Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yingbin Xue | -- | 3914 | 2022-08-25 06:43:00 | | | |
| 2 | Sirius Huang | Meta information modification | 3914 | 2022-08-26 02:32:29 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Xue, Y.; Zhu, S.; Schultze-Kraft, R.; Liu, G.; Chen, Z. Metabolisms Responsive to Nutrient Deficiencies in Crops. Encyclopedia. Available online: https://encyclopedia.pub/entry/26471 (accessed on 06 June 2026).
Xue Y, Zhu S, Schultze-Kraft R, Liu G, Chen Z. Metabolisms Responsive to Nutrient Deficiencies in Crops. Encyclopedia. Available at: https://encyclopedia.pub/entry/26471. Accessed June 06, 2026.
Xue, Yingbin, Shengnan Zhu, Rainer Schultze-Kraft, Guodao Liu, Zhijian Chen. "Metabolisms Responsive to Nutrient Deficiencies in Crops" Encyclopedia, https://encyclopedia.pub/entry/26471 (accessed June 06, 2026).
Xue, Y., Zhu, S., Schultze-Kraft, R., Liu, G., & Chen, Z. (2022, August 25). Metabolisms Responsive to Nutrient Deficiencies in Crops. In Encyclopedia. https://encyclopedia.pub/entry/26471
Xue, Yingbin, et al. "Metabolisms Responsive to Nutrient Deficiencies in Crops." Encyclopedia. Web. 25 August, 2022.
Copy Citation
Crop growth and yield often face sophisticated environmental stresses, especially the low availability of mineral nutrients in soils, such as deficiencies of nitrogen, phosphorus, potassium, and others. Thus, it is of great importance to understand the mechanisms of crop response to mineral nutrient deficiencies, as a basis to contribute to genetic improvement and breeding of crop varieties with high nutrient efficiency for sustainable agriculture.
metabolome
crop metabolism
nutrient deficiency
metabolites
1. N Deficiency
As N is one of the most important macronutrients for crop growth and development, its deficiency severely decreases crop biomass, inhibits chlorophyll content, and disrupts photosynthesis and photorespiration, ultimately limiting crop yield [1][2][3]. A series of physiological and molecular mechanisms underlying crop adaptation to N deficiency have been demonstrated, such as coordinating carbon (C) and N metabolisms, regulating root architecture, modulating phytohormone signaling, enhancing N uptake and translocation, and accumulating stress tolerance-related compounds [4][5][6][7][8][9]. Since total N content and crop growth are affected by N limitation, metabolome analysis has been performed to identify N deficiency responsive metabolites and metabolic pathways, dissecting the adaptive mechanisms through regulation of metabolic profiles in many crops, such as rice (Oryza sativa), maize (Zea mays), wheat (Triticum aestivum), barley (Hordeum vulgare), soybean (Glycine max), tomato (Solanum lycopersicum), and rapeseed (Brassica napus) [10][11][12][13][14][15][16][17]. Many of the identified DAMs can be integrated into specific metabolic pathways regulated by low-N stress (Figure 1).
N deficiency has been shown to significantly decrease photosynthetic rate [14][18]. Several photosynthesis-related genes and proteins have been found to be downregulated by N deficiency [10][19], which is closely related to the accumulation of carbohydrates under N deprivation [20]. A variety of sugars, including fructose, galactose, glucose, sucrose, and maltose, are markedly increased in N-deficient leaves of barley according to metabolome analysis [21]. A similar result has been reported in apple leaves, where several carbohydrates related to C metabolism, such as glucose-6-P, fructose-6-P, and glycerate-3-P, are increased by N deficiency [18]. The accumulation of carbohydrates is believed to act as a key signal to fine-tune the decrease in photosynthesis in plant leaves during N limitation. Consistent with the reduction in photosynthesis, the tricarboxylic acid (TCA) cycle is also inhibited in leaves during low-N stress [10][18][22]. For example, intermediate metabolites involved in the TCA cycle, such as 2-oxoglutarate, citrate, isocitrate, succinate, fumarate, and malate, are decreased in tomato leaves under N-deficient conditions [23].
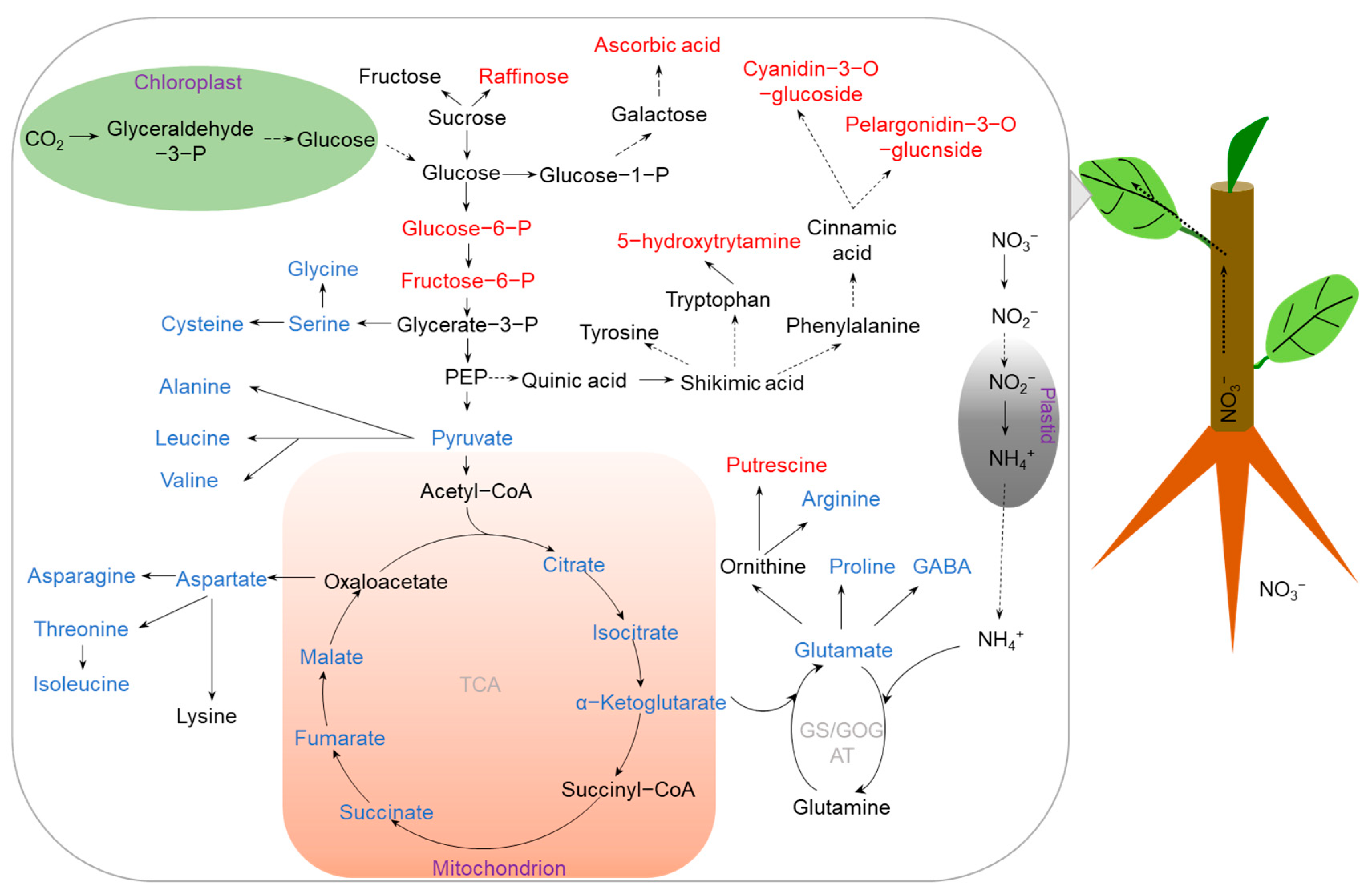

Figure 1. Metabolic pathways changes in crop leaves under N deficiency. N deficiency inhibits the N assimilation pathway and TCA cycle, resulting in large decreases in amino acids, while it accumulates tolerance-related metabolites for reactive oxygen species (ROS) scavenging. The accumulated and reduced metabolites are marked in red and blue, respectively. Abbreviations: P (phosphate), PEP (phosphoenolpyruvic acid), GABA (γ-aminobutyric acid), TCA (tricarboxylic acid), GS/GOGAT (glutamine synthetase/glutamate synthetase).
It has been demonstrated that N deficiency is bound to affect N metabolism. For example, the concentrations of free amino acids were decreased by 12.5% in leaves of rice exposed to low-N treatment [14]. In maize, a set of amino acids, such as glutamate, asparagine, alanine, serine, and glycine, were all decreased in leaves under N deficiency [10]. Similarly, under N-deficient conditions, most amino acids, including aspartic acid, lysine, glycine, threonine, asparagine, and glutamine, were decreased in barley leaves [21]. Interestingly, the decreased amino acid metabolites may be attributed to the downregulated glutamine synthetase (GS) and glutamine aminotransferase (GOGAT) genes, which are involved in the conversion of ammonium to amino acids [18]. Furthermore, integrated analyses of transcriptomics and metabolomics in rice showed that the NADH-dependent glutamate synthase (OsGLT1) gene is downregulated by N deficiency, which is consistent with the decreases in glutamate concentration [14]. Similar results have also been obtained in leaves of soybean and tomato where amino acids were decreased by N deficiency [11][15]. The reduction in amino-acid levels under low-N stress is considered as an energy-saving strategy. On the basis of the above results, it is reasonable to propose that a crop can adjust the balance between C and N metabolism to avoid metabolic inefficiencies and maintain crop growth under N deprivation.
Similar to other abiotic stresses, N deficiency also causes the generation of reactive oxygen species (ROS), resulting in lipid peroxidation and triggering oxidative stress in crops if not well scavenged. For example, the content of H2O2 is significantly increased in apple leaves subjected to low-N treatment, generating oxidative stress [18]. In addition to induce the activities of antioxidant enzymes to eliminate ROS damage, the other adaptive change that alleviates oxidative stress during N deficiency in plants is the accumulation of stress tolerance-related metabolites. Among these metabolites, galactinol, raffinose, sugar alcohols, ascorbic acid, and polyamines have been considered as ROS scavengers [24][25]. It was reported that ascorbic acid, putrescine, and 5-hydroxytryptamine were greatly accumulated in barley shoots [21], which are beneficial for the tolerance of barley to low-N stress. Secondary metabolites are proposed to be important resistance substances produced by plants during long-term adaptation to environmental stress. Metabolome studies showed that flavonoid-related metabolites, such as cinnamic acid, dihydroquercetin, pelargonidin-3-O-glucoside, and cyanidin-3-O-glucoside, were increased by N deficiency in apple [18], which is likely to protect cells from oxidative stress damage. Furthermore, under N limitation, β-alanine levels were found to be increased in leaves of rapeseed [17]. Increases in the levels of osmoprotectants, such as β-alanine, proline, and γ-aminobutyric acid (GABA), are generally associated with enhanced low-N stress tolerance in plants [26], but the exact roles of these metabolites in different crops remain to be investigated.
Another strategy for increasing low-N stress tolerance can be achieved by promoting root elongation under N deficiency. Thus, metabolic profile changes in roots can reveal the mechanisms underlying adaptation of a crop to N deficiency. For example, plant hormones are found to play an important role in regulating root growth under low-N stress [17]. The concentrations of gibberellic acid (GA) in rapeseed roots were significantly increased under N deficiency, which may contribute to promoting root growth [17]. In addition to phytohormones, increasing C partitions to roots is also necessary to increase root growth [27][28]. In contrast to leaves, the levels of metabolites involved in the TCA cycle were increased in apple and soybean roots under N deficiency [15][18], which may promote root growth through enhancing energy accumulation under N-deficient conditions. On the contrary, the contents of alanine, aspartic acid, isoleucine, serine, and threonine were found to be decreased in low-N-tolerant soybean roots, indicating that low-N-tolerant soybean may adapt to N deficiency by reducing energy consumption [15]. Malate, related to the TCA cycle, was found to be increased in roots under N deficiency [18]. Since dehydrogenation of malate is accompanied by the generation of NADH, which is an important antioxidant, the increased malate concentration in roots is considered as an adaptive mechanism of plant tolerance to N deficiency by an enhanced antioxidant status [18]. An additional study in soybean showed that the accumulation of malate in roots could also stimulate nitrate uptake under N deficiency [29]. Furthermore, secondary metabolites, such as salicylic acid (SA) and catechol, were increased in soybean roots under N deprivation [15]. SA was found to be involved in increasing N use efficiency of isolated cucumber (Cucumis sativus) cotyledons [30]. Moreover, the shikimate metabolic pathway-related compounds phenylalanine, shikimic acid, SA, naringin, and neohesperidin also increased in soybean roots during N deficiency [15], which may contribute to the synthesis of aromatic amino acids, plant hormones, and a variety of important active secondary metabolites, increasing tolerance to stress conditions [31][32]. Furthermore, the levels of raffinose and galactitol in roots were higher than those in shoots of barley [21]; the authors concluded that roots were more affected by low-N stress than shoots. A comparison of amino-acid metabolites in common soybean with the low-N-tolerant soybean genotype Tongyu06311 showed that proline was accumulated in roots of the low-N-tolerant soybean genotype Tongyu06311, which is probably beneficial for soybean adapted to low-N stress [15]. Thus, metabolism adjustments are essential for crops in response to N deficiency.
2. P Deficiency
P is a key component of nucleic acids, proteins, and membrane lipids, and it is essential for many biological processes in plants [33][34][35]. Low P availability in soils is a major constraint for crop production. In past decades, there have been large advances in dissecting the mechanisms of plant adaptation to P deficiency including physiological and biochemical responses. Plants have developed a variety of adaptive strategies, such as changing root architecture and morphology, increasing the secretion of organic acids, and developing a bypass pathway for recycling internal P [19][36][37]. Metabolome analysis has also been widely conducted to investigate the metabolite-based low-P tolerance mechanisms in crops, such as soybean, quinoa (Chenopodium quinoa), common bean (Phaseolus vulgaris), tomato, and oats (Avena sativa) [11][38][39][40][41][42]. To date, numerous metabolites have been identified to be involved in the responses of crops to P deficiency. The DAMs can be integrated into specific pathways associated with lipids, flavonoids, amino acids, and nucleotide metabolisms, shedding light on the changes in crop responses to low-P stress (Figure 2). These findings provide major insights into understanding the mechanisms of low-P stress tolerance through metabolic modulation.
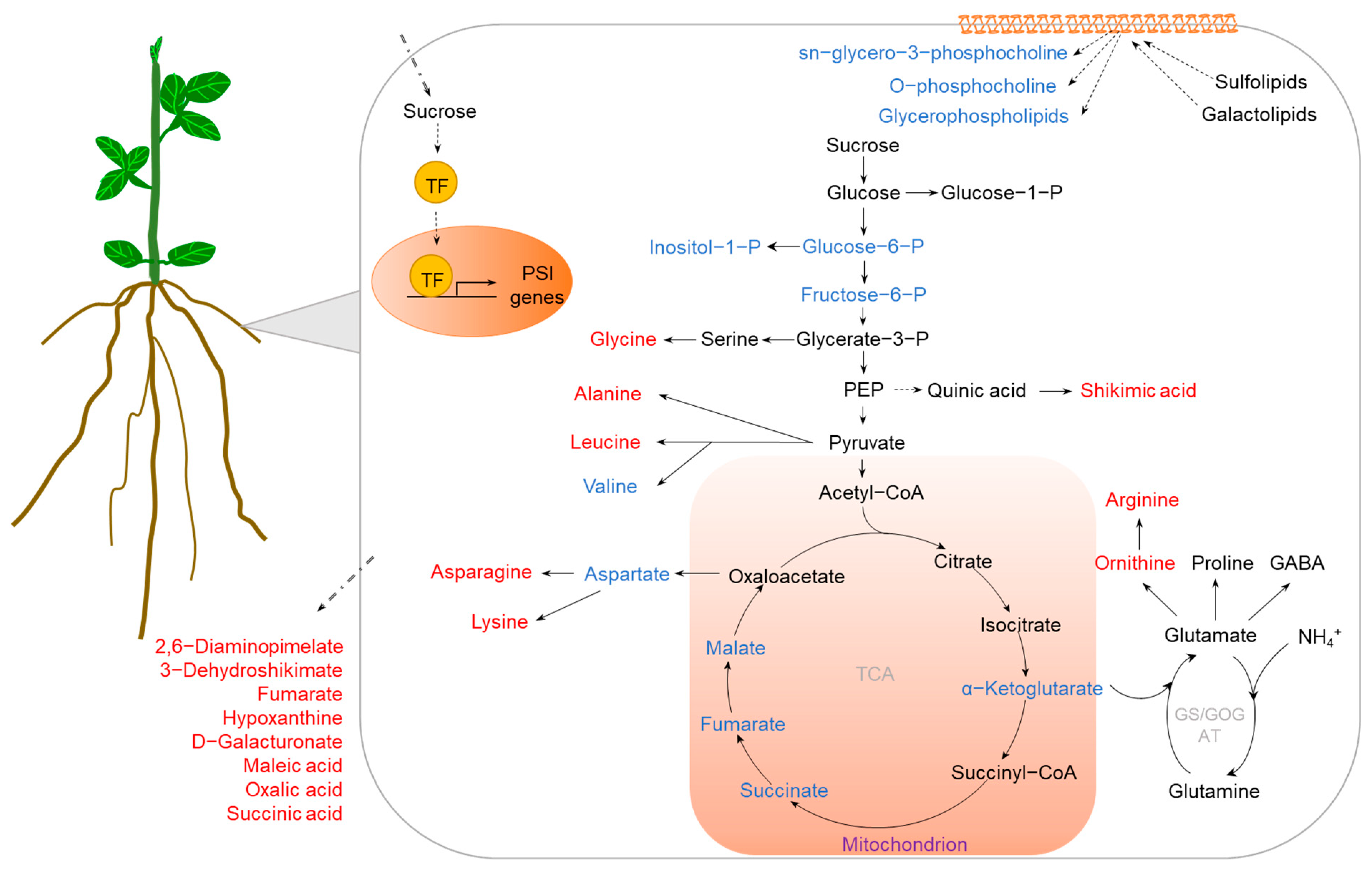
Figure 2. Metabolic pathways regulated by P deficiency in crop roots. The specific changes in metabolites involve organic acids and phospholipids, which may be related to external Pi acquisition and internal P reutilization, respectively. The accumulated and reduced metabolites are marked in red and blue, respectively. Abbreviations: PSI (phosphate starvation-induced), P (phosphate), PEP (phosphoenolpyruvic acid), GABA (γ-aminobutyric acid), TCA (tricarboxylic acid), GS/GOGAT (glutamine synthetase/glutamate synthetase).
Modifying root growth and increasing the root-to-shoot ratio are key adaptive mechanisms to enhance phosphate (Pi) acquisition efficiency for plants under low-P stress. Transcriptomic and proteomic analyses have been conducted to identify key genes or proteins involved in the regulation of root architecture and morphology in response to P deficiency [41]. Metabolites involved in root development regulation have also been identified through a metabolomic approach [11][38][40][41]. Both C and N metabolisms have been reported to be modulated in response of crops to P deficiency. Most amino-acid metabolites, including asparagine, lysine, histidine, ornithine, isoleucine, leucine, and arginine, were found to be accumulated in P-deprived roots of several crops, such as common bean, tomato, and soybean [11][38][41]. Furthermore, it was found that the increase in amino-acid concentration may be due to the upregulation of protein degradation-related genes and the downregulation of protein synthesis-related genes under P deficiency [43][44]. During low-P stress, plants can increase C distribution to the root system, thereby increasing the root-to-shoot ratio and regulating the root system morphology. Significant increases in maltose, sucrose, raffinose, and 6-kestose were observed in barley roots under 17 days of low-P treatment [45]; the authors considered this an adaptive mechanism of plants by promoting root growth through regulating C allocation. In addition, sugar has been documented to be an important sensor for the Pi starvation response; the expression of phosphate starvation-induced (PSI) genes was found to be regulated by sugar limitation [46]. Thus, increases in sugar levels in roots may induce the expression of PSI genes, regulating plant growth under low-P stress. However, further characterization of sugar and PSI genes is needed to confirm their exact roles in low-P stress tolerance via regulating C allocation in plants.
In addition to root growth regulation for acquiring Pi, crop roots can exudate organic acids into the rhizosphere to promote solubilization of fixed Pi [17][47]. It has been found that organic acids have an important role in the response of plants to Pi starvation. For example, metabolome analysis of the exudates from rice roots revealed that organic acids, such as 2,6-diaminopimelate, 3-dehydroshikimate, fumarate, hypoxanthine, and d-galacturonate, were increased by P deficiency [13], which may contribute to the mobilization of insoluble soil P, as suggested by the authors. Furthermore, significant increases in the exudation of malic, oxalic, and succinic acids were observed in the P-efficient wheat genotype RAC875 [48]. On the other hand, metabolome analysis has shown that internal organic acids in roots are also affected by P deficiency. The levels of organic acids, such as tartaric acid and 2,4-dihydroxybutanoic acid, in roots of common bean were found to be decreased during low-P stress [39]. Similar results were also obtained in barley roots exposed to low-P treatment, where the levels of several organic acids, including α-ketoglutarate, succinate, fumarate, and malate, were reduced [45]. Therefore, organic acids secreted to the rhizosphere may lead to the reduction in organic acids in roots under P deficiency. An increase in organic acid exudation from roots is one of the important physiological mechanisms for crops increasing Pi utilization from soils.
On the other hand, promoting the remobilization of internal P resources, such as phosphorylated metabolites, nucleic acids, and phospholipids, which are well known as the largest P pool in plants [49], is necessary for crop adaptation to P deficiency. Under P-limited conditions, the levels of phosphorylated metabolites were reported to be decreased in soybean roots, including sn-glycero-3-phosphocholine, O-phosphocholine, deoxyribose 5-phosphate, O-phosphorylethanolamine, and dl-glyceraldehyde 3-phosphate [41]. Similar results were also found in oats where glucose-6-phosphate and myo-inositol phosphate were dramatically decreased in P-deficient roots [40]. Moreover, nucleotides, such as adenosine 3′-monophosphate, inosine 5′-monophosphate, guanosine 5′-monophosphate, uridine 5′-diphospho-D-glucose, guanosine monophosphate, adenosine 5′-monophosphate, deoxyribose 5-phosphate, cytidine 5′-monophosphate, uridine 5′-monophosphate, and guanosine 3′,5′-cyclic monophosphate, were decreased by Pi starvation in soybean roots [41]. Decreases in nucleic acid concentration were also observed in white lupin under Pi starvation [50]. The regulation of the synthesis and/or degradation of nucleotides is likely to help a crop cope with P deficiency. Recently, a key gene, DNA polymerase delta 1 (DPD1), involved in organelle DNA degradation for improving P use efficiency, was characterized in Arabidopsis [51]. Several DPD1 homologs in soybean were also found to be upregulated in roots under P deficiency [41]. These results support the hypothesis that changes in nucleotide metabolism are beneficial for increasing internal P remobilization, thereby improving P utilization efficiency. Furthermore, lipid-related metabolites such as glycerophospholipids were found to be decreased in responses of crops to P deficiency [41][52]. For example, in soybean roots, sn-glycero-3-phosphocholine, O-phosphocholine, and several glycerophospholipids, all of which are involved in remodeling membrane lipids, were decreased under P-deficient conditions [41]. Replacing phospholipids with sulfolipids or galactolipids in bio-membranes can also help plant tolerance to low-P stress; this deserves further investigation.
In contrast to roots, increased accumulation of sucrose, maltose, raffinose, and 6-kestose was observed mainly in shoots of barley growing under moderately P-deficient conditions [45], indicating that barley roots are less sensitive to Pi starvation. Furthermore, amino acids in legume nodules are also significantly affected by P deficiency. For example, five out of 10 amino-acid metabolites were decreased, whereas three out of 10 amino-acid metabolites were increased in nodules of common bean [39]. N metabolism-related metabolites, including spermidine, putrescine, urea, glycine, serine, glutamine, and threonine, were reduced in nodules of common bean under P deficiency, which may lead to a decrease in symbiotic nitrogen fixation [39]. However, the mechanism of metabolite changes in nodules under low-P stress requires to be studied further.
3. K Deficiency
Among the macronutrients, K plays essential roles in plant growth and development as a major cation or as a cofactor of various enzymes. Unlike N and P, K is not a part of organic compounds, but plays important roles in many physiological and biochemical processes, such as enzyme activation, ion homeostasis, osmoregulation, and protein synthesis [53][54]. Generally, the availability of K in soils is limited, which has become a limiting factor for sustainable production of cultivated crops [55]. Recently, metabolomic approaches have been applied to dissect the mechanism of crop tolerance to K deficiency; examples include tomato (Solanum lycopersicum), sunflower (Helianthus annuus), barley (Hordeum vulgare), rapeseed (Brassica napus), and peanut (Arachis hypogaea) [11][21][56][57][58][59]. Many of the identified DAMs can be integrated into specific metabolic pathways regulated by K deficiency stress (Figure 3).
It is generally believed that carbohydrate metabolism not only is an important energy source for plants, but also plays a vital role in protein and lipid metabolisms [60]. Increases in the content of sugars, such as glucose, sucrose, and fructose, are suggested to be associated with plants in response to various stresses, including K deficiency [61]. Sugar levels have been reported to be increased in both leaves and roots of barley under K deficiency [21][57]. Accumulation of sucrose was also found in tomato roots under low-K stress [11]. Furthermore, low-K-tolerant barley genotypes seemed to accumulate more sugars in both leaves and roots than low-K-sensitive barley genotypes [57], indicating that increasing sugar accumulation is critical for barley adaptation to low-K stress. In addition, sucrose is an important signaling molecule that is transferred from leaves to roots, regulating root growth in response to nutrient stress [10][39]. Since K is involved in the loading of sucrose to the phloem, availability of K seriously affects the transport of sucrose from leaves to roots [62][63]. Therefore, under K-deficient conditions, sucrose in roots is not only an important substance for low-K tolerance, but also a key indicator to screen crops for tolerance to K limitation. It has been documented that N metabolism is affected by K deficiency; according to metabolome analysis, amino acids in leaves and roots of barley were increased during K limitation [57]. Metabolomic analysis also showed that tryptophan, guanidineacetic acid, asparagine, alanine, ornithine, and histidine were all increased in K-deficient wheat roots, while citric acid, glutamic acid, and GABA were decreased [64]. Interestingly, most of the increased amino acids were positively charged, whereas the negatively charged amino acids were reduced in both leaves and roots of barley [57]. Since K deficiency could lead to electric charge imbalance, it is important to maintain charge balance in plant cells to cope with low-K stress. The phenylpropanoid metabolic pathway is one of the most important secondary metabolic pathways in plants [65]. Within this pathway, l-phenylalanine can be catalyzed into trans-Cinnamic acid, which is a key substrate for the synthesis of flavonoids, lignin, and alkaloids [66]. Metabolome analysis revealed that, under K-deficient conditions, l-phenylalanine levels in a low-K-tolerant barley genotype were higher than those in a low-K-sensitive barley genotype [57], suggesting that regulation of the phenylpropanoid metabolic pathway can contribute to barley coping with low-K stress.
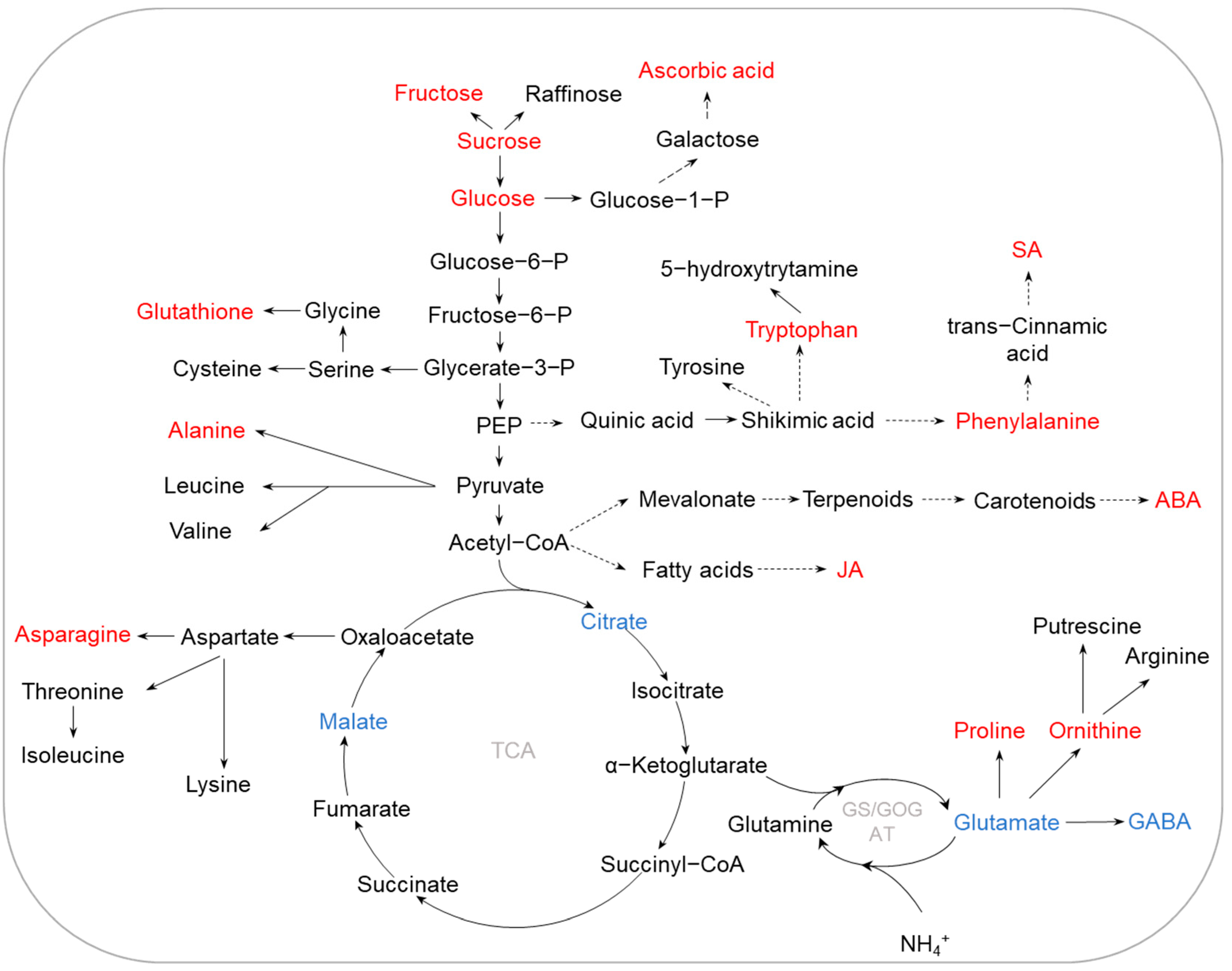

Figure 3. Metabolic pathways regulated by K deficiency in crops. K deficiency affects diverse pathways, including N metabolism, TCA cycle, glycolysis, shikimic acid pathway, and secondary metabolic pathways, accumulating stress-tolerant metabolites and phytohormones. The accumulated and reduced metabolites are marked in red and blue, respectively. Abbreviations: SA (salicylic acid), JA (jasmonic acid), ABA (abscisic acid), GABA (γ-aminobutyric acid), TCA (tricarboxylic acid), GS/GOGAT (glutamine synthetase/glutamate synthetase).
K deficiency also causes an excess accumulation of reactive oxygen species (ROS), resulting in oxidative stress in plants [67]. Thus, increasing the concentration of antioxidant metabolites is a vital stress tolerance strategy for plants dealing with K deprivation. The accumulation of compatible solutes, such as proline, soluble sugars, amino acids, and polyols, plays an important role in osmotic adjustment [68]. Among them, proline is regarded as an important antioxidant for stress tolerance [69]. There is evidence that K deficiency increases the concentration of proline in both leaves and roots of barley; for example, leaves of the low-K-tolerant cultivar XZ153 contained higher proline levels than those of the sensitive cultivar XZ141 [57]. Similarly, increases in proline concentration were observed in K-deprived leaves and roots of peanut [59]. In addition, ascorbic acid is an important antioxidant protecting cell membrane permeability [70]. The concentration of ascorbic acid in barley roots was found to be increased during low-K stress, especially in the low-K-tolerant cultivar XZ153. Furthermore, ascorbic acid concentrations were increased in leaves of the low-K-tolerant barley cultivar XZ153, but decreased in the low-K-sensitive barley cultivar XZ141 [57]. In addition, glutathione is also a key antioxidant involved in scavenging ROS via the GSH-ascorbate cycle [71]. Metabolome analysis showed that the content of glutathione was increased in roots of the low-K-tolerant wheat cultivar KN9204 but not in low-K-sensitive cultivar BN207 [64]. Thus, it is reasonable to propose that antioxidant metabolites, such as proline, ascorbic acid, and glutathione, are important metabolites for crop adaptation to K deficiency, although further investigation is required.
Phytohormones are small endogenous signaling molecules that participate in regulating plant growth and development in various life stages and stress conditions. Metabolites related to phytohormones, such as abscisic acid (ABA), jasmonic acid (JA), and SA, are regulated by K deficiency. ABA is well known as a stress signal in response to drought, salinity, and nutrient limitation [72]. It can maintain the water relation by regulating stomatal conductance and plant metabolism [73]. JA is involved in abiotic stress through activation of antioxidant systems, synthesis of amino acids and sugars, and regulation of stomatal opening and closing [74]. SA is involved in protecting membrane integrity and modulating abundance of protein associated with secondary metabolites [59]. It was shown that, in both leaves and roots of peanut, K deficiency increased the levels of ABA [59]. Similarly, JA concentration in leaves of peanut also increased during low-K stress [59]. Unlike ABA and JA, SA concentration increased in leaves of peanut but decreased in roots under K-limited conditions [59]. Therefore, considering the importance of phytohormones in plant growth, it is reasonable to suggest that ABA, JA, SA, and other phytohormones are important molecules for low-K stress tolerance.
4. Other Nutrient Deficiencies
Despite the advances in identifying various metabolites and metabolic pathways responding to N, P, and K deficiency, little attention has been given to metabolic changes in response of crops to deficiencies of other essential nutrients, such as magnesium (Mg), iron (Fe), zinc (Zn), sulfur (S), and boron (B).
Mg is an important component of chlorophyll and a cofactor for enzymes participating in many physiological processes [75]. It has been reported that Mg deficiency leads to large differentiated metabolic processes in source and sink tissues. For example, Mg deficiency led to leaf-specific accumulation of amino-acid metabolites in soybean, such as phenylalanine, asparagine, leucine, isoleucine, glycine, glutamine, and serine; in contrast, root-specific depletion of pyruvic acid, citrate, 2-keto-glutaric acid, succinic acid, fumaric acid, and malate were observed under Mg deficiency [76]. Mg deficiency also impaired C allocation in soybean, as reflected by significant increases in carbohydrates, such as starch, sucrose, glucose, and fructose in leaves, and moderate decreases in sucrose and starch in roots [76]. These results suggest that reprogramming of distinct C and N metabolisms may occur in the response of soybean leaves and roots to Mg limitation.
Fe is the fourth most common element in the Earth’s crust, and it is easily fixed into insoluble Fe3+ precipitates, leading to low availability for plants [77]. Fe limitation affects several metabolic processes, such as photosynthesis and respiration, as well as leads to an increase in ROS [78]. In rice, glycolysis and respiration-related metabolites, such as 3-P-glycerate, 3-P-glycerate derivatives, branched-chain amino acids, and pyruvate derivatives, were found to be increased in roots during low-Fe stress [78]. Furthermore, an increase in phytosiderophore 2′-deoxymugineic acid was observed in rice roots under Fe deficiency [78]. These results suggest that changes in C and energy metabolisms and increasing 2′-deoxymugineic acid secretion are important adaptive mechanisms of rice dealing with Fe deficiency. In addition, in leaves of the betel palm (Areca catechu), significant increases in naringenin, butin, and hesperetin but decreases in xanthohumol, purine, and N-p-coumaroylspermidine were observed under Fe deficiency [79], suggesting that regulating biosynthesis of flavonoids and flavonols is an important adaptive strategy for the betel palm in response to Fe deficiency.
In tea (Camellia sinensis) plants, Zn deficiency reduced the contents of two secondary metabolites, four carbohydrate metabolites, and four nitrogenous metabolites in leaves [80], indicating that tea plants respond to Zn-deficient stress through regulating carbohydrate, nitrogenous, and secondary metabolisms. Recently, several secondary metabolites, such as sesquiterpene lactones, caffeoyl derivatives, caffeic acid hexose, 5-caffeoylquinic acid, quercetin, and luteolin glucoside derivatives, were found to be regulated by S deficiency in leaves of lettuce (Lactuca sativa) [81]. Furthermore, in alfalfa (Medicago sativa), B deficiency increased the accumulation of sugars and phenolic compounds in flowers and seeds, respectively, which may cause abscission or abortion of reproductive organs [82].
Although the results above provide some useful information on the changes in metabolic profiles of crops in response to deficiencies of Mg, Zn, Fe, S, and B, more studies in these areas are needed to increase the understanding of the metabolic mechanisms of crop adaptation.
References
- Lu, C.; Zhang, J. Photosynthetic CO2 assimilation, chlorophyll fluorescence and photoinhibition as affected by nitrogen deficiency in maize plants. Plant Sci. 2000, 151, 135–143.
- Cruz, J.L.; Mosquim, P.R.; Pelacani, C.R.; Araujo, W.L.; DaMatta, F.M. Photosynthesis impairment in cassava leaves in response to nitrogen deficiency. Plant Soil 2003, 257, 417–423.
- Li, H.; Hu, B.; Chu, C. Nitrogen use efficiency in crops: Lessons from Arabidopsis and rice. J. Exp. Bot. 2017, 68, 2477–2488.
- Xu, G.; Fan, X.; Miller, A.J. Plant nitrogen assimilation and use efficiency. Annu. Rev. Plant Biol. 2012, 63, 153–182.
- Amtmann, A.; Armengaud, P. Effects of N, P, K and S on metabolism: New knowledge gained from multi-level analysis. Curr. Opin. Plant Biol. 2009, 12, 275–283.
- Kant, S.; Bi, Y.M.; Rothstein, S.J. Understanding plant response to nitrogen limitation for the improvement of crop nitrogen use efficiency. J. Exp. Bot. 2011, 62, 1499–1509.
- Guo, T.; Xuan, H.; Yang, Y.; Wang, L.; Wei, L.; Wang, Y.; Kang, G. Transcription analysis of genes encoding the wheat root transporter NRT1 and NRT2 families during nitrogen starvation. J. Plant Growth Regul. 2014, 33, 837–848.
- Lv, X.; Zhang, Y.; Hu, L.; Zhang, Y.; Zhang, B.; Xia, H.; Du, W.; Fan, S.; Kong, L. Low nitrogen stress stimulates lateral root initiation and nitrogen assimilation in wheat: Roles of phytohormone signaling. J. Plant Growth Regul. 2020, 40, 436–450.
- Sun, X.; Chen, F.; Yuan, L.; Mi, G. The physiological mechanism underlying root elongation in response to nitrogen deficiency in crop plants. Planta 2020, 251, 84.
- Schlüter, U.; Mascher, M.; Colmsee, C.; Scholz, U.; Brautigam, A.; Fahnenstich, H. Maize source leaf adaptation to nitrogen deficiency affects not only nitrogen and carbon metabolism but also control of phosphate homeostasis. Plant Physiol. 2012, 160, 1384–1406.
- Sung, J.; Lee, S.; Lee, Y.; Ha, S.; Song, B.; Kim, T.; Waters, B.M.; Krishnan, H.B. Metabolomic profiling from leaves and roots of tomato (Solanum lycopersicum L.) plants grown under nitrogen, phosphorus or potassium-deficient condition. Plant Sci. 2015, 241, 55–64.
- Quan, X.; Qian, Q.; Ye, Z.; Zeng, J.; Han, Z.; Zhang, G. Metabolic analysis of two contrasting wild barley genotypes grown hydroponically reveals adaptive strategies in response to low nitrogen stress. J. Plant Physiol. 2016, 206, 59–67.
- Tawaraya, K.; Horie, R.; Wagatsuma, T.; Saito, K.; Oikawa, A. Metabolite profiling of shoot extract, root extract, and root exudate of rice under nitrogen and phosphorus deficiency. Soil Sci. Plant Nutr. 2018, 64, 312–322.
- Xin, W.; Zhang, L.; Zhang, W.; Gao, J.; Yi, J.; Zhen, X.; Li, Z.; Zhao, Y.; Peng, C.; Zhao, C. An integrated analysis of the rice transcriptome and metabolome reveals differential regulation of carbon and nitrogen metabolism in response to nitrogen availability. Int. J. Mol. Sci. 2019, 20, 2349.
- Liu, Y.; Xue, Y.; Xie, B.; Zhu, S.; Lu, X.; Liang, C.; Tian, J. Complex gene regulation between young and old soybean leaves in responses to manganese toxicity. Plant Physiol. Biochem. 2020, 155, 231–242.
- Wang, Y.; Wang, D.; Tao, Z.; Yang, Y.; Gao, Z.; Zhao, G.; Chang, X. Impacts of nitrogen deficiency on wheat (Triticum aestivum L.) grain during the medium filling stage: Transcriptomic and metabolomic comparisons. Front. Plant Sci. 2021, 12, 674433.
- Shen, X.; Yang, L.; Han, P.; Gu, C.; Li, Y.; Liao, X.; Qin, L. Metabolic profiles reveal changes in the leaves and roots of rapeseed (Brassica napus L.) seedlings under nitrogen deficiency. Int. J. Mol. Sci. 2022, 23, 5784.
- Sun, T.; Zhang, J.; Zhang, Q.; Li, X.; Li, M.; Yang, Y.; Zhou, J.; Wei, Q.; Zhou, B. Integrative physiological, transcriptome, and metabolome analysis reveals the effects of nitrogen sufficiency and deficiency conditions in apple leaves and roots. Environ. Exp. Bot. 2021, 192, 104633.
- Liang, C.; Tian, J.; Liao, H. Proteomics dissection of plant responses to mineral nutrient deficiency. Proteomics 2013, 13, 624–636.
- Sugiura, D.; Betsuyaku, E.; Terashima, I. Interspecific differences in how sink-source imbalance causes photosynthetic downregulation among three legume species. Ann. Bot. 2019, 123, 715–726.
- Zhao, H.; Ni, S.; Cai, S.; Zhang, G. Comprehensive dissection of primary metabolites in response to diverse abiotic stress in barley at seedling stage. Plant Physiol. Biochem. 2021, 161, 54–64.
- Li, M.X.; Xu, J.S.; Wang, X.X.; Fu, H.; Zhao, M.L.; Wang, H.; Shi, L.X. Photosynthetic characteristics and metabolic analyses of two soybean genotypes revealed adaptive strategies to low-nitrogen stress. J. Plant Physiol. 2018, 229, 132–141.
- Urbanczyk-Wochniak, E.; Fernie, A.R. Metabolic profiling reveals altered nitrogen nutrient regimes have diverse effects on the metabolism of hydroponically-grown tomato (Solanum lycopersicum) plants. J. Exp. Bot. 2005, 56, 309–321.
- Keunen, E.; Peshev, D.; Vangronsveld, J. Plant sugars are crucial players in the oxidative challenge during abiotic stress: Extending the traditional concept. Plant Cell Environ. 2013, 36, 1242–1255.
- Shi, H.; Chan, Z. Improvement of plant abiotic stress tolerance through modulation of the polyamine pathway. J. Integr. Plant Biol. 2014, 56, 114–121.
- Ashraf, M.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216.
- Hermans, C.; Hammond, J.P.; White, P.J.; Verbruggen, N. How do plants respond to nutrient shortage by biomass allocation? Trends Plant Sci. 2006, 11, 610–617.
- Feng, B.; Xu, C.Y. Advance of studies on the mechanism of effect of light on carbon and nitrogen allocation in plants. J. Jilin Agr. Sci. 2014, 39, 18–22.
- Touraine, B.; Muller, B.; Grignon, C. Effect of phloem-translocated malate on NO3− uptake by roots of intact soybean plants. Plant Physiol. 1992, 99, 1118–1123.
- Singh, P.K.; Chaturvedi, V.K. Effects of salicylic acid on seedling growth and nitrogen use efficiency in cucumber (Cucumis sativus L.). Giorn. Bot. Ital. 2010, 146, 302–308.
- Dai, H.; Xiao, C.N.; Liu, H.B.; Hao, F.H.; Tang, H.R. Combined NMR and LC-DAD-MS analysis reveals comprehensive metabonomic variations for three phenotypic cultivars of Salvia miltiorrhiza bunge. J. Proteome Res. 2010, 9, 1565–1578.
- Becerra-Moreno, A.; Redondo-Gil, M.; Benavides, J.; Nair, V.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. Combined effect of water loss and wounding stress on gene activation of metabolic pathways associated with phenolic biosynthesis in carrot. Front. Plant Sci. 2015, 6, 837.
- Zhou, M.; Zhu, S.; Mo, X.; Guo, Q.; Li, Y.; Tian, J.; Liang, C. Proteomic analysis dissects molecular mechanisms underlying plant responses to phosphorus deficiency. Cells 2022, 11, 651.
- Chiou, T.J.; Lin, S.I. Signaling network in sensing phosphate availability in plants. Annu. Rev. Plant Biol. 2011, 62, 185–206.
- Lambers, H. Phosphorus acquisition and utilization in plants. Annu. Rev. Plant Biol. 2022, 73, 17–42.
- Ajmera, I.; Hodgman, T.C.; Lu, C. An integrative systems perspective on plant phosphate research. Genes 2019, 10, 139.
- Mo, X.; Liu, G.; Zhang, Z.; Lu, X.; Liang, C.; Tian, J. Mechanisms underlying soybean response to phosphorus deficiency through integration of omics analysis. Int. J. Mol. Sci. 2022, 23, 4592.
- Hernández, G.; Ramírez, M.; Valdés-López, O.; Tesfaye, M.; Graham, M.A.; Czechowski, T.; Schlereth, A.; Wandrey, M.; Erban, A.; Cheung, F.; et al. Phosphorus stress in common bean: Root transcript and metabolic responses. Plant Physiol. 2007, 144, 752–767.
- Hernández, G.; Valdés-López, O.; Ramírez, M.; Goffard, N.; Weiller, G.; Aparicio-Fabre, R.; Fuentes, S.I.; Erban, A.; Kopka, J.; Udvardi, M.K.; et al. Global changes in the transcript and metabolic profiles during symbiotic nitrogen fixation in phosphorus-stressed common bean plants. Plant Physiol. 2009, 151, 1221–1238.
- Wang, Y.; Lysøe, E.; Armarego-Marriott, T.; Erban, A.; Paruch, L.; van Eerde, A.; Bock, R.; Liu-Clarke, J. Transcriptome and metabolome analyses provide insights into root and root-released organic anion responses to phosphorus deficiency in oat. J. Exp. Bot. 2018, 69, 3759–3771.
- Mo, X.; Zhang, M.; Liang, C.; Cai, L.; Tian, J. Integration of metabolome and transcriptome analyses highlights soybean roots responding to phosphorus deficiency by modulating phosphorylated metabolite processes. Plant Physiol. Biochem. 2019, 139, 697–706.
- Wang, Q.; Guo, Y.; Huang, T.; Zhang, X.; Zhang, P.; Xie, H.; Liu, J.; Li, L.; Kong, Z.; Qin, P. Transcriptome and metabolome analyses revealed the response mechanism of quinoa seedlings to different phosphorus stresses. Int. J. Mol. Sci. 2022, 23, 4704.
- Wu, P.; Ma, L.; Hou, X.; Wang, M.; Wu, Y.; Liu, F.; Deng, X.W. Phosphate starvation triggers distinct alterations of genome expression in Arabidopsis roots and leaves. Plant Physiol. 2003, 132, 1260–1271.
- Misson, J.; Raghothama, K.G.; Jain, A.; Jouhet, J.; Block, M.A.; Bligny, R.; Ortet, P.; Creff, A.; Somerville, S.; Rolland, N.; et al. A genome-wide transcriptional analysis using Arabidopsis thaliana Affymetrix gene chips determined plant responses to phosphate deprivation. Proc. Natl. Acad. Sci. USA 2005, 102, 11934–11939.
- Huang, C.Y.; Roessner, U.; Eickmeier, I.; Genc, Y.; Callahan, D.L.; Shirley, N.; Langridge, P.; Bacic, A. Metabolite profiling reveals distinct changes in carbon and nitrogen metabolism in phosphate-deficient barley plants (Hordeum vulgare L.). Plant Cell Physiol. 2008, 49, 691–703.
- Liu, J.; Samac, D.A.; Bucciarelli, B.; Allan, D.L.; Vance, C.P. Signaling of phosphorus deficiency-induced gene expression in white lupin requires sugar and phloem transport. Plant J. 2005, 41, 257–268.
- Dong, D.; Peng, X.; Yan, X. Organic acid exudation induced by phosphorus deficiency and/or aluminum toxicity in two contrasting soybean genotypes. Physiol. Plant. 2004, 122, 190–199.
- Nguyen, V.L.; Palmer, L.; Roessner, U.; Stangoulis, J. Genotypic variation in the root and shoot metabolite profiles of wheat (Triticum aestivum L.) indicate sustained, preferential carbon allocation as a potential mechanism in phosphorus efficiency. Front. Plant Sci. 2019, 10, 995.
- Veneklaas, E.J.; Lambers, H.; Bragg, J.; Finnegan, P.M.; Lovelock, C.E.; Plaxton, W.C.; Price, C.A.; Scheible, W.; Shane, M.W.; White, P.J.; et al. Opportunities for improving phosphorus-use efficiency in crop plants. New Phytol. 2012, 195, 306–320.
- Müller, J.; Gödde, V.; Niehaus, K.; Zörb, C. Metabolic adaptations of white lupin roots and shoots under phosphorus deficiency. Front. Plant Sci. 2015, 6, 1014.
- Takami, T.; Ohnishi, N.; Kurita, Y.; Iwamura, S.; Ohnishi, M.; Kusaba, M.; Mimura, T.; Sakamoto, W. Organelle DNA degradation contributes to the efficient use of phosphate in seed plants. Nat. Plants 2018, 4, 1044–1055.
- Plaxton, W.C.; Tran, H.T. Metabolic adaptations of phosphate-starved plants. Plant Physiol. 2011, 156, 1006–1015.
- Pettigrew, W.T. Potassium influences on yield and quality production for maize, wheat, soybean and cotton. Physiol. Plant 2008, 133, 670–681.
- Hafsi, C.; Debez, A.; Abdelly, C. Potassium deficiency in plants: Effects and signaling cascades. Acta Physiol. Plant 2014, 36, 1055–1070.
- Wang, Y.; Wu, W.H. Genetic approaches for improvement of the crop potassium acquisition and utilization efficiency. Curr. Opin. Plant Biol. 2015, 25, 46–52.
- Cui, J.; Abadie, C.; Carroll, A.; Lamade, E.; Tcherkez, G. Responses to K deficiency and waterlogging interact via respiratory and nitrogen metabolism. Plant Cell Environ. 2019, 42, 647–658.
- Zeng, J.; Quan, X.; He, X.; Cai, S.; Ye, Z.; Chen, G.; Zhang, G. Root and leaf metabolite profiles analysis reveals the adaptive strategies to low potassium stress in barley. BMC Plant Biol. 2018, 18, 187.
- Hu, W.; Lu, Z.; Meng, F.; Li, X.; Cong, R.; Ren, T.; Lu, J. Potassium modulates central carbon metabolism to participate in regulating CO2 transport and assimilation in Brassica napus leaves. Plant Sci. 2021, 307, 110891.
- Patel, M.; Fatnani, D.; Parida, A.K. Potassium deficiency stress tolerance in peanut (Arachis hypogaea) through ion homeostasis, activation of antioxidant defense, and metabolic dynamics: Alleviatory role of silicon supplementation. Plant Physiol. Biochem. 2022, 182, 55–75.
- Rolland, F.; Baena-Gonzalez, E.; Sheen, J. Sugar sensing and signaling in plants: Conserved and novel mechanisms. Annu. Rev. Plant Biol. 2006, 57, 675–709.
- Rosa, M.; Prado, C.; Podazza, G.; Interdonato, R.; González, J.A.; Hilal, M.; Prado, F.E. Soluble sugars-metabolism, sensing and abiotic stress: A complex network in the life of plants. Plant Signal Behav. 2009, 4, 388–393.
- Armengaud, P.; Sulpice, R.; Miller, A.J.; Stitt, M.; Amtmann, A.; Gibon, Y. Multilevel analysis of primary metabolism provides new insights into the role of potassium nutrition for glycolysis and nitrogen assimilation in Arabidopsis roots. Plant Physiol. 2009, 150, 772–785.
- White, P.J.; Karley, A.J. Potassium. In Cell Biology of Metals and Nutrients; Hell, R., Mendel, R.R., Eds.; Springer: Berlin, Germany, 2010; pp. 199–224.
- Zhao, Y.; Sun, R.; Liu, H.; Liu, X.; Xu, K.; Xiao, K.; Xue, C. Multi-omics analyses reveal the molecular mechanisms underlying the adaptation of wheat (Triticum aestivum L.) to potassium deprivation. Front. Plant Sci. 2020, 11, 588994.
- Dixon, R.A.; Achnine, L.; Kota, P.; Liu, C.J.; Reddy, M.S.; Wang, L. The phenylpropanoid pathway and plant defence—A genomics perspective. Mol. Plant Pathol. 2002, 3, 371–390.
- Zhang, X.; Liu, C.J. Multifaceted regulations of gateway enzyme phenylalanine ammonia-lyase in the biosynthesis of phenylpropanoids. Mol. Plant 2015, 8, 17–27.
- Hernandez, M.; Fernandez-Garcia, N.; Garcia-Garma, J.; Rubio-Asensio, J.S.; Rubio, F.; Olmos, E. Potassium starvation induces oxidative stress in Solanum lycopersicum L. roots. J. Plant Physiol. 2012, 169, 1366–1374.
- Khan, M.N.; AlSolami, M.A.; Basahi, R.A.; Siddiqui, M.H.; Al-Huqail, A.A.; Abbas, Z.K.; Siddiqui, Z.H.; Ali, H.M.; Khan, F. Nitric oxide is involved in nano-titanium dioxide-induced activation of antioxidant defense system and accumulation of osmolytes under water-deficit stress in Vicia faba L. Ecotoxicol. Environ. Saf. 2020, 190, 110152.
- Szabados, L.; Savouré, A. Proline: A multifunctional amino acid. Trends Plant Sci. 2010, 15, 89–97.
- Shalata, A.; Neumann, P.M. Exogenous ascorbic acid (vitamin C) increases resistance to salt stress and reduces lipid peroxidation. J. Exp. Bot. 2001, 52, 2207–2211.
- Zagorchev, L.; Seal, C.E.; Kranner, I.; Odjakova, M. A central role for thiols in plant tolerance to abiotic stress. Int. J. Mol. Sci. 2013, 14, 7405–7432.
- Peuke, A.D.; Jeschke, W.D.; Hartung, W. Flows of elements, ions and abscisic acid in Ricinus communis and site of nitrate reduction under potassium limitation. J. Exp. Bot. 2002, 53, 241–250.
- Kim, Y.H.; Khan, A.L.; Waqas, M.; Shim, J.K.; Kim, D.H.; Lee, K.Y.; Lee, I.J. Silicon application to rice root zone influenced the phytohormonal and antioxidant responses under salinity stress. J. Plant Growth Regul. 2014, 33, 137–149.
- Wang, J.; Song, L.; Gong, X.; Xu, J.; Li, M. Function of jasmonic acid in plant regulation and response to abiotic stress. Int. J. Mol. Sci. 2020, 21, 1446.
- Verbruggen, N.; Hermans, C. Physiological and molecular responses to magnesium nutritional imbalance in plants. Plant Soil. 2013, 368, 87–99.
- Yang, N.; Jiang, J.; Xie, H.; Bai, M.; Xu, Q.; Wang, X.; Yu, X.; Chen, Z.; Guan, Y. Metabolomics reveals distinct carbon and nitrogen metabolic responses to magnesium deficiency in leaves and roots of soybean . Front. Plant Sci. 2017, 8, 2091.
- Kim, S.A.; Guerinot, M.L. Mining iron: Iron uptake and transport in plants. FEBS Lett. 2007, 581, 2273–2280.
- Selby-Pham, J.; Lutz, A.; Moreno-Moyano, L.T.; Boughton, B.A.; Roessner, U.; Johnson, A.A.T. Diurnal changes in transcript and metabolite levels during the iron deficiency response of rice. Rice 2017, 10, 14.
- Li, J.; Cao, X.; Jia, X.; Liu, L.; Cao, H.; Qin, W.; Li, M. Iron deficiency leads to chlorosis through impacting chlorophyll synthesis and nitrogen metabolism in Areca catechu L. Front. Plant Sci. 2021, 12, 710093.
- Zhang, Y.; Wang, Y.; Ding, Z.; Wang, H.; Song, L.; Jia, S.; Ma, D. Zinc stress affects ionome and metabolome in tea plants. Plant Physiol. Biochem. 2017, 111, 318–328.
- Abdalla, M.A.; Li, F.; Wenzel-Storjohann, A.; Sulieman, S.; Tasdemir, D.; Mühling, K.H. Comparative metabolite profile, biological activity and overall quality of three lettuce (Lactuca sativa L., Asteraceae) cultivars in response to sulfur nutrition. Pharmaceutics 2021, 13, 713.
- Chen, L.; Xia, F.; Wang, M.; Wang, W.; Mao, P. Metabolomic analyses of alfalfa (Medicago sativa L. cv. ‘Aohan’) reproductive organs under boron deficiency and surplus conditions. Ecotoxicol. Environ. Saf. 2020, 202, 111011.
More
Information
Subjects:
Biochemistry & Molecular Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.5K
Revisions:
2 times
(View History)
Update Date:
26 Aug 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





