Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Najeeb Ur Rehman | -- | 2789 | 2022-07-21 07:31:08 | | | |
| 2 | Beatrix Zheng | + 5 word(s) | 2794 | 2022-07-21 09:45:50 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Shah, M.; Mubin, S.; Hassan, S.S.U.; Tagde, P.; Ullah, O.; Rahman, M.H.; Al-Harrasi, A.; Rehman, N.U.; Murad, W. Biological Activities of Genus Scutellaria. Encyclopedia. Available online: https://encyclopedia.pub/entry/25372 (accessed on 26 March 2026).
Shah M, Mubin S, Hassan SSU, Tagde P, Ullah O, Rahman MH, et al. Biological Activities of Genus Scutellaria. Encyclopedia. Available at: https://encyclopedia.pub/entry/25372. Accessed March 26, 2026.
Shah, Muddaser, Sidra Mubin, Syed Shams Ul Hassan, Priti Tagde, Obaid Ullah, Md. Habibur Rahman, Ahmed Al-Harrasi, Najeeb Ur Rehman, Waheed Murad. "Biological Activities of Genus Scutellaria" Encyclopedia, https://encyclopedia.pub/entry/25372 (accessed March 26, 2026).
Shah, M., Mubin, S., Hassan, S.S.U., Tagde, P., Ullah, O., Rahman, M.H., Al-Harrasi, A., Rehman, N.U., & Murad, W. (2022, July 21). Biological Activities of Genus Scutellaria. In Encyclopedia. https://encyclopedia.pub/entry/25372
Shah, Muddaser, et al. "Biological Activities of Genus Scutellaria." Encyclopedia. Web. 21 July, 2022.
Copy Citation
Scutellaria (Lamiaceae) comprises over 360 species. Based on its morphological structure of calyx, also known as Skullcap, it is herbaceous by habit and cosmopolitan by habitat. The species of Scutellaria are widely used in local communities as a natural remedy. The genus contributed over three hundred bioactive compounds mainly represented by flavonoids and phenols, chemical ingredients which serve as potential candidates for the therapy of various biological activities.
natural products
antimicrobial
antioxidant
anticancer: anti-inflammatory
enzyme inhibitory activity
analgesic activity
1. Antimicrobial Capabilities
The antibiotic resistance by the microbes persuaded the researchers to devise a new alternative and effective antimicrobial agents which are much more effective with less adverse effects. The finding related to the essential oils of Scutellaria reveals the significant potential to act as an antimicrobial agent [1]. In addition to that, the essential oils of the Scutellaria contain bioactive ingredients such as eugenol, linalool, and other long-chain alcohols which can resist microbes. The finding of Shah et al. [2] reflected that the oils of the Scutellaria edelbergii have appreciable potential to act as an antimicrobial agent while the same plant various fractions offered appreciable antibacterial significance [3]. The data stated by Yu et al. [4] also provide the researchers with information that the EOs of S. barbata has significant potential against the screened microbes K. pneumoniae, S. maltophila, S. aureus, E. faecalis, S. marcescens, S. flexneri, C. freundii, S. paratyphi-A, S. simulans, S. heamolyticus, E. coli, P. aeruginosa, S. epidermidis, S. liquefaciens, C. tropicalis, S. typhi, and C. albicans using agar well diffusion assay. As per their findings, the essential oil had a strong bactericidal effect; S. epidermidis was perhaps the maximum resistance to the concentrated abstraction 29 mm inhibition zones and 0.77 mg/mL MBC), while C. albicans was the least (7–9 mm and 24.50 mg/mL MBC) [5]. The literature also reflected that the essential oils of S. strigillosa had more antimicrobial activity against Gram-positive bacteria and fungus than Gram-negative bacteria and fungus, as stated by Shen et al. [5]. Pant et al. [6] discovered the antibacterial activity of S. grossa essential oils against K. pneumonia, E. faecalis, B. subtilis, and S. enterica [5]. Skaltsa et al. [7] confirmed that essential oils obtained in Greece from S. rupestris and S. sieberi had rational antibacterial activity compared to Staphylococcus aureus and B. cereus [5]. Gousiadou et al. [8] discovered that after exposure to high rates of linalool and nerolidol, the essential oil of S. albida subsp albida was highly active against S. aureus, E. coli, P. aeruginosa, B. subtilis, and S. cerevisiae [5]. Dereboylu et al. [9] examined Scutellaria plant species and observed with significant resistance against numerous human pathogenic microbes [10]). Yi Nan et al. [11] discussed total flavones and antimicrobial activity in S. baicalensis. Yu et al. [12] discussed Chinese herbal medicine additives in aquaculture. Leung et al. investigated the antibacterial effects of nanoparticles synthesized from S. baicalensis [13]. According to the spectrum effect relationships between ultra-performance chromatography and E.coli B, incubation with S. baicalensis presented significant resistance against E.coli, as evaluated by Leach et al. [14].
2. Enzyme Inhibitory Potential
Acetylcholinesterase (AChE) inhibitors have recently been shown to be an effective clinical strategy for preserving acetylcholine levels and improving cholinergic activity, as stated by Colovic et al. [15]. Inhibiting AChE and Butyrylcholinesterase (BChE) has developed into a standard method for treating the symptoms of neurodegenerative diseases like Alzheimer’s disease [16]. A-amylase and a-glucosidase are two other key glycaemic control enzymes (Shah et al. [17]). The finding of new and reverse tyrosinase enzyme inhibitors, on the other hand, has enabled scientists to develop more accurate Parkinson’s disease prevention measures. The tyrosine-to-dopaquinone conversion catalyzed by tyrosinase may cause neurotoxicity, which has been connected to Parkinson’s disease [18]. Apart from antioxidant activity, aqueous concentrated abstractions of both plants had the lowest inhibitory activity for AChE, a-amylase, and tyrosinase; however, aqueous concentrated abstractions had the highest a-glucosidase inhibition (2.95 and 2.78 mmol/g concentrated abstraction for S. orientalis and S. salviifolia, respectively). The aqueous concentrated abstractions of both plants did not affect BChE inhibition. Concentrated abstractions of S. orientalis and S. salviifolia, on the other hand, inhibited AChE (1.37 and 1.69 mg GALAE/g concentrated abstraction, respectively), BChE (1.76 and 1.67 mg GALAE/g concentrated abstraction, respectively), and a-amylase (0.50 and 0.65 mmol ACAE/g concentrated abstraction, respectively) [19][20].
3. Anti-Fungal Significance
The increase in the complications caused by the fungus and resistance to the marketed available drugs also leads scientists and herbalists to find out new and effective ways to overcome the fungal complications [21]. Previous research has shown that some species of Scutellaria and their bioactive ingredients can resist fungal growth [22][23]. The n-Hexane extracted crude oils also have the capacity to resist fungal growth as stated in the literature of Shah et al. [2]. Scutellaria also contain bioactive groups such as alkaloid, which is used to break down peptidoglycan stability and degrade fungal cell walls. Another antifungal active compound found in natural herb plants is triterpenoid saponins [24][25]. According to Katzung et al. [26], topical antifungal agents and oral antifungal agents from the azole class can be used to treat candidiasis. According to Ghannoum and Rice [27], the azole antifungal inhibits 14-lanosterol demethylase in the ergosterol synthesis pathway. According to Lyon, Karatela, and Sunay, fluconazole has been identified as an antifungal agent that is effective against the majority of Candida isolates in 2010. Many researchers have recently investigated the case to investigate traditional medicine [28][29].
4. Anticancer Implication of Genus Scutellaria
Scutellaria has anti-metastatic, anti-proliferative, anti-invasion, anti-angiogenic, and apoptosis effects in vitro as well as in vivo [30]. The major constituents of Scutellaria baicalensis are wogonin, baicalein, and baicalin [31]. These phytochemicals are not only cytostatic but also cytotoxic to various human tumor cell lines in vitro and inhibit tumor growth in vivo. Most importantly, they show almost no or minor toxicity to normal epithelial and normal peripheral blood and myeloid cells [32]. The antitumor functions of these flavones are largely due to their abilities to scavenge oxidative radicals, attenuate NF-κB activity, inhibit several genes important for regulation of the cell cycle, suppress COX-2 gene expression, and prevent viral infections. The tumor-selectivity of Wogonin has recently been demonstrated to be due to its ability to differentially modulate the oxidation-reduction status of malignant vs. normal lymphocytic cells and to preferentially induce phospholipase Cγ1, a key enzyme involved in Ca2+ signaling, through H2O2 signaling in malignant lymphocytes. Numerous studies have also shown that Wogonin achieves its anticancer effects by modulating a variety of molecular pathways. The major molecular pathways by which it exerts its antitumor effects are reactive oxygen species (ROS), calcium, NF-B, tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), and tumor necrosis factor-alpha, [33] all of which participate in the both intrinsic mitochondria-mediated and extrinsic receptor-mediated pathways [34]. Apart from activating Bax/Bak protein and caspase-8/caspase-9/caspase-3, Wogonin plays a critical function in inhibiting tumor angiogenesis produced by lipopolysaccharide (LPS) or hydrogen peroxide (H2O2) through the PI3K/AKT/NF-B pathway. Yin et al. examined wogonin to determine its anticancer impact on breast cancer cells. They found that Wogonin might reduce AKT signaling, limit tumor angiogenesis, and finally inhibit tumor development [35]. The toxicity profile of Scutellaria baicalensis is given in Table 1.
Table 1. Toxicity and side effects of Scutellaria baicalensis.
| Compound/Extraction | Cell Lines/Animal | Dose/MTC |
|---|---|---|
| EESB | Mice | LD50 = 39.60 g/kg |
| Baicalin | Embryonic stem cell | IC50 = 135.9 mg/L |
| EESB | Rat | 2500 mg/kg |
| Wogonin | Mice | LD50 = 286.15 mg/kg |
Abbreviations—MTC: minimal toxic concentration, EESB: ethanol extracts of Scutellaria baicalensis.
He et al. [36] stated that wogonin can stop the cell cycle in HCT116 cells in the G1 phase in depends on its dosage by inhibiting the Wnt/β-catenin signaling pathway. Baicalein (5,6,7-trihydroxyflavone) is a flavonoid chemical ingredient mainly isolated from S. baicalensis roots. Baicalein is widely used as an anti-inflammatory and anti-cancer agent in Korean and Chinese herbal medicine (Lee et al. [37]). Kim et al. [38] have researched baicalein’s anticancer activity on HCT116 human colon cancer cells and its tumor-preventive potential in mice with colitis-associated cancer. They used azoxymethane (AOM) and dextran sulphate sodium (DSS) to develop colon cancers in mice and examined the impact of baicalein on tumor formation. Baicalein treatment of HCT116 cells inhibited cell growth and induced apoptotic cell death in a concentration-dependent manner. Apoptosis was determined by morphological alterations and poly (ADP-ribose) polymerase cleavage. Baicalein also inhibited NF-kB activation through PPAR-γ activation. These findings suggest that baicalein’s anti-inflammatory actions may be mediated through PPAR-γ activation. Finally, baicalein treatment dramatically reduced the frequency of tumor growth associated with inflammation and data show that baicalein may be a possibility for preventing colon carcinogenesis linked with inflammation.
5. Anti-Inflammatory Potential
Inflammation is a complication that leads to numerous other pathological disorders. Some Scutellaria species are used as a local remedy to treat inflammation. The dominant compounds such as scutellarin, baicalin, alkaloids, saponins, tannins, and glycosides have considerable in vivo pharmacological capacities to cure inflammation, relieve pain, and scavenge the free radicals (Liu et al. [39]). S. baicalensis has been found to be a prominent factor to release of oxidative stress and cure inflammation, as described in the literature stated by Huang et al. [40]. S. edelbergii has been used for a long time to treat inflammation, which is further validated by the investigation of the screening of S. edelbergii in crude extract and sub-fractions and n-hexane extracted crude oils, which possess significant potential to overcome inflammation, as stated in the literature of Shah et al. [2][3]. The EtOAc fraction was the most effective presented (54%) inhibition in comparison to the other examined fractions. They employed Diclofenac sodium as a control, which inhibited inflammation induced by carrageenan by 74% in the experimental mice.
Dogan et al. [41] described the therapeutic role of S. brevibracteata in the treatment of inflammation. They sought to decipher the underlying molecular pathways behind stomach inflammatory processes using network pharmacology and molecular docking analysis. The researchers performed gene enrichment analysis and target screening. Nitric oxide (NO) and interleukin-6 (IL-6) cytokines were used for experimental validation in LPS-stimulated RAW 264.7 cells. Additionally, antioxidant activity was determined by examining the radical scavenging effects of various radicals. The isolated compounds were associated with a total of 144 targets, 26 of which were associated with chosen inflammatory targets. The HIF1 signaling pathway and the TNF signaling pathway were identified as being implicated in inflammation by the gene enrichment analysis. Additionally, the researchers designated AKT1, TNF, EGFR, and COX2 as priority targets according to their 26 frequent protein–protein interactions. At 100 and 200 µg/mL, the extract suppressed NO and IL-6 production, respectively, while the flavonoid-rich fraction exhibited considerable anti-inflammatory effects through NO and IL-6 production at 50 and 100 µg/Ml respectively. When combined with the results of network analysis and literature, it is believed that the anti-inflammatory effects of extracts, fractions, and pure compounds were achieved by lowering NO and IL-6 levels via regulation of the NF-B pathway or by lowering NO production via suppression of iNOS via the HIF-1 pathway. The extract and fractions exhibited anti-inflammatory activity that was comparable to that of S. baicalensis, a plant widely utilized for its anti-inflammatory properties. Joshee N et al. [42] have highlighted that the genus Scutellaria plant species are a prevalent component of Eastern and traditional American medicine. Skullcap is a perennial plant native to North America that is a member of the genus Scutellaria. The genus is widespread in the Northern Hemisphere, with almost 400 species. Numerous species are endangered, imperiled, or uncommon. Habitat damage, urbanization, and poor seed set are only a few of the factors contributing to the decline of numerous skullcap populations. Numerous skullcaps feature brilliant, lovely flowers that make them excellent decorative plants. Skullcap is an anti-inflammatory, antispasmodic, emmenagogue, nervine, sedative, and powerful tonic used in alternative medicine. At Fort Valley State University, authors have built a germplasm collection and maintained populations in the greenhouse and by micropropagation. The researchers have achieved great progress in the fields of micropropagation, transformation for desired gene transfer, hairy root induction, and flavonoids, which were observed through HPLC analysis, which is effective for glioma cell lines. According to Mamadalieva NZ et al. [43], the genus Scutellaria is represented in Uzbekistan by 32 species, which are used in traditional medicine to treat epilepsy, inflammation, allergies, chorea, and nervous stress. Jia et al. [44] demonstrated that the herbal remedy baicalin induces autophagy in the macrophage cell line Raw 264.7 and results in enhanced Mtb destruction. Additionally, baicalin reduced Mtb-induced activation of the NLRP3 inflammasome and subsequent production of inflammasome-derived IL-1β. To elucidate the molecular processes of baicalin, the researchers studied the signaling pathways involved in autophagy. Baicalin lowered phosphorylated protein kinase B (p-Akt) and phosphorylated mammalian target of rapamycin (p-mTOR) at Ser473 and Ser2448, respectively, but did not affect p38, JNK, or ERK phosphorylation in Raw264.7 or primary peritoneal macrophages. Additionally, baicalin inhibited NF-kB activity. Finally, immunofluorescence experiments revealed that baicalin increased the co-localization of the inflammasome with the autophagosome, suggesting that this may be the underlying mechanism for the autophagic degradation impact on inflammasome activation. Together, baicalin strongly activates autophagy in Mtb-infected macrophages through the PI3K/Akt/mTOR route rather than the MAPK pathway. Additionally, baicalin suppressed the PI3K/Akt/NF-kB signaling pathway, and both activations of autophagy and inhibition of NF-kB contribute to restricting the NLRP3 inflammasome and consequent generation of the pro-inflammatory cytokine IL-1β. They conclude that baicalin is a promising antimycobacterial and anti-inflammatory agent that may serve as a fresh option for the development of new adjunct medications targeting HDT for potential therapy improvement. Therapeutic applications of baicalin are shown in Figure 1.
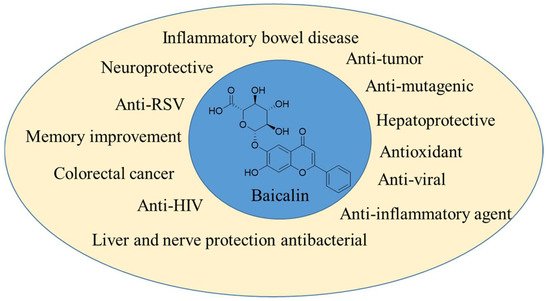
Figure 1. Therapeutic applications of baicalin.
Scutellaria galericulata has been used to isolate two novel flavanone glycosides, dubbed Scugalerosides A and B (1-2). Their chemical structures, including their exact configurations, were determined by an in-depth analysis of physical data. Two novel compounds demonstrated anti-inflammatory activity in vitro, inhibiting the release of -glucuronidase from polymorphonuclear leukocytes of rats by 43.7 and 45.1 percent, respectively, at 10 μM as stated by Xiao et al. [45]
Han QT et al. [46] have isolated two new flavanone glucuronate esters, named Scumoniliosides A and B along with two known flavonoid glucuronate esters, 5,4′-dihydroxyflavonoid-7-O-β-d-glycuronate methyl ester, and 5,4′-dihydroxyflavonoid-7-O-β-d-glycuronate butyl ester, from the ethanolic extract of the whole plant of S. moniliorrhiza. Additionally, their chemical structures were confirmed using integrated spectroscopic techniques, and in vitro studies revealed that four compounds exhibited anti-inflammatory activity, with inhibition rates of -glucuronidase release from rat polymorphonuclear leukocytes ranging from 43.5 to 48.1% at 10 μM. Four undescribed flavonoid alkaloids, as two pairs of enantiomers, were initially isolated as a racemate from the whole plant of S. moniliorrhiza. Utilizing chiral HPLC, four isomers, named Scumonilines A-D, were successfully separated, and their chemical structures, including absolute configurations, were established by mass as well as NMR spectroscopy and CD technique. In vitro, four flavonoid alkaloids showed anti-inflammatory activities, with IC50 values against the release of β-glucuronidase from polymorphonuclear leukocytes of rats being in the range 5.16–5.85 μΜ. Moreover, four compounds were evaluated for their inhibitory activities against aldose reductase and gave IC50 values in the range 2.29–3.03 μΜ. Previously uncharacterized flavonoid alkaloids were first extracted as a racemate from the full plant of Scutellaria moniliorrhiza. Four isomers of Scumonilines A-D were effectively separated using chiral HPLC, and their chemical structures, including their absolute configurations, were established using mass, NMR, and CD spectroscopy. Four flavonoid alkaloids demonstrated anti-inflammatory activity in vitro, with IC50 values of 5.16–5.8 μM. against the release of β-glucuronidase from polymorphonuclear leukocytes of rats. Additionally, four drugs were tested for their inhibitory activity against aldose reductase, with IC50 values ranging between 2.29 and 3.03 μM [47].
6. Analgesic Significance
Some species of the genus Scutellaria have the appreciable capacity to assuage pain locally which was further authenticated by the study of Shah et al. [2][3] using various fractions and n-Hexane extracted oils of S. edelbergii. The screening presented by [48] also reflected that the species of the selected genus can allay pain. The Baicalin from the roots of Scutellaria baicalensis (S. baicalensis) and (+)-catechin from the heartwoods of Acacia catechu (A. catechu) have been used in both over-the-counter joint care dietary supplements and a portion of prescription medical food. Baicalin and catechin anti-inflammatory activity has been reported [49], but there is less work on their analgesic effect. Therefore, author Yimam et al. [50] have used three regularly used animal models of pain to assess the analgesic effect of UP446, a standardized bioflavonoid composition including baicalin and catechins. The antinociceptive efficacy of UP446 oral dosages of (150 mg/kg and 100 mg/kg body weight) was evaluated using carrageenan-induced paw edema, the formalin test, and abdominal constriction tests. Each test employed ibuprofen as a reference chemical. Pretreatment with UP446 at a 150 mg/kg oral dose decreased hypersensitivity to pain by 39.5% in carrageenan-induced hyperalgesia mice. Similarly, a single oral administration of UP446 at a concentration of 100 mg/kg inhibited pain sensitivity by 58% and 71.9%, respectively, in writhing and formalin tests. These results imply that UP446’s standardized anti-inflammatory bioflavonoid content might potentially be used to reduce nociception [51]. Various chemical ingredients reported from the genus Scutellaria have some important significance as given in Figure 2.
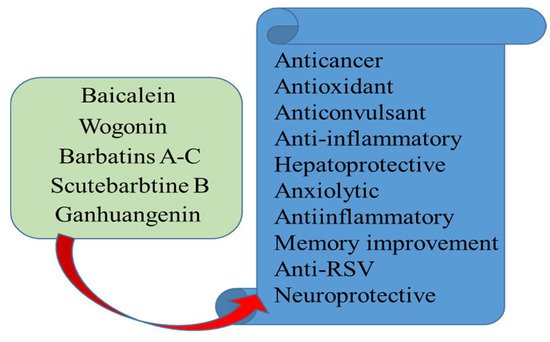
Figure 2. Medicinal uses of the major compounds isolated from Scutellaria species.
References
- Hajiasgharzadeh, K.; Somi, M.H.; Sadigh-Eteghad, S.; Mokhtarzadeh, A.; Shanehbandi, D.; Mansoori, B.; Mohammadi, A.; Doustvandi, M.A.; Baradaran, B. The dual role of alpha7 nicotinic acetylcholine receptor in inflammation-associated gastrointestinal cancers. Heliyon 2020, 6, e03611.
- Shah, M.; Murad, W.; Ur Rehman, N.; Mubin, S.; Al-Sabahi, J.N.; Ahmad, M.; Zahoor, M.; Ullah, O.; Waqas, M.; Ullah, S. GC-MS analysis and biomedical therapy of oil from n-hexane fraction of Scutellaria edelbergii Rech. f.: In vitro, in vivo, and in silico approach. Molecules 2021, 26, 7676.
- Shah, M.; Murad, W.; Ur Rehman, N.; Halim, S.A.; Ahmed, M.; Rehman, H.; Zahoor, M.; Mubin, S.; Khan, A.; Nassan, M.A. Biomedical applications of Scutellaria edelbergii Rech. f.: In vitro and in vivo approach. Molecules 2021, 26, 3740.
- Yu, J.; Lei, J.; Yu, H.; Cai, X.; Zou, G. Chemical composition and antimicrobial activity of the essential oil of Scutellaria Barb. Phytochem. 2004, 65, 881–884.
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 177.
- Pant, C.C.; Melkani, A.B.; Mohan, L.; Dev, V. Composition and antibacterial activity of essential oil from Scutellaria grossa Wall ex Benth. Nat. Prod. Res. 2012, 26, 190–192.
- Skaltsa, H.D.; Lazari, D.M.; Kyriazopoulos, P.; Golegou, S.; Triantaphyllidis, S.; Sokovic, M.; Kypriotakis, Z. Composition and antimicrobial activity of the essential oils of Scutellaria sieberia Benth. and Scutellaria rupestris Boiss. et Heldr. ssp. adenotricha (Boiss. et Heldr.) Greuter et Burdet from Greece. J. Essent. Oil Res. 2005, 17, 232–235.
- Gousiadou, C.; Karioti, A.; Heilmann, J.; Skaltsa, H. Iridoids from Scutellaria albida ssp. albida. Phytochemistry 2007, 68, 1799–1804.
- Dereboylu, A.; Sarikahya, N.; Sengonca, N.; Kirmizigul, S.; Yasa, I.; Gucel, S.; Guvensen, A. Glandular trichomes morphology, chemical composition and antimicrobial activity of the essential oil of three endemic Scutellaria taxa (Lamiaceae). Asian J. Chem. 2012, 24, 4911.
- Kim, H.I.; Hong, S.H.; Ku, J.M.; Lim, Y.S.; Lee, S.J.; Song, J.; Kim, T.Y.; Cheon, C.; Ko, S.-G. Scutellaria radix promotes apoptosis in non-small cell lung cancer cells via induction of AMPK-dependent autophagy. Am. J. Chin. Med. 2019, 47, 691–705.
- Nan, Y.; Yuan, L.; Zhou, L.; Niu, Y. Study on the optimization of the technology for the extraction and purification of total flavone in Scutellaria baicalensis and its antibacterial activity. Afr. J. Microbiol. Res. 2011, 5, 5689–5696.
- Yu, T.-T.; Guo, K.; Chen, H.-C.; Lan, C.-Z.; Wang, J.; Huang, L.-L.; Wang, X.-H.; Zhang, Z.; Gao, S. Effects of traditional Chinese medicine Xin-Ji-Er-Kang formula on 2K1C hypertensive rats: Role of oxidative stress and endothelial dysfunction. BMC Complement. Altern. Med. 2013, 13, 173.
- Leung, K.C.-F.; Seneviratne, C.J.; Li, X.; Leung, P.C.; Lau, C.B.S.; Wong, C.-H.; Pang, K.Y.; Wong, C.W.; Wat, E.; Jin, L. Synergistic antibacterial effects of nanoparticles encapsulated with Scutellaria baicalensis and pure chlorhexidine on oral bacterial biofilms. Nanomaterials 2016, 6, 61.
- Leach, F.S. Anti-microbial properties of Scutellaria baicalensis and Coptis chinensis, two traditional Chinese medicines. Biosci. Horizon. 2011, 4, 119–127.
- Rong, X.; Jiang, L.; Qu, M.; ul Hassan, S.S.; Liu, Z. Enhancing therapeutic efficacy of donepezil by combined therapy: A comprehensive review. Curr. Pharm. Des. 2020, 27, 332–344.
- Shrestha, D.; Sharma, P.; Adhikari, A.; Mandal, A.K.; Verma, A. A Review on Nepalese medicinal plants used traditionally as alpha-amylase and alpha-glucosidase inhibitors against diabetes mellitus. Curr. Trad. Med. 2021, 7, 63–72.
- Shah, M.; Rahman, H.; Khan, A.; Bibi, S.; Ullah, O.; Ullah, S.; Ur Rehman, N.; Murad, W.; Al-Harrasi, A. Identification of α-Glucosidase Inhibitors from Scutellaria edelbergii: ESI-LC-MS and Computational Approach. Molecules 2022, 27, 1322.
- Behl, T.; Kaur, G.; Bungau, S.; Jhanji, R.; Kumar, A.; Mehta, V.; Zengin, G.; Brata, R.; ul Hassan, S.S.; Fratila, O. Distinctive evidence involved in the role of endocannabinoid signalling in parkinson’s disease: A Perspective on associated therapeutic interventions. Int. J. Mol. Sci. 2020, 21, 6235.
- Wei, L.; Dai, Q.; Zhou, Y.; Zou, M.; Li, Z.; Lu, N.; Guo, Q. Oroxylin A sensitizes non-small cell lung cancer cells to anoikis via glucose-deprivation-like mechanisms: C-Src and hexokinase II. Biochim. Biophy. Acta 2013, 1830, 3835–3845.
- Liu, Y.; Wang, X.; Li, W.; Xu, Y.; Zhuo, Y.; Li, M.; He, Y.; Wang, X.; Guo, Q.; Zhao, L. Oroxylin A reverses hypoxia-induced cisplatin resistance through inhibiting HIF-1α mediated XPC transcription. Oncogene 2020, 39, 6893–6905.
- Joshee, N.; Mentreddy, S.; Yadav, A.K. Mycorrhizal fungi and growth and development of micropropagated Scutellaria integrifolia plants. Indus. Crops Prod. 2007, 25, 169–177.
- Da, X.; Nishiyama, Y.; Tie, D.; Hein, K.Z.; Yamamoto, O.; Morita, E. Antifungal activity and mechanism of action of Ou-gon (Scutellaria root extract) components against pathogenic fungi. Sci. Rep. 2019, 9, 1–12.
- Cole, M.D.; Bridge, P.D.; Dellar, J.E.; Fellows, L.E.; Cornish, M.C.; Anderson, J.C. Antifungal activity of neo-clerodane diterpenoids from Scutellaria. Phytochemistry 1991, 30, 1125–1127.
- Al-Alwan, L.A.; Chang, Y.; Baglole, C.J.; Risse, P.-A.; Halayko, A.J.; Martin, J.G.; Eidelman, D.H.; Hamid, Q. Autocrine-regulated airway smooth muscle cell migration is dependent on IL-17–induced growth-related oncogenes. J. Allergy Clin. Immunol. 2012, 130, 977–985.
- Al-Alwan, L.A.; Chang, Y.; Mogas, A.; Halayko, A.J.; Baglole, C.J.; Martin, J.G.; Rousseau, S.; Eidelman, D.H.; Hamid, Q. Differential roles of CXCL2 and CXCL3 and their receptors in regulating normal and asthmatic airway smooth muscle cell migration. J. Immunol. 2013, 191, 2731–2741.
- Katzung, B.G.; Parmley, W.W. Drugs used in heart failure. Basic Clin. Pharmacol. 2010, 24, 212–227.
- Ji, X.; Li, J.; Xu, L.; Wang, W.; Luo, M.; Luo, S.; Ma, L.; Li, K.; Gong, S.; He, L. IL4 and IL-17A provide a Th2/Th17-polarized inflammatory milieu in favor of TGF-β1 to induce bronchial epithelial-mesenchymal transition (EMT). Int. J. Clin. Exp. Pathol. 2013, 6, 1481.
- Glevitzky, I.; Dumitrel, G.A.; Glevitzky, M.; Pasca, B.; Otrisal, P.; Bungau, S.; Cioca, G.; Pantis, C.; Popa, M. Statistical analysis of the relationship between antioxidant activity and the structure of flavonoid compounds. Rev. Chim. 2019, 70, 3103–3107.
- Xie, Y.G.; Zhao, X.C.; ul Hassan, S.S.; Zhen, X.Y.; Muhammad, I.; Yan, S.K.; Yuan, X.; Li, H.L.; Jin, H.Z. One new sesquiterpene and one new iridoid derivative from Valeriana amurensis. Phytochem. Lett. 2019, 32, 6–9.
- EghbaliFeriz, S.; Taleghani, A.; Tayarani-Najaran, Z. Scutellaria: Debates on the anticancer property. Biomed. Pharmacother. 2018, 105, 1299–1310.
- Li-Weber, M. New therapeutic aspects of flavones: The anticancer properties of Scutellaria and its main active constituents Wogonin, baicalein and baicalin. Cancer Treat. Rev. 2009, 35, 57–68.
- Parajuli, P.; Joshee, N.; Rimando, A.M.; Mittal, S.; Yadav, A.K. In vitro antitumor mechanisms of various Scutellaria extracts and constituent flavonoids. Planta Med. 2009, 75, 41–48.
- Kim, Y.O.; Leem, K.; Park, J.; Lee, P.; Ahn, D.-K.; Lee, B.C.; Park, H.K.; Suk, K.; Kim, S.Y.; Kim, H. Cytoprotective effect of Scutellaria baicalensis in CA1 hippocampal neurons of rats after global cerebral ischemia. J. Ethnopharmacol. 2001, 77, 183–188.
- Chen, C.-C.; Kao, C.-P.; Chiu, M.-M.; Wang, S.-H. The anti-cancer effects and mechanisms of Scutellaria barbata D. Don on CL1–5 lung cancer cells. Oncotarget 2017, 8, 109340.
- Yin, X.; Zhou, J.; Jie, C.; Xing, D.; Zhang, Y. Anticancer activity and mechanism of Scutellaria barbata extract on human lung cancer cell line A549. Life Sci. 2004, 75, 2233–2244.
- He, L.; Lu, N.; Dai, Q.; Zhao, Y.; Zhao, L.; Wang, H.; Li, Z.; You, Q.; Guo, Q. Wogonin induced G1 cell cycle arrest by regulating Wnt/β-catenin signaling pathway and inactivating CDK8 in human colorectal cancer carcinoma cells. Toxicology 2013, 312, 36–47.
- Lee, W.; Ku, S.-K.; Bae, J.-S. Anti-inflammatory effects of Baicalin, Baicalein, and Wogonin in vitro and in vivo. Inflammation 2015, 38, 110–125.
- Kim, D.H.; Hossain, M.A.; Kang, Y.J.; Jang, J.Y.; Lee, Y.J.; Im, E.; Yoon, J.-H.; Kim, H.S.; Chung, H.Y.; Kim, N.D. Baicalein, an active component of Scutellaria baicalensis Georgi, induces apoptosis in human colon cancer cells and prevents AOM/DSS-induced colon cancer in mice. Int. J. Oncol. 2013, 43, 1652–1658.
- Liu, H.-L.; Kao, T.-H.; Shiau, C.-Y.; Chen, B.-H. Functional components in Scutellaria barbata D. Don with anti-inflammatory activity on RAW 264.7 cells. J. Food Drug Anal. 2018, 26, 31–40.
- Huang, W.-H.; Lee, A.-R.; Yang, C.-H. Antioxidative and anti-inflammatory activities of polyhydroxyflavonoids of Scutellaria baicalensis Georgi. Biosci. Biotech. Biochem. 2006, 70, 2371–2380.
- Dogan, Z.; Telli, G.; Tel, B.C.; Saracoglu, I. Scutellaria brevibracteata Stapf and active principles with anti-inflammatory effects through regulation of NF-κB/COX-2/iNOS pathways. Fitoterapia 2022, 158, 105159.
- Parajuli, P.; Joshee, N.; Chinni, S.; Rimando, A.; Mittal, S.; Sethi, S.; Yadav, A. Delayed growth of glioma by Scutellaria flavonoids involve inhibition of Akt, GSK-3 and NF-κB signaling. J. Neurooncol. 2011, 101, 15–24.
- Mamadalieva, N.Z.; Ovidi, E.; Triggiani, D.; Yuldasheva, N.K.; Ul’chenko, N.T.; Glushenkova, A.I.; Tiezzi, A. Lipid fraction from Scutellaria ramosissima tested on the microtubular array of cancer cell models. Arch. Biomed. Sci. 2014, 2, 11–17.
- Jia, X.; Zhang, C.; Bao, J.; Wang, K.; Tu, Y.; Wan, J.-B.; He, C. Flavonoids from Rhynchosia minima root exerts anti-inflammatory activity in lipopolysaccharide-stimulated RAW 264.7 cells via MAPK/NF-κB signaling pathway. Inflammopharmacology 2020, 28, 289–297.
- Xiao, K.; Han, Q.-T.; Zhang, L.; Dai, S.-J. Two new flavanone glycosides from Scutellaria galericulata with anti-inflammatory activities. Phytochem. Lett. 2017, 20, 151–154.
- Han, Q.-T.; Ren, Y.; Li, G.-S.; Xiang, K.-L.; Dai, S.-J. Flavonoid alkaloids from Scutellaria moniliorrhiza with anti-inflammatory activities and inhibitory activities against aldose reductase. Phytochemistry 2018, 152, 91–96.
- Zhang, L.; Ravipati, A.S.; Koyyalamudi, S.R.; Jeong, S.C.; Reddy, N.; Smith, P.T.; Bartlett, J.; Shanmugam, K.; Münch, G.; Wu, M.J. Antioxidant and anti-inflammatory activities of selected medicinal plants containing phenolic and flavonoid compounds. J. Agricul. Food Chem. 2011, 59, 12361–12367.
- Yin, B.; Li, W.; Qin, H.; Yun, J.; Sun, X. The use of Chinese Skullcap (Scutellaria baicalensis) and its extracts for sustainable animal production. Animals 2021, 11, 1039.
- Yimam, M.; Zhao, Y.; Ma, W.; Jia, Q.; Do, S.-G.; Shin, J.-H. 90-day oral toxicity study of UP446, a combination of defined extracts of Scutellaria baicalensis and Acacia catechu, in rats. Food Chem. Toxicol. 2010, 48, 1202–1209.
- Yimam, M.; Brownell, L.; Hodges, M.; Jia, Q. Analgesic effects of a standardized bioflavonoid composition from Scutellaria baicalensis and Acacia catechu. J. Diet. Suppl. 2012, 9, 155–165.
- Burnett, B.; Jia, Q.; Zhao, Y.; Levy, R. A medicinal extract of Scutellaria baicalensis and Acacia catechu acts as a dual inhibitor of cyclooxygenase and 5-lipoxygenase to reduce inflammation. J. Med. Food 2007, 10, 442–451.
More
Information
Subjects:
Primary Health Care
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
983
Revisions:
2 times
(View History)
Update Date:
21 Jul 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





