Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Adriana Laca Perez | -- | 1317 | 2023-05-10 12:25:23 | | | |
| 2 | Dean Liu | Meta information modification | 1317 | 2023-05-11 07:12:55 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Sánchez, M.; Laca, A.; Laca, A.; Díaz, M. Cocoa Bean Shell. Encyclopedia. Available online: https://encyclopedia.pub/entry/44101 (accessed on 08 May 2026).
Sánchez M, Laca A, Laca A, Díaz M. Cocoa Bean Shell. Encyclopedia. Available at: https://encyclopedia.pub/entry/44101. Accessed May 08, 2026.
Sánchez, Marta, Amanda Laca, Adriana Laca, Mario Díaz. "Cocoa Bean Shell" Encyclopedia, https://encyclopedia.pub/entry/44101 (accessed May 08, 2026).
Sánchez, M., Laca, A., Laca, A., & Díaz, M. (2023, May 10). Cocoa Bean Shell. In Encyclopedia. https://encyclopedia.pub/entry/44101
Sánchez, Marta, et al. "Cocoa Bean Shell." Encyclopedia. Web. 10 May, 2023.
Copy Citation
Cocoa bean shell (CBS) is one of the main solid wastes derived from the chocolate industry. This residual biomass could be an interesting source of nutrients and bioactive compounds due to its high content in dietary fibres, polyphenols and methylxanthines.
cocoa bean shell
cocoa by-products
bioactive compounds
1. Chemical and Nutritional Composition of CBS
CBS is the fibrous external tegument and constitutes about 10–20% of the total cocoa bean weight [1][2][3]. CBS is recovered during the cocoa bean roasting process at the chocolate factory, i.e., for every kg of chocolate produced, almost 100 g of CBS is obtained [4][5][6]. It is estimated that each year, between 700 and 900 thousand tons of CBS are produced worldwide, approximately 300 of which are generated in Europe. The accumulation of this by-product represents an important disposal problem in terms of economic and environmental issues, which is further accentuated by new legal regulations [6].
Although CBS is generally considered and treated as a residue, it has been reported that its nutritional composition does not differ too much from that of cocoa beans. CBS nutritional composition has been deeply investigated in different research works with the aim of extracting carbohydrates, dietary fibres, phenolic compounds (mainly epicatechin and catechin) and methylxanthines, such as theobromine. Table 1 summarizes the composition of CBS according to literature data, which varies depending on the origin and the processing of cocoa fruit.
Table 1. Chemical and nutritional composition of CBS, all data, except moisture, expressed on a dry weight basis.
| Content | References | |
|---|---|---|
| Moisture | 4–13.1 g/100 g | [5][7][8][9] |
| Ash | 6.0–9.1 g/100 g | [7][8][10] |
| Carbohydrates | 13.2–70.3 g/100 g | [5][9][11][12][13] |
| Proteins | 18.2–27.4 g/100 g | [7][9][13][14][15] |
| Lipids | 2.3–6.5 g/100 g | [5][7][16][17] |
| Dietary fibres | 13.8–65.6 g/100 g | [5][7][9] |
| Total phenolic content | 22–100 mg GAE/g | [8][10][18][19][20] |
| Total flavonoid content | 7.5–21.8 mg RU/g1.6–43.9 mg CE/g | [19][20][21][22][23] |
| Total tannin content | 2.3–25.3 mg CE/g | [19][20] |
| Flavanols | ||
| Catechin | 0.8–5.7 mg/g | [5][24][25][26] |
| Epicatechin | 0.6–30 mg/g | [5][24][25][26][27] |
| Procyanidin B1 | 0.5–0.8 mg/g | [28] |
| Procyanidin B2 | 0.2–1.4 mg/g | [28][29] |
| Methylxanthines | ||
| Theobromine | 0.6–13.5 mg/g | [5][10][29][30] |
| Caffeine | 0.1–1.1 mg/g | [10][31] |
| Theophylline | 0.1–0.3 mg/g | [31] |
GAE: gallic acid equivalent; RU: rutin equivalent; CE: catechin equivalent.
It has been reported that the moisture content of CBS, which is determined by the roasting process, normally ranges from 4 to 8 g/100 g, values considered acceptable for CBS storage. However, in some cases, moisture levels can reach up to 13 g/100 g [9]. In this sense, CBS has been described as a considerably hygroscopic material; therefore, storage at moisture levels over 15% could lead to mould growth [7]. According to the literature, the ash content is in the range of 6 to 9 g/100 g of CBS, which is a parameter also influenced by the roasting process and is relatively similar among varieties. CBS contains a higher ash content than other fruit residues, such as apple pomace (0.5 g/100 g) and orange peel (2.6 g/100 g) [32][33]. Osundahunsi et al., 2007 [34] reported that sodium and potassium are the main components of CBS ash.
The fat content is quite low in CBS compared to cocoa beans, which accounts for almost 50% (dry weight). It has been reported that the content of lipids in CBS can be reduced by 40% after the roasting process [9]. The chemical and physical characteristics of CBS lipids are similar to cocoa butter fat, except for the acidity levels, which are higher in CBS (9% oleic acid) than in cocoa butter (1.7% oleic acid) due to the hydrolysis of triacylglycerols [29]. The saponifiable fraction of CBS is mainly formed by oleic, capric, palmitic and stearic fatty acids, whereas the unsaponifiable fraction is composed of phytosterols, specifically stigmasterol [35]. From a health perspective, it has been demonstrated that the consumption of cocoa increases high-density cholesterol and reduces low-density cholesterol because of its fatty acid content [36].
The carbohydrate content in CBS varies between 13.2 and 70.3 g/100 g (Table 1), mainly constituting glucose (17%), galactose (3%), mannose (3%), xylose (1.2%) and arabinose (1.7%) [1][10]. Carbohydrates are one of the most important macronutrients in cocoa beans since these compounds are responsible for the flavour during the fermentation and roasting processes. Additionally, it has been demonstrated that roasted CBS contains more carbohydrates than unroasted CBS, which is associated with the transfer of sugars toward the outer shell during roasting [9]. Furthermore, it has been reported that some CBS carbohydrates, such as feruloylated oligosaccharides, could improve the intestinal microbiota due to their prebiotic characteristics [33]. With respect to the protein content, during fermentation, an increase of up to 30% can be achieved in CBS. However, roasting processes result in a reduction in the protein content of CBS. According to the literature, proteins constitute between 18.2 and 27.4% of CBS. Furthermore, it is known that just approximately 1% of the total protein in CBS remains in a free condition, while 90% is firmly bound to oxidized polyphenols found in the shell [29][37].
CBS has been reported to be an important source of dietary fibre (13.8–65.6 g/100 g) with a small fat content (2–7 g/100 g). The amount of dietary fibre in CBS also depends on the roasting process. Certainly, it has been proved that, in roasted cocoa shells, Maillard reaction compounds and tannin or protein complexes are responsible for an increment in fibre content [38]. Insoluble dietary fibre accounts for 80% of the total dietary fibre of CBS extracts and almost half of the total dry weight. According to the literature, the main components of this CBS insoluble fibre are uronic acid and glucose, followed by galactose, xylose, mannose and arabinose to a lesser extent. This composition indicates that the cell wall mainly consists of cellulose with lower amounts of hemicellulose and pectin [39]. It has been reported that cocoa fibre has physicochemical and antioxidant properties, which make it an interesting alternative for the preparation of low-calorie and high-fibre cocoa products [38].
Along with dietary fibre, CBS polyphenols are the most studied and interesting compounds since they are responsible for biofunctional properties attributed to this by-product, such as its antidiabetic, anticarcinogenic and anti-inflammatory effects [40]. Flavonoids, one of the most remarkable groups of phenolic compounds, have been widely investigated due to their extensive bioactive properties, including cardio-protective, anti-oxidation and anti-inflammatory activities [41]. The reviewed literature describes that the total phenolic content (TPC), total flavonoid content (TFC) and total tannin content (TTC) in CBS range between 22 and 100 mg GAE/g CBS, 1.6 and 44 mg CE/g CBS and 2.3 and 25.3 CE/g CBS, respectively. The number of phenolic compounds in CBS extracts varies depending on several factors, such as origin, variety, extraction procedure (technologies, conditions, solvents…) and the presence of other bioactive compounds [20][42][43]. Flavanols have been identified as the major class of flavonoids in CBS, mainly including catechin (0.8–5.7 mg/g CBS), epicatechin (0.6–30 mg/g CBS) and procyanidin B2 (0.2–1.4 mg/g CBS). These polyphenols are not essential for short-term well-being, but it has been suggested that a long-term intake of them could contribute to health benefits, such as those related to cognitive function [44]. Other flavonoids also detected in CBS include kaempferol, quercetin and anthocyanins [6][20][40].
Theobromine and caffeine are the main methylxanthines, also known as alkaloids, found in CBS in quantities that fluctuate from 0.6 to 2.7 mg/g of CBS and 0.1 to 0.1–1.1 mg/g of CBS, respectively. Both alkaloids are related to physiological effects on the cardiovascular, gastrointestinal, respiratory, renal and central nervous systems in addition to their anticarcinogen and diuretic properties [35][43]. However, it is well known that excessive consumption of methylxanthines, particularly caffeine, is related to tachycardia, kidney dysfunction and other disorders. In addition to theobromine and caffeine, theophylline is another methylxanthine detected in CBS extracts, but at lower concentrations (0.1–0.3 mg/g CBS) or even at trace levels [31][42]. Methylxanthines, along with polyphenols, contribute to the characteristic bitter taste of cocoa and its derivatives, such as chocolate [45].
CBS can also contain some toxic compounds and/or antinutrients, such as biogenic amines, trypsin inhibitors and phytic acid. However, they are at concentrations so low (for example, the amount of phytic acid is below 0.6 g/100 g CBS) that this does not affect the use of CBS as a potential source of nutrition [33][46].
2. CBS Valorization
Recently, CBS valorization has been investigated to find novel applications for this by-product, including its use in food formulation, the obtention of biofuels, the extraction of bioactive compounds and employment as an adsorbent. Figure 2 shows an overview of different potential applications of CBS.
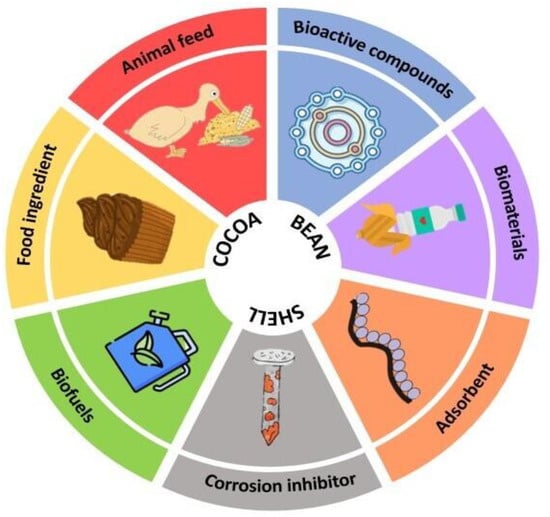
Figure 2. Overview of CBS alternatives of valorization.
References
- Sánchez, M.; Laca, A.; Laca, A.; Díaz, M. Cocoa Bean Shell as Promising Feedstock for the Production of Poly(3-Hydroxybutyrate) (PHB). Appl. Sci. 2023, 13, 975.
- Hashimoto, J.C.; Lima, J.C.; Celeghini, R.M.S.; Nogueira, A.B.; Efraim, P.; Poppi, R.J.; Pallone, J.A.L. Quality Control of Commercial Cocoa Beans (Theobroma cacao L.) by Near-Infrared Spectroscopy. Food Anal. Methods 2018, 11, 1510–1517.
- Ho, V.T.T.; Zhao, J.; Fleet, G. Yeasts Are Essential for Cocoa Bean Fermentation. Int. J. Food Microbiol. 2014, 174, 72–87.
- Afrane, G.; Ntiamoah, A. Use of Pesticides in the Cocoa Industry and Their Impact on the Environment and the Food Chain. In Pesticides in the Modern World-Risks and Benefit; InTechOpen: London, UK, 2011.
- Botella-Martínez, C.; Lucas-Gonzalez, R.; Ballester-Costa, C.; Pérez-álvarez, J.Á.; Fernández-López, J.; Delgado-Ospina, J.; Chaves-López, C.; Viuda-Martos, M. Ghanaian Cocoa (Theobroma cacao L.) Bean Shells Coproducts: Effect of Particle Size on Chemical Composition, Bioactive Compound Content and Antioxidant Activity. Agronomy 2021, 11, 401.
- Cantele, C.; Rojo-Poveda, O.; Bertolino, M.; Ghirardello, D.; Cardenia, V.; Barbosa-Pereira, L.; Zeppa, G. In Vitro Bioaccessibility and Functional Properties of Phenolic Compounds from Enriched Beverages Based on Cocoa Bean Shell. Foods 2020, 9, 715.
- Rojo-Poveda, O.; Barbosa-Pereira, L.; Mateus-Reguengo, L.; Bertolino, M.; Stévigny, C.; Zeppa, G. Effects of Particle Size and Extraction Methods on Cocoa Bean Shell Functional Beverage. Nutrients 2019, 11, 867.
- Mellinas, A.C.; Jiménez, A.; Garrigós, M.C. Optimization of Microwave-Assisted Extraction of Cocoa Bean Shell Waste and Evaluation of Its Antioxidant, Physicochemical and Functional Properties. LWT 2020, 127, 109361.
- Agus, B.A.P.; Mohamad, N.N.; Hussain, N. Composition of Unfermented, Unroasted, Roasted Cocoa Beans and Cocoa Shells from Peninsular Malaysia. J. Food Meas. Charact. 2018, 12, 2581–2589.
- Grillo, G.; Boffa, L.; Binello, A.; Mantegna, S.; Cravotto, G.; Chemat, F.; Dizhbite, T.; Lauberte, L.; Telysheva, G. Cocoa Bean Shell Waste Valorisation; Extraction from Lab to Pilot-Scale Cavitational Reactors. Food Res. Int. 2019, 115, 200–208.
- Adamafio, N.A. Theobromine Toxicity and Remediation of Cocoa Byproducts: An Overview. J. Biol. Sci. 2013, 7, 570–576.
- Martínez, R.; Torres, P.; Meneses, M.A.; Figueroa, J.G.; Pérez-Álvarez, J.A.; Viuda-Martos, M. Chemical, Technological and in Vitro Antioxidant Properties of Cocoa (Theobroma cacao L.) Co-Products. Food Res. Int. 2012, 49, 39–45.
- Vojvodić, A.; Komes, D.; Vovk, I.; Belščak-Cvitanović, A.; Bušić, A. Compositional Evaluation of Selected Agro-Industrial Wastes as Valuable Sources for the Recovery of Complex Carbohydrates. Food Res. Int. 2016, 89, 565–573.
- Mancini, G.; Papirio, S.; Lens, P.N.L.; Esposito, G. Anaerobic Digestion of Lignocellulosic Materials Using Ethanol-Organosolv Pretreatment. Environ. Eng. Sci. 2018, 35, 953–960.
- Fakhlaei, R.; Rozzamri, A.; Hussain, N. Composition, Color and Antioxidant Properties of Cocoa Shell at Different Roasting Temperatures. Food Res. Int. 2020, 4, 585–593.
- Belščak-Cvitanović, A.; Vojvodić, A.; Bušić, A.; Keppler, J.; Steffen-Heins, A.; Komes, D. Encapsulation Templated Approach to Valorization of Cocoa Husk, Poppy and Hemp Macrostructural and Bioactive Constituents. Ind. Crops Prod. 2018, 112, 402–411.
- Sandoval, A.J.; Barreiro, J.A.; De Sousa, A.; Valera, D.; López, J.V.; Müller, A.J. Composition and Thermogravimetric Characterization of Components Ov Venezuelan Fermented and Dry Trinitario Cocoa Beans (Theobroma cacao L.): Whole Beans, Peeled Beans and Shells. Rev. Técnica La Fac. Ing. 2019, 42, 38–47.
- Becerra, L.D.; Quintanilla-Carvajal, M.X.; Escobar, S.; Ruiz, R.Y. Correlation between Color Parameters and Bioactive Compound Content during Cocoa Seed Transformation under Controlled Process Conditions. Food Biosci. 2023, 53, 102526.
- Nsor-Atindana, J.; Zhong, F.; Mothibe, K.J.; Bangoura, M.L.; Lagnika, C. Quantification of Total Polyphenolic Content and Antimicrobial Activity of Cocoa (Thebroma cacao L.) Bean Shells. Pakistan J. Nutr. 2012, 11, 672–677.
- Barbosa-Pereira, L.; Guglielmetti, A.; Zeppa, G. Pulsed Electric Field Assisted Extraction of Bioactive Compounds from Cocoa Bean Shell and Coffee Silverskin. Food Bioprocess Technol. 2018, 11, 818–835.
- Švarc-Gajić, J.; Brezo-Borjan, T.; Dzedik, V.; Rodrigues, F.; Morais, S.; Delerue-Matos, C. ESG Approach in the Valorization of Cocoa (Theobroma cacao) by-Products by Subcritical Water: Application in the Cosmetic Industry. Sustain. Chem. Pharm. 2023, 31, 100908.
- Jokić, S.; Nastić, N.; Vidović, S.; Flanjak, I.; Aladić, K.; Vladić, J. An Approach to Value Cocoa Bean By-Product Based on Subcritical Water Extraction and Spray Drying Using Different Carriers. Sustainability 2020, 12, 2174.
- Yusof, A.H.M.; Abd Gani, S.S.; Zaidan, U.H.; Halmi, M.I.E.; Zainudin, B.H. Optimization of an Ultrasound-Assisted Extraction Condition for Flavonoid Compounds from Cocoa Shells (Theobroma cacao) Using Response Surface Methodology. Molecules 2019, 24, 711.
- Aranaz, P.; Romo-Hualde, A.; Navarro-Herrera, D.; Zabala, M.; López-Yoldi, M.; González-Ferrero, C.; Gil, A.G.; Martínez, J.A.; Vizmanos, J.L.; Milagro, F.I.; et al. Low Doses of Cocoa Extract Supplementation Ameliorate Diet-Induced Obesity and Insulin Resistance in Rats. Food Funct. 2019, 10, 4811–4822.
- Lemarcq, V.; Monterde, V.; Tuenter, E.; Van de Walle, D.; Pieters, L.; Sioriki, E.; Dewettinck, K. Flavor Diversification of Dark Chocolate Produced through Microwave Roasting of Cocoa Beans. LWT 2022, 159, 113198.
- Valadez-Carmona, L.; Ortiz-Moreno, A.; Ceballos-Reyes, G.; Mendiola, J.A.; Ibáñez, E. Valorization of Cacao Pod Husk through Supercritical Fluid Extraction of Phenolic Compounds. J. Supercrit. Fluids 2018, 131, 99–105.
- Caprioli, G.; Fiorini, D.; Maggi, F.; Nicoletti, M.; Ricciutelli, M.; Toniolo, C.; Prosper, B.; Vittori, S.; Sagratini, G. Nutritional Composition, Bioactive Compounds and Volatile Profile of Cocoa Beans from Different Regions of Cameroon. Int. J. Food Sci. Nutr. 2016, 67, 422–430.
- Papillo, V.A.; Locatelli, M.; Travaglia, F.; Bordiga, M.; Garino, C.; Coïsson, J.D.; Arlorio, M. Cocoa Hulls Polyphenols Stabilized by Microencapsulation as Functional Ingredient for Bakery Applications. Food Res. Int. 2019, 115, 511–518.
- Okiyama, D.C.G.; Soares, I.D.; Cuevas, M.S.; Crevelin, E.J.; Moraes, L.A.B.; Melo, M.P.; Oliveira, A.L.; Rodrigues, C.E.C. Pressurized Liquid Extraction of Flavanols and Alkaloids from Cocoa Bean Shell Using Ethanol as Solvent. Food Res. Int. 2018, 114, 20–29.
- Pavlović, N.; Jokić, S.; Jakovljević, M.; Blažić, M.; Molnar, M. Green Extraction Methods for Active Compounds from Food Waste—Cocoa Bean Shell. Foods 2020, 9, 140.
- Bartella, L.; Di Donna, L.; Napoli, A.; Siciliano, C.; Sindona, G.; Mazzotti, F. A Rapid Method for the Assay of Methylxanthines Alkaloids: Theobromine, Theophylline and Caffeine, in Cocoa Products and Drugs by Paper Spray Tandem Mass Spectrometry. Food Chem. 2019, 278, 261–266.
- Boluda-Aguilar, M.; García-Vidal, L.; del Pilar González-Castañeda, F.; López-Gómez, A. Mandarin Peel Wastes Pretreatment with Steam Explosion for Bioethanol Production. Bioresour. Technol. 2010, 101, 3506–3513.
- Younes, A.; Li, M.; Karboune, S. Cocoa Bean Shells: A Review into the Chemical Profile, the Bioactivity and the Biotransformation to Enhance Their Potential Applications in Foods. Crit. Rev. Food Sci. Nutr. 2022, 2, 1–25.
- Osundahunsi, O.F.; Bolade, M.K.; Akinbinu, A.A. Effect of Cocoa Shell Ash as an Alkalizing Agent on Cocoa Products. J. Appl. Sci. 2007, 7, 1674–1678.
- Rojo-poveda, O.; Barbosa-pereira, L.; Zeppa, G.; St, C. Cocoa Bean Shell—A By-Product with Nutritional Properties and Biofunctional Potential. Nutrients 2020, 12, 1123.
- González-Alejo, F.A.; Barajas-Fernández, J.; Olán-Acosta, M.D.L.Á.; Lagunes-Gálvez, L.M.; García-Alamilla, P. Supercritical Fluid Extraction of Fat and Caffeine with Theobromine Retention in the Cocoa Shell. Processes 2019, 7, 385.
- Bentil, J.A.; Dzogbefia, V.P.; Alemawor, F. Enhancement of the Nutritive Value of Cocoa (Theobroma cacao) Bean Shells for Use as Feed for Animals through a Two-Stage Solid State Fermentation with Pleurotus Ostreatus and Aspergillus Niger. Int. J. Appl. Miicrobiology Biotechnol. Res. 2015, 3, 20–30.
- Redgwell, R.; Trovato, V.; Merinat, S.; Curti, D.; Hediger, S.; Manez, A. Dietary Fibre in Cocoa Shell: Characterisation of Component Polysaccharides. Food Chem. 2003, 81, 103–112.
- Balentić, J.P.; Ačkar, Đ.; Jokić, S.; Jozinović, A.; Babić, J.; Miličević, B.; Šubarić, D.; Pavlović, N. Cocoa Shell: A By-Product with Great Potential for Wide Application. Molecules 2018, 23, 1404.
- Soares, T.F.; Oliveira, M.B.P.P. Cocoa By-Products: Characterization of Bioactive Compounds and Beneficial Health Effects. Molecules 2022, 27, 1625.
- Zhu, J.; Chen, C.; Zhang, B.; Huang, Q. The Inhibitory Effects of Flavonoids on α-Amylase and α-Glucosidase. Crit. Rev. Food Sci. Nutr. 2020, 60, 695–708.
- Jokić, S.; Gagić, T.; Knez, E.; Ubarić, D.; Kerget, M. Separation of Active Compounds from Food By-Product (Cocoa Shell) Using Subcritical Water Extraction. Molecules 2018, 23, 1408.
- Hernández-Hernández, C.; Morales-Sillero, A.; Fernández-Bolaños, J.; Bermúdez-Oria, A.; Morales, A.A.; Rodríguez-Gutiérrez, G. Cocoa Bean Husk: Industrial Source of Antioxidant Phenolic Extract. J. Sci. Food Agric. 2019, 99, 325–333.
- Aprotosoaie, A.C.; Luca, S.V.; Miron, A. Flavor Chemistry of Cocoa and Cocoa Products-An Overview. Compr. Rev. Food Sci. Food Saf. 2016, 15, 73–91.
- Neshatdoust, S.; Saunders, C.; Castle, S.M.; Vauzour, D.; Williams, C.; Butler, L.; Lovegrove, J.A.; Spencer, J.P.E. High-Flavonoid Intake Induces Cognitive Improvements Linked to Changes in Serum Brain-Derived Neurotrophic Factor: Two Randomised, Controlled Trials. Nutr. Health Aging 2016, 4, 81–93.
- Pavlović, N.; Miškulin, M.; Aladić, K.; Jokić, S. Cocoa Bean Shell—A Promising By-Product Rich in Bioactive Compounds. Food Health Dis. 2019, 8, 116–122.
More
Information
Subjects:
Green & Sustainable Science & Technology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.6K
Revisions:
2 times
(View History)
Update Date:
11 May 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





