphenols | fruits, vegetables, cereals (Coffee, tomato, rice, etc.) | [27][28][29] |
| Salicilic/cinnamic acid | Aloe vera plant | [30] |
| Tocopherols/FFA | Argania Spinosa oil | [31] |
| Chitin | Crustaceans, fungi, insects | [32] |
| Lignin | Agro and forestry waste | [32] |
| Vitamin b and C, minerals | Potato | [33] |
| Cannabinoids, terpenes | Cannabis sativa | [34] |
| Minerals, proteins | Microalgae | [35][36] |
4. Structure of Skin
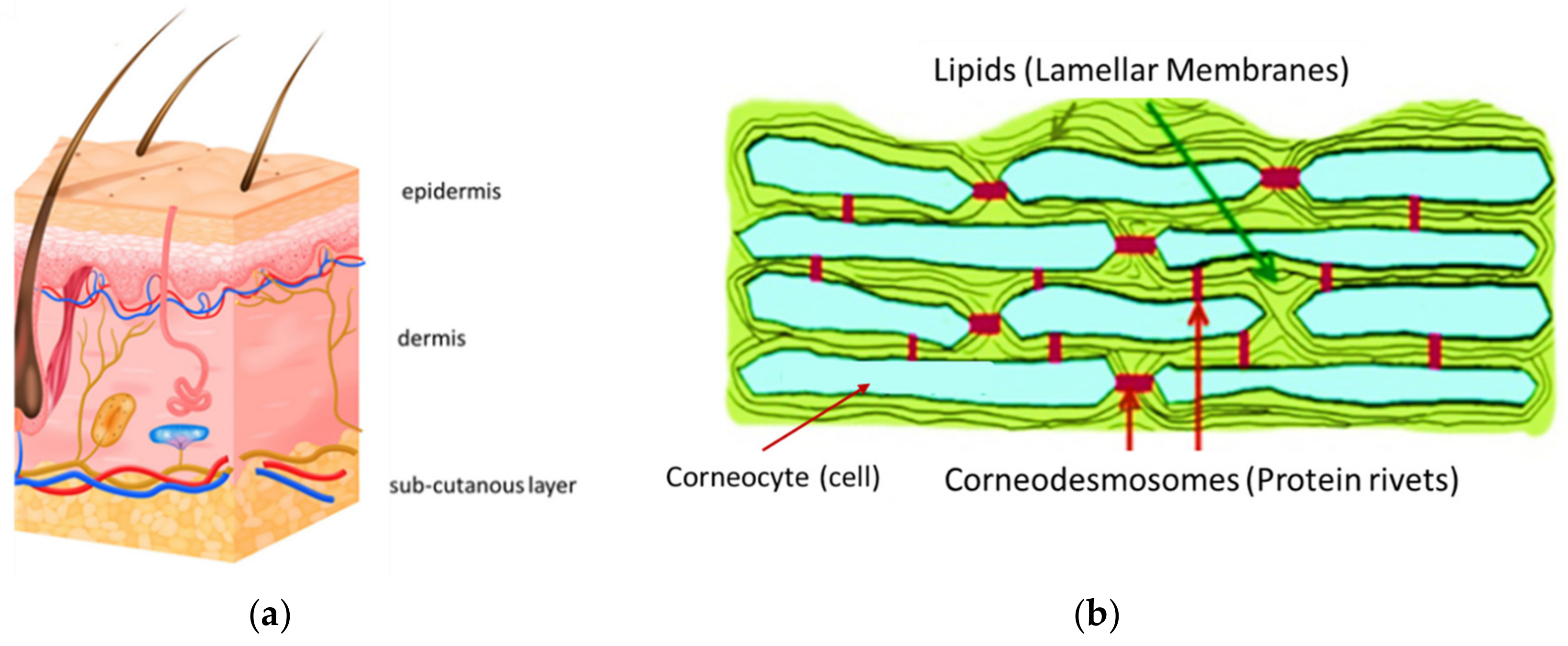
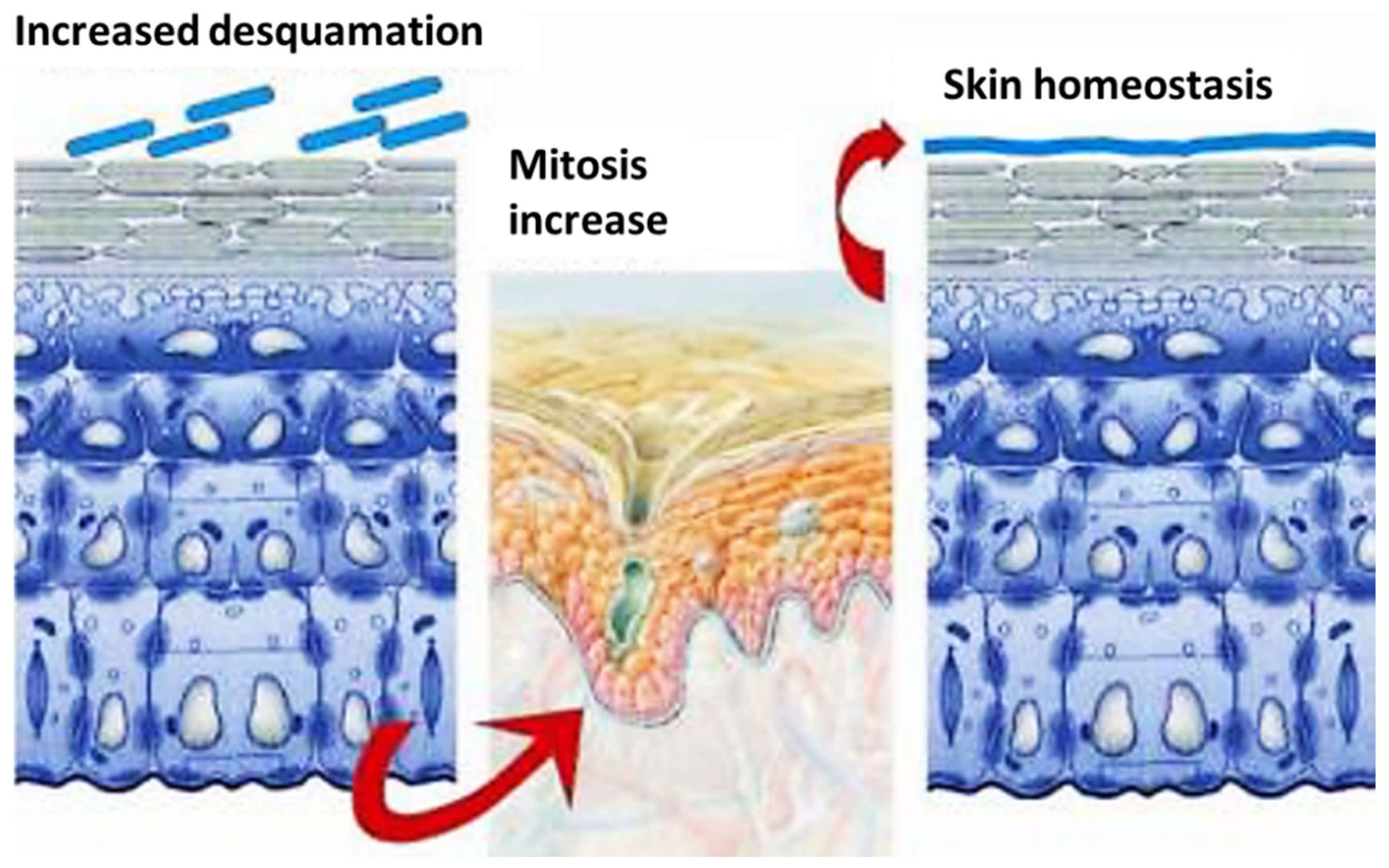
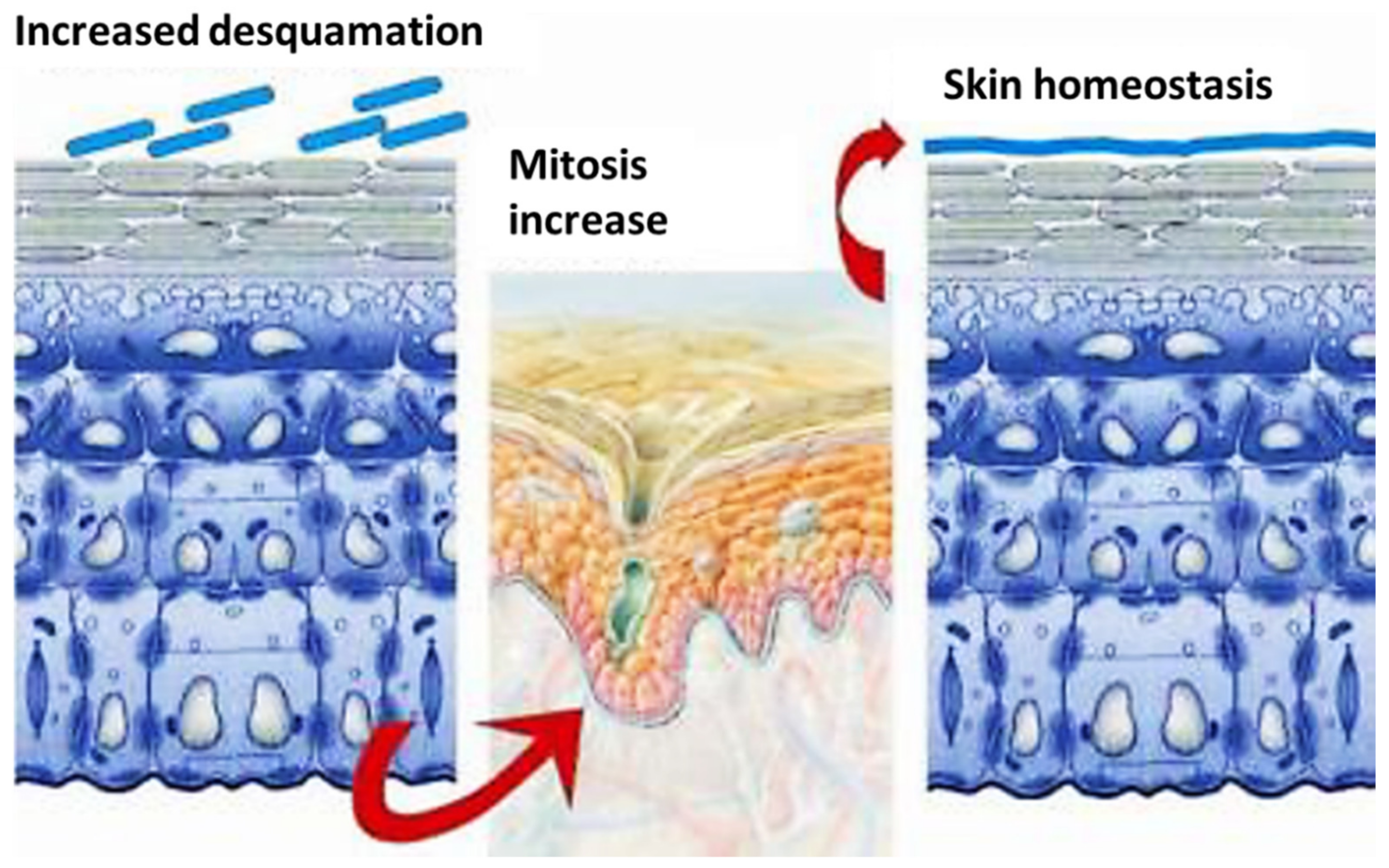
5. Skin Penetration and Supposed Mechanism of Action of Carriers/Active Ingredients
It is necessary to remember the different skin penetration modalities, achievable through three pathways -intercellular, intracellular and trans-follicular. They depend on the barrier disruption area and frequency of application, the degree of friction or rubbing of the product onto its surface and naturally to the composition of carrier and the selected active ingredients (Figure 3) [43][44][45]. The primary purpose of the carrier, in fact, is to enable the cosmetic active ingredients to be conveniently spread upon the skin for their better penetration through its layers. Consequently, composition and physicochemical characteristics of the carriers represent an important instrumental role of the products’ activity, which depends principally on the scaffold porous material used to make the carrier structure. The product effectiveness and safeness, in fact, are determined by the intrinsic activity of the selected active ingredients and the capacity of the carrier to deliver them at the right skin or mucous membrane layers [43][44][45].
The carrier, mimicking the skin ECM, may perform its essential functions, such as anchorage, proliferation and migration of the cells [46][47][48]. Thus, for example, the right porosity of the carrier-tissue permits growth and migration of the cells, which, through its pores, may have not only the necessary oxygen and nutrients but also the possibility to eliminate the waste materials [46]. This is the reason why many scientific research studies have been published during the last years on the topic of carriers and nanocarriers, including micro/nano emulsions, liposomes, lipid nanoparticles and polymeric nanocomposites, all realized with the aim to overcome the skin’ penetration problems [44][45][46][47][48][49][50][51].
Regarding the delivery of nutraceuticals, they can be administered by the same water-soluble carrier-tissues through the trans-mucosal sublingual route [52]. The oral mucosa, in fact, is characterized by a high permeability and capability to bypass the first-pass metabolism of liver. Thus, the active ingredients may slowly diffuse into the systemic circulation in a relatively short time, avoiding their distraction by the gastric juices [53][54][55]. Therefore, the necessity to realize specialized nano-carriers able to traverse this thick web, evading adhesion to the sticky mucin fibers [56][57][58]. Just for trying to better understand the problem, it is interesting to remark that mucosa, covered by an adhesive gel, the mucus, is composed of a density network of natural mucin polymers [52][53][54][59]. These macromolecules, interspersed with a variety of glycoproteins made by about 10 carbohydrate side chains, create a selective barrier to the diffusion of other macromolecular systems across mucosal surfaces [43][57].
6. Skin and Mucous Membrane: Penetration, Safeness and Effectiveness and the Ambiguity of the Rules
References
- Statista. Beauty & Personal Care-Worldwide, 2022 Statista Market Forecast. Available online: https://www.statista.com/outlook/cmo/beauty-personal-care/worldwide (accessed on 22 February 2022).
- Westbrook, G.; Angus, A. Top 10 Global Consumer Trends 2021, 2022 Euromonitor International. Available online: https://go.euromonitor.com/white-paper-EC-2022-Top-10-Global-Consumer-Trends.html?utm_source=website&utm_medium=website&utm_campaign=CT_22_01_18_WP_Top_10_GCT_2022_EN (accessed on 11 January 2023).
- Key, B.; Kohl, A.-K.; Elflein, J.; Puri-Mirza, A.; Sapun, P.; Cherowbrien, J. Market Size of Cosmetic Ingredients Worldwide 2016–2025. Statista Research Department. 2022. Available online: https://www.statista.com/statistics/627786/market-size-of-cosmetic-ingredients-worldwide/ (accessed on 22 February 2022).
- Beck, G.; Villena, K. Beauty in Recovery: Going Green and Clean, 2021 Euromonitor International. Available online: https://go.euromonitor.com/webinar-bpc-210622-beauty_state_of_play.html?utm_source=Press_release&utm_medium=PR&utm_campaign=WB_21_06_22_REC_BeautyStateofPlay (accessed on 13 January 2022).
- Villena, K. Clean Labels and Holistic Skin Wellness: The Crossover Between Skin Care and Dermatologicals, 2022 Cosmoprof, Las Vegas, Euromonitor International. Available online: https://cosmoprofnorthamerica.com/clean-labels-and-holistic-skin-wellness-the-crossover-between-skin-care-and-dermatologicals/ (accessed on 13 January 2022).
- Ross, G. A Perspective on the Safety of Cosmetic Products: A Position Paper of The American Council on Science and Health. Int. J. Toxicol. 2006, 25, 269–277.
- Morganti, P. Natural Products Work in Multiple Ways. In Nutritional Cosmetics. Beauty from Within; Tabor, A., Blair, R., Eds.; William Andrew Publishes: Norwich, NY, USA, 2009; pp. 95–1111.
- Kim, S.; Lee, Y. Why do women want to be beautiful? A qualitative study proposing a new “human beauty values” concept. PLoS ONE 2018, 13, e0201347.
- Fortune, B. Dietary Supplements Market Size, Share & COVID-19 Impact Analysis, by Type, Form Regional Forecasts, 2031–2028; Market Research Report; Fortune Business Insight: Pune, India, 2020.
- Feng, C. Substantial Innovation in the Cosmetic Industry-Obstacles, Contributing Factors and Strategies. Ph.D. Thesis, Minnesota University, Minneapolis, MN, USA, 2016.
- Jindal, S.; Kwek, S.; McDougall, A. Global Beauty and Personal Care Trends 2030. Mintel Report. 2020. Available online: https://www.mintel.com/beauty-trends-2030 (accessed on 11 January 2023).
- Hojnik, J.; Ruzzier, M.; Ruzzier, M.K. Transition towards Sustainability: Adoption of Eco-Products among Consumers. Sustainability 2019, 11, 4308.
- Byrd, A.L.; Belkaid, Y.; Segre, J.A. The human skin microbiome. Nat. Rev. Microbiol. 2018, 16, 143–155.
- Carlberg, C.; Molnár, F. (Eds.) Mechanisms of Gene Regulation; Springer: Berlin/Heidelberg, Germany, 2014.
- Khan, F.A. (Ed.) Biotechnology Fundamentals; CRC Press: Boca Raton, FL, USA, 2012; p. 6.
- Morganti, P.; Gagliardini, A.; Morganti, G. Nanochitin and nanolignin: Activity and effectiveness. In Biofunctional Textiles for an Aging Skin; Morganti, P., Ed.; Lambert Academic Publishing: Chisinau, Moldova, 2022; pp. 333–370.
- Ibrahim, M.M.; Nair, A.B.; Aldhubiab, B.E.; Shehata, B.E.A.A.T.M. Hydrogels and Their Combination with Liposomes, Niosomes, or Transfersomes for Dermal and Transdermal Drug Delivery. Liposomes 2017, 155–186.
- Bartelds, R.; Nematollahi, M.H.; Pols, T.; Stuart, M.C.A.; Pardakhty, A.; Asadikaram, G.; Poolman, B. Niosomes, an alternative for liposomal delivery. PLoS ONE 2018, 13, e0194179.
- Ge, X.; Wei, M.; He, S.; Yuan, W.-E. Advances on non-ionic surfactant vesicles (Niosomes) and their applications in drug delivery. Pharmaceutics 2019, 11, 55.
- Chaudhri, N.; Soni, G.C.; Prajapati, S.K. Nanotechnology: An advance tool for nanoCosmetics Preparation. Int. J. Pharma. Res. Rev. 2015, 4, 28–40.
- Soares, M.; Vittorino, C.; Sousa, J.; Pais, A. Skin permeation: Challenges and opportunities. Rev. Ciencias Farm. Basic. Appl. 2015, 21, 2698–2712.
- Draelos, Z.D. Skin-Lightening Challenges. In Cosmetically Active Ingredients: Recent Advances; Kozlowski, A., Ed.; Allured Books: Carol Stream, IL, USA, 2011; pp. 337–346. ISBN 978-1-932633-87-0.
- Long, V. Aloe Vera in Dermatology—The Plant of Immortality. JAMA Dermatol. 2016, 152, 1364.
- Sumaiyah; Leisyah, B.M. The effect of antioxidant of grapeseed oil as skin anti-aging in nanoemulsion and emulsion preparations. Rasayan J. Chem. 2019, 12, 1185–1194.
- Carneiro, B.M.; Batista, M.N.; Braga, A.C.S.; Nogueira, M.L.; Rahal, P. The green tea molecule EGCG inhibits Zika virus entry. Virology 2016, 496, 215–218.
- Bukhari, S.N.A.; Roswandi, N.L.; Waqas, M.; Habib, H.; Hussain, F.; Khan, S. Hyaluronic acid, a promising Skin rejuvenating biomedicine: A review of Recent updates and preClinical and Clinical investigations on Cosmetic and nutri-cosmetic effects. Int. J. Biol. Macromol. 2018, 120, 1682–1695.
- Toschi, T.G.; Cardenia, V.; Bonaga, G. Coffee silver skin: Characterization, possible uses, and safety aspects. J. Agric. Food Chem. 2014, 62, 10836–10844.
- Razak, D.L.A.; Rashid, N.Y.A.; Jamaluddin, A.; Sharifudin, S.A.; Kahar, A.A.; Long, K. Cosmeceutical potentials and bioactive compounds of rice bran fermented with single and mix culture of Aspergillus oryzae and Rhizopus oryzae. J. Saudi Soc. Agric. Sci. 2017, 16, 127–134.
- Chaudhary, P.; Sharma, A.; Singh, B.; Nagpal, A.K. Bioactivities of phytochemicals present in tomato. J. Food Sci. Technol. 2018, 55, 2833–2849.
- Itrat, M.; Zarnigar. Aloe Vera: A review of its clinical effectiveness. Int. Res. J. Pharm. 2013, 4, 8.
- Khong, N.M.H.; Chan, K.W. Cap 24. Biological activities of Argan oil: Evidence from in vivo studies. In Multiple Biological Activities of Unconventional Seed Oils; Abdalbasit Mariod, A., Ed.; Elsevier: Amsterdam, The Netherlands, 2012.
- Morganti, P.; Morganti, G.; Memic, A.; Coltelli, M.B.; Hong, D.-C. The new renaissance of beauty and wellness through the green economy. Trends Text. Fash. Des. 2021, 4, 000185.
- Wu, D. Recycle Technology for Potato Peel Waste Processing: A Review. Procedia Environ. Sci. 2016, 31, 103–107.
- Radwan, M.M.; Chandra, S.; Gul, S.; Elsohly, M.A. Cannabinoids, phenolics, terpenes and alkaloids of cannabis. Molecules 2021, 26, 2774.
- Kobayashi, J. d-Amino Acids and Lactic Acid Bacteria. Microorganisms 2019, 7, 690.
- Koyande, A.K.; Chen, K.W.; Rambadu, K.; Tao, Y.; Chu, D.T.; Show, P. Micro aNLae: A potential alternative to health supplementation for humans. Food Sci. Hum. Wellness 2019, 8, 16–24.
- Montero-Rivière, N.A. Structure and function of skin. In Toxicology of Skin; Monteiro-Riviere, N.A., Ed.; CRC Press: Boca Raton, FL, USA, 2010; pp. 1–18.
- Robinson, M.; Visscher, M.; Laruffa, A.; Wickett, R. Natural moisturizing factors (NMF) in the stratum corneum (SC) 1. Effects of lipid extraction and soaking. J. Cosmet. Sci. 2010, 61, 13–22.
- Harding, C.R. The stratum corneum: Structure and function in health and disease. Dermatol. Ther. 2004, 17, 6–15.
- Yi, S.; Ding, F.; Gong, L.; Gu, X. Extracellular Matrix Scaffolds for Tissue Engineering and Regenerative Medicine. Curr. Stem Cell Res. Ther. 2017, 12, 233–246.
- Malafaya, P.B.; Silva, G.A.; Reis, R.L. Natural–origin polymers as carriers and scaffolds for biomolecules and cell delivery in tissue engineering applications. Adv. Drug Deliv. Rev. 2007, 59, 207–233.
- Shearan, A.; Garcia, A.J. Nanoscale engineering of extracellular matrix-mimetic bioadhesive surfaces and implants for tissue engineering. Biochim. Biophys. Acta 2011, 1810, 350–360.
- Morganti, P.; Lanzone, A.; Tiberi, L. A new diffusion system through the mucous membranes. J. Appl. Cosmetol. 1998, 16, 45–50.
- Wiechers, J.W. Skin Delivery: What it. In Skin Delivery Systems; Wichers, J.W., Ed.; Allured Publishing Corporation: Carol Stream, IL, USA, 2008; pp. 1–21.
- Kim, B.; Cho, H.-E.; Moon, S.H.; Ahn, H.-J.; Bae, S.; Cho, H.-D.; An, S. Transdermal delivery systems in cosmetics. Biomed. Dermatol. 2020, 4, 1–12.
- Alexander, A.; Dwivedi, S.; Ajazuddin, T.K.; Saraf, S.; Tripathi, D.K. Approaches for break the barriers of drug permeation through transdermal drug Delivery. J. Control. Release 2012, 164, 26–40.
- Liu, C.; Xia, Z.; Czermuszka, J. Design and development of three-dimensional scaffolds for Tissue-engineering. Chem. Eng. Res. Design 2007, 85, 1051–1064.
- Sundararajan, V.; Madihally, S.V.; Marrhew, H.W. Porous Chitosan scaffolds for tissue Engineering. Biomaterials 1999, 29, 1133–1142.
- Geckil, H.; Xu, F.; Zhang, X.; Moon, S.; Demirci, U. Engineering hydrogels as extracellular matrix mimics. Nanomedicine 2010, 5, 469–484.
- Pavlou, P.; Siamidi, A.; Varvaresou, A.; Vlachou, M. Skin Care Formulations and Lipid Carriers as Skin Moisturizing Agents. Cosmetics 2021, 8, 89.
- Euliss, L.E.; DuPont, J.A.; Gratton, S.; DeSimone, J. Imparting size, shape and composition control of materials for Nanomedicine. ChemInform 2007, 38, 1095–1104.
- Motwani, K.G.; Lipworth, B.J. Clinical pharmacokinetics drug administered buccally and sublingually. Clin. Pharm. 1991, 21, 83–94.
- Barang, N.; Sharma, J. Sublingual mucosa as a route for systemic drug delivery. Int. J. Pharm. Pharm. 2011, 3 (Suppl. 2), 18–22.
- Sing, H.N.; Joshi, A.; Toor, A.P.; Verna, G. Drug delivery: Advancements and Challenges. In Nanostructures for Drug Delivery; Andronescu, E., Grumezescu, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 865–886.
- Jung, T.; Kamm, W.; Breitenbach, A.; Kaiserling, E.; Xiao, J.X.; Kissel, T. Biodegradable Nanoparticles for oral delivery of peptides: Is there a role for polymers to affect mucosal uptake? Eur. J. Pharm. Biopharm. 2000, 50, 147–160.
- Madhav, N.V.S.; Shakya, A.K.; Shakya, P.; Singh, K. Orotransmucal drug delivery systems: A review. J. Control. Release 2009, 140, 2–11.
- Lai, S.K.; Wang, Y.-Y.; Hanes, J. Mucus-penetrating nanoparticles for drug and gene delivery to mucosal tissues. Adv. Drug Deliv. Rev. 2009, 61, 158–171.
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751–760.
- Morganti, P.; Morganti, G.; Colao, C. Biofunctional textiles for AGING Skin. Biomedicines 2019, 7, 51.
- Mawazi, S.M.; Ann, J.; Othman, N.; Khan, J.; Alolayan, S.O.; Al Thagfan, S.S.; Kaleemullah, M. A Review of Moisturizers; History, Preparation, Characterization and Applications. Cosmetics 2022, 9, 61.
- Mintel. Beauty & Personal Care, Mintel-Trends 2025. Available online: mintel.com (accessed on 18 March 2021).
- Jarret, A. (Ed.) The Mucous Membranes Vol 6 of Pathophysiology of the Skin; Academic Press: London, UK, 1980.
- Kaaber, S. The permeability and barrier functions of the oral mucosa with respect to water and electrolytes. Studies of the transport of water, sodium and potassium through the human mucosal surface in vivo. Acta Odontol. Scand. Suppl. 1974, 32, 3–47.
- McHugh, D.; Gil, J. Senescence and aging: Causes consequences, and therapeutic avenues. J. Clin. Biol. 2018, 217, 66–77.
- Morganti, P. The photoprotective activity of nutraceuticals. Clin. Dermatol. 2009, 27, 166–174.
- Matejuk, A. Skin Immunity. Arch. Immunol. Ther. Exp. 2018, 66, 45–54.
- Vranesić-Bender, D. The role of nutraceuticals in anti-aging medicine. Acta Clin. Croat. 2010, 49, 537–544.
- Perez-Sanchez, A.; Barrajon-Catalan, E.; Herranz-Lopez, M.; Micol, V. Nutraceuticals for skin care: A comprehensive review of human clinical studies. Nutrients 2018, 10, 403.
- Morganti, P.; Fabrizi, G.; Palombo, P.; Palombo, M.; Guarneri, F.; Cardillo, A.; Morganti, G. New chitin complexes and their anti-aging activity from inside out. J. Nutr. Health Aging 2011, 16, 242–245.
- Abdulaziz, Z.A.; Mohd-Nasir, H.; Ahmad, A.; Setapar, S.H.M.; Lee Peng, W.; Chuo, S.C. Role of nanotechnology for design and development of cosmeceutical application: In make up and skin care. Front. Chem. 2019, 7, 739.
- Salvioni, L.; Morelli, L.; Ochoa, E.; Labra, M.; Fiandra, V.; Palugan, L. The emerging role of nanotechnology in skin care. Adv. Colloid Interface Sci. 2021, 293, 102437.
- Alkilani, A.Z.; McCrudden, M.T.C.; Donnelly, R.F. Transdermal Drug Delivery: Innovative Pharmaceutical Developments Based on Disruption of the Barrier Properties of the Stratum Corneum. Pharmaceutics 2015, 7, 438–470.
- Barnes, T.M.; Mijaljica, D.; Townley, J.P.; Spada, F.; Harrison, I.P. Vehicles for Drug Delivery and Cosmetic Moisturizers: Review and Comparison. Pharmaceutics 2021, 13, 2012.
- EU. Cosmetic Products Regulation 1223/2009 Art 2.1.a; European Union: Brussels, Belgium, 2009.
- Morganti, P. Where cosmeceuticals and nutraceuticals are going. J. Appl. Cosmetol. 2002, 20, 259–274.
- Morganti, P. Reflections on cosmetics, cosmeceuticals and nutraceuticals. Clin. Dermatol. 2008, 26, 318–320.
- Draelos, Z.D. (Ed.) Cosmeceuticals, 3rd ed.; Elsevier: Berlin, Germany, 2014.
- Morganti, P.; Paglialunga, S. EU borderline cosmetic products review of current regulatory status. Clin. Dermatol. 2008, 26, 392–397.
- Ita, K. Transdermal drug delivery: Progress and challenges. J. Drug Deliv. Sci. Technol. 2014, 24, 245–250.





