
Video Upload Options
Lodging is one of the most chronic restraints of the maize-soybean intercropping system, which causes a serious threat to agriculture development and sustainability. In the maize-soybean intercropping system, shade is a major causative agent that is triggered by the higher stem length of a maize plant. Many morphological and anatomical characteristics are involved in the lodging phenomenon, along with the chemical configuration of the stem. Due to maize shading, soybean stem evolves the shade avoidance response and resulting in the stem elongation that leads to severe lodging stress. However, the major agro-techniques that are required to explore the lodging stress in the maize-soybean intercropping system for sustainable agriculture have not been precisely elucidated yet.
1. Introduction
Climatic change and population explosions are the major threats to food security in the future [1][2]. It has been projected that, by 2050, the present world population will be enhanced by up to 30%, which will make the world population nine billion or more people [3]. This problem can only be solved through multiple cropping systems to fulfill the food demand and supply requirements which leads to sustainable agriculture [4]. The maize-soybean intercropping is one of the most important systems that plays a key role in the sustainability of food production systems [5]. The maize-soybean intercropping system has great importance among the legume-cereal intercropping systems because of its maximum yield and efficient use of resources [6][7]. Initially, it was developed in the South-Western region of China and now it is progressing throughout the globe. It is estimated that from the total cropped area of 182.3 million hectares, about 83% area is used for intercropping in Africa [8]. In China, half of the total grain yield is gained through multiple cropping systems [9]. The maize-soybean intercropping system has been adopted in different parts of China due to its maximum production and land use efficiency [7]. The biggest challenge of this century has to be met by China to boost the production of cereals by approximately 600 Mt by 2030 to achieve food security [7]. In another study, it is reported that annually 2.8–3.4 × 107 ha area was grown under the intercropping system in China [10]. Over 667 thousand hectares of soybean are being intercropped with maize in the south-west of China [11][12].
Although this system has many advantages, in this intercropping system the spatial light pattern affects the growth of soybean due to the shading of maize plants during the co-growth period [13]. In order to capture more light, the soybean plants increase their heights and this phenomenon is called shade avoidance. Shade avoidance causes several morpho-physiological changes such as low photosynthetic activities, increased intermodal length, decreased stem diameter and higher rate of lodging [14]. Previous studies revealed that continuous and periodic prevailing of shade had decreased the total grain production, approximately 25–45% [15][16]. However, the climatic factors such as continuous storms and heavy rainfall comprise about 8% and 19% lodging to crops, respectively [17][18]. Many studies focused on the monocropping conditions of the soybean, fertilizer regulation and lodging resistant cultivars [19][20]. In addition, higher N application rates could enhance the lodging threat due to excessive canopy growth and stem elongation [21]. It is also noticed that excessive canopy growth decreases the light interception which in turn elongates the stem length [22][23]. Chen et al. [24] briefed that higher application of basal N decreased the lignin content in internodes. Moreover, it has been seen that high planting densities also reduced the lignin content in stem which leads to weaker stem and hence causes the plant lodging [25].
2. Agro-Techniques for the Management of Lodging Stress
2.1. Genetic Manipulation for Increasing Lodging Resistance
The poor resistance to lodging could reduce the soybean yield potential. Previously, independent studies have indicated a significant number of observations of quantitative trait loci (QTL) for lodging resistance [26]. A recent investigation on the integration of QTLs in the lodging resistance of soybean indicated the four QTLs which resulting in the two considerable QTL integrations on chromosomes 6 and 19. Their finding could be useful to increase the lodging resistance in soybean. Their results find a strong and pleiotropic relationship between the lodging resistance and QTL integration on chromosome 6 [27]. Several genes and their QTLs revealed the resistance to lodging and its related traits have been reported in rice (4CL gene family), wheat and barley [28][29][30]. Along with conventional breeding, we have to focus on the identification of lodging-resistance genes especially for cereal crops [31]. Now, identification and transformation of desired genes are much easier because of recent advances in breeding, genomics and biotechnology, which eventually help to increase crop productivity [32]. So, the transformation of lodging susceptible genes to lodging resistance genotypes has the potential to increase the grain yield of cereal crops under lodging prone zones [33]. The population based studies have been reported for QTL mapping in cereals and exhibited lodging resistance especially in wheat [34]. The QTLs study of phenotypic traits which are directly associated with lodging resistance in cereals had already been reported like plant stalk strength, pith diameter, culm diameter and culm wall thickness in wheat [35].
These QTLs have significant effects on lodging but further validation is needed by fine mapping or other advanced techniques. Currently, some of them have already been validated, for instance, in winter wheat a dominant Rht5 gene related to dwarfism in plant height, has been marked on chromosome 3BS linked with molecular marker Xbarc102 [36]. The recent studies revealed that this region on chromosome 6A showed higher phenotypic variations for plant height and could be used further for QTL mapping studies [37][38]. Rht3 is another dwarfing gene in wheat which shortened plant height up to 59% but has not been used in commercial varieties [39]. Rht5, a plant hormone (GA)-responsive gene for dwarfism, which significantly shortened the plant height up to 55% without having negative effects over coleoptile length and seedling vigor [40][41]. On the other hand, some researchers also reported that Rht5 has negative effect over flowering time and delayed by 4.8–14% in a thermal environment [42][43]. In addition, there is still an attention required from molecular actors to identify the sequence of candidate genes through QTLs intervals, map-based cloning to enhance the lodging resistance in soybean.
2.2. Proper Sowing Time and Planting Density
Sowing time and planting density are both key factors in the maize-soybean intercropping system that affect the lodging resistance and yield of soybean crop [44]. Sowing time is a crucial factor that enhanced the competition within the species which ultimately reduced the crop yield [45]. Therefore, the selection of an optimal sowing time is vital perspective to enhance the lodging resistance and yield under the maize-soybean intercropping system [46]. It has been observed that delay sowing significantly decreased the risk of lodging by shortening the internodal length, plant height and center of gravity point and by increasing the cell wall thickness, diameter and grain filling period [47]. For instance, only two weeks late sowing could reduce up to 30% threat of lodging in wheat [48]. Under the maize-soybean intercropping the tall stature crops adversely affected the short stature crops at the both vegetative and reproductive stages as compared to relay intercropping in which the adverse effect could only be observed at vegetative stage [7]. However, in intercropping systems, the competition between the intercropped species could be decreased by fluctuating the planting time [49]. Most importantly, under relay intercropping systems, the crop sown first have more competition than the second crop [50]. On the other hand, under the relay intercropping system, selecting an optimal planting time could reduce the co-growth duration among the intercropped species and adverse effect of first sown crop could also be minimized on the second grown crop [51]. Furthermore, it is shown that fluctuating the planting time, 50 days of co-growth period of maize and soybean in relay intercropping system enhanced the crop growth rate 17–64% as compared to 70–90 days of co-growth period. In maize-soybean relay intercropping the soybean production was recorded maximum 2.11 t/ha at 50 days co-growth duration of maize and soybean [49].
High plant populations have been used extensively to increase crop production [52]. Under high planting densities, mutual shading of plants disturbs the light environment of crop which reduced the photosynthetic activities and carbohydrates accumulation in the stem that leads to lodging easily [25][53]. It has been witnessed that planting densities were negatively correlated with the lodging resistance of stem; however, high plant population had promoted the lodging and lower grain production [53][54][55][56]. In addition, optimum planting densities could expand the structure of the plant population and provide a better light environment, enhanced photosynthetic rate and ultimately increased the lodging resistance of stem [57][58]. Furthermore, it was found that in strip intercropping optimum planting densities that is, 17 and 20 plants/m2 had significantly enhanced the stem diameter by 4.3% and 6%, respectively as compared to 25 plants/m2 planting density. Most specifically, the plant height was decreased by 6.2% and 9.4% at 17 and 20 plants/m2, respectively than that of 25 plants/m2 planting density. In addition, decreasing the planting densities such as 17 and 20 plants/m2 could decrease the lodging percentage by 50.3% and 19.3%, respectively as compared to 25 plants/m2 planting density [59].
2.3. Efficient Use of Fertilizers
Nitrogen (N), phosphorus (P) and potassium (K) are the important macro-elements, vital for crop growth and development [60]. N fertilizer application rate and time of application had also a significant effect on the lodging resistance of crops [61][62]. It has been described that high nitrogen rate had reduced the diameter of basal or lower internode that cause lodging [63]. On the other hand, the low application of nitrogen had enhanced the concentrations of water soluble carbohydrates as in the middle internodes 21% and in the basal internodes 42% than that of high nitrogen rate [64]. Several researchers suggested that a high rate of nitrogen application promoted vegetative growth and decreased the root anchorage in the soil and stem secondary cell wall composition (lignin content) which resulted in lodging of the crops [25][58][65]. A higher rate of nitrogen application had reduced the activities of lignin related enzymes (PAL, POD, CAD and 4CL) and lignin deposition in the cell wall which decreased the lodging resistance [24]. Berry et al. [66] also observed that a low rate of N had reduced the height of plant and also proliferated the stem diameter and cell wall width which turns in high stem strength. Another study had found that increasing the amount of N in wheat crop had progressively increased the cell wall constituents (lignin and cellulose content) and afterward it was decreased gradually [65]. Many recent studies showed that lodging resistance of crop could be improved at the price of yield sacrificing by minimizing the N application rate and rescheduling the application of N fertilizers [66][67]. Furthermore, the relation between N and K has a fundamental role in improving crop grain production and quality [63][67]. Increasing the level of K+ along with elevated NH4+ could decrease the stem cell wall thickness. For an instance, the application of a high level of N and P fertilizers in the absence of K decreased the 30%–35% grain production in rice due to lodging [68]. However, with the application of K, the stem mechanical strength could be increased [60]. An equal application rate of N and K had significantly promoted the root growth and enhanced the root anchorage which resulted in lodging resistance [69].
The optimum level of K+ nutrition to plants was positively correlated with lignin accumulation into the vascular bundles and cells of sclerenchyma of plant cell wall which ultimately enhanced the stem diameter and lodging resistance [70]. In the same way, it has been noticed that K+ considerably inhibited the adverse effects of a higher level of NH4+ which in turns amplified the 20–27% of stem mechanical strength in wheat [63]. However, K+ has a pivotal role in the process of photosynthesis and metabolic activities of carbohydrates production in plants [71][72]. However, appropriate fertilization of nitrogen could enhance the lignin content in the basal internode and improved the stem lodging resistance [24][73][74]. Conclusively, there is a still gap in nitrogen fertilization of soybean under maize-soybean intercropping; henceforth a more attention is required from agronomists and plant breeders for rescheduling the rate and application of nitrogen fertilizer which play a major role in lodging resistance and crop final production.
Previous experiments have revealed that the efficiency of intercrops to absorb nitrogen (N) is more than the sole-cropping, however, the total uptake of N by intercropped soybean and wheat is greater than the total of the sole crops [75][76]. According to an estimation, soybean can fix nitrogen about 39 to 182 kg N ha−1 [77]. It has been noticed that under high nitrogen conditions legumes are usually shaded by the maize which results in shade avoidance response and low grain production [78]. These adverse effects of cereal crops on legumes can be alleviated by fluctuating the sowing date [79][80]. A recent meta-analysis explained the land and fertilizer nitrogen use efficiency of intercropped maize and soybean [81]. They concluded that maize-soybean intercropping system has greater potential to attain high land equivalent ratio (LER) and fertilizer nitrogen equivalent ratio (FNER) by utilizing the optimum levels of nitrogen inputs. Whereas further studies are needed to pinpoint the optimum combinations of sowing configuration, planting dates and fertilizer rate and time to attain the high yields by reducing the lodging stress.
2.4. Development of Lodging Resistant Cultivars
Agronomists characterized the soybean cultivars into three sets depending on the genotypes response towards lodging resistance: highly, moderately and susceptible cultivars [82]. In some previously conducted experiments, different cultivars of soybean were grown under the maize-soybean intercropping system to distinguish the more suitable and lodging resistance cultivars. In a previous experiment, four recombinant inbred lines (B3, B15, B23 and B24) of Nandou-12 (that is shade tolerant and widely grown in maize-soybean intercropping system of China) and Nan 032-4 (that is shade susceptible cultivar) in were used [83], the lignin content in stem and lodging resistance index of B23 and B24 was significantly higher than that of B3 and B15 under both monocropping and intercropping systems. Furthermore, another experiment was conducted in which three cultivars were selected on the base of their response to lodging and shade stress [19]. A shade susceptible cultivar (Nan 032-4), a moderate cultivar to shade and lodging tolerance (Jiuyuehang) and shade tolerance and lodging resistance cultivar (Nandou-12) [19]. Their findings revealed that Nandou-12 had more accumulation of lignin in stem and high enzymatic activities of lignin related enzymes (PAL, 4CL, CAD and POD), hence more lodging resistance as compared to Nan 032-4 and Jiuyuehang cultivars under both monocropping and intercropping systems (Figure 1). Along with these research outcomes, there is a dire need to work out on the soybean cultivars which are well suited for intercropping systems with greater lodging resistance.
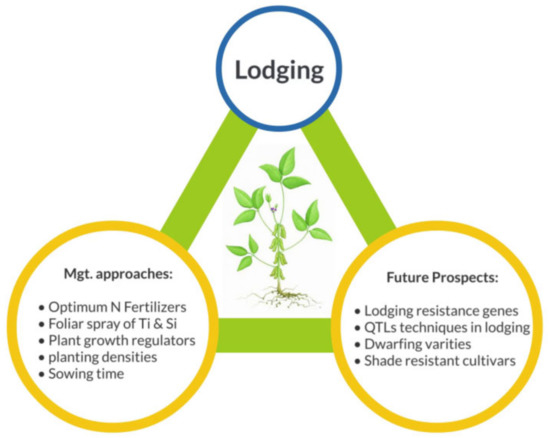
Figure 1. An overview of the most suitable approaches including molecular breeding techniques, agronomical managements and chemical controls to mitigate the lodging stress in maize-soybean intercropping.
2.5. Role of Silicon and Titanium
Si plays a pivotal role in the growth and development of plants as a beneficial micronutrient. A significant impact of silicon has been observed on the plant height, internodal length and stem strength and lodging tolerance [84][85]. It was found that the application of Si could be dispersed from third to the fourth internode, which result in enhancement of lodging resistance [86]. A previous study revealed that Si had enhanced the thickness of the cell wall and the vascular bundles size in the rice stem [87]. With increase in the amount of Si application, the cellulose and hemicellulose content are increased which contribute in the cell wall formation of rice stem [88]. However, it was also identified that Si acts as ligands binding with hemicellulose in the rice cell walls [89][90]. A recent research showed that appropriate concentrations of Si could improve the enzymatic activity of lignin related enzymes and also prompted the gene expression of related enzymes [91]. It also revealed that Si content also increased the lignin content in the stem cell wall and promoted the lodging resistance of soybean under low light conditions. Moreover, Si also accelerated the photosynthetic activities, stomatal conductance and increased the chlorophyll content of tobacco under cadmium stress [92]. Wang et al. [93] observed that Si element could amplify the photosynthetic rate by altering the leaf structure and the content of chlorophyll in rice plants. In addition, Si element can change the leaf anatomy to capture more light and enhance the light interception in the plant’s leaves and to improve the vascular bundle sheath and sclerenchyma tissues, which help in lodging resistance [94][95]. Another research has concluded that Si could be used as fuel to ignite the process of lignification and silicification for the cell wall and collenchyma cells thickness that increases the development of keratinocyte and cellulose contents resulting in lodging tolerance [96].
On the other hand, the biological importance and role of titanium (Ti) in growth and development has been studied for decades but still, it is not recognized as an essential nutrient for the plants. However, recent research revealed that optimum concentration of Ti improves the leaf chloroplast structure, total biomass, chlorophyll fluorescence, root architecture, RuBisCO enzyme activity and total chlorophyll content of soybean plants under low light conditions [97]. In addition, how Ti affects the lignin content is not elaborated yet under low light conditions. Therefore, in future RNAseq transcriptional studies, proteomics and genomic profiling should be done to gain deeper insights into the effects and benefits of Si and Ti on soybean stem strength, lodging resistance under the maize-soybean intercropping system.
2.6. Chemical Approaches
Foliar application of plant growth regulators at the suitable growth stage of a crop can enhance the stem mechanical strength, reduce the plant height and inhibit lodging [98][99]. Plant growth hormones that stop the biosynthesis of GA are being widely used in high input cropping systems to decrease straw content and also increase the lodging resistance [98]. Many plant growth regulators were extensively utilized to minimize lodging index through shortening the plant height and to obtain a stable grain production [99][100]. The most common growth regulators that inhibit the GA production have been used to decrease the growth of stem are the onium type elements, which have Chlormequate chloride (2-chloroethyl-N, N, N-trimethyl-ammonium chloride, CCC) and Mepiquat-Cl. Some other plant growth regulators which comprise N heterocycles for example, triazoles and imidazoles could also be used to minimize the lodging risk [101]. The application of paclobutrazol had considerably increased the lignin content in the stem cell wall and its function is closely related to enzymes present in the basal second internode [102]. It had also enhanced the stem diameter, internode filling degree and cell wall thickness with increasing the lodging resistance [103]. The Paclobutrazol chemical (PP333) could prompt the enzymatic activities of lignin related enzymes that is, tyrosine ammonia-lyase (TAL), phenylalanine ammonia-lyase (PAL) and cinnamyl alcohol dehydrogenase (CAD) and ultimately proliferated the lignin content and lodging resistance [104]. A plant growth regulator (Trinexapac-ethyl) has reduced the height of the plant in wheat [105][106]. In addition, some other chemicals are needed to be introduced which are capable of enhancing the lodging resistance in intercropped soybean to get better results.
3. Future Prospects
In the maize-soybean intercropping system, shade has a drastic effect on the normal growth and development of soybean at both vegetative and reproductive stages. Shading of maize disturbs the microclimate of soybean which results in shade avoidance response of soybean (stem elongation) and finally causes lodging. The influence of lodging concerning the reduction of grain yield depends on the types of cultivars and planting geometries. This review mainly focused on the multiple approaches and genetic techniques which would be helpful to control the lodging under intercropping systems (Figure 3). The damages induced by lodging in maize-soybean intercropping could be actively reduced with more advanced crop breeding techniques. We further explored that, lignin and cellulose are the main constituents of plant cell wall which play a vital role in plant vigor against biotic and abiotic factors such as lodging. However, the molecular mechanism of lignin and cellulose formation and their relation with hormones (indole-acetic acid, IAA) that how they affect each other has not been explored thoroughly yet. Therefore, it is necessary to explore the biochemical association of hormones and lignin biosynthesis pathways in the maize-soybean intercropping system.
In addition, researchers have to develop new techniques and tools to modify the lignin content in soybean stem without altering its normal functions. In consequences of natural calamities like high-speed winds and rainfall could damage the crops catastrophically through lodging. Therefore, to escape from these devastating effects of lodging following approaches could be adopted: (i) breeding of soybean cultivars with stronger and harder roots without disturbing the existing root numbers per plant, (ii) suitable agronomic management’s that is, use of lodging resistant cultivars, rescheduling the planting time and density and use of fertilizers. Furthermore, plant growth regulators can also manipulate the height of the plant which helps in lowering the risk of lodging. Optimum levels of N, P, K and Si fertilizers could play a significant role in the maize-soybean intercropping system. Additionally, further studies are required to alter the plant canopy area which is a vital part of modern agronomy techniques and it is usually obtained through the application of balanced nitrogen fertilizers.
References
- Oelbermann, M.; Echarte, L. Evaluating soil carbon and nitrogen dynamics in recently established maize-soyabean inter-cropping systems. Eur. J. Soil Sci. 2011, 62, 35–41.
- Tscharntke, T.; Clough, Y.; Wanger, T.C.; Jackson, L.; Motzke, I.; Perfecto, I.; Vandermeer, J.; Whitbread, A. Global food security, biodiversity conservation and the future of agricultural intensification. Biol. Conserv. 2012, 151, 53–59.
- Rosenberg, M. Current World Population. 2011. Available online: www.about.com (accessed on 1 February 2011).
- Vogel, E.; Meyer, R. Climate Change, Climate Extremes, and Global Food Production—Adaptation in the Agricultural Sector. In Resilience; Elsevier: Amsterdam, The Netherlands, 2018; pp. 31–49.
- Yang, F.; Liao, D.; Fan, Y.; Gao, R.; Wu, X.; Rahman, T.; Yong, T.; Liu, W.; Liu, J.; Du, J.; et al. Effect of narrow-row planting patterns on crop competitive and economic advantage in maize–soybean relay strip intercropping system. Plant Prod. Sci. 2016, 20, 1–11.
- Kermah, M.; Franke, A.C.; Adjei-Nsiah, S.; Ahiabor, B.D.K.; Abaidoo, R.C.; Giller, K.E. Maize-grain legume intercropping for enhanced resource use efficiency and crop productivity in the Guinea savanna of northern Ghana. Field Crops Res. 2017, 213, 38–50.
- Iqbal, N.; Hussain, S.; Ahmed, Z.; Yang, F.; Wang, X.; Liu, W.; Yong, T.; Du, J.; Shu, K.; Yang, W.; et al. Comparative analysis of maize–soybean strip intercropping systems: A review. Plant Prod. Sci. 2019, 22, 131–142.
- Knörzer, H.; Graeff-Hönninger, S.; Guo, B.; Wang, P.; Claupein, W. The rediscovery of intercropping in China: A traditional cropping system for future Chinese agriculture—A review. In Climate Change, Intercropping, Pest Control and Beneficial Microorganisms; Springer: Berlin/Heidelberg, Germany, 2009; pp. 13–44.
- Hong, Y.; Heerink, N.; Zhao, M.; van der Werf, W. Intercropping contributes to a higher technical efficiency in smallholder farming: Evidence from a case study in Gaotai County, China. Agric. Syst. 2019, 173, 317–324.
- Li, L.; Li, S.-M.; Sun, J.-H.; Zhou, L.-L.; Bao, X.-G.; Zhang, H.-G.; Zhang, F.-S. Diversity enhances agricultural productivity via rhizosphere phosphorus facilitation on phosphorus-deficient soils. Proc. Natl. Acad. Sci. USA 2007, 104, 11192–11196.
- Du, J.-B.; Han, T.-F.; Gai, J.-Y.; Yong, T.-W.; Xin, S.; Wang, X.-C.; Feng, Y.; Jiang, L.; Kai, S.; Liu, W.-G. Maize-soybean strip intercropping: Achieved a balance between high productivity and sustainability. J. Integr. Agric. 2018, 17, 747–754.
- Fan, Y.; Chen, J.; Cheng, Y.; Raza, M.A.; Wu, X.; Wang, Z.; Liu, Q.; Wang, R.; Wang, X.; Yong, T. Effect of shading and light recovery on the growth, leaf structure, and photosynthetic performance of soybean in a maize-soybean relay-strip intercropping system. PLoS ONE 2018, 13, e0198159.
- Ahmed, S.; Raza, M.A.; Zhou, T.; Hussain, S.; Khalid, M.H.B.; Feng, L.; Wasaya, A.; Iqbal, N.; Ahmed, A.; Liu, W. Responses of soybean dry matter production, phosphorus accumulation, and seed yield to sowing time under relay intercropping with maize. Agronomy 2018, 8, 282.
- Feng, L.; Raza, M.A.; Li, Z.; Chen, Y.; Khalid, M.H.B.; Du, J.; Liu, W.; Wu, X.; Song, C.; Yu, L. The influence of light intensity and leaf movement on photosynthesis characteristics and carbon balance of soybean. Front. Plant Sci. 2019, 9, 1952.
- Yang, F.; Liao, D.; Wu, X.; Gao, R.; Fan, Y.; Raza, M.A.; Wang, X.; Yong, T.; Liu, W.; Liu, J. Effect of aboveground and belowground interactions on the intercrop yields in maize-soybean relay intercropping systems. Field Crops Res. 2017, 203, 16–23.
- Raza, M.A.; Feng, L.Y.; van Der Werf, W.; Iqbal, N.; Khalid, M.H.B.; Chen, Y.K.; Wasaya, A.; Ahmed, S.; Din, A.M.U.; Khan, A. Maize leaf-removal: A new agronomic approach to increase dry matter, flower number and seed-yield of soybean in maize soybean relay intercropping system. Sci. Rep. 2019, 9, 1–13.
- Gardiner, B.; Berry, P.; Moulia, B. Wind impacts on plant growth, mechanics and damage. Plant Sci. 2016, 245, 94–118.
- Wassmann, R.; Jagadish, S.; Sumfleth, K.; Pathak, H.; Howell, G.; Ismail, A.; Serraj, R.; Redona, E.; Singh, R.; Heuer, S. Regional vulnerability of climate change impacts on Asian rice production and scope for adaptation. Adv. Agron. 2009, 102, 91–133.
- Liu, W.; Deng, Y.; Hussain, S.; Zou, J.; Yuan, J.; Luo, L.; Yang, C.; Yuan, X.; Yang, W. Relationship between cellulose accumulation and lodging resistance in the stem of relay intercropped soybean [Glycine max (L.) Merr.]. Field Crops Res. 2016, 196, 261–267.
- Yamaguchi, N.; Sayama, T.; Yamazaki, H.; Miyoshi, T.; Ishimoto, M.; Funatsuki, H. Quantitative trait loci associated with lodging tolerance in soybean cultivar ‘Toyoharuka’. Breed. Sci. 2014, 64, 300–308.
- Mizuta, K.; Araki, H.; Takahashi, T. Shifting timing of intensive nitrogen topdressing later to the stem-elongation phase reduced lower internodes length and lodging risk of wheat. Plant Prod. Sci. 2020, 1–9.
- Nagashima, H.; Hikosaka, K. Plants in a crowded stand regulate their height growth so as to maintain similar heights to neighbours even when they have potential advantages in height growth. Ann. Bot. 2011, 108, 207–214.
- Hubbart, S.; Smillie, I.R.; Heatley, M.; Swarup, R.; Foo, C.C.; Zhao, L.; Murchie, E.H. Enhanced thylakoid photoprotection can increase yield and canopy radiation use efficiency in rice. Commun. Biol. 2018, 1, 1–12.
- Chen, X.; Wang, J.; Wang, Z.; Li, W.; Wang, C.; Yan, S.; Li, H.; Zhang, A.; Tang, Z.; Wei, M. Optimized nitrogen fertilizer application mode increased culms lignin accumulation and lodging resistance in culms of winter wheat. Field Crops Res. 2018, 228, 31–38.
- Zheng, M.; Chen, J.; Shi, Y.; Li, Y.; Yin, Y.; Yang, D.; Luo, Y.; Pang, D.; Xu, X.; Li, W. Manipulation of lignin metabolism by plant densities and its relationship with lodging resistance in wheat. Sci. Rep. 2017, 7, 1–12.
- Wang, D.; Graef, G.L.; Procopiuk, A.M.; Diers, B.W. Identification of putative QTL that underlie yield in interspecific soybean backcross populations. Theor. Appl. Genet. 2004, 108, 458–467.
- Hwang, S.; Lee, T.G. Integration of lodging resistance QTL in soybean. Sci. Rep. 2019, 9, 6540.
- Berry, P.; Berry, S. Understanding the genetic control of lodging-associated plant characters in winter wheat (Triticum aestivum L.). Euphytica 2015, 205, 671–689.
- Yadav, S.; Singh, U.M.; Naik, S.M.; Venkateshwarlu, C.; Ramayya, P.J.; Raman, K.A.; Sandhu, N.; Kumar, A. Molecular mapping of QTLs associated with lodging resistance in dry direct-seeded rice (Oryza sativa L.). Front. Plant Sci. 2017, 8, 1431.
- Yano, K.; Ookawa, T.; Aya, K.; Ochiai, Y.; Hirasawa, T.; Ebitani, T.; Takarada, T.; Yano, M.; Yamamoto, T.; Fukuoka, S. Isolation of a novel lodging resistance QTL gene involved in strigolactone signaling and its pyramiding with a QTL gene involved in another mechanism. Mol. Plant 2015, 8, 303–314.
- Liu, F.; Wang, P.; Zhang, X.; Li, X.; Yan, X.; Fu, D.; Wu, G. The genetic and molecular basis of crop height based on a rice model. Planta 2018, 247, 1–26.
- Varshney, R.K.; Bansal, K.C.; Aggarwal, P.K.; Datta, S.K.; Craufurd, P.Q. Agricultural biotechnology for crop improvement in a variable climate: Hope or hype? Trends Plant Sci. 2011, 16, 363–371.
- Wu, W.; Ma, B.L. A new method for assessing plant lodging and the impact of management options on lodging in canola crop production. Sci. Rep. 2016, 6, 31890.
- Cui, F.; Li, J.; Ding, A.; Zhao, C.; Wang, L.; Wang, X.; Li, S.; Bao, Y.; Li, X.; Feng, D. Conditional QTL mapping for plant height with respect to the length of the spike and internode in two mapping populations of wheat. Theor. Appl. Genet. 2011, 122, 1517–1536.
- Hai, L.; Guo, H.; Xiao, S.; Jiang, G.; Zhang, X.; Yan, C.; Xin, Z.; Jia, J. Quantitative trait loci (QTL) of stem strength and related traits in a doubled-haploid population of wheat (Triticum aestivum L.). Euphytica 2005, 141, 1–9.
- Ellis, M.; Rebetzke, G.; Azanza, F.; Richards, R.; Spielmeyer, W. Molecular mapping of gibberellin-responsive dwarfing genes in bread wheat. Theor. Appl. Genet. 2005, 111, 423–430.
- Griffiths, S.; Simmonds, J.; Leverington, M.; Wang, Y.; Fish, L.; Sayers, L.; Alibert, L.; Orford, S.; Wingen, L.; Snape, J. Meta-QTL analysis of the genetic control of crop height in elite European winter wheat germplasm. Mol. Breed. 2012, 29, 159–171.
- Spielmeyer, W.; Hyles, J.; Joaquim, P.; Azanza, F.; Bonnett, D.; Ellis, M.; Moore, C.; Richards, R. A QTL on chromosome 6A in bread wheat (Triticum aestivum) is associated with longer coleoptiles, greater seedling vigour and final plant height. Theor. Appl. Genet. 2007, 115, 59–66.
- Shearman, V.J. Changes in the Yield Limiting Processes Associated with the Genetic Improvement of Wheat; University of Nottingham: Nottingham, UK, 2001.
- Rebetzke, G.; Ellis, M.; Bonnett, D.; Mickelson, B.; Condon, A.; Richards, R. Height reduction and agronomic performance for selected gibberellin-responsive dwarfing genes in bread wheat (Triticum aestivum L.). Field Crops Res. 2012, 126, 87–96.
- Daoura, B.G.; Chen, L.; Du, Y.; Hu, Y.-G. Genetic effects of dwarfing gene Rht-5 on agronomic traits in common wheat (Triticum aestivum L.) and QTL analysis on its linked traits. Field Crops Res. 2014, 156, 22–29.
- Daoura, B.G.; Chen, L.; Hu, Y.-G. Agronomic traits affected by dwarfing gene’Rht-5’in common wheat (‘Triticum aestivum’ L.). Aust. J. Crop Sci. 2013, 7, 1270.
- Chen, L.; Yang, Y.; Cui, C.; Lu, S.; Lu, Q.; Du, Y.; Su, R.; Chai, Y.; Li, H.; Chen, F. Effects of Vrn-B1 and Ppd-D1 on developmental and agronomic traits in Rht5 dwarf plants of bread wheat. Field Crops Res. 2018, 219, 24–32.
- Huang, C.; Liu, Q.; Li, H.; Li, X.; Zhang, C.; Zhang, F. Optimised sowing date enhances crop resilience towards size-asymmetric competition and reduces the yield difference between intercropped and sole maize. Field Crops Res. 2018, 217, 125–133.
- Egbe, O. Effects of plant density of intercropped soybean with tall sorghum on competitive ability of soybean and economic yield at Otobi, Benue State, Nigeria. J. Cereals Oilseeds 2010, 1, 1–10.
- Zhou, T.; Du, Y.; Ahmed, S.; Liu, T.; Ren, M.; Liu, W.; Yang, W. Genotypic differences in phosphorus efficiency and the performance of physiological characteristics in response to low phosphorus stress of soybean in southwest of China. Front. Plant Sci. 2016, 7, 1776.
- Dai, X.; Wang, Y.; Dong, X.; Qian, T.; Yin, L.; Dong, S.; Chu, J.; He, M. Delayed sowing can increase lodging resistance while maintaining grain yield and nitrogen use efficiency in winter wheat. Crop J. 2017, 5, 541–552.
- Spink, J.; Semere, T.; Sparkes, D.; Whaley, J.; Foulkes, M.; Clare, R.; Scott, R. Effect of sowing date on the optimum plant density of winter wheat. Ann. Appl. Biol. 2000, 137, 179–188.
- Ahmed, S.; Raza, M.A.; Yuan, X.; Du, Y.; Iqbal, N.; Chachar, Q.; Soomro, A.A.; Ibrahim, F.; Hussain, S.; Wang, X.; et al. Optimized planting time and co-growth duration reduce the yield difference between intercropped and sole soybean by enhancing soybean resilience toward size-asymmetric competition. Food Energy Secur. 2020, 9.
- Yu, Y.; Stomph, T.-J.; Makowski, D.; van der Werf, W. Temporal niche differentiation increases the land equivalent ratio of annual intercrops: A meta-analysis. Field Crops Res. 2015, 184, 133–144.
- Zhang, F.; Li, L. Using competitive and facilitative interactions in intercropping systems enhances crop productivity and nutrient-use efficiency. Plant Soil 2003, 248, 305–312.
- Raza, M.A.; Feng, L.Y.; Iqbal, N.; Ahmed, M.; Chen, Y.K.; Khalid, M.H.B.; Din, A.M.U.; Khan, A.; Ijaz, W.; Hussain, A. Growth and development of soybean under changing light environments in relay intercropping system. PeerJ 2019, 7, e7262.
- Song, D.; Tariq, A.; Pan, K.; Khan, S.U.; Saleh, T.A.; Gong, S.; Zhang, A.; Wu, X. Influence of planting distance and density on the yield and photosynthetic traits of sweet potato (Ipomoea balatas L.) under an intercropping system with walnut (Juglans regia) saplings. Soil Tillage Res. 2020, 196, 104484.
- Wang, C.; Wu Ruan, R.; Hui Yuan, X.; Hu, D.; Yang, H.; Li, Y.; Lin Yi, Z. Effects of nitrogen fertilizer and planting density on the lignin synthesis in the culm in relation to lodging resistance of buckwheat. Plant Prod. Sci. 2015, 18, 218–227.
- Sher, A.; Khan, A.; Ashraf, U.; Liu, H.H.; Li, J.C. Characterization of the effect of increased plant density on canopy morphology and stalk lodging risk. Front. Plant Sci. 2018, 9, 1047.
- Fan, Y.; Wang, Z.; Liao, D.; Raza, M.A.; Wang, B.; Zhang, J.; Chen, J.; Feng, L.; Wu, X.; Liu, C. Uptake and utilization of nitrogen, phosphorus and potassium as related to yield advantage in maize-soybean intercropping under different row configurations. Sci. Rep. 2020, 10, 1–10.
- Xiang, D.B.; Zhao, G.; Wan, Y.; Tan, M.L.; Song, C.; Song, Y. Effect of planting density on lodging-related morphology, lodging rate, and yield of tartary buckwheat (Fagopyrum tataricum). Plant Prod. Sci. 2016, 19, 479–488.
- Khan, S.; Anwar, S.; Kuai, J.; Noman, A.; Shahid, M.; Din, M.; Ali, A.; Zhou, G. Alteration in yield and oil quality traits of winter rapeseed by lodging at different planting density and nitrogen rates. Sci. Rep. 2018, 8, 1–12.
- Cheng, B.; Raza, A.; Wang, L.; Xu, M.; Lu, J.; Gao, Y.; Qin, S.; Zhang, Y.; Ahmad, I.; Zhou, T.; et al. Effects of Multiple Planting Densities on Lignin Metabolism and Lodging Resistance of the Strip Intercropped Soybean Stem. Agronomy 2020, 10, 1177.
- Zaman, U.; Ahmad, Z.; Farooq, M.; Saeed, S.; Ahmad, M.; Wakeel, A. Potassium fertilization may improve stem strength and yield of Basmati rice grown on nitrogen-fertilized soils. Pak. J. Agric. Sci. 2015, 52, 437–443.
- Zhang, M.; Wang, H.; Yi, Y.; Ding, J.; Zhu, M.; Li, C.; Guo, W.; Feng, C.; Zhu, X. Effect of nitrogen levels and nitrogen ratios on lodging resistance and yield potential of winter wheat (Triticum aestivum L.). PLoS ONE 2017, 12, e0187543.
- Mariani, L.; Ferrante, A. Agronomic management for enhancing plant tolerance to abiotic stresses—Drought, salinity, hypoxia, and lodging. Horticulturae 2017, 3, 52.
- Kong, L.; Sun, M.; Wang, F.; Liu, J.; Feng, B.; Si, J.; Zhang, B.; Li, S.; Li, H. Effects of high NH+ 4 on K+ uptake, culm mechanical strength and grain filling in wheat. Front. Plant Sci. 2014, 5, 703.
- Pan, J.; Zhao, J.; Liu, Y.; Huang, N.; Tian, K.; Shah, F.; Liang, K.; Zhong, X.; Liu, B. Optimized nitrogen management enhances lodging resistance of rice and its morpho-anatomical, mechanical, and molecular mechanisms. Sci. Rep. 2019, 9, 1–13.
- Wei, F.-Z.; Li, J.-C.; Wang, C.-Y.; Qu, H.-J.; Shen, X.-S. Effects of nitrogenous fertilizer application model on culm lodging resistance in winter wheat. Acta Agron. Sin. 2008, 34, 1080–1085.
- Berry, P.; Griffin, J.; Sylvester-Bradley, R.; Scott, R.; Spink, J.; Baker, C.; Clare, R. Controlling plant form through husbandry to minimise lodging in wheat. Field Crops Res. 2000, 67, 59–81.
- Peake, A.; Bell, K.; Carberry, P.; Poole, N.; Raine, S. Vegetative nitrogen stress decreases lodging risk and increases yield of irrigated spring wheat in the subtropics. Crop Pasture Sci. 2016, 67, 907–920.
- Yousaf, M.; Li, J.; Lu, J.; Ren, T.; Cong, R.; Fahad, S.; Li, X. Effects of fertilization on crop production and nutrient-supplying capacity under rice-oilseed rape rotation system. Sci. Rep. 2017, 7, 1–9.
- Bhiah, K.; Guppy, C.; Lockwood, P.; Jessop, R. Effect of potassium on rice lodging under high nitrogen nutrition. In Proceedings of the Brisbane (AU): 19th World Congress of Soil Science “Soil Solution for a Changing World, Brisbane, Australia, 1–6 August 2010; pp. 1–6.
- Kong, E.; Liu, D.; Guo, X.; Yang, W.; Sun, J.; Li, X.; Zhan, K.; Cui, D.; Lin, J.; Zhang, A. Anatomical and chemical characteristics associated with lodging resistance in wheat. Crop J. 2013, 1, 43–49.
- Wang, M.; Zheng, Q.; Shen, Q.; Guo, S. The critical role of potassium in plant stress response. Int. J. Mol. Sci. 2013, 14, 7370–7390.
- Hafsi, C.; Debez, A.; Abdelly, C. Potassium deficiency in plants: Effects and signaling cascades. Acta Physiol. Plant. 2014, 36, 1055–1070.
- Luo, Y.; Ni, J.; Pang, D.; Jin, M.; Chen, J.; Kong, X.; Li, W.; Chang, Y.; Li, Y.; Wang, Z. Regulation of lignin composition by nitrogen rate and density and its relationship with stem mechanical strength of wheat. Field Crops Res. 2019, 241, 107572.
- Pitre, F.E.; Cooke, J.E.; Mackay, J.J. Short-term effects of nitrogen availability on wood formation and fibre properties in hybrid poplar. Trees 2007, 21, 249–259.
- Yong, T.; Liu, X.; Yang, F.; Song, C.; Wang, X.; Liu, W.; Su, B.; Zhou, L.; Yang, W. Characteristics of nitrogen uptake, use and transfer in a wheat-maize-soybean relay intercropping system. Plant Prod. Sci. 2015, 18, 388–397.
- Li, L.; Sun, J.; Zhang, F.; Li, X.; Yang, S.; Rengel, Z. Wheat/maize or wheat/soybean strip intercropping: I. Yield advantage and interspecific interactions on nutrients. Field Crops Res. 2001, 71, 123–137.
- Peoples, M.; Brockwell, J.; Herridge, D.; Rochester, I.; Alves, B.; Urquiaga, S.; Boddey, R.; Dakora, F.; Bhattarai, S.; Maskey, S. The contributions of nitrogen-fixing crop legumes to the productivity of agricultural systems. Symbiosis 2009, 48, 1–17.
- Yang, F.; Huang, S.; Gao, R.; Liu, W.; Yong, T.; Wang, X.; Wu, X.; Yang, W. Growth of soybean seedlings in relay strip intercropping systems in relation to light quantity and red: Far-red ratio. Field Crops Res. 2014, 155, 245–253.
- Yu, Y.; Makowski, D.; Stomph, T.J.; van der Werf, W. Robust Increases of Land Equivalent Ratio with Temporal Niche Differentiation: A Meta-Quantile Regression. Agron. J. 2016, 108, 2269–2279.
- Yu, Y.; Stomph, T.-J.; Makowski, D.; Zhang, L.; van der Werf, W. A meta-analysis of relative crop yields in cereal/legume mixtures suggests options for management. Field Crops Res. 2016, 198, 269–279.
- Xu, Z.; Li, C.; Zhang, C.; Yu, Y.; van der Werf, W.; Zhang, F. Intercropping maize and soybean increases efficiency of land and fertilizer nitrogen use; A meta-analysis. Field Crops Res. 2020, 246, 107661.
- Upadhya, D.; Dhakal, R.; Khadka, K.; Rana, S.; Acharya, P.; Rana, R.; Chaudhary, P. Local knowledge on climate-induced traits in rice for improving crop yield, food security and climate resilience. Int. Agric. Innov. Res. J. 2016, 5, 385–396.
- Hussain, S.; Liu, T.; Iqbal, N.; Brestic, M.; Pang, T.; Mumtaz, M.; Shafiq, I.; Li, S.; Wang, L.; Gao, Y.; et al. Effects of lignin, cellulose, hemicellulose, sucrose and monosaccharide carbohydrates on soybean physical stem strength and yield in intercropping. Photochem. Photobiol. Sci. 2020, 19, 462–472.
- Waseem, M.; Ahmad, R.; Randhawa, M.A.; Aziz, T.; Mahmood, N. Influence of silicon application on blast incidence and lodging resistance in rice. J. Agric. Res. 2016, 54, 435–443.
- Fallah, A. Studies effect of silicon on lodging parameters in rice plant under hydroponics culture in a greenhouse experiment. In Proceedings of the Silicon in Agriculture Conference, Wild Coast Sun, South Africa, 26–31 October 2008.
- Salman, D.; Morteza, S.; Dariush, Z.; Abbas, G.M.; Reza, Y.; Ehsan, G.D.; Reza, N.A. Application of nitrogen and silicon rates on morphological and chemical lodging related characteristics in rice (Oryza sativa L.) at North of Iran. J. Agric. Sci. 2012, 4, 12.
- Kim, S.G.; Kim, K.W.; Park, E.W.; Choi, D. Silicon-induced cell wall fortification of rice leaves: A possible cellular mechanism of enhanced host resistance to blast. Phytopathology 2002, 92, 1095–1103.
- Zhang, J.; Zou, W.; Li, Y.; Feng, Y.; Zhang, H.; Wu, Z.; Tu, Y.; Wang, Y.; Cai, X.; Peng, L. Silica distinctively affects cell wall features and lignocellulosic saccharification with large enhancement on biomass production in rice. Plant Sci. 2015, 239, 84–91.
- Yu, J.-K.; Graznak, E.; Breseghello, F.; Tefera, H.; Sorrells, M.E. QTL mapping of agronomic traits in tef [Eragrostis tef (Zucc) Trotter]. BMC Plant Biol. 2007, 7, 30.
- He, C.; Wang, L.; Liu, J.; Liu, X.; Li, X.; Ma, J.; Lin, Y.; Xu, F. Evidence for ‘silicon’within the cell walls of suspension-cultured rice cells. New Phytol. 2013, 200, 700–709.
- Hussain, S.; Shuxian, L.; Mumtaz, M.; Shafiq, I.; Iqbal, N.; Brestic, M.; Shoaib, M.; Sisi, Q.; Li, W.; Mei, X.; et al. Foliar application of silicon improves stem strength under low light stress by regulating lignin biosynthesis genes in soybean (Glycine max (L.) Merr.). J. Hazard. Mater. 2020, 401, 123256.
- Yingang, L.; Jun, M.; Ying, T.; Junyu, H.; Christie, P.; Lingjia, Z.; Wenjie, R.; Zhang, M.; Shiping, D. Effect of silicon on growth, physiology, and cadmium translocation of tobacco (Nicotiana tabacum L.) in cadmium-contaminated soil. Pedosphere 2018, 28, 680–689.
- Wang, Y.; Zhang, B.; Jiang, D.; Chen, G. Silicon improves photosynthetic performance by optimizing thylakoid membrane protein components in rice under drought stress. Environ. Exp. Bot. 2019, 158, 117–124.
- Rehman, B. Silicon elicited varied physiological and biochemical responses in Indian mustard (Brassica juncea): A concentration dependent study. Israel J. Plant Sci. 2016, 63, 158–166.
- Hernandez-Apaolaza, L. Can silicon partially alleviate micronutrient deficiency in plants? A review. Planta 2014, 240, 447–458.
- Dorairaj, D.; Ismail, M.R.; Sinniah, U.R.; Kar Ban, T. Influence of silicon on growth, yield, and lodging resistance of MR219, a lowland rice of Malaysia. J. Plant Nutr. 2017, 40, 1111–1124.
- Hussain, S.; Iqbal, N.; Brestic, M.; Raza, M.A.; Pang, T.; Langham, D.R.; Safdar, M.E.; Ahmed, S.; Wen, B.; Gao, Y.; et al. Changes in morphology, chlorophyll fluorescence performance and Rubisco activity of soybean in response to foliar application of ionic titanium under normal light and shade environment. Sci. Total Environ. 2019, 658, 626–637.
- Xu, C.; Gao, Y.; Tian, B.; Ren, J.; Meng, Q.; Wang, P. Effects of EDAH, a novel plant growth regulator, on mechanical strength, stalk vascular bundles and grain yield of summer maize at high densities. Field Crops Res. 2017, 200, 71–79.
- Berry, P.; Sterling, M.; Spink, J.; Baker, C.; Sylvester-Bradley, R.; Mooney, S.; Tams, A.; Ennos, A. Understanding and reducing lodging in cereals. Adv. Agron. 2004, 84, 215–269.
- Rajala, A. Plant Growth Regulators to Manipulate Cereal Growth in Northern Growing Conditions. 2003. Available online: https://helda.helsinki.fi/bitstream/handle/10138/20698/plantgro.pdf?sequence=1 (accessed on 2 June 2012).
- Bhaskaruni, S.V.; Maddila, S.; Gangu, K.K.; Jonnalagadda, S.B. A Review on multi-component green synthesis of N-containing heterocycles using mixed oxides as heterogeneous catalysts. Arabian J. Chem. 2020, 13, 1142–1178.
- Kamran, M.; Ahmad, I.; Wu, X.; Liu, T.; Ding, R.; Han, Q. Application of paclobutrazol: A strategy for inducing lodging resistance of wheat through mediation of plant height, stem physical strength, and lignin biosynthesis. Environ. Sci. Pollut. Res. 2018, 25, 29366–29378.
- Okuno, A.; Hirano, K.; Asano, K.; Takase, W.; Masuda, R.; Morinaka, Y.; Ueguchi-Tanaka, M.; Kitano, H.; Matsuoka, M. New approach to increasing rice lodging resistance and biomass yield through the use of high gibberellin producing varieties. PLoS ONE 2014, 9, e86870.
- Shah, L.; Yahya, M.; Shah, S.M.A.; Nadeem, M.; Ali, A.; Ali, A.; Wang, J.; Riaz, M.W.; Rehman, S.; Wu, W. Improving lodging resistance: Using wheat and rice as classical examples. Int. J. Mol. Sci. 2019, 20, 4211.
- Leśniowska-Nowak, J.; Nowak, M.; Zapalska, M.; Dudziak, K.; Kowalczyk, K. Influence of CCC and trinexapac-ethyl on the expression of genes involved in gibberellic biosynthesis and metabolism pathway in isogenic line with Rht12 dwarfing gene. Acta Sci. Polonorum. Hortorum Cultus 2017, 16, 141–151.
- Espindula, M.; Rocha, V.; Grossi, J.; Souza, M.; Souza, L.; Favarato, L. Use of growth retardants in wheat. Planta Daninha 2009, 27, 379–387.




