Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Shashi Kant Bhatia | -- | 4229 | 2023-04-26 05:10:41 | | | |
| 2 | Rita Xu | -3 word(s) | 4226 | 2023-04-26 05:23:52 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Ahuja, V.; Bhatt, A.K.; Ravindran, B.; Yang, Y.; Bhatia, S.K. Syngas Fermentation to Bio-Alcohols. Encyclopedia. Available online: https://encyclopedia.pub/entry/43492 (accessed on 24 May 2026).
Ahuja V, Bhatt AK, Ravindran B, Yang Y, Bhatia SK. Syngas Fermentation to Bio-Alcohols. Encyclopedia. Available at: https://encyclopedia.pub/entry/43492. Accessed May 24, 2026.
Ahuja, Vishal, Arvind Kumar Bhatt, Balasubramani Ravindran, Yung-Hun Yang, Shashi Kant Bhatia. "Syngas Fermentation to Bio-Alcohols" Encyclopedia, https://encyclopedia.pub/entry/43492 (accessed May 24, 2026).
Ahuja, V., Bhatt, A.K., Ravindran, B., Yang, Y., & Bhatia, S.K. (2023, April 26). Syngas Fermentation to Bio-Alcohols. In Encyclopedia. https://encyclopedia.pub/entry/43492
Ahuja, Vishal, et al. "Syngas Fermentation to Bio-Alcohols." Encyclopedia. Web. 26 April, 2023.
Copy Citation
Biomass gasification produces syngas, mainly comprised of CO and H2 along with H2S, CO2, N2, and tar compounds. Inorganic carbon present in syngas as CO and CO2 can be utilized for the production of several value-added chemicals including ethanol, higher alcohols, fuels, and hydrogen using microbial sysytem.
biomass gasification
syngas utilization
microbial fermentation
1. Introduction
Increased energy requirements and fuel consumption have raised environmental pollution and lowered air quality. During the inter-season period, burning agricultural residues (left out after harvesting) is a common practice that not only releases pollutant gases but also aerosols [1]. It has been found that biomass burning contributed around 10–70% of PM 2.5, and concerning health, PM 2.5 and PM 10 are among the most lethal pollutants [2]. Biomass burning contributes significantly, but it is not the main source of air pollution. Transportation, industries’ operations including construction, power plants, and indoor emissions are also among the top air polluters [3][4]. Industrial operations alone contribute 23% SO2, 15% CO, and 14% PM 2.5. On the other hand, transportation contributed 53% NOx, 18% CO, 17% PM 2.5, and 13% PM 10 while stubble burning shared only 14% CO and 12% PM 2.5 [2].
Reduced soil fertility and disturbed nutrient cycle are other major ill effects of air pollution [5][6][7]. Stubble burning raises the temperature of the soil and eliminates beneficial microorganisms. Reduction in microbial diversity affects the nutrient cycle [8][9] and the availability of nutrients for plants which ultimately deteriorates agricultural productivity. Besides agricultural productivity, air pollution also leads to injury to leaves and grains, affects metabolism and enzyme activity, and promotes discoloration, chlorosis, and necrosis [2]. Air pollution exposure leads to skin and eye irritation, respiratory distress and neurological and cardiovascular disorders. Chronic exposure can lead to permanent health ailments like respiratory disorders, Chronic Obstructive Pulmonary Disease, and even cancer [2][10].
Air pollution not only leads to biodiversity loss and global temperature rise but also to the loss of GDP. An increase in global temperature by 3.2 °C can consume 18% of the worldwide GDP [11]. To control the financial loss and deterioration of the environment, the Paris agreement targets climate change and aims at limiting the global temperature rise by controlling GHG emissions well, and a standard temperature rise has been set to keep it within 2 °C (1.5 °C preferred) [12]. The Intergovernmental Panel on Climate Change suggested a preferred global warming level of 1.5 °C which can be accomplished only by lowering the global CO emission by at least 45% by 2030 and subsequently to net-zero emission by 2050. Transportation and industrial sectors have been identified as prime GHG emitters comprising 16% and 27% of the total emissions [11].
The severity of air pollution incidents has fueled the need for carbon-negative fuels, industrial processes, and the capture of already present CO/CO2, which would aid in global decarbonisation. A transition toward renewable fuel/energy sources and utilization of renewable biomass from agricultural and carbon-rich waste as feed to carbon-negative fuels and other commodity chemicals also accelerate decarbonisation [13]. The carbon capture and storage (CCS) techniques are focused on the specific capturing of CO2 from environmental air and industrial gaseous discharge using physical, chemical, or biological approaches and storing it till further use. The storage time relies on the method used for storage. The major hindrances to the selection of methods among various approaches employed for CCS are energy-intensiveness and associated cost [14].
Nowadays, another approach is getting much more attention than the conventional carbon capture approach: syngas production and fermentation. Syngas/synthesis gas is a mixture of H2/CO produced from the gasification of conventional fuels and hydrocarbons, and its reforming in the presence of oxygen, air, steam, or mixtures that react with the carbon source at elevated temperatures [15]. In this process, renewable biomass/residues from agricultural fields are getting special consideration for syngas production. Basically, syngas is an intermediate product as well as raw material for a variety of commodity chemicals and small organic compounds like organic acids, alcohols, hydrogen, jet fuels, alkenes, etc., via catalytic hydrogenation at higher temperatures and pressures [11][15] or biocatalysts [16] like Clostridium kluyveri [17], Clostridium autoethanogenum [18], and Clostridium carboxidivorans [19]. Most chemical catalysis referred to the use of metal-based catalysts like Ni [20], Fe-Ca oxides [21], Co, and Mo [22], but the conversion operates at a much higher temperature which makes the process costly and energy intensive. In comparison, microbial fermentation can work at mild conditions and efficiently transform it into commercial products like organic acid by C. carboxidivorans P7 [19], Acetobacterium, and Desulfovibrio dominated mixed culture [23], butanol by C. carboxidivorans [24], and ethanol by Clostridium ljungdahlii [25][26].
Microorganisms have shown potential for the utilization of CO as a carbon source and/or electron donor followed by their utilization via various metabolic pathways. For example, anaerobes follow the ‘Wood-Ljungdahl pathway’ [27] and sequester it into acetyl-CoA while, under an aerobic environment, carboxydotrophs used molecular oxygen for its oxidation to CO2 followed by sequestration via pentose phosphate [28]. There is another group, chemolithoautotrophs, which oxidized CO by carbon monoxide dehydrogenase to CO2 [29]. For all these pathways, acetyl-CoA is the junction point and its further utilisation depends upon the growth conditions prevailing. The pathways determining the end products are under the control of gene expression hence genetic modification might contribute to improve the fermentation rate and product formation.
Syngas fermentation represents an opportunity to transform waste gases into commercially viable products rather than leaving them to deteriorate the environment. The available literature also stated that inorganic as well as organic carbon can be converted into commodity products [30][31]. According to the Scopus database, a total of 439 articles (353 research articles and 86 review articles) have been published since 2014 on syngas production and its valorization using chemical and fermentation methods. The United State alone has published 98 articles, followed by China (86), Germany (56), South Korea (46), Spain (37), and the Netherlands (34) (Figure 1 Visualization network of countries involved in this area of research). A recently published article by Maki-Arvela discusses the use of various catalysts for syngas conversion into aviation fuel [32]. In another article, Liu et al. discussed different catalytic routes for ethanol production from syngas [33]. Very few articles have focused on the production of energy-related products from syngas via microbial fermentation. There is a review article focused on a broad aspect of biochemicals and biofuel production from syngas fermentation [30]. The recent trends in syngas fermentation for the production of ethanol and other energy-related products are summarized. Various parameters that affect the syngas fermentation process are discussed and techno-economic aspects are also evaluated for industrial as well as social acceptance of the process. The integration of processes with the currently available industrial process is emphasized to minimize pollution and make processes greener and more cost-effective (Figure 2).
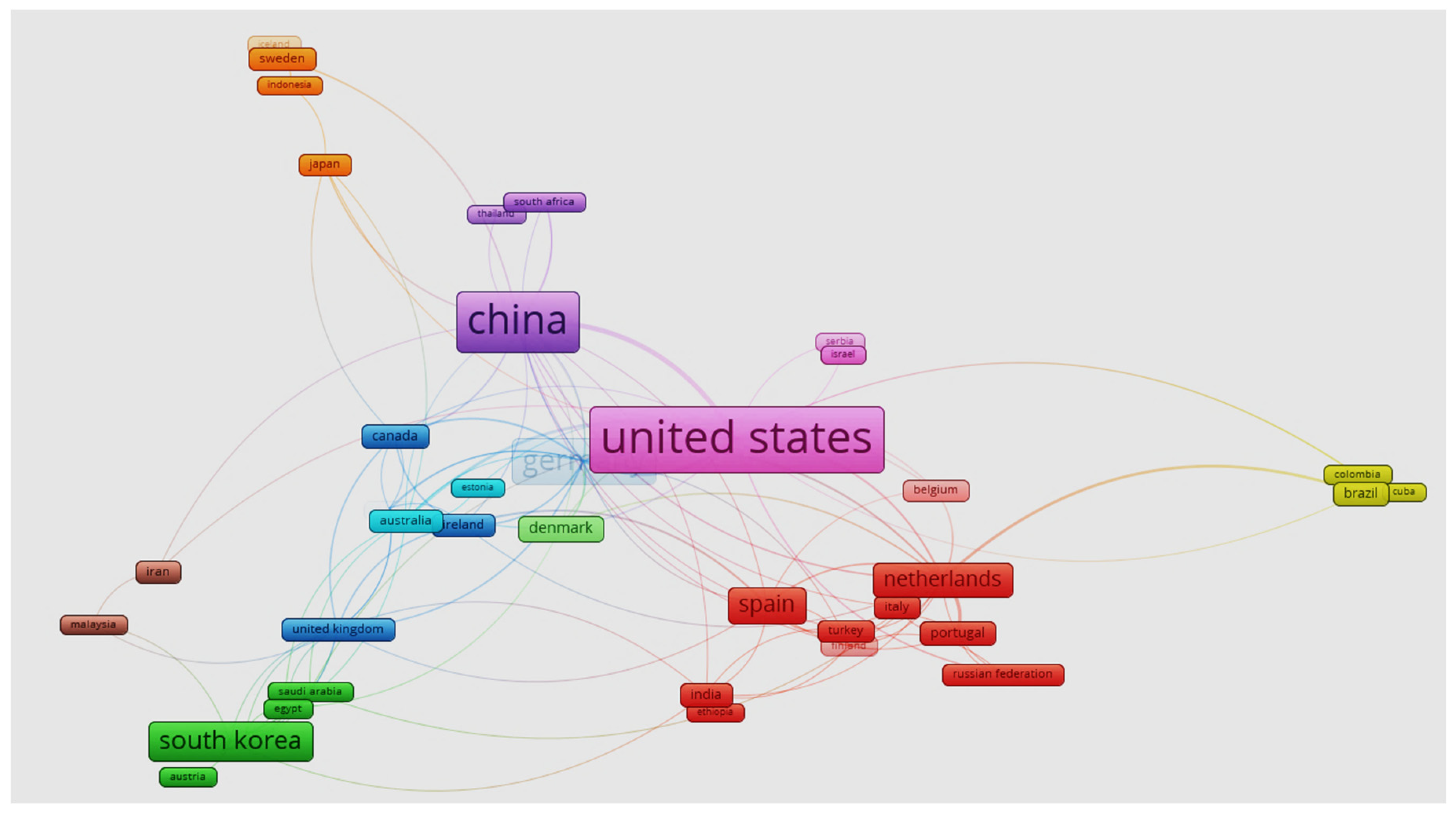
Figure 1. Co-occurrence mapping based on the publication number (minimum number of co-occurrences was fixed at 5). Map was generated using Vosviewer.
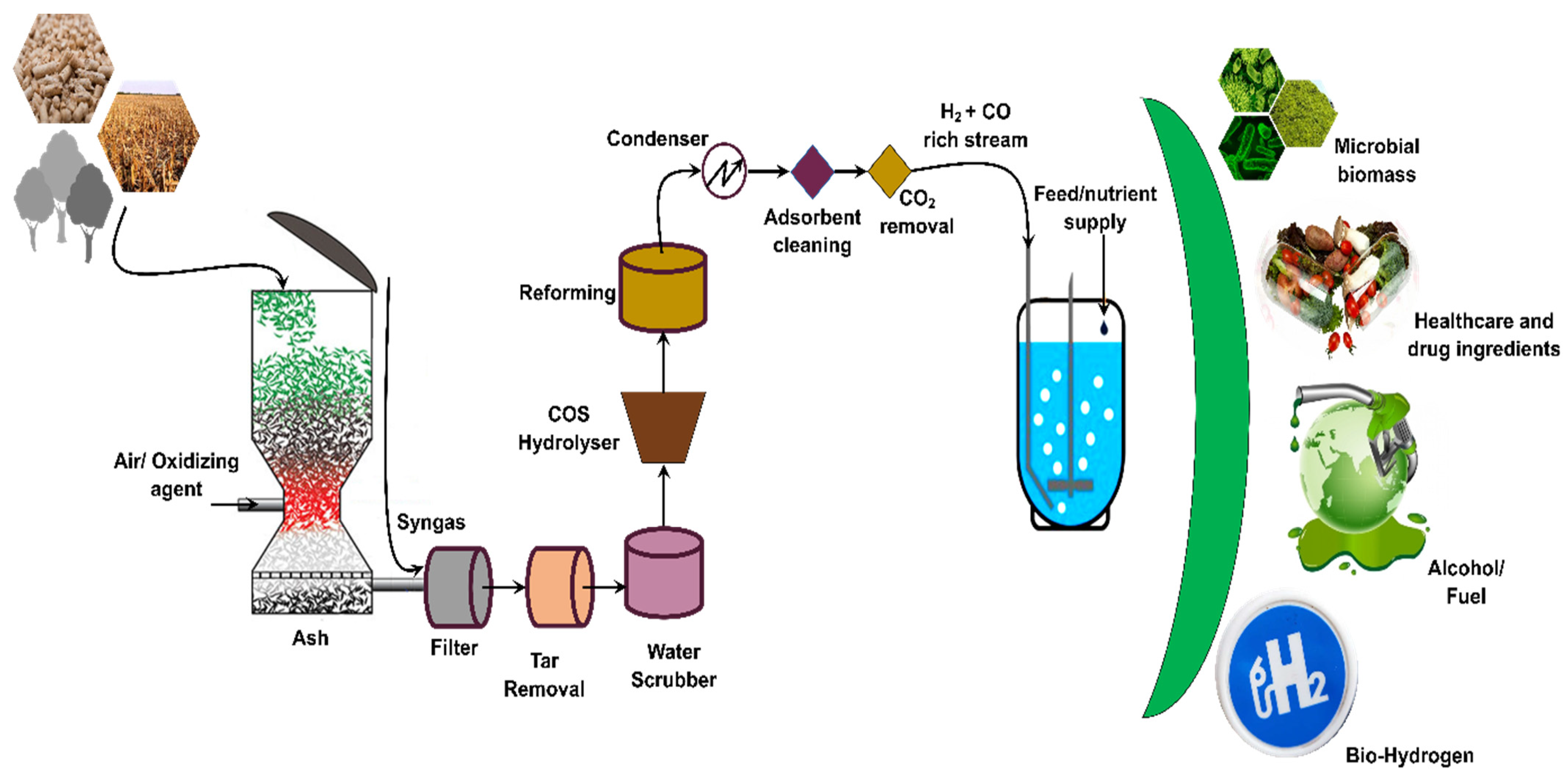
Figure 2. Process diagram showing syngas production technologies, pretreatment, and fermentation into various products.
2. Technologies for Syngas Production Pretreatment and Transformation
Syngas is mainly comprised of CO and H2 as major components along with CO2 and H2O. It is a common product of biomass/fuel gasification with/without a catalyst. Usually, the process begins with a solid feedstock like coal or biological biomass including agricultural or microbial sources but may also include heavy oil. Before processing/gasification, biomass is dried and pulverized (if needed) for better results followed by gasification at a high temperature (800–1000 °C) and pressure of 1–20 bar [34]. The gasification process can be expressed in a reaction as follows:
Biomass + O2 → CO + H2 + CO2 + H2O + CH4
At the beginning of the gasification process, the solid carbon present in biomass/feed is partially oxidized in the presence of air/O2/steam/or their mixture. The product is mostly represented by methane with a minor fraction of hydrocarbons like ethane and ethylene. The composition of gas may vary with the type of gasifiers, feedstock, and operational parameters, and product gas also carries some hydrocarbons and tar compounds like benzene, naphthalenes, and toluene. Besides ashes and char are also among the major by-products [34].
Xie and colleagues integrated catalytic pyrolysis and gasification for syngas production. Different types of nickel-based catalysts were evaluated along with pyrolysis temperature and gasification temperature. A temperature of 750 °C during pyrolysis and gasification was found good for higher syngas production with high-quality char. At 750 °C, char produced syngas with steam. A maximum syngas yield of 3.29 N m3/kg feed was attained at 850 °C, but the catalyst got deactivated. At a higher temperature range, ‘Ni’ formed Mg0.4Ni0.6O, and the size of Ni0 crystallite size increased [20].
Nourbakhsh and colleagues conducted Gibbs free energy minimization-based thermodynamic analysis for syngas utilization followed by experimental validation with ultra-rich methane/oxygen mixtures in an inert porous reactor via thermal-assisted partial oxidation. For the analysis, the equivalence ratio and the thermal load was considered the major factor. From the experimentation, the establishment of adiabatic conditions and heat loss abatement enhanced the syngas yield, and maximum syngas production (69.5%) was achieved at an equivalence ratio of 2.5 with 8 kW energy investment [35]. Chen et al. used CO2 as feed for direct conversion to syngas via the electrocatalytic process. From the process and scaleup point of view, the requirement of large current density, high cell voltage efficiency, and the design of efficient catalysts are critical points. Oxide-derived Cu nanowires with CuO heterostructures offered 90% CO2 reduction with H2/CO ratios of 1, 2, and 3 along with 50% energy efficiency [36]. Hu et al. developed Fe-Ca oxides and employed them as oxygen carriers during syngas production to improve the hydrogen content via chemical looping gasification of rice straw as feed. The assessment was focused on the Fe/Ca ratio, reaction temperature, and oxygen carrier recyclability. Different combinations of Fe/Ca form calcium ferrites (Ca2Fe2O5 and CaFe2O4) in which Fe and Ca were uniformly distributed. In comparison to control, hydrogen production was greatly enhanced due to steam chemical looping gasification as during looping steam re-oxidizes iron. The maximum yield of H2 (23.07 mmol/g feed) was achieved with a Fe/Ca ratio of 1:1 which forms Ca2Fe2O5 (oxygen carrier) at 800 °C. Rice husk is rich in silica which got accumulated on the catalyst during the process and destroys the Fe-Ca native structure after three cycles [21].
Gur and Canbaz conducted underground coal gasification (UCG) lab-scale experiment trials in the presence of hydrogen. The model was evaluated with a 2D-UCG computational fluid dynamics model considering syngas characteristics, formation of a cavity, reaction rate, and active zones. A two-stage gasification method was employed using lignite in a prism-shaped reactor in the presence of oxygen and steam (flow rate 5 m3/h). The final product/syngas has 40 vol% hydrogens with a calorific value of 8 MJ/Nm3 [37]. Dang et al. employed thermodynamics and biomass reaction kinetics for syngas production analysis. Data analysis identified the initial volatile composition from pyrolysis as a critical point for final product distribution while gasification temperature determines syngas composition and yield. Additionally, the moisture content in the feed, equivalence ratio, and steam-to-biomass ratio are also crucial parameters. Multivariable analysis-based optimization led to a maximum yield of 78.6 vol% at 900 °C from biomass feed with 30% moisture content, with an ER of 0.23 and a steam-to-biomass ratio of 0.21 [38].
Syngas is produced from the thermal process which not only has a high temperature but also carries particulate matter and tar compounds (depending upon the sources). Studies have suggested that syngas processing may become one of the necessary steps before fermentation as it might improve the microorganism’s activities. Cleaning is a multistage process in which raw gases pass through water-trayed columns to eliminate fine char and ash particulate. Besides wet filters, cyclones, sieve filters, or candle filters are also used. In this stage, fine particulates, chlorides, ammonia salts, and traces of H2S are removed [39][40]. After scrubbing, gas is passed through sulfided, activated carbon beds which removed a major fraction (>90%) of the mercury and heavy metal from syngas [41][42]. Syngas usually contains acid gases including H2S, carbonyl sulfide ‘COS’, and CO2 near the ambient temperature of 37.7 °C. Acid gases are removed by passing the gas from regenerative solvents like methanol, dimethyl ether, methyl diethanolamine, rectisol, etc., in an absorber column [43].
Wilson et al. compared twenty zeolites, three activated carbons, two activated aluminas, and two silica gels for CO2 and CO separation at 30 °C. As the concentration of CO and CO2 changes with production methods and process conditions, different types of adsorbents may be needed for syngas cleaning. Among twenty-seven samples, high-density silica gel and H-Y-type zeolites were most effective in CO2 removal from syngas. This was due to the shape of their isotherms which offered higher CO2 adsorption capacities contributed by Na+ exchange in zeolites at low pressures [44]. Monir and colleagues compared the effect of syngas processing and the removal of tarring compounds on microorganisms during a syngas fermentation trial for ethanol production. Among two syngas streams used, one was used as it is and and one was processed by passing through methanol and acetone (to remove tar compounds) followed by a cotton filter (to remove particulate matter). Syngas impurities in the untreated stream have an adverse effect on cell growth. Until the second day, bacterial growth was rapid followed by a stationary phase. Till the 11th day, growth appeared to be steady, then bacteria were not even visible. In the case of treated syngas, a lag phase was observed in bacterial growth during which bioethanol was produced. Comparative analysis revealed that cell growth in the treated stream was 500 times higher in comparison to the untreated one [45]. Liakakou et al. also processed the syngas produced from beech wood and lignin-rich feedback (received from biorefinery). Both the syngas contained tar compounds and sulfur-containing compounds. The gas was cleaned by a series of cleaning chambers equipped with host gas filters, activated carbon, OLGA, and TNO cleaning systems along with a CoMoO catalyst-based desulfurization unit. After cleaning, the gas streams were used for microbial fermentation for acetate and ethanol production [22].
Cali and colleagues integrated an air-blown gasifier of 5 MWth with a wet scrubber for syngas production and in-line cleaning. The whole unit is combined with a wastewater management system. Gasifier converted stone pine and eucalyptus feed to syngas with a conversion efficiency of 79–80%. In the cleaning unit system, tar and dust particles were removed via wet cleaning followed by water scrubbing. From here on, the outlet passed to waste management and regeneration systems like oil skimmers and activated carbon-based absorbent systems to regenerate the water followed by its reutilisation [46].
Chatrattanawet et al. simulated the syngas production followed by its cleaning using AspenPlus™ software [47]. It was found that syngas yield increased with the rise in temperature and achieved an optimum yield at 750 °C and a molar flow rate of ∼149 kmol/h. The air gasification unit offered a higher syngas molar flow rate (A/B: 1.309) than steam-air gasification. Gasification was followed by absorption-based cleaning to capture CO2 and H2S using monoethanolamine. The cleaning process was operated at 40 bars of column pressure with 10 trays. As a result, H2S content reduced to >0.1 mg/m3 at monoethanolamine with all kinds of gasification systems comprised of steam, air, and steam-air having molar flow rates of 325, 450, and 465 kmol/h, respectively [47]. Frilund and colleagues developed a hybrid activated carbon and ZnO-based adsorbent for syngas cleaning. It was reported that the adsorbent successfully removed almost all syngas contaminants of biomass origin and the results were better than the wet scrubbing system [48].
2.1. Chemical-Based Methods for Syngas Conversion into Bio-Alcohol
Syngas (H2/CO) can act as ideal feedstock for the production of numerous chemicals used as fuel/fuel additives and polymers. Chemical catalysis is one of the preferred methods used for syngas conversion [49]. However, this relies upon heterogenous or heavy metal catalysts for the purpose. Among various chemicals, ethanol and butanol are among the major products that can be employed as fuel/fuel additives and act as carriers for hydrogen in fuel cells.
Spivey and Egbebi developed a process for ethanol production from biomass-derived syngas under thermodynamically practicable operating conditions, i.e., 350 °C at 30 bar. Under the given conditions, selective conversion was very low (<10%), and ethanol production was further lowered if methane was also allowed as a product. Maximum ethanol production was achieved with an Rh-based catalyst [50]. Metal-based hybrid catalyst (Cu-ZnO-metal loaded ZSM-5) was evaluated for the conversion of syngas to gasoline-ranged hydrocarbons at 543 K in near-critical n-hexane. Cu-ZnO catalyzed the hydrogenation of CO to methanol and finally to hydrocarbons over the metal-loaded ZSM-5 via dimethyl ether. Among the selected metals, i.e., Pd, Co, Fe, and Cu, hydrocarbon yields of 59% and 64% were obtained with Pd/ZSM-5 and Cu/ZSM-5, respectively, at 5% metal loading. A further increase in Cu loading in ZSM-5 lowered ether and improved hydrocarbon yield. In addition, Cu/ZSM-5 hybrid catalyst was not deactivated for 30 h of the reaction [51].
Kang and colleagues employed triple tandem catalysis for syngas-to-ethanol conversion. The catalyst was comprised of potassium-modified ZnO–ZrO2+ modified zeolite mordenite + Pt–Sn/SiC. These three catalysts work in a single reactor but in a sequential manner as K+–ZnO–ZrO2 catalysed the hydrogenation of syngas to methanol, mordenite fraction carboxylate methanol to acetic acid, and Pt–Sn/SiC further hydrogenate it to ethanol. The maximum syngas-to-ethanol conversion was reported with 90% selectivity [49]. Kaithal et al. reported the conducive effect of alcohol for CO hydrogenation to methanol under the catalysis of molecular manganese complex [Mn(CO)2Br[HN(C2H4PiPr2)2]] ([HN(C2H4PiPr2)2] = MACHO-iPr). The complex attained the turnover number of 4023 with a turnover frequency of 857/h in the presence of EtOH/toluene as solvent at 150 °C, 5/50 bar (pCO/H2), in 8–12 h. The reaction attained a selectivity of >99% under optimum conditions without accumulation of formate ester [52].
It is clear from the above examples that chemical catalysis offered very high selectivity and tenability for syngas conversion to alcohols and other products, but like other processes, it also operates at a very high-temperature range, from 350–600 °C, and pressure. In addition, the cost of catalysts is also very high in the case of Rh, Pd, Pt, etc., which need regeneration after regular intervals. The process also employed solvents which may be hazardous. Overall, technical feasibility, process cost, and environmental suitability make it more challenging to find some eco-friendly alternatives. In order to make the conversion process sustainable and cost-effective over chemical catalysis, biological sequestrations have been adopted. This approach emphasized the utilization of major components of syngas, i.e., CO, H2, and in some cases COx and NOx as well in regular metabolic pathways of microorganisms via assimilation into various value-added chemicals and microbial biomass.
2.2. Biological Sequestration and Syngas Fermentation
Microorganisms import component gases (CO, H2, and CO2) from syngas and metabolic enzymes oxidize/reduce these and finally use them for regular metabolism. It has been found that in anaerobes the Wood-Ljungdahl pathway is critical for the sequestration of CO as well as CO2 while aerobic microorganisms utilize two different mechanisms. chemolithoautotrophs oxidize CO to CO2 in the presence of carbon monoxide dehydrogenase while another group of microorganisms, i.e., carboxydotrophs, utilizes molecular oxygen and oxidizes CO. The resultant CO2 is sequestered to the regular metabolic pathway via the pentose pathway and leads to glycolysis and the TCA cycle.
The Wood-Ljungdahl pathway can be classified into the carbonyl phase and methyl phase pathway [27]. Both the branches are connected at the acetyl-CoA junction, and from here, the successive reactions determine the fate of the molecules like the synthesis of the biomass or some other value-added chemicals including alcohol, acetates, acids, etc. (Figure 3).
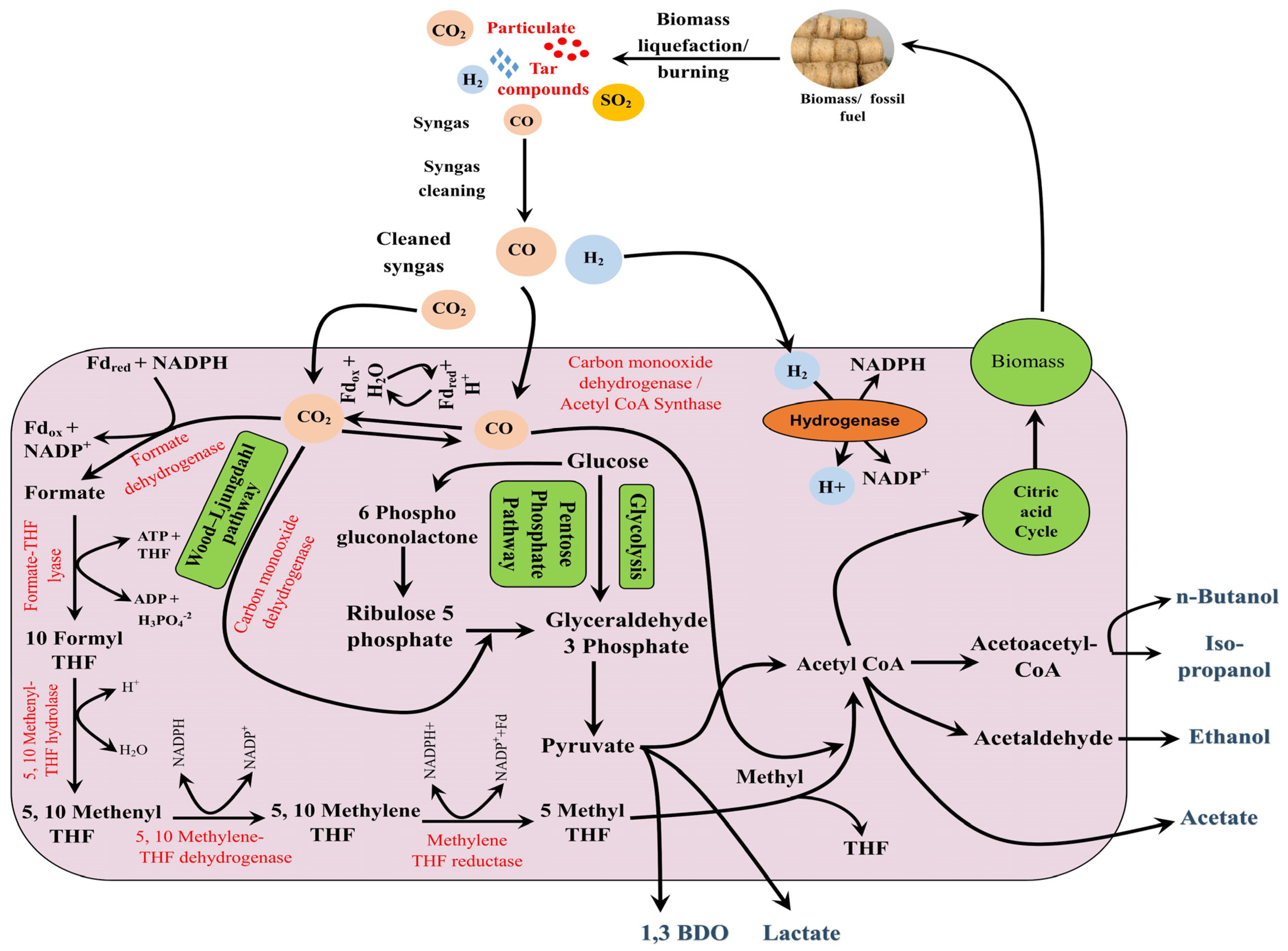
Figure 3. Sequestration of syngas and production of microbial metabolites.
Anaerobic bacteria follow the ‘Wood-Ljungdahl pathway’ for the utilization of CO and CO2 sequestration. For syngas fermentation sequestration hold higher values, as syngas is rich in CO. Carbon monoxide dehydrogenase enzyme catalyzed the interconversion of CO to CO2 reversibly [53]. Researchers have reported the CO−dehydrogenase complex (CODH) from Methanosarcina thermophila [54], Hydrogenophaga pseudoflava [55], Oligotropha carboxidovorans [56], Thermoanaerobacter kivui [57], etc. This enzyme is known to catalyze the assimilation reaction among CO, CH3 moiety, and coenzyme-A (CoA) for the synthesis of acetyl−CoA [54][58]. The other one, i.e., methyl branch, represents the utilization of CO2 via a multistep pathway that generates methyl moieties. CO2 is assimilated into formate by formate dehydrogenase and successively reduced to a methyl group. Methyl radicle is bound to the tetrahydropterin coenzyme and then assimilated to acetyl-CoA together with CO [59].
Under aerobic conditions also, some of the microorganisms like chemolithoautotrophs and aerobic carboxydotrophs possess the potential to utilize carbon monoxide as the sole source. For chemolithoautotrophs, CO acts as a sole source of carbon, as well as electrons, and in the presence of carbon monoxide, dehydrogenase enzyme CO oxidizes to CO2 [29]. While for aerobic carboxydotrophs, CO is oxidized by utilizing molecular oxygen. From here, carbon monoxide can be sequestered via the pentose phosphate pathway [28].
These pathways are themselves under the expression of related genes; hence, genetic recombination and overexpression of genes would enhance the tolerance of microorganisms and fermentation rate. Insilico analysis of C. ljungdahlii of the genome was conducted by genome-scale reconstruction and the OptKnock computational framework to identify gene knockouts, followed by metabolic engineering for the overproduction of ethanol, and the production of native products like butanol and butyrate along with the increase in the production of native products including lactate and 2,3−butanediol. In the native strain C. ljungdahlii iHN, the 637 genes identified contributed to 698 metabolites and 785 reactions. The OptKnock-derived strategies were combined with a spatiotemporal metabolic model considering the syngas bubble column reactor to overcome the drawbacks of decreased growth. The two-stage methodology fabricated a new C. ljungdahlii engineered strain which has increased product synthesis under realistic syngas fermentation conditions with the supply of CO and H2 uptake rates of 35 and 50 mmol/gDW/h. The analysis also revealed that only 201 genes and 331 reactions were critical among 637 genes and 785 reactions [60]. With an aim of higher butanol and ethanol production, four genes, i.e., adhE2, aor, and fnr were considered for metabolic engineering. These genes were dedicated for acetyl−CoA/butyl−CoA to acetaldehyde/butaldehyde and acetaldehyde/butaldehyde to ethanol/butanol (adhE2), acetate/butyrate to acetaldehyde/butaldehyde (aor), and regeneration of NADH at the cost of FdH2 (fnr), respectively. Cells were engineered with Escherichia coli as a donor as well as host, i.e., DH5α as host for plasmid amplification and CA434 as a donor cell for conjugation-assisted transfer. Overexpression of different genes has affected the metabolism differently, but all the engineered strains have higher alcohol production from glucose than the wild. In the presence of the aor gene, strains were able to reassimilate CO2 even during heterotrophic growth. ‘adhE2’-overexpressed strains have ∼50% higher ethanol production, while the combination of adhE2 and fnr improved the butanol production by ∼18% as well as ethanol by ∼22%. The strains exhibiting higher alcohol production were able to re-assimilate acid [61].
Lauer et al. used a metabolic engineering approach for the overproduction of butanol and hexanol. A gene cluster of 17.9-kb carrying 13 genes from C. acetobutylicum (for hexanol) and C. kluyveri (butanol) was inserted into C. ljungdahlii via conjugation resulted in a butanol yield of 1075 mg/L (butanol) and 133 mg/L (hexanol) from fructose. In the case of a gaseous substrate (comprised of 80% H2 and 20% CO2), the yield of butanol and hexanol were 174 mg/L and 15 mg/L, respectively. The insertion of the gene cluster expressed all 13 enzymes encoded by the cluster. In the next step, a first-round selection marker was eliminated using CRISPR/Cas9, and a 7.8 kb gene cluster with 6 genes from C. carboxidivorans was further inserted that resulted in the hexanol and butanol titers of 251 mg/L and 158 mg/L, respectively, from the gaseous substrate. Further scale-up of fermentation to 2 L resulted in increased titer to 109 mg/L and 393 mg/L for butanol and hexanol, respectively [62]. Genetic and metabolic engineering have opened the doors to the industrial application of microorganisms with unending opportunities. It not only improved the efficiency of native fermentation products but also added new products to the metabolites.
Syngas fermentation into bio-alcohol: Syngas fermentation is based on the microbial utilization of gases and sequestration into value-added products and microbial biomass. Its kinetics and process dynamics are different from the conventional submerged fermentation as it solely relies upon the solubility of the gaseous phase into medium followed by its utilization [54]. Among all the microbial products, alcohol, especially ethanol, has its own market as it can be used in beverages and healthcare, besides fuels. Maintenance of anaerobic conditions during the production phase is the major prerequisite for alcohol/ethanol production. Syngas fermentation itself creates a similar environment without any preprocessing and pretreatment of feedstock (as in the case of biomass) [55]. Richter and colleagues reported that ethanol production by C. ljungdahlii from syngas fermentation was mainly controlled and regulated by thermodynamics and not by the expression of responsible enzymes. Acetogenic bacteria exhibited two different physiological phases: the growth phase (acidogenesis for acid/acetate production) and the starvation phase (solventogenesis for ethanol production). Both stages were maintained at equilibrium for acetate and ethanol production, respectively, in a sequential bioreactor. Protein profiling with around 2000 proteins from both stages revealed that nutrient-limited conditions are responsible for the transition from acidogenesis to solventogenesis without any change in the enzyme population in the central energy metabolism, as enzymes responsible for ethanol production were remain present in abundance even during acidogenesis. Thermodynamic modeling recognized reduced cofactors and acetic acid as saturation reactants as well as switching points. As soon as intracellular undissociated acetic acid touched the threshold limit, bacterial cells diverted the surplus reducing equivalents for ethanol production. During syngas fermentation, reducing equivalents cannot be diverted for biomass production, and the CO-rich syngas supply was still high. Nutrient availability and pH can be used as switching points and can aid in attaining the desired level of solventogenesis [63].
References
- Kaskaoutis, D.G.; Kumar, S.; Sharma, D.; Singh, R.P.; Kharol, S.K.; Sharma, M.; Singh, A.K.; Singh, S.; Singh, A.; Singh, D. Effects of Crop Residue Burning on Aerosol Properties, Plume Characteristics, and Long-Range Transport over Northern India. J. Geophys. Res. Atmos. 2014, 119, 5424–5444.
- Abdurrahman, M.I.; Chaki, S.; Saini, G. Stubble Burning: Effects on Health & Environment, Regulations and Management Practices. Environ. Adv. 2020, 2, 100011.
- Sharma, S.K.; Agarwal, P.; Mandal, T.K.; Karapurkar, S.G.; Shenoy, D.M.; Peshin, S.K.; Gupta, A.; Saxena, M.; Jain, S.; Sharma, A.; et al. Study on Ambient Air Quality of Megacity Delhi, India During Odd–Even Strategy. Mapan 2017, 32, 155–165.
- Govindaraju, M.; Fowmitha Banu, J.; Senthamil selvi, S.; Goel, M. CO2 Sequestration Through Phytoremediation Techniques with Special Emphasis on Urban Forestry to Mitigate Climate Change Impact. In Climate Change and Green Chemistry of CO2 Sequestration; Green Energy and Technology; Goel, M., Satyanarayana, T., Sudhakar, M., Agrawal, D.P., Eds.; Springer: Singapore, 2021; pp. 263–271. ISBN 9789811600296.
- Al-Taai, S.H.H. Soil Pollution—Causes and Effects. IOP Conf. Ser. Earth Environ. Sci. 2021, 790, 012009.
- von Schneidemesser, E.; Driscoll, C.; Rieder, H.E.; Schiferl, L.D. How Will Air Quality Effects on Human Health, Crops and Ecosystems Change in the Future? Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2020, 378, 20190330.
- Wei, W.; Wang, Z. Impact of Industrial Air Pollution on Agricultural Production. Atmosphere 2021, 12, 639.
- Singh, J.; Singhal, N.; Singhal, S.; Sharma, M.; Agarwal, S.; Arora, S. Environmental Implications of Rice and Wheat Stubble Burning in North-Western States of India. In Advances in Health and Environment Safety; Springer Transactions in Civil and Environmental Engineering; Siddiqui, N.A., Tauseef, S.M., Bansal, K., Eds.; Springer: Singapore, 2018; pp. 47–55. ISBN 978-981-10-7121-8.
- Jain, N.; Bhatia, A.; Pathak, H. Emission of Air Pollutants from Crop Residue Burning in India. Aerosol Air Qual. Res. 2014, 14, 422–430.
- Saggu, G.S.; Mittal, S.K.; Agarwal, R.; Beig, G. Epidemiological Study on Respiratory Health of School Children of Rural Sites of Malwa Region (India) During Post-Harvest Stubble Burning Events. Mapan 2018, 33, 281–295.
- Bolívar Caballero, J.J.; Zaini, I.N.; Yang, W. Reforming Processes for Syngas Production: A Mini-Review on the Current Status, Challenges, and Prospects for Biomass Conversion to Fuels. Appl. Energy Combust. Sci. 2022, 10, 100064.
- Höhne, N.; Kuramochi, T.; Warnecke, C.; Röser, F.; Fekete, H.; Hagemann, M.; Day, T.; Tewari, R.; Kurdziel, M.; Sterl, S.; et al. The Paris Agreement: Resolving the Inconsistency between Global Goals and National Contributions. Clim. Policy 2017, 17, 16–32.
- Patrizio, P.; Fajardy, M.; Bui, M.; Dowell, N.M. CO2 Mitigation or Removal: The Optimal Uses of Biomass in Energy System Decarbonization. Iscience 2021, 24, 102765.
- Ahuja, V. Carbon Bio-Capturing System for Environment Conservation. In Advances in Carbon Capture and Utilization; Energy, Environment, and Sustainability; Pant, D., Kumar Nadda, A., Pant, K.K., Agarwal, A.K., Eds.; Springer: Singapore, 2021; pp. 99–126. ISBN 9789811606373.
- Medrano-García, J.D.; Ruiz-Femenia, R.; Caballero, J.A. Multi-Objective Optimization of a Carbon Dioxide Utilization Superstructure for the Synthesis of Formic and Acetic Acid. In Computer Aided Chemical Engineering; 28 European Symposium on Computer Aided Process Engineering; Friedl, A., Klemeš, J.J., Radl, S., Varbanov, P.S., Wallek, T., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; Volume 43, pp. 1419–1424.
- Calvo, D.C.; Luna, H.J.; Arango, J.A.; Torres, C.I.; Rittmann, B.E. Determining Global Trends in Syngas Fermentation Research through a Bibliometric Analysis. J. Environ. Manag. 2022, 307, 114522.
- Steinbusch, K.J.J.; Hamelers, H.V.M.; Plugge, C.M.; Buisman, C.J.N. Biological Formation of Caproate and Caprylate from Acetate: Fuel and Chemical Production from Low Grade Biomass. Energy Environ. Sci. 2010, 4, 216–224.
- Haas, T.; Krause, R.; Weber, R.; Demler, M.; Schmid, G. Technical Photosynthesis Involving CO2 Electrolysis and Fermentation. Nat. Catal. 2018, 1, 32–39.
- Oh, H.J.; Ko, J.K.; Gong, G.; Lee, S.-M.; Um, Y. Production of Hexanol as the Main Product Through Syngas Fermentation by Clostridium Carboxidivorans P7. Front. Bioeng. Biotechnol. 2022, 10, 850370.
- Xie, Q.; Kong, S.; Liu, Y.; Zeng, H. Syngas Production by Two-Stage Method of Biomass Catalytic Pyrolysis and Gasification. Bioresour. Technol. 2012, 110, 603–609.
- Hu, Q.; Shen, Y.; Chew, J.W.; Ge, T.; Wang, C.-H. Chemical Looping Gasification of Biomass with Fe2O3/CaO as the Oxygen Carrier for Hydrogen-Enriched Syngas Production. Chem. Eng. J. 2020, 379, 122346.
- Liakakou, E.T.; Infantes, A.; Neumann, A.; Vreugdenhil, B.J. Connecting Gasification with Syngas Fermentation: Comparison of the Performance of Lignin and Beech Wood. Fuel 2021, 290, 120054.
- Xiang, Y.; Luo, H.; Liu, G.; Zhang, R. Improvement of Organic Acid Production with Sulfate Addition during Syngas Fermentation Using Mixed Cultures. Water Cycle 2022, 3, 26–34.
- Phillips, J.R.; Atiyeh, H.K.; Tanner, R.S.; Torres, J.R.; Saxena, J.; Wilkins, M.R.; Huhnke, R.L. Butanol and Hexanol Production in Clostridium Carboxidivorans Syngas Fermentation: Medium Development and Culture Techniques. Bioresour. Technol. 2015, 190, 114–121.
- Schulz, S.; Molitor, B.; Angenent, L.T. Acetate Augmentation Boosts the Ethanol Production Rate and Specificity by Clostridium Ljungdahlii during Gas Fermentation with Pure Carbon Monoxide. Bioresour. Technol. 2023, 369, 128387.
- Schulz, S. Tailoring Clostridium Ljungdahlii for Improved Ethanol Production by Genetic Engineering of the Aldehyde: Ferredoxin Oxidoreductase (AOR) and Chemostat Fermentation. Ph.D. Thesis, Universität Tübingen, Tübingen, Germany, 2024. Available online: https://ub01.uni-tuebingen.de/xmlui/handle/10900/133662 (accessed on 10 January 2023).
- Bengelsdorf, F.R.; Straub, M.; Dürre, P. Bacterial Synthesis Gas (Syngas) Fermentation. Environ. Technol. 2013, 34, 1639–1651.
- Siebert, D.; Eikmanns, B.J.; Blombach, B. Exploiting Aerobic Carboxydotrophic Bacteria for Industrial Biotechnology. In One-Carbon Feedstocks for Sustainable Bioproduction; Advances in Biochemical Engineering/Biotechnology; Zeng, A.-P., Claassens, N.J., Eds.; Springer International Publishing: Cham, Switzerland, 2022; pp. 1–32. ISBN 978-3-031-06854-6.
- Tirado-Acevedo, O.; Chinn, M.S.; Grunden, A.M. Production of Biofuels from Synthesis Gas Using Microbial Catalysts. Adv. Appl. Microbiol. 2010, 70, 57–92.
- Sun, X.; Atiyeh, H.K.; Huhnke, R.L.; Tanner, R.S. Syngas Fermentation Process Development for Production of Biofuels and Chemicals: A Review. Bioresour. Technol. Rep. 2019, 7, 100279.
- Phillips, J.R.; Huhnke, R.L.; Atiyeh, H.K. Syngas Fermentation: A Microbial Conversion Process of Gaseous Substrates to Various Products. Fermentation 2017, 3, 28.
- Mäki-Arvela, P.; Aho, A.; Simakova, I.; Yu. Murzin, D. Sustainable Aviation Fuel from Syngas through Higher Alcohols. ChemCatChem 2022, 14, e202201005.
- Liu, G.; Yang, G.; Peng, X.; Wu, J.; Tsubaki, N. Recent Advances in the Routes and Catalysts for Ethanol Synthesis from Syngas. Chem. Soc. Rev. 2022, 51, 5606–5659.
- El-Nagar, R.A.; Ghanem, A.A.; El-Nagar, R.A.; Ghanem, A.A. Syngas Production, Properties, and Its Importance; IntechOpen: London, UK, 2019; ISBN 978-1-78984-581-5.
- Nourbakhsh, H.; Rahbar Shahrouzi, J.; Ebrahimi, H.; Zamaniyan, A.; Jafari Nasr, M.R. Experimental and Numerical Study of Syngas Production during Premixed and Ultra-Rich Partial Oxidation of Methane in a Porous Reactor. Int. J. Hydro. Energy 2019, 44, 31757–31771.
- Chen, P.; Jiao, Y.; Zhu, Y.-H.; Chen, S.-M.; Song, L.; Jaroniec, M.; Zheng, Y.; Qiao, S.-Z. Syngas Production from Electrocatalytic CO2 Reduction with High Energetic Efficiency and Current Density. J. Mater. Chem. A 2019, 7, 7675–7682.
- Gür, M.; Canbaz, E.D. Analysis of Syngas Production and Reaction Zones in Hydrogen Oriented Underground Coal Gasification. Fuel 2020, 269, 117331.
- Dang, Q.; Zhang, X.; Zhou, Y.; Jia, X. Prediction and Optimization of Syngas Production from a Kinetic-Based Biomass Gasification Process Model. Fuel Process. Technol. 2021, 212, 106604.
- Guan, X.; Hewitt, A.; Peng, W.; Vimalchand, P.; Nelson, M.; Pinkston, T.; Madden, D. Particulate Control Devices in Kemper County IGCC Project. Energy Rep. 2019, 5, 969–978.
- Kurella, S.; Meikap, B.C. Removal of Fly-Ash and Dust Particulate Matters from Syngas Produced by Gasification of Coal by Using a Multi-Stage Dual-Flow Sieve Plate Wet Scrubber. J. Environ. Sci. Health Part A 2016, 51, 870–876.
- Frilund, C.; Hiltunen, I.; Simell, P. Activated Carbons for Syngas Desulfurization: Evaluating Approaches for Enhancing Low-Temperature H2S Oxidation Rate. Chem. Eng. 2021, 5, 23.
- Molino, G.; Gandiglio, M.; Fiorilli, S.; Lanzini, A.; Drago, D.; Papurello, D. Design and Performance of an Adsorption Bed with Activated Carbons for Biogas Purification. Molecules 2022, 27, 7882.
- Mokhatab, S.; Poe, W.A.; Mak, J.Y. Chapter 3—Basic Concepts of Natural Gas Processing. In Handbook of Natural Gas Transmission and Processing, 3rd ed.; Mokhatab, S., Poe, W.A., Mak, J.Y., Eds.; Gulf Professional Publishing: Boston, MA, USA, 2015; pp. 123–135. ISBN 978-0-12-801499-8.
- Wilson, S.M.W.; Kennedy, D.A.; Tezel, F.H. Adsorbent Screening for CO2/CO Separation for Applications in Syngas Production. Sep. Purif. Technol. 2020, 236, 116268.
- Monir, M.U.; Aziz, A.A.; Khatun, F.; Yousuf, A. Bioethanol Production through Syngas Fermentation in a Tar Free Bioreactor Using Clostridium Butyricum. Renew. Energy 2020, 157, 1116–1123.
- Calì, G.; Deiana, P.; Bassano, C.; Meloni, S.; Maggio, E.; Mascia, M.; Pettinau, A. Syngas Production, Clean-Up and Wastewater Management in a Demo-Scale Fixed-Bed Updraft Biomass Gasification Unit. Energies 2020, 13, 2594.
- Chatrattanawet, N.; Authayanun, S.; Saebea, D.; Patcharavorachot, Y. Syngas Production from Sugarcane Leftover Gasification Integrated with Absorption Process for Green Liquid Production. J. Clean. Prod. 2019, 235, 519–534.
- Frilund, C.; Tuomi, S.; Kurkela, E.; Simell, P. Small- to Medium-Scale Deep Syngas Purification: Biomass-to-Liquids Multi-Contaminant Removal Demonstration. Biomass Bioenergy 2021, 148, 106031.
- Kang, J.; He, S.; Zhou, W.; Shen, Z.; Li, Y.; Chen, M.; Zhang, Q.; Wang, Y. Single-Pass Transformation of Syngas into Ethanol with High Selectivity by Triple Tandem Catalysis. Nat. Commun. 2020, 11, 827.
- Spivey, J.J.; Egbebi, A. Heterogeneous Catalytic Synthesis of Ethanol from Biomass-Derived Syngas. Chem. Soc. Rev. 2007, 36, 1514–1528.
- Ma, T.; Imai, H.; Yamawaki, M.; Terasaka, K.; Li, X. Selective Synthesis of Gasoline-Ranged Hydrocarbons from Syngas over Hybrid Catalyst Consisting of Metal-Loaded ZSM-5 Coupled with Copper-Zinc Oxide. Catalysts 2014, 4, 116–128.
- Kaithal, A.; Werlé, C.; Leitner, W. Alcohol-Assisted Hydrogenation of Carbon Monoxide to Methanol Using Molecular Manganese Catalysts. JACS Au 2021, 1, 130–136.
- Westerholm, M.; Moestedt, J.; Schnürer, A. Biogas Production through Syntrophic Acetate Oxidation and Deliberate Operating Strategies for Improved Digester Performance. Appl. Energy 2016, 179, 124–135.
- Abbanat, D.R.; Ferry, J.G. Synthesis of Acetyl Coenzyme A by Carbon Monoxide Dehydrogenase Complex from Acetate-Grown Methanosarcina Thermophila. J. Bacteriol. 1990, 172, 7145–7150.
- Reginald, S.S.; Lee, H.; Fazil, N.; Sharif, B.; Lee, M.; Kim, M.J.; Beyenal, H.; Chang, I.S. Control of Carbon Monoxide Dehydrogenase Orientation by Site-Specific Immobilization Enables Direct Electrical Contact between Enzyme Cofactor and Solid Surface. Commun. Biol. 2022, 5, 390.
- Ritacca, A.G.; Rovaletti, A.; Moro, G.; Cosentino, U.; Ryde, U.; Sicilia, E.; Greco, C. Unraveling the Reaction Mechanism of Mo/Cu CO Dehydrogenase Using QM/MM Calculations. ACS Catal. 2022, 12, 7336–7343.
- Jain, S.; Katsyv, A.; Basen, M.; Müller, V. The Monofunctional CO Dehydrogenase CooS Is Essential for Growth of Thermoanaerobacter Kivui on Carbon Monoxide. Extremophiles 2021, 26, 4.
- Biester, A.; Marcano-Delgado, A.N.; Drennan, C.L. Structural Insights into Microbial One-Carbon Metabolic Enzymes Ni-Fe-S-Dependent Carbon Monoxide Dehydrogenases and Acetyl-CoA Synthases. Biochemistry 2022, 61, 2797–2805.
- de Souza, Y.P.A.; Rosado, A.S. Chapter 19—Opening the Black Box of Thermophilic Autotrophic Bacterial Diversity. In Microbial Diversity in the Genomic Era; Das, S., Dash, H.R., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 333–343. ISBN 978-0-12-814849-5.
- Chen, J.; Henson, M.A. In Silico Metabolic Engineering of Clostridium Ljungdahlii for Synthesis Gas Fermentation. Met. Eng. 2016, 38, 389–400.
- Cheng, C.; Li, W.; Lin, M.; Yang, S.-T. Metabolic Engineering of Clostridium Carboxidivorans for Enhanced Ethanol and Butanol Production from Syngas and Glucose. Bioresour. Technol. 2019, 284, 415–423.
- Lauer, I.; Philipps, G.; Jennewein, S. Metabolic Engineering of Clostridium Ljungdahlii for the Production of Hexanol and Butanol from CO2 and H2. Microb. Cell. Fact. 2022, 21, 85.
- Richter, H.; Molitor, B.; Wei, H.; Chen, W.; Aristilde, L.; Angenent, L.T. Ethanol Production in Syngas-Fermenting Clostridium Ljungdahlii Is Controlled by Thermodynamics Rather than by Enzyme Expression. Energy Environ. Sci. 2016, 9, 2392–2399.
More
Information
Subjects:
Biotechnology & Applied Microbiology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.5K
Revisions:
2 times
(View History)
Update Date:
26 Apr 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





