Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Anita Klaus | -- | 3173 | 2022-11-08 11:19:01 | | | |
| 2 | Beatrix Zheng | Meta information modification | 3173 | 2022-11-09 03:08:37 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Ahmad, N.; Vunduk, J.; Klaus, A.; Dahlan, N.Y.; Ghosh, S.; Muhammad-Sukki, F.; Dufossé, L.; Bani, N.A.; Wan-Mohtar, W.A.A.Q.I. Medicinal Mushrooms as Natural Food Dyes. Encyclopedia. Available online: https://encyclopedia.pub/entry/33521 (accessed on 23 May 2026).
Ahmad N, Vunduk J, Klaus A, Dahlan NY, Ghosh S, Muhammad-Sukki F, et al. Medicinal Mushrooms as Natural Food Dyes. Encyclopedia. Available at: https://encyclopedia.pub/entry/33521. Accessed May 23, 2026.
Ahmad, Nurfadzilah, Jovana Vunduk, Anita Klaus, Nofri Yenita Dahlan, Soumya Ghosh, Firdaus Muhammad-Sukki, Laurent Dufossé, Nurul Aini Bani, Wan Abd Al Qadr Imad Wan-Mohtar. "Medicinal Mushrooms as Natural Food Dyes" Encyclopedia, https://encyclopedia.pub/entry/33521 (accessed May 23, 2026).
Ahmad, N., Vunduk, J., Klaus, A., Dahlan, N.Y., Ghosh, S., Muhammad-Sukki, F., Dufossé, L., Bani, N.A., & Wan-Mohtar, W.A.A.Q.I. (2022, November 08). Medicinal Mushrooms as Natural Food Dyes. In Encyclopedia. https://encyclopedia.pub/entry/33521
Ahmad, Nurfadzilah, et al. "Medicinal Mushrooms as Natural Food Dyes." Encyclopedia. Web. 08 November, 2022.
Copy Citation
Dyes are widely used in high-tech sectors, including food and energy. Natural dyes are more environment-friendly than synthetic dyes and may have medicinal benefits. Fungi are a natural source of dye that can be substituted for plants. For example, medicinal mushrooms offer a wide range of safe organic dyes that may be produced instantly, inexpensively, and in large quantities. Meanwhile, medicinal mushroom dyes may provide a less expensive choice for photovoltaic (PV) technology due to their non-toxic and environmentally friendly qualities.
medicinal mushroom
fungal dyes
food colourants
1. Medicinal Mushroom Dyes
Dyes have always been essential in various fields of human activity, dating back to prehistoric times [1]. In addition to pigments extracted from numerous plants, mushrooms were also a common material for dyeing fabrics due to the wide range of vivid colours [2]. However, during the development of human society, the need for pigments grew steadily, and their purpose was very diverse. Dyes obtained from natural sources were no longer sufficient to meet all needs, which in addition to costly extraction and instability led to the development of synthetic dyes during the 1800s. Nevertheless, modern society is trying to return to natural sources of pigments, which is of particular interest in the food industry, as synthetic dyes lead to many harmful health effects, including the carcinogenic [3].
Mushrooms are a vibrant but still insufficiently researched natural source of dyes. Their pigmentation can vary depending on age, and some species are subject to discoloration when the tissue is damaged. Chromophores contain valuable organic compounds that play multiple roles in the life of mushroom, such as protection from harmful UV radiation, antimicrobial action, and attracting insects to spread their spores [4]. Melanin is a pigment that protects us from the effects of the environment [5]. Flavins as cofactors of enzymes [1] or carotenoids necessary to prevent the harmful effects of photooxidation [6] are just some of the pigments that directly affect a number of biological activities.
The pigments present in mushrooms differ from those that are dominant in plants, hence they do not contain any chlorophylls and anthocyanins. In contrast, some species contain betalains and terpenoids, among which carotenoids are common [4]. According to Lin and Xu [7], four main pathways of biosynthesis of the most important mushroom pigments have been described to date:
-
Polyketide synthetic pathway; Antrodia cinnamomea [8]—allowing the formation of melanin, flavins, ankaflavin, quinones, and azaphilones. Aromatic ketides or fatty acids are formed via this pathway. Cyclisation reactions and partial reductions lead to stabilisation of the growing chain in the synthesis of aromatic ketides. In the case of fatty acid synthesis, the carbonyl groups of the chain are reduced prior to binding to the next C2 group. This process produces tetra-, hepta-, octa-, and higher number of aromatic ketides as well as fatty acid-derived compounds;
-
Shikimate pathway; Agaricus bisporus [9]—the key intermediates of shikimic and chorismic acids are used to manufacture the important amino acids tyrosine, tryptophan, and phenylalanine. Various important building components of many colours found in fungi, such as benzoic, arylpyruvic, and cinnamic acids, are produced from tyrosine and phenylalanine precursors. Tyrosine is crucial in the phylum Basidiomycota because it is a precursor of betalaine, a pigment found solely in the genera Hygrocybe and Amanita;
-
Terpenoid synthetic pathway; Hypsizygus marmoreus [10]—significant for the formation of carotenoids belonging to terpenoids. The condensation routes of C5 isoprene units and terpenoid synthesis are triggered during carotenoids production. A common precursor to terpenoid formation is isopentenyl pyrophosphate, which is formed by mevalonate pathway;
-
Nitrogen-containing metabolite pathway; Agaricus bisporus [11]—ensure the formation of various fungal pigments.
The Agrocybe cylindracea fruiting body contains two indole pigments with pronounced free radical scavenging potential as well as distinct inhibitory activity on lipid peroxidation. Chalciporus piperatus produces chalciporone, a type of 2H-azepine alkaloid pigment, which has a protective role against insects and other predators. Pycnoporus cinnabarinus is a rich source of cinnabarinic acid, a red pigment that acts antibacterially against the genus Streptococcus and is derived from precursors 3-hydroxyanthranilic acid that undergo oxidative dimerisation.
In recent decades, the biological properties of therapeutic mushrooms have been widely studied [12][13]. Although the specific biological effects of pigments remain understudied, research confirms the potential use of these important compounds in formulating health-promoting drugs that could be used to treat many disorders, such as cardiovascular disease, Alzheimer’s disease, and infectious diseases [7]. Various pigments derived from some medicinal mushrooms are presented in Table 1.
Table 1. Pigment-producing medicinal mushrooms and their respective pigments.
| Mushroom Species | Family | Key Uses | Bioactive Pigment | Ref. |
|---|---|---|---|---|
| Agaricus bisporus | Agaricaceae | Bruising-tolerant | γ-glutaminyl-4-hydroxybenzene | [14] |
| Agrocybe cylindracea | Strophariaceae | Immunity Immunity |
6-hydroxy-1H-indole-3-carboxaldehyde, 6-hydroxy-1H-indole-3-acetamide |
[7] |
| Albatrellus confluens | Albatrellaceae | Anti-cancer | albatrellin | [15] |
| Albatrellus flettii | Albatrellaceae | Anti-cancer | grifolin, neogrifolin, confluentin | [16] |
| Auricularia auricula | Auriculariaceae | Antibiofilm Antioxidant |
melanin pheomelanin |
[17] [18] |
| Boletus pseudocalopus | Boletaceae | Anti-cancer | grifolin derivatives 1–3 | [19] |
| Cantharelluus cibarius | Cantharellaceae | Antihypertensive | lycopene and β-carotene | [20] |
| Cerioporus squamosus | Polyporaceae | Blood regulation | melanin | [21] |
| Chalciporus piperatus | Boletaceae | Bio deterrent | chalciporone | [7] |
| Cordyceps farinosa | Cordycipitaceae | Protecion | anthraquinone derivative | [22] |
| Fomes fomentarius | Polyporaceae | Anti-tumour | melanin | [21] |
| Inonotus hispidus | Hymenochaetaceae | Antiviral | melanin | [21] |
| Inonotus obliquus | Hymenochaetaceae | Antioxidant | melanin, inotodiol | [23] |
| Lactarius spp. | Russulaceae | Antimicrobial | azulenes | [24] |
| Lactarius subvellereus | Russulaceae | Anti-cancer | subvellerolactone B, subvellerolactone D, subvellerolactone E, |
[25] |
| Laetiporus sulphureus | Fomitopsidaceae | Antioxidant | laetiporic acid A, 2-dehydro-3-deoxylaetiporic acid A, laetiporic acids B, laetiporic acids C |
[26] |
| Lentinus brumalis | Polyporaceae | Not available | melanin | [21] |
| Ophiocordyceps unilateralis | Ophiocordycipitaceae | Antimalarial | Erythrostominone, 3,5,8–TMON, deoxyerythrostominone, deoxyerythrostominol, 4–O–methyl, erythrostominone, epierythrostominol, naphthoquinones |
[24] |
| Pleurotus citrinopileatus | Pleurotaceae | Antioxidant | eumelanin, phaeomelanin | [27] |
| Pleurotus cornucopiae | Pleurotaceae | Antioxidant | eumelanin, phaeomelanin | [27] |
| Pleurotuis djamor | Pleurotaceae | Antioxidant | eumelanin, phaeomelanin | [27] |
| Pycnoporus cinnabarinus | Polyporaceae | Antibacterial | cinnabarinic acid | [7] |
| Termitomyces albuminosus | Agaricaceae | Antioxidant | melanin | [28] |
| Trametes versicolor | Polyporaceae | Breast cancer | melanin | [21] |
| Tuber melanosporum | Tuberaceae | Anti-fatigue | melanin | [29] |
Carotenoids are a diverse set of pigments found throughout nature; not only are they a crucial component of plants that aid in photosynthesis, but their presence has also been proven in over 200 species of fungi [30]. Fungi are hypothesised to be protected by these pigments from UV radiation and oxidative damage. Carotenoids have an aliphatic polyene chain, which is made up of eight isoprene units and contains conjugated double bonds that absorb light, giving them their characteristic yellow, orange or red colour [31]. They benefit human health by acting as precursors to vitamin A as well as slowing down and preventing aging-related changes, such as retinal degeneration and cataracts. They also lower the risk of coronary heart disease and certain types of tumours. Their ability to scavenge free radicals and act as antioxidants has positive impacts on health [7]. The presence of two carotenoid pigments in significant amounts, lycopene and β-carotene, as well as their contribution to the antioxidant potential of the methanolic mushroom extract Cantharelluus cibarius (which has a distinct yellow-orange colour) was confirmed by Kozarski et al. [20].
Melanins are also very common pigments in fungi [32]. These are in fact polyphenolic and/or polyindole heterogeneous polymers that usually give a brown, black or dark green colour. The black extremophilous fungus Cryomyces antarcticus, which was discovered on the outside walls of the International Space Station and was initially isolated from the Antarctic desert, is an excellent example. The wide distribution of melanised fungi indicates that pigmentation allows them to survive in very hostile conditions prevailing in cold and hot deserts as well as in areas contaminated with ionising radiation and toxic metals [33]. As excellent scavengers of reactive nitrogen species as well as reactive oxygen species, melanins protect cells from the damaging effects of free radicals [7]. The pathway of melanin synthesis in mushrooms involves the conversion of benzoquinone from a γ-glutaminyl-4-hydroxybenzene (GHB) precursor by tyrosinase. Weijn et al. [14] discovered the presence of GHB as the most abundant phenolic component in the fruiting body and spores of Agaricus bisporus. One of the highly melanised medicinal mushrooms, Inonotus obliquus, also known as Chaga, contains bioactive phenolic components, such as melanin as well as triterpenoids (e.g., inotodiol) [34]. As Lin and Xu [7] stated, research to date indicate a significant contribution of these pigments to health improvement, among other things due to their hypoglycemic ability and antiproliferative effect on some cancer cell lines. The high content of melanin has also been confirmed in the medicinal mushroom Auricularia auricula, which has a proven potential in the treatment of various health disorders. This pigment may affect the antioxidant capacity as well as inhibit quorum sensing, causing the prevention of biofilm formation [18]. The presence of several pigments in one genus has also been proven, and the final colour of the mushroom depends on the quantities and relative ratios of individual pigments. The colours of the caps Pleurotus citrinopileatus, P. cornucopiae, and P. djamor are yellow, gray, and pink, respectively. In all three species, the confirmed presence of melanin consist of eumelanin and pheomelanin, the interrelationships of which result in a corresponding colour [27].
There are many other types of pigments that are an integral part of medicinal mushrooms and affect their beneficial effects on health. Due to the abundance and variety of pigments, medicinal mushrooms are also potentially important sources of food colourants. Mushroom pigments could provide visual appeal to food and contribute to improving health due to their beneficial effect on many body functions.
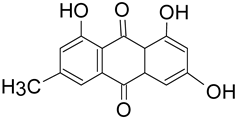
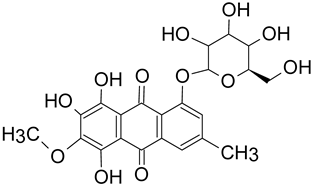
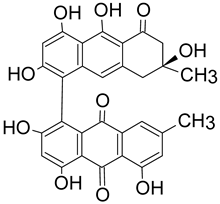
There are key chemical structures found in pigment producing medicinal mushrooms, such as azulenes from Lactarius indigo blue mushroom (Figure 1a,b) as well as other common Lactarius deliciosious and Ganoderma theaecolum. These azulene derivates are identified as guaiane sesquiterpenes, such as 7-(1-hydroxyethyl)-4-methyl-1-azulenecarboxaldehyde, 7-isopropenyl-4-methyl-1-azulenecarboxylic acid, 4-methyl-7-(1-methylethenyl)azulene-1-carbaldehyde, and 7-(1-hydroxy-1-methylethyl)-4-methylazulene-1-carbaldehyde. Meanwhile, anthraquinonens (Figure 1c,d) are found in the red coloured Cortinarius sanguineus [35], such as Emodin, dermocybin-1-β-D-glucopyranoside, Icterinoidin B1, Austrocolorin A1, and Hypericin (Table 2).
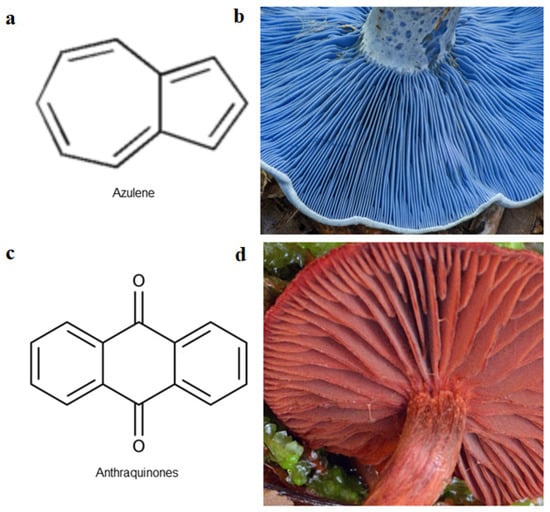
Figure 1. Chemical structures found in pigment-producing medicinal mushrooms. (a) chemical structure of azulene; (b) Lactarius indigo containing azulenes; (c) chemical skeleton of anthraquinones; (d) Cortinarius sanguineus containing anthraquinone dye derivatives.
Table 2. Anthraquinone dyes derivatives from the common Cortinarius sanguineus.
| Compounds Name | Molecular Weight [g/mol] and Formula | Chemical Structure | Species |
|---|---|---|---|
| Emodin | 270.0528 C15O5H10 |
 |
Cortinarius sanguineus, C. semisanguineus |
| Dermocybin-1-β-D-glucopyranoside | 478.1111 C22O12H22 |
 |
C. vitiosus, C. sanguineus, C. semisanguineus |
| Icterinoidin B1 | 528.1056 C29H20O10 |
 |
C. icterinoides |
| Austrocolorin A1 | 602.2152 C34H34O10 |
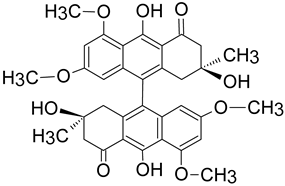 |
Dermocybe sp. WAT 26641 |
| Hypericin | 504.0845 C30H16O8 |
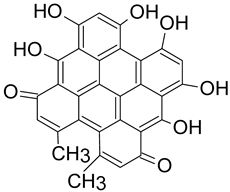 |
C. austrovenetus |
Melanine derivatives are commonly found in Xylaria polymorpha (Figure 2a) and Auricularia auricula (Figure 2b), such as GHB-melanin (green), Eumelanins (pink), Pyomelanins (red), and Allomelanins (blue). The antecedents for the four types of fungal melanin are simplified in Figure 2. GHB-melanins are produced in the body by a sequence of events involving tyrosinase. The L-Dopa pathway, which uses tyrosine as a precursor, produces eumelanins. Pyomelanins, such as tyrosine, have a tyrosine precursor but are eventually made from HGA. 1,8-DHN is the source of allomelanins [36].
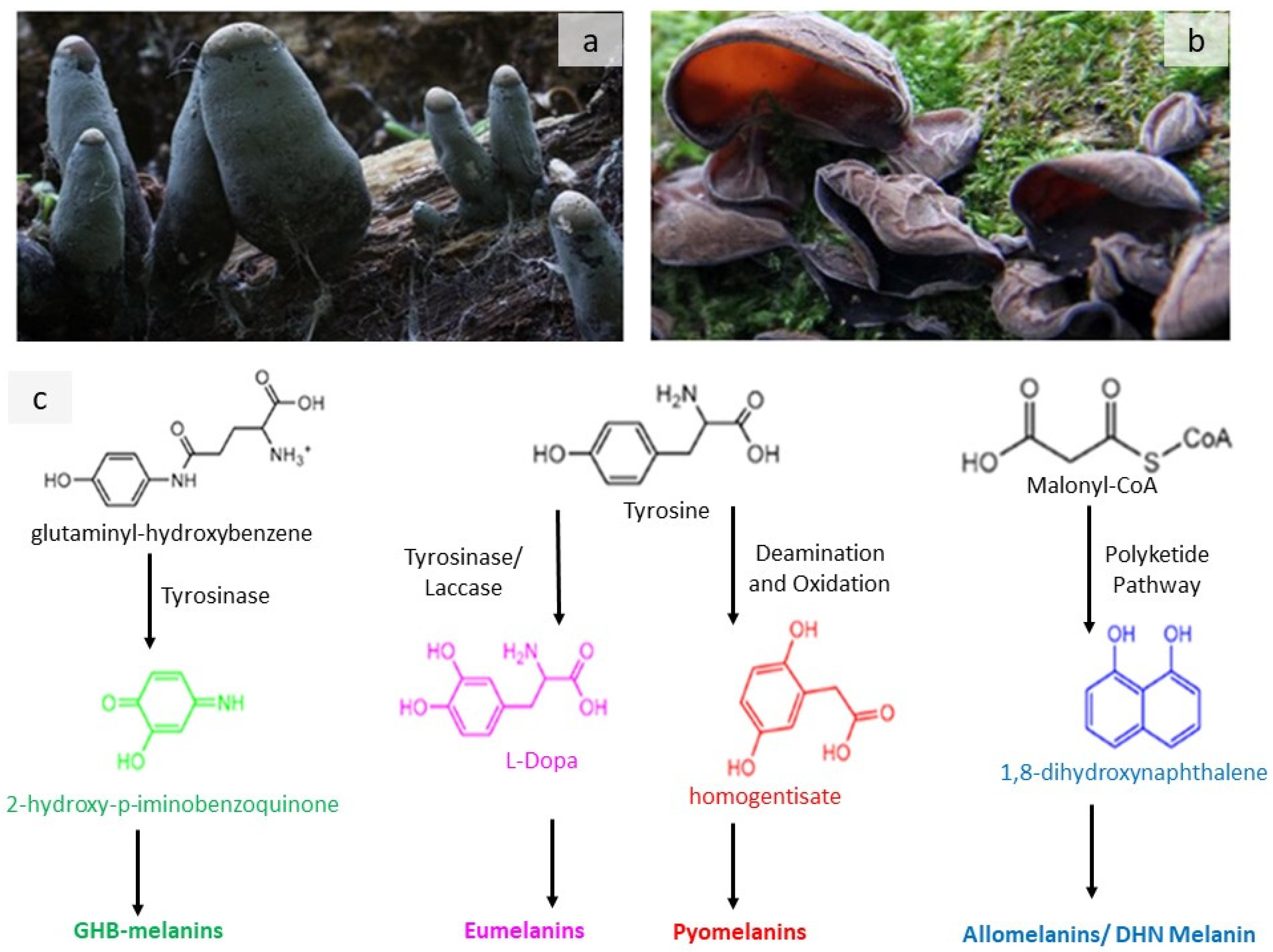
Figure 2. Simplified depiction of the four basic types of fungal melanin precursors. (a) Xylaria polymorpha containing melanins; (b) Auricularia auricula containing melanins; (c) Biosynthesis scheme. The figure is improvised based on a previous work [36].
2. Medicinal Mushroom Pigments in Food Production
Apart from being the raw material for nutraceuticals, medicinal mushrooms are increasingly explored as functional foods, whether alone or as an additive [37]. Because of its antioxidant, anti-inflammatory, anti-obesity, hepatoprotective, antibacterial, and antidiabetic action, mushroom components are believed to provide health advantages [12][38]. However, introducing any new compound into the already established food product can affect its physicochemical and sensorial characteristics, such as cohesiveness, chewiness, volume, texture, smell, taste, aroma, and colour. Each has its significance, which contributes to the overall acceptancy, and colour is among the most important [39]. Thus, studies dealing with novel food products with mushrooms have also examined their influence on colour. Some of the several categories of foods in which mushrooms have been used as functional ingredients and for the colour are summarised in Figure 3.
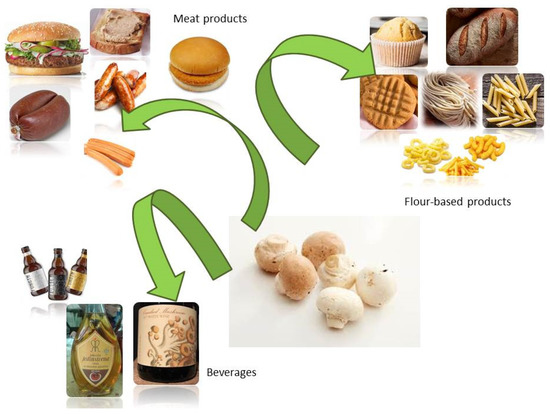
Figure 3. Mushroom-associated food products.
Bread and pasta are staple and popular foods worldwide, hence it is no wonder that much of the contemporary research effort is going into this sector. Mushrooms, mainly the commercial species, such as white button, oyster, black ear mushroom, and shiitake, were added as a supplement in bread making in concentrations ranging from 2.5% to 20%. The addition of mushroom flour (dried and powdered fruit bodies or mycelium) caused a decrease in crumb and crust lightness, which was attributed to the flour colour and Maillard reaction as well as caramelisation. These loaves of bread were generally less acceptable among panelists, and the recommendation was to use up to 5% of mushroom powder in white bread recipes. However, in a study by Sulieman et al. [40], the darker crumb of mushroom supplemented bread was marked as positive since the new formulation was compared with the gluten-free type of bread, which is usually lighter. Ulzijargal et al. [41] experimented with some less common mushroom species as additives in bread production, such as Anthrodia camphorata, Hericium erinaceus, and Phellinus linteus. They concluded that colour changes in final products were the result of pigments present in mushroom mycelium as well as oxidation and caramelisation during thermal treatment. In the case of noodles, the addition of mushroom powder caused a decrease in lightness and yellowness, while the redness factor (obtained by instrumental colour assessment) increased. Ganoderma lucidum caused the most significant colour change in noodles [42]. Colour was not the most significant sensory attribute in the case of noodles, but the addition of mushroom powder affected other characteristics, such as texture and mouthfeel, hence the adequate concentration of mushroom supplement should not exceed 5% [43]. The same was concluded in the case of muffins, sponge cake and cookies enriched with mushrooms. The introduction of this component affected the decrease in product’s lightness [44][45][46].
Meat products are the second large group of foods in which mushrooms have been used as a source of flavour or preservative, especially due to their antioxidant properties [20]. Gencelep [47] explored the effect of Agaricus bisporus powder on colour properties of a Turkish traditional meat product, sucuk. All instrumental colour parameters (lightness, redness, and yellowness) were affected, meaning that the addition of a white button mushroom caused the darker colour of the product’s surface and cross-section. Pérez Montes et al. [48] reviewed research on meat products recently supplemented with different mushrooms. In the case of burgers, patties, sausages, frankfurters, nuggets, and salami, the addition of mushrooms decreased the lightness. However, during a several-month storage period, mushroom components preserved or retarded colour change due to antioxidant activity. Wan-Mohtar et al. [49] prepared a chicken patty with Pleurotus sapidus stems, which affected the lightness of this product. The authors speculated that the colour of the patty originated from the colour of the mushroom powder supported by polysaccharide caramelisation during cooking. Likewise, Cerón-Guevara et al. [50] developed liver pâté supplemented with A. bisporus and Pleurotus ostreatus. In this case, the dark colour originating with the button mushroom addition affected lower colour scores. These pâtés differed greatly from the control because colour is among the most important sensory characteristic appreciated by consumers of this type of product. Stephan et al. [51] added P. sapidus mycelium to a vegan boiled sausage analog system. The mycelium was cultivated on apple pomace and isomaltose molasses. The product cultivated on molasses appeared very similar to meat products with a slightly more pronounced yellowish to an orange tone. However, the researchers proposed the adjustment of raw material colour by using clean label dye.
Several research groups have prepared extruded snacks using mushroom powder. The conclusion is that the higher the percentage of this additive, the darker the final product. Still, the colour contributed to the Maillard reaction and to caramelisation during cooking [52]. Pecic et al. [53] reported the potential use of G. lucidum in spirit fabrication when the addition of 40 g/L of this mushroom (fruit body) into grain brandy affected the colour of the brandy after seven days, similar to that of an 11 year old plum brandy in sessile oak and an 18 year old in mulberry cask. The colour was strongly correlated with total phenolic compounds. Higher concentrations of mushroom added to brandy increased the yellow and red colour. In another study, Reishi mushrooms significantly influenced the colour of Shiraz wine when added prior to or after fermentation [54]. This study’s panellists thought the wines containing 4 g/L of mushroom (added after fermentation) were darker in colour than that of the control wine. Beer made with mushroom extracts was also investigated, and while visual appearance and colour influenced other sensory aspects, this attribute has not been investigated [55]. Encapsulated G. lucidum extract added to beer, on the other hand, generated a more prominent and darker hue, making it more appealing to customers [56].
Except for beverages, medicinal mushrooms were never intended as a colourant. On the contrary, this healthy ingredient was included to improve storage quality to retard oxidative changes, or for its nutritional and physiological benefits. Modern consumers are ever more aware of artificial dye issues and demand the use of colourants of natural origin [57]. These new pigments derived from plants, micro-organisms or animals are expected to be safe, healthy, and functional. Surprisingly, none of the mushroom pigments has reached the market yet because they have not been examined as a food colourant. The only exception is melanin from Auricularia auricula. Wu et al. [58] characterised and examined its physicochemical and stability in the presence of salt, sugar, light, and high temperature. Melanin from this commercially cultivated medicinal mushroom may be a healthy colour for the food industry due to its antioxidative, antibiofilm, quorum sensing, and radical scavenging effect. However, it is still in the research phase. The three pigments laetiporic acid A, B, and C, isolated from Laetiporus sulphurous by Davoli et al. [26] are oxygen and light-stable during several months. Considering that this mushroom species is also edible, there is a potential use of its pigments as food colourants with GRAS status. Besides melanin and laetiporic acids, mushrooms produce black, brown, red, orange, yellow, violet, green, and blue pigments [4] and Lagashetti et al. [59]. Such a wide array of compounds, especially blue which are rare in nature [39], have not been widely studied.
Altogether, novel food products with a darker colour have received lower sensory scores with a lower acceptancy among panelists, which were all compared with a lighter colour control, such as functional bread compared with commercial white bread. The sensory scores for bread supplemented with mushroom flour were naturally low since mushroom powder, rich in melanin, proteins, amino acids, and polysaccharides, is originally darker and undergoes the Maillard reaction and caramelisation during thermal processing. Moreover, studies were performed mainly with commercial species, such as white button or oyster mushroom, which contain melanin. It would be wise to compare functional bread with bread prepared with different coloured wheat, composite flours or wholemeal flours. Adulteration with flour of a lesser value, or wheat flour used in wholemeal bread is another problem encountered in bread production. In Serbia, the bread is expected to be of dark colour, which is considered to be a healthier option, and is usually adulterated with white bread flour while the dark colour is obtained by adding a discrete amount of cocoa powder. This can be eliminated by using mushroom-derived pigments or powders which will also provide taste and functional characteristics to the final product. Furthermore, mushroom species rich in orange, yellow, and red pigments might be functional colourants used to develop novel bread types (Figure 4).

Figure 4. Yellow-dye mushrooms used in bread making.
References
- Rao, M.P.N.; Xiao, M.; Li, W.-J. Fungal and Bacterial Pigments: Secondary Metabolites with Wide Applications. Front. Microbiol. 2017, 8, 1113.
- Yusuf, M.; Shabbir, M.; Mohammad, F. Natural colorants: Historical, processing and sustainable prospects. Nat. Prod. Prod. Prod. Bioprospecting 2017, 7, 123–145.
- Ntrallou, K.; Gika, H.; Tsochatzis, E. Analytical and Sample Preparation Techniques for the Determination of Food Colorants in Food Matrices. Foods 2020, 9, 58.
- Velíšek, J.; Cejpek, K. Pigments of higher fungi—A review. Czech J. Food Sci. 2011, 29, 87–102.
- Dufossé, L.; Fouillaud, M.; Caro, Y.; Mapari, S.A.; Sutthiwong, N. Filamentous fungi are large-scale producers of pigments and colorants for the food industry. Curr. Opin. Biotechnol. 2014, 26, 56–61.
- Gmoser, R.; Ferreira, J.A.; Lennartsson, P.R.; Taherzadeh, M.J. Filamentous ascomycetes fungi as a source of natural pigments. Fungal Biol. Biotechnol. 2017, 4, 1–25.
- Lin, L.; Xu, J. Fungal Pigments and Their Roles Associated with Human Health. J. Fungi 2020, 6, 280.
- Yu, P.-W.; Chang, Y.-C.; Liou, R.-F.; Lee, T.-H.; Tzean, S.-S. pks63787, a Polyketide Synthase Gene Responsible for the Biosynthesis of Benzenoids in the Medicinal Mushroom Antrodia cinnamomea. J. Nat. Prod. 2016, 79, 1485–1491.
- Yang, X.; Yang, K.; Wang, X.; Wang, Y.; Zhao, Z.; Meng, D. Transcriptomic analysis reveals the mechanism of bacterial disease resistance of postharvest button mushroom (Agaricus bisporus). Physiol. Mol. Plant Pathol. 2022, 122, 101903.
- Min, B.; Kim, S.; Oh, Y.-L.; Kong, W.-S.; Park, H.; Cho, H.; Jang, K.-Y.; Kim, J.-G.; Choi, I.-G. Genomic discovery of the hypsin gene and biosynthetic pathways for terpenoids in Hypsizygus marmoreus. BMC Genom. 2018, 19, 789.
- Baars, J.J.P.; Camp, H.J.M.O.D.; Hermans, J.M.H.; Mikeš, V.; Van Der Drift, C.; Van Griensven, L.J.L.D.; Vogels, G.D. Nitrogen assimilating enzymes in the white button mushroom Agaricus bisporus. Microbiology 1994, 140, 1161–1168.
- Klaus, A.; Wan-Mohtar, W.A.A.Q.I.; Nikolić, B.; Cvetković, S.; Vunduk, J. Pink oyster mushroom Pleurotus flabellatus mycelium produced by an airlift bioreactor—The evidence of potent in vitro biological activities. World J. Microbiol. Biotechnol. 2021, 37, 1–14.
- Ma, G.; Yang, W.; Zhao, L.; Pei, F.; Fang, D.; Hu, Q. A critical review on the health promoting effects of mushrooms nutraceuticals. Food Sci. Hum. Wellness 2018, 7, 125–133.
- Weijn, A.; Berg-Somhorst, D.B.P.M.V.D.; Slootweg, J.C.; Vincken, J.-P.; Gruppen, H.; Wichers, H.; Mes, J.J. Main Phenolic Compounds of the Melanin Biosynthesis Pathway in Bruising-Tolerant and Bruising-Sensitive Button Mushroom (Agaricus bisporus) Strains. J. Agric. Food Chem. 2013, 61, 8224–8231.
- Yang, X.-L.; Qin, C.; Wang, F.; Dong, Z.-J.; Liu, J.-K. A New Meroterpenoid Pigment from the Basidiomycete Albatrellus confluens. Chem. Biodivers. 2008, 5, 484–489.
- Yaqoob, A.; Li, W.M.; Liu, V.; Wang, C.; Mackedenski, S.; Tackaberry, L.E.; Massicotte, H.B.; Egger, K.N.; Reimer, K.; Lee, C.H. Grifolin, neogrifolin and confluentin from the terricolous polypore Albatrellus flettii suppress KRAS expression in human colon cancer cells. PLoS ONE 2020, 15, e0231948.
- Bin, L.; Wei, L.; Xiaohong, C.; Mei, J.; Mingsheng, D. In vitro antibiofilm activity of the melanin from Auricularia auricula, an edible jelly mushroom. Ann. Microbiol. 2012, 62, 1523–1530.
- Zou, Y.; Hu, W.; Ma, K.; Tian, M. Physicochemical properties and antioxidant activities of melanin and fractions from Auricularia auricula fruiting bodies. Food Sci. Biotechnol. 2015, 24, 15–21.
- Song, J.; Manir, M.; Moon, S.-S. Cytotoxic Grifolin Derivatives Isolated from the Wild Mushroom Boletus pseudocalopus (Basidiomycetes). Chem. Biodivers. 2009, 6, 1435–1442.
- Kozarski, M.; Klaus, A.; Vunduk, J.; Zizak, Z.; Niksic, M.; Jakovljevic, D.; Vrvic, M.M.; Van Griensven, L.J.L.D. Nutraceutical properties of the methanolic extract of edible mushroom Cantharellus cibarius (Fries): Primary mechanisms. Food Funct. 2015, 6, 1875–1886.
- Tudor, D.; Robinson, S.C.; Cooper, P.A. The influence of pH on pigment formation by lignicolous fungi. Int. Biodeterior. Biodegradation 2013, 80, 22–28.
- Velmurugan, P.; Lee, Y.H.; Nanthakumar, K.; Kamala-Kannan, S.; Dufossé, L.; Mapari, S.A.; Oh, B.T. Water-soluble red pigments from Isaria farinosa and structural characterization of the main colored component. J. Basic Microbiol. 2010, 50, 581–590.
- Lee, J.-H.; Hyun, C.-K. Insulin-Sensitizing and Beneficial Lipid-Metabolic Effects of the Water-Soluble Melanin Complex Extracted from Inonotus obliquus. Phytother. Res. 2014, 28, 1320–1328.
- Caro, Y.; Venkatachalam, M.; Lebeau, J.; Fouillaud, M.; Dufossé, L. Pigments and Colorants from Filamentous Fungi. Fungal Metab. 2017, 25, 499–568.
- Kim, K.H.; Noh, H.J.; Choi, S.U.; Park, K.M.; Seok, S.-J.; Lee, K.R. Lactarane sesquiterpenoids from Lactarius subvellereus and their cytotoxicity. Bioorganic Med. Chem. Lett. 2010, 20, 5385–5388.
- Davoli, P.; Mucci, A.; Schenetti, L.; Weber, R.W. Laetiporic acids, a family of non-carotenoid polyene pigments from fruit-bodies and liquid cultures of Laetiporus sulphureus (Polyporales, Fungi). Phytochemistry 2005, 66, 817–823.
- Zhang, Y.; Wu, X.; Huang, C.; Zhang, Z.; Gao, W. Isolation and identification of pigments from oyster mushrooms with black, yellow and pink caps. Food Chem. 2021, 372, 131171.
- De Souza, R.A.; Kamat, N.M.; Nadkarni, V.S. Purification and characterisation of a sulphur rich melanin from edible mushroom Termitomyces albuminosus Heim. Mycology 2018, 9, 296–306.
- Harki, E.; Talou, T.; Dargent, R. Purification, characterisation and analysis of melanin extracted from Tuber melanosporum Vitt. Food Chem. 1997, 58, 69–73.
- Dufossé, L.; Galaup, P.; Yaron, A.; Arad, S.M.; Blanc, P.; Murthy, K.N.C.; Ravishankar, G.A. Microorganisms and microalgae as sources of pigments for food use: A scientific oddity or an industrial reality? Trends Food Sci. Technol. 2005, 16, 389–406.
- Barredo, J.L.; García-Estrada, C.; Kosalkova, K.; Barreiro, C. Biosynthesis of Astaxanthin as a Main Carotenoid in the Heterobasidiomycetous Yeast Xanthophyllomyces dendrorhous. J. Fungi 2017, 3, 44.
- Zhou, Z.-Y.; Liu, J.-K. Pigments of fungi (macromycetes). Nat. Prod. Rep. 2010, 27, 1531–1570.
- Pacelli, C.; Cassaro, A.; Maturilli, A.; Timperio, A.M.; Gevi, F.; Cavalazzi, B.; Stefan, M.; Ghica, D.; Onofri, S. Multidisciplinary characterization of melanin pigments from the black fungus Cryomyces antarcticus. Appl. Microbiol. Biotechnol. 2020, 104, 6385–6395.
- Wold, C.W.; Gerwick, W.H.; Wangensteen, H.; Inngjerdingen, K.T. Bioactive triterpenoids and water-soluble melanin from Inonotus obliquus (Chaga) with immunomodulatory activity. J. Funct. Foods 2020, 71, 104025.
- Räisänen, R. Fungal colorants in applications–focus on Cortinarius species. Color. Technol. 2019, 135, 22–31.
- Mattoon, E.; Cordero, R.; Casadevall, A. Fungal Melanins and Applications in Healthcare, Bioremediation and Industry. J. Fungi 2021, 7, 488.
- Reis, F.S.; Martins, A.; Vasconcelos, M.H.; Morales, P.; Ferreira, I.C.F.R. Functional foods based on extracts or compounds derived from mushrooms. Trends Food Sci. Technol. 2017, 66, 48–62.
- Wan-Mohtar, W.A.A.Q.I.; Taufek, N.M.; Yerima, G.; Rahman, J.; Thiran, J.P.; Subramaniam, K.; Sabaratnam, V. Effect of bioreactor-grown biomass from Ganoderma lucidum mycelium on growth performance and physiological response of red hybrid tilapia (Oreochromis sp.) for sustainable aquaculture. Org. Agric. 2020, 11, 327–335.
- Neves, M.I.L.; Silva, E.K.; Meireles, M.A.A. Natural blue food colorants: Consumer acceptance, current alternatives, trends, challenges, and future strategies. Trends Food Sci. Technol. 2021, 112, 163–173.
- Sulieman, A.A.; Zhu, K.-X.; Peng, W.; Shoaib, M.; Obadi, M.; Hassanin, H.A.M.; Alahmad, K.; Zhou, H.-M. Assessment of rheological, physicochemical, and staling characteristics of gluten-free dough and bread containing Agaricus bisporus polysaccharide flour and inulin. J. Food Meas. Charact. 2018, 12, 2032–2044.
- Ulziijargal, E.; Yang, J.-H.; Lin, L.-Y.; Chen, C.-P.; Mau, J.-L. Quality of bread supplemented with mushroom mycelia. Food Chem. 2013, 138, 70–76.
- Szydłowska-Tutaj, M.; Złotek, U.; Combrzyński, M. Influence of addition of mushroom powder to semolina on proximate composition, physicochemical properties and some safety parameters of material for pasta production. LWT 2021, 151, 112235.
- Parvin, R.; Farzana, T.; Mohajan, S.; Rahman, H.; Rahman, S.S. Quality improvement of noodles with mushroom fortified and its comparison with local branded noodles. NFS J. 2020, 20, 37–42.
- Olawuyi, I.F.; Lee, W.Y. Quality and antioxidant properties of functional rice muffins enriched with shiitake mushroom and carrot pomace. Int. J. Food Sci. Technol. 2019, 54, 2321–2328.
- Salehi, F.; Kashaninejad, M.; Asadi, F.; Najafi, A. Improvement of quality attributes of sponge cake using infrared dried button mushroom. J. Food Sci. Technol. 2016, 53, 1418–1423.
- Wan-Mohtar, W.A.A.Q.I.; Mahmud, N.; Supramani, S.; Ahmad, R.; Zain, N.A.M.; Hassan, N.A.M.; Peryasamy, J.; Halim-Lim, S.A. Fruiting-body-base flour from an oyster mushroom—A waste source of antioxidative flour for developing potential functional cookies and steamed-bun. AIMS Agric. Food 2018, 3, 481–492.
- Kurt, A.; Gençcelep, H. Enrichment of meat emulsion with mushroom (Agaricus bisporus) powder: Impact on rheological and structural characteristics. J. Food Eng. 2018, 237, 128–136.
- Pérez-Montes, A.; Rangel-Vargas, E.; Lorenzo, J.M.; Romero, L.; Santos, E.M. Edible mushrooms as a novel trend in the development of healthier meat products. Curr. Opin. Food Sci. 2020, 37, 118–124.
- Wan-Mohtar, W.A.A.Q.I.; Halim-Lim, S.A.; Kamarudin, N.Z.; Rukayadi, Y.; Rahim, M.H.A.; Jamaludin, A.A.; Ilham, Z. Fruiting-body-base flour from an Oyster mushroom waste in the development of antioxidative chicken patty. J. Food Sci. 2020, 85, 3124–3133.
- Cerón-Guevara, M.I.; Santos, E.M.; Lorenzo, J.M.; Pateiro, M.; Bermúdez-Piedra, R.; Rodríguez, J.A.; Castro-Rosas, J.; Rangel-Vargas, E. Partial replacement of fat and salt in liver pâté by addition of Agaricus bisporus and Pleurotus ostreatus flour. Int. J. Food Sci. Technol. 2021, 56, 6171–6181.
- Stephan, A.; Ahlborn, J.; Zajul, M.; Zorn, H. Edible mushroom mycelia of Pleurotus sapidus as novel protein sources in a vegan boiled sausage analog system: Functionality and sensory tests in comparison to commercial proteins and meat sausages. Eur. Food Res. Technol. 2017, 244, 913–924.
- Keerthana, K.; Anukiruthika, T.; Moses, J.; Anandharamakrishnan, C. Development of fiber-enriched 3D printed snacks from alternative foods: A study on button mushroom. J. Food Eng. 2020, 287, 110116.
- Pecić, S.; Nikićević, N.; Veljović, M.; Jardanin, M.; Tešević, V.; Belović, M.; Nikšić, M. The influence of extraction parameters on physicochemical properties of special grain brandies with Ganoderma lucidum. License 2015, 22, 181–189.
- Nguyen, A.N.; Capone, D.L.; Johnson, T.E.; Jeffery, D.W.; Danner, L.; Bastian, S.E. Volatile Composition and Sensory Profiles of a Shiraz Wine Product Made with Pre- and Post-Fermentation Additions of Ganoderma lucidum Extract. Foods 2019, 8, 538.
- Vunduk, J.; Veljović, S. Macrofungi in the Production of Alcoholic Beverages Beer, Wine, and Spirits. In Advances in Macrofungi; CRC Press: Boca Raton, FL, USA, 2021; pp. 108–141.
- Belscak-Cvitanovic, A.; Nedovic, V.; Salevic, A.; Despotovic, S.; Komes, D.; Niksic, M.; Bugarski, B.; Leskosek-Cukalovic, I. Modification of functional quality of beer by using microencapsulated green tea (Camellia sinensis L.) and Ganoderma mushroom (Ganoderma lucidum L.) bioactive compounds. Chem. Ind. Chem. Eng. Q. 2017, 23, 457–471.
- Martins, N.; Roriz, C.L.; Morales, P.; Barros, L.; Ferreira, I.C. Food colorants: Challenges, opportunities and current desires of agro-industries to ensure consumer expectations and regulatory practices. Trends Food Sci. Technol. 2016, 52, 1–15.
- Wu, Z.; Zhang, M.; Yang, H.; Zhou, H.; Yang, H. Production, physico-chemical characterization and antioxidant activity of natural melanin from submerged cultures of the mushroom Auricularia auricula. Food Biosci. 2018, 26, 49–56.
- Lagashetti, A.C.; Dufossé, L.; Singh, S.K.; Singh, P.N. Fungal Pigments and Their Prospects in Different Industries. Microorganisms 2019, 7, 604.
More
Information
Subjects:
Mycology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.6K
Revisions:
2 times
(View History)
Update Date:
09 Nov 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





