| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Seongho Ryu | + 3097 word(s) | 3097 | 2020-10-13 08:33:34 |
Video Upload Options
Extracellular vesicles (EVs) are 50–300 nm vesicles secreted by eukaryotic cells. They can carry cargo (including miRNA) from the donor cell to the recipient cell. miRNAs in EVs can change the translational profile of the recipient cell and modulate cellular morphology. This endogenous mechanism has attracted the attention of the drug-delivery community in the last few years. EVs can be enriched with exogenous therapeutic miRNAs and used for treatment of diseases by targeting pathological recipient cells. However, there are some obstacles that need to be addressed before introducing therapeutic miRNA-enriched EVs in clinics.
1. Introduction
The past few decades have seen enormous research in the field of extracellular vesicles (EVs). EVs produced by cells are divided into broad categories of exosomes, microvesicles, and apoptotic bodies. This categorization depends upon their origin in cell. This review mainly points out to ‘exosome’ related studies while referring to them as EVs (unless otherwise mentioned). Exosomes are small vesicles produced endogenously by almost all types of cells for intercellular communication. Their ability to carry their cargo, specifically nucleic acids, i.e., mRNA and miRNA to the recipient cells, has attracted the attention of the drug-delivery community[1] [2]. For this paper, words such as ‘extracellular vesicles’, ‘exosomes’, ‘RNA’, ‘miRNA’, ‘treatment’, ‘non-coding RNA’, ‘therapy’, were used to search in “PubMed” and “google scholars”. Generally, articles from the last ten years were preferentially included.
Previously, synthetic liposomes and nanovesicles have been used to deliver therapeutic molecules, including miRNA, to cure certain diseases. However, the efforts to make them biocompatible and safe in every aspect have not been met with absolute success. In this regard, intrinsic EVs may provide immunologically safer options as carriers of therapeutic molecules, specifically miRNA [1] [2]. Two important features for considering EVs as potential option for drug-delivery are their compositional properties and nano-mechanical properties. Compositional properties include their surface lipid/protein content and capacity to carry specific molecules in their lumen. Moreover, nano-mechanical properties include size and colloidal ability, which are important in EV-cell interaction. Compositional properties have been studied extensively; however, comparatively little attention has been given to the nano-mechanical features until now. Therefore, this article only covers the aspect of compositional characteristics of EVs as a therapeutic carrier [3].
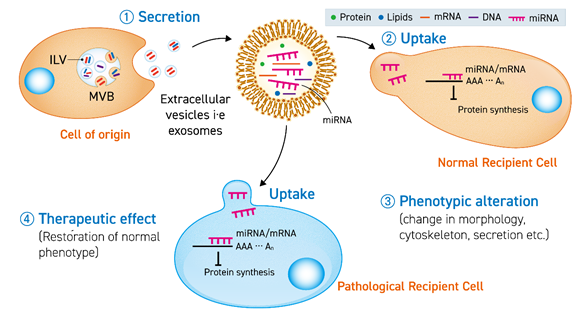
Studies with endogenous EVs have led to the identification of miRNAs in their cargo. These EVs can be used to restore normal phenotype in in-vitro diseased models (Figure 1). Exploiting this natural mechanism, efforts have been made to isolate EVs from cell media and body fluids to enrich them with therapeutic miRNAs. However, despite the rapid expansion of investigations regarding miRNA-enriched EVs, there have been only a few human clinical trials reported to date as evidence of treating pathologies. One of the reasons behind the lack of clinical studies is insufficient pre-clinical investigations in animals. The in-vivo studies, which are the precursor to human trials, should make sure the availability of: (1) higher yield of purified exosomes, (2) well-defined and optimized conditions to maintain exosome-secreting cells for reproducible results, (3) an optimized number of doses and quantity of variations of immune systems in subjects. Extensive efforts should be focused on developing and engineering EVs specifically for in-vivo administration.
Several technical limitations and gaps in our knowledge may become a possible hurdle for therapeutic miRNA enriched EVs to reach clinics. To overcome such obstacles, numerous methods to successfully obtain clinical-grade exosomes and manipulating them to target specific cells/tissues have been developed [4]. Moreover, engineering these EVs to load specific therapeutic miRNAs has achieved special attention in the EV-community and drug-delivery experts. Until recently, a number of investigational studies have focused on the endogenous mechanisms of miRNA loading/sorting in EVs, to pave the way of engineering miRNA-based EV carriers for alleviating diseases using in-vitro and in-vivo models. However, there is still an unsettled debate about whether the miRNAs are sorted/loaded actively or passively in endogenous systems. The factors responsible for determining the choice of active or passive loading have just begun to be studied. Uncovering this can provide important information to develop an improved protocol for engineering miRNA-enriched EV therapies in the future.
Figure 1. The secreted extracellular vesicle (EV) (1) carrying miRNA is uptaken by a normal cell (2) or pathological cell to cause a phenotypic change (3) or therapeutic effect (4) by translational inhibition of mRNA in the recipient cells. ILV stands for intraluminal vesicles and MVB stands for multivesicular bodies.
To the best of our knowledge, the subject of therapeutic miRNA-enriched EVs has not been extensively and exclusively reviewed previously. However, there have been some reviews summarizing the intrinsic sorting mechanisms of miRNA in exosomes [5]. Moreover, O’ Brien et al. published a review covering all non-coding/coding RNAs in EVs (including mRNA, tRNA fragments, snoRNA, ln-RNA) but the focus to miRNA is limited [6].
2. Principal Concepts about Therapeutic miRNA-Enriched EV
2.1. Extracellular Vesicles
Extracellular vesicles are small vesicles enclosed by phospholipid-bilayer, secreted by eukaryotic cells. They are found in body fluids, such as blood, tears, sweat, ascites, cerebrospinal fluid, etc. In laboratory conditions, they can be obtained from the culture media of cell lines [1] [2]. The term ‘extracellular vesicles’ refer to a broad category of vesicles, which are further divided on the basis of their biogenesis pathway, i.e., exosomes, microvesicles, and apoptotic bodies. Exosomes are formed by inward budding of multivesicular bodies (MVBs). The size of microvesicles is variable, reaching up to 1 micrometer, produced by outward budding of the cell membrane, whereas apoptotic bodies are produced from the cell membrane’s fragmentation when cells undergo apoptosis. Usually, EVs within size range 50–300 nm are used for studies referring to them as ‘exosomes’ [7]. However, due to lack of sufficient biomarkers and overlap in size-range, it is difficult to discriminate between the types of vesicles.
Extracellular vesicles (EVs) carry biomolecules, such as mRNA, proteins, miRNA, metabolites, and have the ability to deliver them to cells along short and long distances. Blood can be used as a medium of transport to deliver EVs over long distances. Their ability to travel in blood and pour their content to recipient cells with functional integrity make them excellent candidates for drug-delivery. Moreover, due to Cluster of Differentiation-47 (CD-47) expression on the EV surface, they can avoid mononuclear phagocytic system in blood [8]. Another important aspect to consider EVs for a drug-carrying vehicle is their similar clearance rate to synthetic liposomes [9]. Synthetic liposomes may accumulate in the spleen and liver depending upon their surface ligands and composition. A similar strategy of surface modification can be applied to EVs to target specific tissues and avoid off-target accumulation [10]. Moreover, due to the intrinsic nature of EVs, they lack the risk of immune rejection as posed by synthetic liposomes. The ideal drug-delivery EV should be able to recognize particular recipient cells. EVs recognize recipient cell surface through their lipids and membrane proteins. The EV surface lipids and membrane proteins can be manipulated to engineer suitable EV-based therapies for special purposes. For example, EVs for neurodegenerative disorders should be optimized to cross the blood–brain barrier (BBB) by engineering them to recognize the recipient cell [11][12]. Secondly, EVs can be loaded with a variety of molecules. To the best of our knowledge, most studies about EV-based therapies utilize miRNA or siRNA as a therapeutic substance encapsulated in EV [13]. The endogenous machinery to sort miRNA can be exploited to manufacture EV-based miRNA therapies. However, there still remains a difficulty in identifying methods to obtain purified homogenous EVs, which can be used in actual clinical settings. In short, it is crucial to understand the benefits of EV-miRNA therapies over the synthetic systems and identify the key challenges that need to be overcome before these EV-miRNA therapies can be available commercially.
2.2. miRNA
miRNAs are small non-coding, single-stranded molecules, discovered for the first time in Caenorhabditis elegans in 1993 [14]. They are used for inhibiting translation by either blocking mRNA or causing degradation of mRNA [15], based upon complementarity of mRNA-miRNA sequence. During miRNA biogenesis, RNA Pol II transcribes primary miRNA (~150 nt). Drosha cleaves primary-miRNA into precursor miRNA (~70 nt), which is exported to the cytosol. Dicer catalyzes precursor-miRNA into mature miRNA (~25 nt). Further, miRNA can be recruited by RNA-induced Silencing Complex (RISC) for silencing mRNA through binding to its 3’ UTR, 5’UTR, or intron [15]. The mechanism through which miRNA is able to silence mRNA is similar to siRNA-mediated silencing, which makes them good candidates for EV mediated therapies. miRNA is small in size and low in weight so it can be transfected efficiently in cells generally and in EVs specifically. Moreover, pathologies such as cancer, inflammation etc., exhibit alteration in the levels of miRNAs, either in upregulation or downregulation. Thus, administering exogenous miRNAs via EVs to restore the normal levels may alleviate the disorders. miRNAs were found as cargo (along with other proteins, lipids, RNA, etc.) in exosomes secreted from T cells to target immune-presenting cells, for influencing translation. This emphasized the presence of the physiological importance of miRNA in exosomes [16]. However, mechanistic details of packaging miRNA within EV remains elusive with a debate going on about whether the sorting is a passive or active phenomenon.
Since miRNAs can remain stable when they are encapsulated in EVs, they are able to travel long distance in body fluids, e.g., blood, without being degraded by extracellular nucleases. The miRNA remains intact functionally in the recipient cells. Therefore, it makes them a good candidate for EV-based therapies. The potential of these miRNA and related molecules are a big hope for future EV-based therapies, despite the fact that, currently, the problem of off-target effects and specific recognition of target mRNA, is still unresolved.
3. Progress in miRNA-Enriched EV Therapies
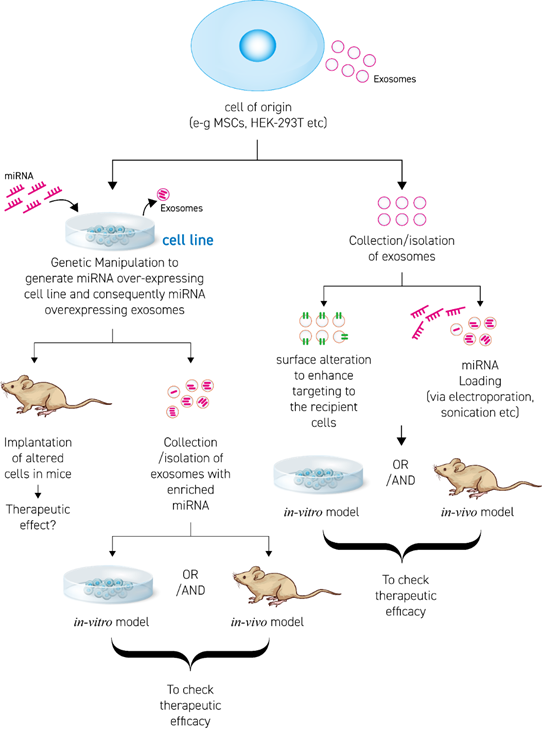
Enrichment/loading of miRNA in EVs is accomplished by two approaches (Figure 2). The first approach is producing a cell line over-expressing (o/e) the desired therapeutic miRNA. The o/e cell line then exhibits a high concentration of miRNA in their cytosol, followed by EV secretion with enclosed therapeutic miRNA. The second approach is isolating EVs from source (cell line or body fluids) and loading them with the miRNA of choice by using chemical or physical methods/techniques. In Figure 2, some aspects of designing appropriate EV-based miRNA therapies are discussed.
Figure 2. Scheme to develop and engineer therapeutic-miRNA EV based therapies.
3.1. Transfection
Since it is well established that enhancing concentration of miRNA in cytosol may increase their passive loading in EVs, it is possible to transfect miRNA of choice into cells to design EV-therapy. One of the prerequisites of transfection is the selection of appropriate cell type. Until now, Mesenchymal Stem Cells (MSCs) and Adipose derived stem cells (ADSCs) are used commonly as ‘biofactories’ to produce EV with loaded miRNA of choice [17].
MSCs can be obtained from various sources, e.g., bone marrow, adipose tissue, umbilical cord, etc. They are naturally involved in the local maintenance and homeostasis of these microenvironments. The tissue-source of MSCs determine the kind of exosomes obtained from it. Overall, MSCs are easy to isolate and expand in-vitro. Therefore, their secreted exosomes can be good candidates for therapeutic purpose [16]. An example is bone marrow derived MSC transfected with miR-29b for healing injured spinal cord in rats via exosomes encapsulated miR-29b [18].
However, it is difficult to use MSCs to obtain high yield of EVs for therapeutic purpose. Moreover, in personalized therapies, obtaining MSCs from aged subjects is difficult due to a lower number of MSCs in their bodies. This implies that the cell system should be chosen while keeping in mind the purpose of miRNA-loading, including the disease under consideration, the communication dynamics between EV-producing cells and the recipient cell, EV-secretion rate, and the ability of EVs to uptake exogenous therapeutic miRNAs [19].
Other cell systems using the same principle are developed for large yields. One example is represented by immortalized CEVEC’s amniocytic production line (CAP) cells. CAP cells were able to produce functional exosomes, and were also able to combine active and passive mechanisms of miRNA loading for EV-enrichment (miR-493 and miR-744). Both contained motifs in 3p strand, which helped them to load in EVs. Further, they can unload their cargo in ovarian cancer cells (SKOV3), thus, down-regulating mRNA targets [20]. Additionally, the immortalized cell line from tumor cells can provide high yield of EVs, but cannot be used for therapeutic purpose due to safety issues [19].
Kojima et al. has reported HEK-293T and a human mesenchymal stem cell system that exploits exosome biogenesis, RNA packaging, secretion, targeting, delivering into cytosol used for treating Parkinson’s disease in in-vitro and in-vivo models. However, the system was used to assess functional mRNA delivery rather than miRNA [21]. Improvement in these cell systems may provide options for transfecting cells to secrete therapeutic-miRNA enriched EVs.
Electroporation is also used for transfection to introduce miRNA in EVs. The process involves using voltage and pulse to open up pores in EVs so that nucleic acids, such as miRNA, can get entry to vesicles, followed by healing of these pores [22]. The miRNA-enriched EVs can be used to target the recipient cells to cure diseases. However, EVs from different sources require different voltage/pulse due to different membrane protein composition. Moreover, there are some problems to the process of electroporation: (i) contamination of reagent [8], (ii) aggregation of EVs [23], (iii) aggregation of miRNA [23], (iv) leakage of endogenous cargo when pores in EVs open. Leakage of endogenous cargo, including miRNA and proteins during electroporation, may cause functional abnormalities when the cargo is delivered to the recipient cells. Nevertheless, the need of specialized equipment makes it difficult to perform. Pomatto et al. addressed the problems by introducing cel-39, miR-451a and miR-31-5p in human plasma-derived EVs through electroporation, followed by successful functional delivery in tumor endothelial cells (TEC). These miRNAs remained functional in hepatocellular carcinoma cell lines and induced apoptosis [24]. In addition to small RNAs (including siRNA and miRNA), miRNA mimics and miRNA inhibitors can be loaded in EVs via electroporation. There are chances that the yield of miRNA loaded in EVs would be over-estimated because of aggregation. Future technical improvements are required to provide solutions to the problems stated and refine the technique for future interventions [25].
3.2. Surface Modification and Membrane Proteins
Proteins on the surface of exosomes are generally representative of parental cells. These proteins include tetraspanins (CD-81, -82, -37, and CD-63), membrane trafficking proteins, cytoskeletal proteins, and two members of Endosomal Sorting Complex Required for Transport (ESCRT) pathway, i.e., Alix and TSG-101 (Tumor Susceptibility Gene 101). The proteins have limited tendency to target specific tissue. Moreover, these proteins allow exosomes to intrinsically accumulate in the liver, kidney, and spleen. Further, they can be excreted out via bile, renal filtration, and reticuloendothelial phagocytosis [26]. Therefore, for enhancing specific targeting and decreasing the clearance rate, it is highly recommended to modify the surface of exosomes. This is possible by modifying surface membrane proteins by direct methods or by genetic alteration of exosome membrane proteins.
Direct modification of exosome surface may occur via non-covalent or covalent methods. The non-covalent method involves mixing protein with exosomes. On the other hand, the covalent method requires attaching a peptide with covalent linkage. Studies have shown covalent attachment of the c(RGDyK) peptide on the exosome surface for specifically targeting avb3 on the cerebral vascular endothelial cell to suppress apoptosis and inflammatory response in ischemia, and deliver curcumin encapsulated as cargo [27]. However, the questions of how valid are these techniques for designing targeted therapies as miRNA-enriched EVs is open for further investigation. Direct methods do not require sophisticated purification. However, both direct methods (i.e., covalent and non-covalent) may have chemical contamination and variable efficiency of modification. Moreover, non-covalent attachment may undergo detachment in physiological conditions [28].
Genetic alteration involves modulating cells to produce a particular protein on the exosome surface. For example, HEK293 cells are genetically altered to produce GE11 peptide (YHWYGYTPQNVI) over-expressed on the secreted exosomes. GE11 is less mitogenic than EGF, therefore they are clinically safer to target cells with EGFR rather than EGF. These GE11 expressing exosomes can bind to EGFR on EGFR-positive breast cancer cells in mice, delivering their let-7 cargo as treatment strategy [29]. Bellavia et al. prepared exosomes from HEK293 cells carrying BCR-ABL siRNA and expressing IL-3L on their surface. These IL-3L expressing exosomes can target IL3-R expressing blast cells in Chronic Myelogenous Leukemia (CML) to induce recovery of CML [30]. Genetic modification produces more uniform population and stable target specificity. It is more expensive than a direct method. Furthermore, it raises safety concerns and, thus, is a disadvantage for clinical applications [29].
3.3. RNA Binding proteins (RBPs)
miRNA sorting in exosomes of endogenous systems is facilitated by RNA binding proteins. RBP can directly bind to miRNA via RBP-recognizing sequences or via unidentified mechanisms. For example, hnRNPA2B1 recognizes GGAG/UGCA motif in miR-198 and miR-601 to sort them in exosomes. Silencing of this protein decreases miRNA in exosomes by 13%, demonstrating its positive regulatory role in exosomal sorting of miR-198 and miR-601. Moreover, it is important to consider the post-translational modification of RBP, e.g., sumoylation [31][32]. Another RBP, i.e. SYNCRIP, associates with miR-3470 and miR-194-2-3p via GGCU, and sorts them in exosomes. Its silencing via shRNA led to decrease in miRNA concentration. SYNCRIP association with miRNAs is ensured via its domain namely, N-terminal unit for RNA Recognition (NURR). Removal of this N-terminus domain hinders miRNA binding and exosomal loading [33]. In addition, RBPs, such as hnRNPH1, might be involved in negatively regulating entry of miRNA in exosomes. Its silencing led to increase in exosomal RNA, leading to the hypothesis that hnRNPH1 may be involved in sequestering miRNA in cytosol [34]. Moreover, indirect recognition of miRNAs by RBP takes place via intermediate protein. An example is adaptor related protein complex-2 mediated interaction of MEX3C and miRNA-451a [34]. Sometimes, more than one RBP are involved in sorting miRNA. For instance caveolin-1 assists sorting of miRNA along with hnRNPA2B1 [35].
Methods used to identify RNA-binding proteins from exosomes may miss some RBPs because of their low quantity in exosomes or due to binding to very few miRNAs. Moreover, systems used to identify RBP (i.e., knock-down and over-expression) cannot distinguish between primary and indirect role of RBP in miRNA sorting. To overcome this problem, studies have demonstrated the use of cell-free system to understand the role of YBX-1 to sort miRNA [36]. Despite the presence of gaps in our knowledge about function of RBPs, it is speculated that these proteins are involved in maintenance of RNA in exosome or detached to target other RNA in recipient cells. However, the mechanistic details are not clear. Further studies are required to integrate all of these concepts about RBP mediated loading of miRNA in exosomes. This will pave the way for utilization of RNA binding proteins in developing and improving miRNA-enriched EV therapies.
References
- Kowal, J.; Arras, G.; Colombo, M.; Jouve, M.; Morath, J.P.; Primdal-Bengtson, B.; Dingli, F.; Loew, D.; Tkach, M.; Théry, C. Proteomic comparison defines novel markers to characterize heterogeneous populations of extracellular vesicle subtypes. Proc. Natl. Acad. Sci. USA 2016, 113, E968–E977.
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7.
- Busatto, S.; Zendrini, A.; Radeghieri, A.; Paolini, L.; Romano, M.; Presta, M.; Bergese, P. The nanostructured secretome. Biomater. Sci. 2019, 17, 39–63.
- Chen, Y.; Lin, E.; Chiou, T.; Harn, H.J. Exosomes in clinical trial and their production in compliance with good manufacturing practice. Tzu Chi Med. J. 2020, 32, 113–120.
- Villarroya-beltri, C.; Baixauli, F.; Gutiérrez-vázquez, C. Seminars in Cancer Biology Sorting it out: Regulation of exosome loading. Semin. Cancer Biol. 2014, 28, 3–13.
- O’Brien, K.; Breyne, K.; Ughetto, S.; Laurent, L.C.; Breakefield, X.O. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat. Rev. Mol. Cell Biol. 2020.
- Elsharkasy, O.M.; Nordin, J.Z.; Hagey, D.W.; de Jong, O.G.; Schiffelers, R.M.; Andaloussi, S.L.; Vader, P. Extracellular vesicles as drug delivery systems: Why and how? Adv. Drug Deliv. Rev. 2020.
- De Jong, O.G.; Kooijmans, S.A.A.; Murphy, D.E.; Jiang, L.; Evers, M. J. W.; Sluijter, J.P.G.; Vader, P.; Schiffelers, R.M. Drug Delivery with Extracellular vesicles: From imagination to innovation. Acc. Chem. Res. 2019, 52, 1761-1770.
- Smyth, T.; Kullberg, M.; Malik, N.; Smith-Jones, P.; Graner, M.; Anchordoquy, T. Biodistributiona and Delivery efficiency of unmodified tumor-derived exosomes. J. Control. Release 2016, 176, 139–148.
- Kooijmans, S.A.A.; Fliervoet, L.A.L.; Van Der Meel, R.; Fens, M.H.A.M.; Heijnen, H.F.G.; Van Bergen En Henegouwen, P.M.P.; Vader, P.; Schiffelers, R.M. PEGylated and targeted extracellular vesicles display enhanced cell specificity and circulation time. J. Control. Release 2016, 224, 77–85.
- Alvarez-Erviti, L.; Seow, Y.; Yin, H.; Betts, C.; Lakhal, S.; Wood, M.J.A. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol. 2011, 29, 341–345.
- Liu, Y.; Li, D.; Liu, Z.; Zhou, Y.; Chu, D.; Li, X.; Jiang, X.; Hou, D.; Chen, X.; Chen, Y.; et al. Targeted exosome-mediated delivery of opioid receptor Mu siRNA for the treatment of morphine relapse. Sci. Rep. 2015, 5, 1–10.
- Shin, H.; Park, S.-J.; Yim, Y.; Kim, J.; Choi, C.; Won, C.; Min, D.-H. Recent Advances in RNA Therapeutics and RNA Delivery Systems Based on Nanoparticles. Adv. Ther. 2018, 1, 1800065.
- Feinbaum, R.; Ambros, V.; Lee, R. The C. elegans Heterochronic Gene lin-4 Encodes Small RNAs with Antisense Complementarity to lin-14. Cell 2004, 116, 843–854.
- Dykxhoorn, D.M.; Novina, C.D.; Sharp, P.A. Killing the messenger: Short RNAs that silence gene expression. Nat. Rev. Mol. Cell Biol. 2003, 4, 457–467.
- Mittelbrunn, M.; Gutiérrez-Vázquez, C.; Villarroya-Beltri, C.; González, S.; Sánchez-Cabo, F.; González, M.Á.; Bernad, A.; Sánchez-Madrid, F. Unidirectional transfer of microRNA-loaded exosomes from T cells to antigen-presenting cells. Nat. Commun. 2011, 2.
- Griswold, M.D.; Oatley, J. Concise Review: Defining Characteristics of Mammalian Spermatogenic Stem cells. Stem Cells 2013, 31, 8–11.
- Yu, T.; Zhao, C.; Hou, S.; Zhou, W.; Wang, B.; Chen, Y. Exosomes secreted from miRNA-29b-modified mesenchymal stem cells repaired spinal cord injury in rats. Braz. J. Med. Biol. Res. 2019, 52, 1–8.
- Bang, O.Y.; Kim, E.H. Mesenchymal Stem Cell-Derived Extracellular Vesicle Therapy for Stroke: Challenges and Progress. Front. Neurol. 2019, 10, 1–11.
- Zeh, N.; Schneider, H.; Mathias, S.; Raab, N.; Kleemann, M.; Schmidt-Hertel, S.; Weis, B.; Wissing, S.; Strempel, N.; Handrick, R.; et al. Human CAP cells represent a novel source for functional, miRNA-loaded exosome production. PLoS ONE 2019, 14, 1–17.
- Kojima, R.; Bojar, D.; Rizzi, G.; El Hamri, G.C.; El-Baba, M.D.; Saxena, P.; Ausländer, S.; Tan, K.R.; Fussenegger, M. Designer exosomes produced by implanted cells intracerebrally deliver therapeutic cargo for Parkinson’s disease treatment. Nat. Commun. 2018, 9.
- Hood, J.L.; Scott, M.J.; Wickline, S.A. Maximizing exosome colloidal stability following electroporation. Anal. Biochem. 2014, 448, 41–49.
- Johnsen, K.B.; Gudbergsson, J.M.; Skov, M.N.; Christiansen, G.; Gurevich, L.; Moos, T.; Duroux, M. Evaluation of electroporation-induced adverse effects on adipose-derived stem cell exosomes. Cytotechnology 2016, 68, 2125–2138.
- Pomatto, M.A.C.; Bussolati, B.; D’Antico, S.; Ghiotto, S.; Tetta, C.; Brizzi, M.F.; Camussi, G. Improved Loading of Plasma-Derived Extracellular Vesicles to Encapsulate Antitumor miRNAs. Mol. Methods Clin. Dev. 2019, 13, 133–144.
- Zhang, D.; Lee, H.; Zhu, Z.; Minhas, J.K.; Jin, Y. Enrichment of selective miRNAs in exosomes and delivery of exosomal miRNAs in vitro and in vivo. Am. J. Physiol. Lung Cell. Mol. Physiol. 2016, 312, L110–L121.
- Xitong, D.; Xiaorong, Z. Targeted therapeutic delivery using engineered exosomes and its applications in cardiovascular diseases. Gene 2016, 575, 377–384.
- Tian, T.; Zhang, H.X.; He, C.P.; Fan, S.; Zhu, Y.L.; Qi, C.; Huang, N.P.; Xiao, Z.D.; Lu, Z.H.; Tannous, B.A.; et al. Surface functionalized exosomes as targeted drug delivery vehicles for cerebral ischemia therapy. Biomaterials 2018, 150, 137–149.
- Hu, Q.; Su, H.; Li, J.; Lyon, C.; Tang, W.; Wan, M.; Hu, T.Y. Clinical applications of exosome membrane proteins. Precis. Clin. Med. 2020, 3, 54–66.
- Ohno, S.I.; Takanashi, M.; Sudo, K.; Ueda, S.; Ishikawa, A.; Matsuyama, N.; Fujita, K.; Mizutani, T.; Ohgi, T.; Ochiya, T.; et al. Systemically injected exosomes targeted to EGFR deliver antitumor microrna to breast cancer cells. Mol. Ther. 2013, 21, 185–191.
- Bellavia, D.; Raimondo, S.; Calabrese, G.; Forte, S.; Cristaldi, M.; Patinella, A.; Memeo, L.; Manno, M.; Raccosta, S.; Diana, P.; et al. Interleukin 3- receptor targeted exosomes inhibit in vitro and in vivo Chronic Myelogenous Leukemia cell growth. Theranostics 2017, 7, 1333–1345.
- Villarroya-Beltri, C.; Gutiérrez-Vázquez, C.; Sánchez-Cabo, F.; Pérez-Hernández, D.; Vázquez, J.; Martin-Cofreces, N.; Martinez-Herrera, D.J.; Pascual-Montano, A.; Mittelbrunn, M.; Sánchez-Madrid, F. Sumoylated hnRNPA2B1 controls the sorting of miRNAs into exosomes through binding to specific motifs. Nat. Commun. 2013, 4, 1–10.
- Lee, H.; Li, C.; Zhang, Y.; Zhang, D.; Otterbein, L.E.; Jin, Y. Caveolin-1 selectively regulates microRNA sorting into microvesicles after noxious stimuli. J. Exp. Med. 2019, 216, 2202–2220.
- Santangelo, L.; Giurato, G.; Cicchini, C.; Montaldo, C.; Mancone, C.; Tarallo, R.; Battistelli, C.; Alonzi, T.; Weisz, A.; Tripodi, M. The RNA-Binding Protein SYNCRIP Is a Component of the Hepatocyte Exosomal Machinery Controlling MicroRNA Sorting. Cell Rep. 2016, 17, 799–808.
- Statello, L.; Maugeri, M.; Garre, E.; Nawaz, M.; Wahlgren, J.; Papadimitriou, A.; Lundqvist, C.; Lindfors, L.; Colle, A.; Sunnerhagen, P.; et al. Identification of RNA-binding proteins in exosomes capable of interacting with different types of RNA: RBP-facilitated transport of RNAs into exosomes. PLoS ONE 2018, 13, e0195969.
- Lu, P.; Li, H.; Li, N.; Singh, R.N.; Bishop, C.E.; Chen, X.; Lu, B. MEX3C interacts with adaptor-related protein complex 2 and involves in miR-451a exosomal sorting. PLoS ONE 2017, 12, 1–25.
- Lin, F.; Zeng, Z.; Song, Y.; Li, L.; Wu, Z.; Zhang, X.; Li, Z.; Ke, X. YBX-1 mediated sorting of miR-133 into derived exosomes to increase fibroblast angiogenesis and MEndoT. Stem Cell Res. Ther. 2019, 10, 263.