
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kacper Szczepski | + 1450 word(s) | 1450 | 2020-10-14 07:48:38 | | | |
| 2 | Camila Xu | -16 word(s) | 1434 | 2020-10-20 11:48:29 | | |
Video Upload Options
Antimicrobial peptides (AMPs), also known as host defense peptides (HDPs), are produced by all living matter as a critical part of the innate immune system [1,2,3]. Their existence was discovered in 1939, the year gramicidin was isolated from the bacteria, Bacillus brevis; some resources, however, claim that the discovery of lysozyme in the 1920s should be treated as the first AMP instance, due to lysozyme’s non enzymatic, bactericidal second mode of action [2].
1. Introduction
As of 2019, over 3000 AMPs have been isolated across all kingdoms [1]. Most of them have variable sequence length—from 10 up to 60 amino acid residues, mainly in L-configuration [1]. AMPs are expressed in many cell types in response to the activation of the Toll-like receptor (TLR) signaling pathway [2]. They are predominantly comprised of two types of amino acids residues—cationic residues such as Arg, Lys, and His, and hydrophobic residues (mainly aliphatic and aromatic) [2]. Both cationic and hydrophobic residues engage in the antimicrobial mechanism of action. Furthermore, AMPs can be classified into three groups, based on their structures (Scheme 1): α-helical, β-sheets, and extended peptides. α-helical peptides are the largest group of AMPs with characteristic qualities such as amphipathic properties, and the ability to possess a tertiary structure with a hinge in the middle of a chain [2][1]. β-sheets peptides feature one to five disulfide bridges that help to stabilize their bioactive conformation. Peptides rich in amino acids such as proline, arginine, tryptophan or glycine usually have a linear structure [2][1].
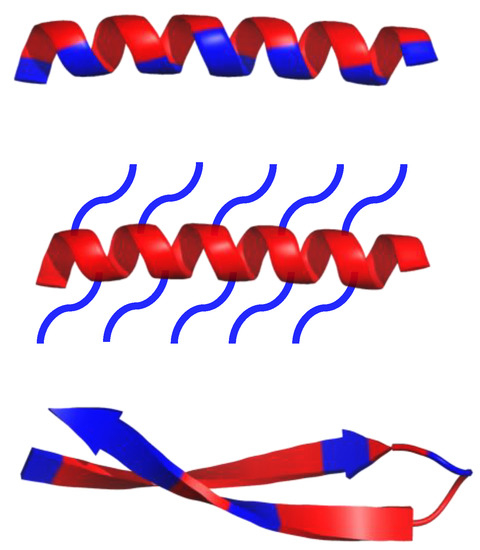
Scheme 1. Schematic representation of α-helical and β-sheets, common secondary structure of amphipathic peptides. Hydrophobic amino acid residues are colored red, while hydrophilic residues are colored blue.
HDPs have a wide range of antibacterial activities, and low susceptibility to antimicrobial resistance [5][6][7][8][9][10][11][12]. Nevertheless, the use of HDPs is limited due to their low resistance to proteases, and their increased cost of preparation [8][13][14]. Different peptides [15][16][17][18], peptoids [19][20][21][22][23], polymethacrylate derivatives [24], polynorbornene derivatives [25], polycarbonate derivatives [26], peptide polymers [27][28][29][30][31][32], and polymers made by controlled living radical polymerization (CLRP) [33] were developed to mimic the antibacterial properties of HDPs, and to ameliorate their shortcomings [34].
The widely developed peptide-drug research has led to the introduction of around 60 FDA approved peptide therapeutics in the market [35]. Over 140 are in clinical trials, and over 500 are in the preclinical stages. Although numerous biomimetic and de novo designed AMPs are at different stages of clinical trials [36][37], only eight AMPs received FDA approval, i.e., daptomycin (approved in 2003) and oritavancin (approved in 2014) [37].
2. Samson's Hair of AMPs
There are three main factors—charge, hydrophobicity and amphipathicity, which determine the activity of AMPs, and they are all related to their intrinsic properties.
The positive charge of AMPs (between 2+ and 9+) is one of the most essential factors responsible for their antibacterial activity [38]. Positively charged AMPs outcompete the binding of native Mg(II) and Ca(II) ions to lipopolysaccharides (LPS), and destabilize the outer membrane of Gram-negative bacteria [39]. Consequently, the destabilized regions enable the peptide to pass into the cell. Once through, AMPs bind to the cytoplasmic membrane and cause depolarization and pore formation, resulting in cell death [40]. In the case of Gram-positive bacteria, AMPs must overcome the outer wall with two main obstacles, peptidoglycan and teichoic acids, before an AMP interacts with the cytoplasmic membrane. AMPs overcome peptidoglycan because it is able to penetrate through the relatively porous peptidoglycan. The porous nature of peptidoglycan makes it easy for small molecules (< 50 kDa) to penetrate through it, and furthermore, it does not possess a negative charge, which could prevent the penetration of positively charged AMPs through it [40][41][42]. To overcome the anionic teichoic acids, they can either act as a ladder that will help positively charged AMPs to travel to the cytoplasmic membrane, or the can act as “cages” that entrap AMPs inside the bacteria, thus reducing their local concentration on the membrane The behaviors of teichoic acids vary based on the type of AMP and the type of bacteria [42]. After crossing the outer wall, AMPs can disrupt the integrity of the cytoplasmic membrane, leading to membrane disruption and dislocation of peripheral membrane proteins [42].
The hydrophobicity of AMPs is another important factor. It is assumed that hydrophobic residues help to insert peptides into the bacterial membrane and further impair the membrane [4][42]. It was also revealed that hydrophobic residues account for 40–60% of all amino acids in AMPs [38]. Moreover, the hydrophobicity is strongly correlated with antimicrobial activity, where increasing the hydrophobicity to a certain level improves antimicrobial activity [1][38]. The relationship between hydrophobicity and antimicrobial activity is also proven to be the case for peptidomimetics such as peptoids, where amino acid side chains are linked to the peptide backbone through the amide nitrogen rather than to the α-carbons [43]. However, a higher hydrophobicity also increases hemolytic activities, resulting in unwanted toxicity to eukaryotic cells [1][38]. Studies with model peptides have shown that a continuous region of 4-6 hydrophobic residues is sufficient to sustain the antimicrobial activity of peptides, while simultaneously reducing the hemolytic activity [44].
The amphipathicity of AMPs, created by the segregation of hydrophobic and polar residues on the opposite sites of the backbone, is another factor related to their activity [1]. Amphipathicity is considered the strongest indicator of AMP activity, and is described by the hydrophobic moment, defined as the vector sum of the hydrophobicity of each amino acid located in the helix [1][45]. The mechanism by, which amphipathicity works is the result of previous factors. The positively charged regions of AMPs enable binding to the anionic phospholipid head groups of the bacterial membrane. Meanwhile, the hydrophobic regions of peptides invade the lipid bilayer and interact with the hydrophobic acyl chains of phospholipids, which results in membrane penetration [45]. Several studies have shown the importance of amphipathicity in the process of AMPs binding to membranes, and the negative impact on activity against Gram-positive bacteria and fungi, when amphipathic character of the peptide was eliminated [13][46][47][48].
3. AMPs: Achilles’ Heel
Despite the recent advancements in AMP research, there are still major challenges to overcome. One of the main obstacles in AMP applications is the proteolytic instability of peptide drugs [49][50]. Peptides are targeted by numerous proteolytic enzymes located in bodily fluids and tissues of the host. Moreover, AMPs can be recognized as an antigen and targeted by host immune system [51]51[52]. One of the countermeasures for their potential immunogenicity could be glycosylating AMPs with a glycosyl profile similar to that of the host. For instance, covering AMPs with polysialic acid (derived from Neisseria meningitidis or E. coli that shares structural mimicry with host cell lectins), or its analogues, can lessen the immunological response [52]. Nevertheless, some bacteria have already developed resistance to AMPs. For example, LL-37, a human antimicrobial peptide, can be digested by proteases synthesized by P. aeruginosa, Enterococcus faecalis, Proteus mirabilis, S. aureus, and S. Pyogenes [50][53]. Synthesizing proteases to target specific antimicrobial peptides is one of the many ways bacteria participate in this evolutionary “arms race”. Other methods used by bacteria to cope with AMPs, including the use of efflux pumps, lowering the binding affinity, or reducing the anionic charge of lipopolysaccharides on their surface, are exhaustively discussed in [50][53].
The high toxicity of AMPs in eukaryotic cells is another one of their shortcomings. This high toxicity can lead to hemolysis, nephrotoxicity, and neurotoxicity [1][55][56][57]. More studies need to focus on the pharmacokinetics and pharmacodynamics of AMPs in order to determine a proper drug dosage that will maintain a balance between positive and negative outcomes [2].
The bioavailability of AMPs is rather low. Peptides are hardly absorbed by the intestinal mucosa, and their pharmacological distribution requires additional financial input. In recent years, much progress has been made in this field—e.g., nano-carriers not only increase the bioavailability, but also lower cytotoxicity and reduce degradation of a compound, leading to increased efficiency [37]. Despite different efforts, the entire process of AMPs design and discovery (costs of synthesis and screening) is relatively high and does not guarantee the expected outcome [58]. The high production cost of AMPs (as for 2006, producing 1 g of peptide using solid-phase peptide synthesis (SPPS) costs USD 100–600) limits the development and testing of new AMPs, even if recent improvements in SPPS lowered the costs of synthesis [8].
AMPs, as other peptide drugs, can induce immune responses and cause allergies [59]. Immunogenicity depends on the host immune system, but is also correlated with the peptide’s dose, duration and frequency of treatment, route of administration, and the patient’s pathologies [60][61][62][63]. Immunogenic response can affect the peptide-drug efficiency and even lead to allergy and/or hypersensitivity [63]. Bering that in mind, it is important to study the immunogenic and allergic activity of AMPs. In vitro tests together with mathematical calculations of allergens similarities, with the use of specific databases of allergenic compounds [64], i.e., http://www.allergome.org, can give nowadays reliable results [65].
References
- Kuppusamy, R.; Willcox, M.; Black, D.S.; Kumar, N. Short cationic peptidomimetic antimicrobials. Antibiotics 2019, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Dou, X.; Song, J.; Lyu, Y.; Zhu, X.; Xu, L.; Li, W.; Shan, A. Antimicrobial peptides: Promising alternatives in the post feeding antibiotic era. Med. Res. Rev. 2019, 39, 831–859. [Google Scholar] [CrossRef] [PubMed]
- Lewies, A.; Du Plessis, L.H.; Wentzel, J.F. Antimicrobial peptides: The Achilles’ heel of antibiotic resistance? Probiotics Antimicrob. Proteins 2019, 11, 370–381. [Google Scholar] [CrossRef] [PubMed]
- Maróti, G.; Kereszt, A.; Kondorosi, E.; Mergaert, P. Natural roles of antimicrobial peptides in microbes, plants and animals. Res. Microbiol. 2011, 162, 363–374. [Google Scholar] [CrossRef]
- Boman, H.G. Antibacterial peptides: Key components needed in immunity. Cell 1991, 65, 205–207. [Google Scholar] [CrossRef]
- Brogden, K.A. Antimicrobial peptides: Pore formers or metabolic inhibitors in bacteria? Nat. Rev. Microbiol. 2005, 3, 238–250. [Google Scholar] [CrossRef]
- Edwards, I.A.; Elliott, A.G.; Kavanagh, A.M.; Blaskovich, M.A.; Cooper, M.A. Structure–Activity and Toxicity Relationships of the Antimicrobial Peptide Tachyplesin-1. ACS Infect. Dis. 2017, 3, 917–926. [Google Scholar] [CrossRef]
- Hancock, R.E.; Sahl, H.-G. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 2006, 24, 1551–1557. [Google Scholar] [CrossRef]
- Lee, E.Y.; Zhang, C.; Di Domizio, J.; Jin, F.; Connell, W.; Hung, M.; Malkoff, N.; Veksler, V.; Gilliet, M.; Ren, P. Helical antimicrobial peptides assemble into protofibril scaffolds that present ordered dsDNA to TLR9. Nat. Commun. 2019, 10, 1–10. [Google Scholar] [CrossRef]
- Ma, Z.; Han, J.; Chang, B.; Gao, L.; Lu, Z.; Lu, F.; Zhao, H.; Zhang, C.; Bie, X. Membrane-active amphipathic peptide WRL3 with in vitro antibiofilm capability and in vivo efficacy in treating methicillin-resistant Staphylococcus aureus burn wound infections. ACS Infect. Dis. 2017, 3, 820–832.
- Mishra, B.; Reiling, S.; Zarena, D.; Wang, G. Host defense antimicrobial peptides as antibiotics: Design and application strategies. Curr. Opin. Chem. Biol. 2017, 38, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Bray, B.L. Large-scale manufacture of peptide therapeutics by chemical synthesis. Nat. Rev. Drug Discov. 2003, 2, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Sieprawska-Lupa, M.; Mydel, P.; Krawczyk, K.; Wójcik, K.; Puklo, M.; Lupa, B.; Suder, P.; Silberring, J.; Reed, M.; Pohl, J. Degradation of human antimicrobial peptide LL-37 by Staphylococcus aureus-derived proteinases. Antimicrob. Agents Chemother. 2004, 48, 4673–4679. [Google Scholar] [CrossRef]
- Choi, S.; Isaacs, A.; Clements, D.; Liu, D.; Kim, H.; Scott, R.W.; Winkler, J.D.; DeGrado, W.F. De novo design and in vivo activity of conformationally restrained antimicrobial arylamide foldamers. Proc. Natl. Acad. Sci. USA 2009, 106, 6968–6973. [Google Scholar] [CrossRef]
- Porter, E.A.; Wang, X.; Lee, H.-S.; Weisblum, B.; Gellman, S.H. Non-haemolytic β-amino-acid oligomers. Nature 2000, 404, 565. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Teng, P.; Sang, P.; She, F.; Wei, L.; Cai, J. γ-AApeptides: Design, structure, and applications. Acc. Chem. Res. 2016, 49, 428–441. [Google Scholar] [CrossRef]
- Wu, H.; Niu, Y.; Padhee, S.; Wang, R.E.; Li, Y.; Qiao, Q.; Bai, G.; Cao, C.; Cai, J. Design and synthesis of unprecedented cyclic γ-AApeptides for antimicrobial development. Chem. Sci. 2012, 3, 2570–2575. [Google Scholar] [CrossRef]
- Bremner, J.B.; Keller, P.A.; Pyne, S.G.; Boyle, T.P.; Brkic, Z.; David, D.M.; Garas, A.; Morgan, J.; Robertson, M.; Somphol, K. Binaphthyl-Based Dicationic Peptoids with Therapeutic Potential. Angew. Chem. Int. Ed. 2010, 49, 537–540. [Google Scholar] [CrossRef]
- Chongsiriwatana, N.P.; Patch, J.A.; Czyzewski, A.M.; Dohm, M.T.; Ivankin, A.; Gidalevitz, D.; Zuckermann, R.N.; Barron, A.E. Peptoids that mimic the structure, function, and mechanism of helical antimicrobial peptides. Proc. Natl. Acad. Sci. USA 2008, 105, 2794–2799. [Google Scholar] [CrossRef]
- Ghosh, C.; Manjunath, G.B.; Akkapeddi, P.; Yarlagadda, V.; Hoque, J.; Uppu, D.S.; Konai, M.M.; Haldar, J. Small molecular antibacterial peptoid mimics: The simpler the better! J. Med. Chem. 2014, 57, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- Olsen, C.A.; Ziegler, H.L.; Nielsen, H.M.; Frimodt-Møller, N.; Jaroszewski, J.W.; Franzyk, H. Antimicrobial, Hemolytic, and Cytotoxic Activities of β-Peptoid–Peptide Hybrid Oligomers: Improved Properties Compared to Natural AMPs. ChemBioChem 2010, 11, 1356–1360. [Google Scholar] [CrossRef] [PubMed]
- Patch, J.A.; Barron, A.E. Helical peptoid mimics of magainin-2 amide. J. Am. Chem. Soc. 2003, 125, 12092–12093. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Poon, Y.F.; Li, W.; Zhu, H.-Y.; Yeap, S.H.; Cao, Y.; Qi, X.; Zhou, C.; Lamrani, M.; Beuerman, R.W. A polycationic antimicrobial and biocompatible hydrogel with microbe membrane suctioning ability. Nat. Mater. 2011, 10, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Ilker, M.F.; Nüsslein, K.; Tew, G.N.; Coughlin, E.B. Tuning the hemolytic and antibacterial activities of amphiphilic polynorbornene derivatives. J. Am. Chem. Soc. 2004, 126, 15870–15875. [Google Scholar] [CrossRef]
- Chin, W.; Zhong, G.; Pu, Q.; Yang, C.; Lou, W.; De Sessions, P.F.; Periaswamy, B.; Lee, A.; Liang, Z.C.; Ding, X. A macromolecular approach to eradicate multidrug resistant bacterial infections while mitigating drug resistance onset. Nat. Commun. 2018, 9, 1–14. [Google Scholar] [CrossRef]
- Chakraborty, S.; Liu, R.; Hayouka, Z.; Chen, X.; Ehrhardt, J.; Lu, Q.; Burke, E.; Yang, Y.; Weisblum, B.; Wong, G.C. Ternary nylon-3 copolymers as host-defense peptide mimics: Beyond hydrophobic and cationic subunits. J. Am. Chem. Soc. 2014, 136, 14530–14535. [Google Scholar] [CrossRef]
- Mowery, B.P.; Lindner, A.H.; Weisblum, B.; Stahl, S.S.; Gellman, S.H. Structure—activity relationships among random nylon-3 copolymers that mimic antibacterial host-defense peptides. J. Am. Chem. Soc. 2009, 131, 9735–9745. [Google Scholar] [CrossRef]
- Qian, Y.-X.; Zhang, D.-F.; Wu, Y.-M.; Chen, Q.; Liu, R.-H. The design, synthesis and biological activity study of nylon-3 polymers as mimics of host defense peptides. ACTA Polym. Sin. 2016, 10, 1300–1311. [Google Scholar]
- Xiong, M.; Lee, M.W.; Mansbach, R.A.; Song, Z.; Bao, Y.; Peek, R.M.; Yao, C.; Chen, L.-F.; Ferguson, A.L.; Wong, G.C. Helical antimicrobial polypeptides with radial amphiphilicity. Proc. Natl. Acad. Sci. USA 2015, 112, 13155–13160. [Google Scholar] [CrossRef]
- Xiong, M.; Han, Z.; Song, Z.; Yu, J.; Ying, H.; Yin, L.; Cheng, J. Bacteria-assisted activation of antimicrobial polypeptides by a random-coil to helix transition. Angew. Chem. Int. Ed. 2017, 56, 10826–10829. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Ma, P.; Xie, J.; Zhang, S.; Xiao, X.; Qiao, Z.; Shao, N.; Zhou, M.; Zhang, W.; Dai, C. Host defense peptide mimicking poly-β-peptides with fast, potent and broad spectrum antibacterial activities. Biomater. Sci. 2019, 7, 2144–2151. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.-K.; Lam, S.J.; Ho, K.K.; Kumar, N.; Qiao, G.G.; Egan, S.; Boyer, C.; Wong, E.H. Rational design of single-chain polymeric nanoparticles that kill planktonic and biofilm bacteria. ACS Infect. Dis. 2017, 3, 237–248. [Google Scholar] [CrossRef]
- Hartlieb, M.; Williams, E.G.; Kuroki, A.; Perrier, S.; Locock, K.E. Antimicrobial Polymers: Mimicking Amino Acid Functionali ty, Sequence Control and Three-dimensional Structure of Host-defen se Peptides. Curr. Med. Chem. 2017, 24, 2115–2140. [Google Scholar] [CrossRef]
- Fosgerau, K.; Hoffmann, T. Peptide therapeutics: Current status and future directions. Drug Discov. Today 2015, 20, 122–128.
- Gong, H.; Zhang, J.; Hu, X.; Li, Z.; Fa, K.; Liu, H.; Waigh, T.A.; McBain, A.; Lu, J.R. Hydrophobic Control of the Bioactivity and Cytotoxicity of de Novo-Designed Antimicrobial Peptides. ACS Appl. Mater. Interfaces 2019, 11, 34609–34620. [Google Scholar] [CrossRef]
- Carratalá, J.V.; Serna, N.; Villaverde, A.; Vázquez, E.; Ferrer-Miralles, N. Nanostructured antimicrobial peptides: The last push towards clinics. Biotechnol. Adv. 2020, 44, 107603. [Google Scholar] [CrossRef]
- Takahashi, D.; Shukla, S.K.; Prakash, O.; Zhang, G. Structural determinants of host defense peptides for antimicrobial activity and target cell selectivity. Biochimie 2010, 92, 1236–1241. [Google Scholar] [CrossRef]
- Clifton, L.A.; Skoda, M.W.; Le Brun, A.P.; Ciesielski, F.; Kuzmenko, I.; Holt, S.A.; Lakey, J.H. Effect of divalent cation removal on the structure of gram-negative bacterial outer membrane models. Langmuir 2015, 31, 404–412. [Google Scholar] [CrossRef]
- Shai, Y. Mode of action of membrane active antimicrobial peptides. Biopolym. Pept. Sci. Sect. 2002, 66, 236–248. [Google Scholar] [CrossRef]
- Malanovic, N.; Lohner, K. Gram-positive bacterial cell envelopes: The impact on the activity of antimicrobial peptides. Biochim. Biophys. Acta Biomembr. 2016, 1858, 936–946. [Google Scholar] [CrossRef] [PubMed]
- Malanovic, N.; Lohner, K. Antimicrobial peptides targeting gram-positive bacteria. Pharmaceuticals 2016, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Andreev, K.; Martynowycz, M.W.; Huang, M.L.; Kuzmenko, I.; Bu, W.; Kirshenbaum, K.; Gidalevitz, D. Hydrophobic interactions modulate antimicrobial peptoid selectivity towards anionic lipid membranes. Biochim. Biophys. Acta BBA Biomembr. 2018, 1860, 1414–1423. [Google Scholar] [CrossRef] [PubMed]
- Blondelle, S.E.; Houghten, R.A. Design of model amphipathic peptides having potent antimicrobial activities. Biochemistry 1992, 31, 12688–12694. [Google Scholar] [CrossRef]
- Eisenberg, D.; Weiss, R.M.; Terwilliger, T.C. The hydrophobic moment detects periodicity in protein hydrophobicity. Proc. Natl. Acad. Sci. USA 1984, 81, 140–144. [Google Scholar] [CrossRef]
- Giangaspero, A.; Sandri, L.; Tossi, A. Amphipathic α helical antimicrobial peptides. A systematic study of the effects of structural and physical properties on biological activity. Eur. J. Biochem. 2001, 268, 5589–5600. [Google Scholar] [CrossRef]
- Iavicoli, P.; Rossi, F.; Lamarre, B.; Bella, A.; Ryadnov, M.G.; Calzolai, L. Modulating charge-dependent and folding-mediated antimicrobial interactions at peptide—lipid interfaces. Eur. Biophys. J. 2017, 46, 375–382. [Google Scholar] [CrossRef]
- Findlay, B.; Zhanel, G.G.; Schweizer, F. Cationic amphiphiles, a new generation of antimicrobials inspired by the natural antimicrobial peptide scaffold. Antimicrob. Agents Chemother. 2010, 54, 4049–4058. [Google Scholar] [CrossRef]
- Otvos, L., Jr.; Wade, J.D. Current challenges in peptide-based drug discovery. Front. Chem. 2014, 2, 62. [Google Scholar] [CrossRef]
- Guilhelmelli, F.; Vilela, N.; Albuquerque, P.; Derengowski, L.; Silva-Pereira, I.; Kyaw, C. Antibiotic development challenges: The various mechanisms of action of antimicrobial peptides and of bacterial resistance. Front. Microbiol. 2013, 4, 353.
- Eckert, R. Road to clinical efficacy: Challenges and novel strategies for antimicrobial peptide development. Future Microbiol. 2011, 6, 635–651. [Google Scholar] [CrossRef] [PubMed]
- Bednarska, N.G.; Wren, B.W.; Willcocks, S.J. The importance of the glycosylation of antimicrobial peptides: Natural and synthetic approaches. Drug Discov. Today 2017, 22, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Schmidtchen, A.; Frick, I.M.; Andersson, E.; Tapper, H.; Björck, L. Proteinases of common pathogenic bacteria degrade and inactivate the antibacterial peptide LL-37. Mol. Microbiol. 2002, 46, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Andersson, D.I.; Hughes, D.; Kubicek-Sutherland, J.Z. Mechanisms and consequences of bacterial resistance to antimicrobial peptides. Drug Resist. Updates 2016, 26, 43–57. [Google Scholar] [CrossRef]
- Gai, Z.; Samodelov, S.L.; Kullak-Ublick, G.A.; Visentin, M. Molecular mechanisms of colistin-induced nephrotoxicity. Molecules 2019, 24, 653. [Google Scholar] [CrossRef]
- Radermacher, S.; Schoop, V.; Schluesener, H. Bactenecin, a leukocytic antimicrobial peptide, is cytotoxic to neuronal and glial cells. J. Neurosci. Res. 1993, 36, 657–662. [Google Scholar] [CrossRef]
- Gordon, Y.J.; Romanowski, E.G.; McDermott, A.M. A review of antimicrobial peptides and their therapeutic potential as anti-infective drugs. Curr. Eye Res. 2005, 30, 505–515. [Google Scholar] [CrossRef]
- Craik, D.J.; Fairlie, D.P.; Liras, S.; Price, D. The future of peptide-based drugs. Chem. Biol. Drug Des. 2013, 81, 136–147. [Google Scholar] [CrossRef]
- Deptuła, M.; Wardowska, A.; Dzierżyńska, M.; Rodziewicz-Motowidło, S.; Pikuła, M. Antibacterial peptides in dermatology–strategies for evaluation of allergic potential. Molecules 2018, 23, 414. [Google Scholar] [CrossRef]
- Corominas, M.; Gastaminza, G.; Lobera, T. Hypersensitivity reactions to biological drugs. J. Investig. Allergol. Clin. Immunol. 2014, 24, 212–225.
- Moussa, E.M.; Panchal, J.P.; Moorthy, B.S.; Blum, J.S.; Joubert, M.K.; Narhi, L.O.; Topp, E.M. Immunogenicity of therapeutic protein aggregates. J. Pharm. Sci. 2016, 105, 417–430. [Google Scholar] [CrossRef] [PubMed]
- Wadhwa, M.; Bird, C.; Dilger, P.; Gaines-Das, R.; Thorpe, R. Strategies for detection, measurement and characterization of unwanted antibodies induced by therapeutic biologicals. J. Immunol. Methods 2003, 278, 1–17. [Google Scholar] [CrossRef]
- Fathallah, A.M.; Bankert, R.B.; Balu-Iyer, S.V. Immunogenicity of subcutaneously administered therapeutic proteins—a mechanistic perspective. AAPS J. 2013, 15, 897–900. [Google Scholar] [CrossRef] [PubMed]
- Mari, A.; Rasi, C.; Palazzo, P.; Scala, E. Allergen databases: Current status and perspectives. Curr. Allergy Asthma Rep. 2009, 9, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Pikuła, M.; Zieliński, M.; Specjalski, K.; Barańska-Rybak, W.; Dawgul, M.; Langa, P.; Jassem, E.; Kamysz, W.; Trzonkowski, P. In vitro evaluation of the allergic potential of antibacterial peptides: Camel and citropin. Chem. Biol. Drug Des. 2016, 87, 562–568.





