
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Agnieszka Ewa Wiącek | + 2637 word(s) | 2637 | 2020-11-10 03:39:22 | | | |
| 2 | Vivi Li | -27 word(s) | 2610 | 2020-11-18 06:45:56 | | |
Video Upload Options
The bioavailability of CsA was matched and enhanced with nanoparticles in the context of the physicochemical properties and the character of its interactions with lipid membranes. This study can suggest the suitability of liposome/microemulsion as promising vehicles for CsA oral or injection delivery. As the most hopeful proved to be formulation with the smaller particle size because can facilitating absorption, but none of the formulations is flawless. However, when safety of the product is assessed, relying on just the particle size cannot be the only criteria.
1. Introduction
Nanotechnology is one of the most promising strategies developed to improve drug delivery systems. Within this field, lipid-based formulations have attracted attention for the administration of poorly soluble bioactive substances, particularly in water and aqueous solutions. The numerous advantages of these nanosystems include biodegradability, biocompatibility, and high physicochemical stability. Their capacity for drug solubilization and cell adhesion are properties that enhance the bioavailability of drugs demonstrating irregular absorption [1][2]. An important parameter of drug delivery systems relates to no or low toxicity of the carrier itself either in vivo or in the environment as a by-product. Therefore, nanoparticles fabricated using an assembly of natural biomolecules, such as lipids, proteins, and carbohydrates are expected to be appropriate. Lipid-based systems are particularly useful for clinical applications. This is closely related to the ability of lipid particles to cross cell membranes directly due to the fact that lipids are one of the basic building blocks of cell membranes [3][4][5][6][7][8]. Drug delivery using lipid-based systems can mimic biological membranes because the translocation of natural cell membrane properties to synthetic systems combines the advantages of the membrane surface and flexibility of material chemistry. An additional benefit is that such a system can escape immune system elimination [5]. Despite the membrane structure, the elasticity of nanoparticles also has an obvious impact on the ability to penetrate across the lipid bilayer as an effect of different morphological responses of hydrophilic and hydrophobic nanoparticles. Studies demonstrated that hydrophilic nanoparticles are flattened in the membrane plane, while hydrophobic nanoparticles are elongated during penetration. These results show the joint effects of elasticity and particle surface hydrophobicity on cellular uptake [1][2][3][4][5][6][7][8]. Effective delivery systems allow for the protecting of drugs from fast degradation, long-term in vivo retention, immune escape, targeted controlled drug release, and give the opportunity to easily cross specific barriers in vivo [3][4][5][6]. Other factors, such as size, surface charge, shape, lipid composition (headgroups, the length of the tail, and even the saturability of the lipid carriers), and elastic and curvature energies may also influence the barrier permeability and transport [7][8]. A lipid bilayer structure allows encapsulating both fat- and water-soluble molecules enabling the delivery of therapeutic agents with different physico-chemical properties. The unique properties of lipid-based systems, including their ability to facilitate extended circulation time, adhesion, and homologous targeting, have led to their various applications in the context of nanomedicine [6][7][8][9].
2. Physico-Chemical Characterization of Typical CsA Systems
2.1. Liposomal CsA Systems Stability
From the point of biopharmaceutics in transplantology, a more efficient and a safe delivery system with less toxicity is needed for immunosuppressive therapy. Cyclosporine is a neutral, high hydrophobic cyclic peptide, and the main component of liposomes are phospholipids. Each phospholipid has an intrinsic liquid crystalline phase. Liposome membranes are fluid above the characteristic individual temperature and are solid below it. Liposomes are widely used in clinical medicine for various purposes, e.g., selective drug delivery and diagnostic vehicles. Liposomes are highly specialized lipid spheres that can preferentially accumulate at sites of tumors, infection, and inflammation. CsA as a lipophilic drug appears to be a model candidate to include into liposomes. Intravenously supplied liposomes of CsA can lower drug side effects, as a result of its avoidance of the kidneys. CsA liposomes are favorably removed by the reticuloendothelial system and tend to accumulate in the spleen. A CsA liposomal carrier provides the possibility to attain the optimal drug level at sites of tissue rejection, as an effect of association of different parameters of the immune response and to decrease the drug exposure to sensitive organs. Studies of various drugs, not only CsA, showed that liposome-encapsulated drugs often have biodistribution and toxicity that are significantly different from the original drug. The particle size distribution of the liposomal formulations with or without CsA was estimated using a laser light scattering technique (the average particle size of the prepared liposome formulations (effective diameter, nm), turbidity (NTU), value of natural pH, polydispersion index (PDI), and zeta potential of exemplary systems). The order of particle size of the obtained CsA nanocarriers was CsA, CaCl2 > CsA, H2O > CsA, KCl, and it was difficult to decrease the particle size to <100 nm through high-pressure homogenization. On the other hand, the PDI of all carriers was near or much lower than 0.3, which can indicate relatively narrow particle-size distribution. CsA liposomes in electrolyte solution had a higher negative charge than CsA liposomes in water. A high zeta potential is necessary to maintain the in vitro stability of nanosuspensions. The liposomes with an absolute value of charge lower than 5 mV are not usually stable; however, the particle size and PDI of all vehicles had no significant changes within 24 h; thus, other mechanisms of stabilization should be considered [2].
2.2. Binding of CsA to Lipid Membranes (BAM Analysis)
The binding of CsA to lipid membranes follows classical hydrophobic effects and is accompanied by a positive enthalpy variation. This is rather uncommon because most peptide-lipid binding reactions require enthalpy-driven binding. Research often assumes that the CsA molecule occupies an area of 260 Å2 in the lipid monolayer. During an increase in the CsA concentration, a model membrane perturbation is observable by changes in the lateral organization in the acyl chain region. As a result, the drug interacts mainly with the part of the fatty acyl chain proximal to the head group of the phospholipids. The characteristics of CsA interactions with biological membranes suggest a drug-dependent increase in the bilayer fluidity at temperatures below the main phase transition, but an increased spatial organization at temperatures above that point. On the other hand, the addition of cholesterol to the model membrane decreased the binding capacity of cyclosporine as a consequence of the acyl chain flexibility. The structure of a bilayer influences the nature and topology of CsA inclusions into monolayer. Different factors, e.g., the composition of the liposomal carriers of cyclosporine, the type of fatty acid chains, and, notably, the lipid head group, strictly affect the pharmacokinetic behavior. These conclusions have been proven on the basis of research described in detail in our earlier articles [2]. Therefore, to achieve the desired drug effect, the type and composition of the lipid formulation used as its carrier should be carefully considered.
The Brewster angle microscopy (BAM) analysis of a pure DPPC monolayer showed small characteristic and irregular domains, which appeared at surface pressure below 10 mN/m (Figure 1). Observed structures are specific for the LE-LC phase transition, and they gradually change along with the progress of compression. After reaching a value of surface pressure of about 20 mN/m, the monolayer was homogenous. On the other hand, a pure CsA monolayer appeared to be homogenous and less condensed than a pure DPPC monolayer, and no domains were observed. The mixed monolayers of DPPC and CsA exhibited the formation of domains but smaller structures were observed if the amount of CsA in the monolayer was higher. Along with the increase in CsA content, the shape of the domains was more circular and they were arranged in points. The πcoll for pure CsA was estimated at 25–26 mN/m and, at the higher surface pressure (about 30 mN/m), small domains appeared. The surface pressure-area isotherms and BAM images were used to investigate the effect of CsA on a model DPPC membrane for different molar ratios of ingredients (DPPC-CsA). Changes in isotherm shapes, their shifts, and also BAM images of domains appearing during the compression process were registered and analyzed. This type of research together with the thorough characterization of different CsA-based formulations may be helpful in better understanding CsA interactions with living cell membranes and should be planned each time for a given patient group because the exact explanation of the drug action requires not only knowing the physico-chemical characteristics but also an in-depth analysis at the molecular level.

Figure 1. Brewster angle microscopy (BAM) images of the mixed monolayers on the water subphase at 10 mN/m (0.15; 0.25; 0.50; and 0.75 indicate the molar fractions of CsA).
2.3. O/W Emulsions of CsA
The formulation of CsA can also be prepared as o/w emulsions. From the literature data, CsA powder can be predominantly amorphous in nature. In Wang et al.’s studies on the basis of SEM images, there were no clear crystalline particles visible. In another investigation, crystalline CsA powders indicated a tetragonal crystal form. As an effect, the kind of emulsion and its stability is strictly dependent on the CsA form. The stability of such systems can be defined on the basis of a regular shape and distributions of droplets or nanoparticles. Instead of micelles and liposomes, drug emulsion may be more convenient regarding the stability. On the other hand, with the increase in the drug/micelle (liposome) ratios, the micelles (liposomes) can become more unstable because the drug can be supersaturated in the core of the micelles. The high chemical potential of the drug in micelles (liposomes) or dissociation of the micelle particles could cause irreversible precipitation of CsA. Pharmacokinetically, the degree of supersaturation is a very important parameter and driving force for drug release because this also potentially affects its precipitation and absorption rates in the gastrointestinal tract.
The CsA suspended in soybean oil and cholesterol with or without the addition of ethanol formed an o/w emulsion where the average droplet sizes were in the range of 500–1500 nm according to the DLS analysis. The thermal behaviors of CsA formulations clarified using DSC showed that crystalline CsA produced a melting endothermic event at 115 °C, amorphous CsA exhibited thermal events at 128 °C, and the transition during the endothermic events might be attributable to a solid-to-liquid transition at over 120 °C. CsA in emulsion formulation might exist in a high-energy amorphous state with a homogeneous molecular interaction between CsA and the lipidic ingredient. In addition, the amorphization of CsA might be effective for improvement of the wettability through a reduction in particle size and the absence of crystallinity. Dollo and co-workers clarified the possible enhancement of dissolution behavior, the dissolution tests for CsA systems were conducted in distilled water. Poor dissolution behaviors were seen in both crystalline and amorphous CsA powders.
2.4. CsA/Chitosan Systems. Transport across Membranes
As was mentioned earlier, studies using mono- or bilayer-imitating cell behavior provide a better prediction of the in vivo absorption of most drugs, including cyclosporine. Liposomes also play an important role in these processes. On the other hand, liposomes used as drug carriers have been shown to be stable when lyophilized in the presence of, for example, disaccharides. Considering this, some studies are carried out for systems with cholesterol, and the effect of antibacterial chitosan has also been studied. The binary or ternary monomolecular films of varying composition make it possible to produce coverage with pre-planned surface properties and check the transport across membranes. An additional component in these systems is the antibacterial chitosan. The prepared mixed layer and charge density dependent chitosan coating can possibly limit the excessive exposure of chitosan to the blood. As a result, a reduction in the effect of blood clot formation is possible. The monolayers morphology and behavior can provide detailed information on the packing, flexibility, and interactions for a better reference to the real conditions in vivo.
This type of hybrid coating allows the design of the structure of defined topography to play a major role in the interactions with living tissues. As a result of that, the problematic mechanisms of the possible immune response can include better recognition. The Langmuir technique permits us to determine the behavior of polypeptide CsA in contact with a model biological membrane built of its two main ingredients, phospholipid DPPC and cholesterol, especially at the pressure of the natural biological membrane (in the range of 25–30 mN/m). Due to the nature of drug, it is not possible to obtain a stable CsA monolayer at such pressure because it collapses near 25 mN/m. Therefore, the behavior of ternary DPPC-Chol-CsA films was examined depending on the drug amount of CsA at the surface pressure of 15 mN/m, regarded as the most suitable for such CsA/lipid-based systems. We found that cyclosporine significantly reduced the fluidity and packing of the tested biological membranes. At low CsA contents, domain formation was observed, and the highest values of excess Gibbs energy of mixing, which suggests repulsion of the film components and their partial miscibility. For higher CsA content, a homogeneous film structure was observed, proving an increase in the intermolecular attraction of the components in the monolayer. Langmuir–Blodgett films (LB) were formed on solid supports with adsorbed chitosan. These investigations are very important in terms of inhibiting platelet aggregation on the surface of, for example, implants. The multilayer hybrid systems technique is useful in the design and preparation of modern materials because it can combine widespread and multi-task utility properties.
2.5. Cyclosporine/Oil Phase/EtOH Formulations
In the present study, novel formulations of CsA employing liposomes or o/w emulsion systems were designed for pharmacologic treatment. There was a marked improvement in the dissolution behavior of a nanoemulsified dispersion CsA compared with that of CsA powder. The obtained systems exhibited high dispersibility and particle distribution, and thus can be suitable not only in transplantation but also for oral absorption or inhalation therapy. The full physicochemical characteristics (Figure 2) can help to obtain the CsA formulation, which might be an efficacious delivery option for immunosuppressive drugs, giving the possibility to provide an optimal local pharmacological action of CsA.
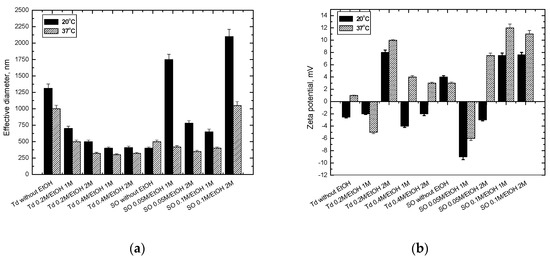
Figure 2. (a) The effective diameter and (b) zeta potential of different CsA formulations at 20 and 37 °C. Abbreviation: SO—soybean oil (0.05 M or 0.1 M), Td n-tetradecane (0.2 M or 0.4 M), EtOH ethanol solution (1 M or 2 M).
3. Summary and Perspectives
Here, we concluded that CsA is a poorly soluble and low absorbable drug that can be prepared easily and reproducibly as positively or negatively charged nanoparticles in the range of size about 200–500 nm. The particle size changes of CsA formulation using cationic polymers (e.g., chitosan), lipid, or oil/water formulations can improve the overall bioavailability. The nature of additives, charges, and their effect on the permeability play an important role in the absorption rate. The improved bioavailability of CsA in systems with the addition of chitosan or phospholipids provides the possibility to use a lower dose of the drug with lower side effects after the therapy. On the other hand, such CsA formulations maximized the bioavailability and gave a reduction of the inter- and intra-individual variability, thereby, improving CsA’s overall efficacy. We demonstrated the ability of the size diameter and zeta potential to be used as a practical and easy method to study CsA liposomal and other colloidal systems, also with nano- and microparticles additions. The zeta potential values of the different modified CsA formulations correlated to the nature of modification.
Our general conclusion is as follows: hybrid formulations can improve the bioavailability of poorly soluble and poorly absorbable drugs. However, to assess the systemic toxic potential, the drug release theoretical investigations should be interpreted with caution as they do not always replicate the in vitro and in vivo conditions. The stability of the prepared systems over a broad pH range and at different temperatures should always be the subject of such studies.
References
- Dai, W.; Guo, Y.; Zhang, H.; Wang, X.; Zhang, Q. Sylysia 350/Eudragit S100 solid nanomatrix as a promising system for oral delivery of cyclosporine A. Int. J. Pharm. 2015, 478, 718–725.
- Wang, K.; Qi, J.; Weng, T.; Tian, Z.; Lu, Y.; Hu, K.; Yin, Z.; Wu, W. Enhancement of oral bioavailability of cyclosporine A: Comparison of various nanoscale drug-delivery systems. Int. J. Nanomed. 2014, 9, 4991–4999.
- Puri, A.; Loomis, K.; Smith, B.; Lee, J.-H.; Yavlovich, A.; Heldman, E.; Blumenthal, R. Lipid-based nanoparticles as pharmaceuticals drug carriers: From concept to clinic. Crit. Rev. Ther. Drug Carrier Syst. 2009, 26, 523–580.
- Guo, Y.; Terrazi, E.; Seemann, R.; Fleury, J.B.; Baulin, V.A. Direct proof of spontaneous translocation of lipid-covered hydrophobic nanoparticles through a phospholipid bilayer. Sci. Adv. 2016, 2, e1600261.
- Li, R.; He, Y.; Zhang, S.; Qin, J.; Wang, J. Cell membrane-based nanoparticles: A new biomimetic platform for tumor diagnosis and treatment. Acta Pharm. Sinica B 2018, 8, 14–22.
- Liu, Y.; Luo, J.; Chen, X.; Liu, W.; Chen, T. Cell membrane coating technology. A promising strategy for biomedical applications. Nano-Micro Lett. 2019, 11, 100.
- Zhang, L.; Chen, H.; Xie, J.; Becton, M.; Wang, X. Interplay of Nanoparticles Rigidity and Its Translocation Ability through Cell Membrane. Phys. Chem. B 2019, 123, 8923–8930.
- Wang, S.; Guo, H.; Li, Y.; Li, X. Penetration of nanoparticles across a lipid bilayer: Effects of particle stiffness and surface hydrophobicity. Nanoscale 2019, 11, 4025–4034.
- Zacheo, A.; Bizzarro, L.; Blasi, L.; Piccirillo, C.; Cardone, A.; Gigli, G.; Ragusa, A.; Quarta, A. Lipid-Based Nanovesicles for Simultaneous Intracellular Delivery of Hydrophobic, Hydrophilic, and Amphiphilic Species. Front. Bioeng. Biotechnol. 2020, 8, 1–17.





