| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Luca Rinaldi | + 692 word(s) | 692 | 2020-09-29 09:49:16 | | | |
| 2 | Vicky Zhou | + 52 word(s) | 744 | 2020-10-10 05:22:32 | | | | |
| 3 | Vicky Zhou | + 52 word(s) | 744 | 2020-10-10 05:22:52 | | | | |
| 4 | Vicky Zhou | -1 word(s) | 743 | 2020-10-27 07:19:59 | | |
Video Upload Options
The human cerebral cortex is asymmetrically organized with hemispheric lateralization pervading nearly all neural systems of the brain. Whether the lack of normal visual development affects hemispheric specialization subserving the deployment of visuospatial attention asymmetries is controversial. In principle, indeed, the lack of early visual experience may affect the lateralization of spatial functions, and the blind may rely on a different sensory input compared to the sighted. In this entry, we thus present a current state-of-the-art synthesis of empirical evidence concerning the effects of visual deprivation on the lateralization of various spatial processes (i.e., including line bisection, mirror symmetry, and localization tasks). Overall, the evidence reviewed indicates that spatial processes are supported by a right hemispheric network in the blind, hence, analogously to the sighted. Such a right-hemisphere dominance, however, seems more accentuated in the blind as compared to the sighted as indexed by the greater leftward bias shown in different spatial tasks. This is possibly the result of the more pronounced involvement of the right parietal cortex during spatial tasks in blind individuals compared to the sighted, as well as of the additional recruitment of the right occipital cortex, which would reflect the cross-modal plastic phenomena that largely characterize the blind brain.
1. Introduction
At first glance, the brain appears to be a symmetrical structure; however, a closer inspection reveals lateralized changes from the subcellular and neurochemical to gross anatomical levels [1]. These laterality patterns are not limited to the level of the brain but pervade as well in traits of human overt behavior such as handedness (the most frequently observed and studied behavioral asymmetry [2]) and spatial asymmetries as measured by line bisection tasks. Lateralization phenomena in the context of the line bisection task have been ascribed to genetic variation with dopaminergic system genes [3][4] or the allelic variation in genes affecting corpus callosum structure [5] associated with the direction (left versus right) and the magnitude of spatial orienting bias. However, investigating the role of environmental factors [6], recent research has suggested that epigenetic regulation contributes to the development of hemispheric asymmetries subserving spatial processing in line bisection tasks [7]. This may indicate that the bias in the line bisection task is likely influenced by multiple genetic, epigenetic, and environmental factors.
Within this theoretical framework, the study of visual deprivation represents a unique model to investigate lateralization phenomena, as experiential factors are clearly different compared to typical development. For instance, whereas blind and sighted individuals are exposed to language (a typical left-lateralized function in the brain [8]) to similar degrees, their spatial abilities (typically recruiting more the right hemisphere [9]) develop based on haptic and auditory input. In principle, the lack of early visual experience may thus affect the lateralization of spatial functions, which in the blind rely on a different sensory input compared to the sighted. Compensatory phenomena occurring at the brain and behavioral levels following visual deprivation have been extensively studied (for reviews see References [10][11][12]). However, how blindness affects the level of hemispheric asymmetry and laterality patterns in spatial processing is an issue that has not reached definite conclusions.
2. Spatial Asymmetries of Blindness
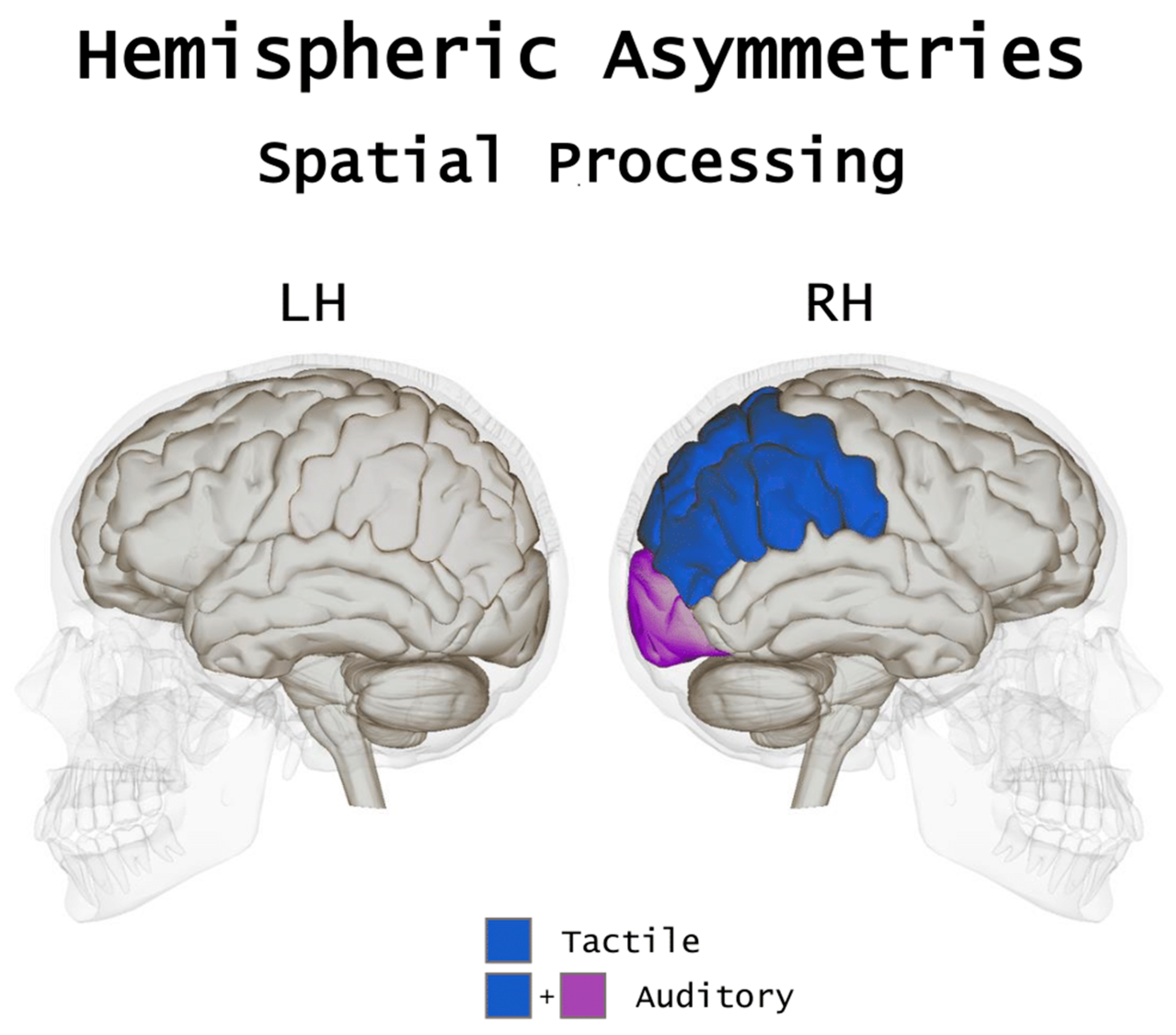
Overall, the debate as to what extent visual experience contributes to a different development of functional asymmetries subserving spatial processing has not reached definite conclusions. Yet, available evidence indicates that the hemispheric dominance for spatial processing seems, overall, largely unaffected by visual loss, as in both sighted and blind individuals’ spatial functions would predominantly rely on the right hemisphere (see Figure 1). Beyond the common lateralization pattern, spatial processes in the blind seem to be supported by a more extended cortical network in the right hemisphere, which would be responsible for the more pronounced leftward bias in spatial tasks as compared to the sighted. Despite right parietal activations in both the blind and sighted brain have been consistently reported across a series of studies in spatial processing, parietal areas of the blind brain would elaborate a larger amount of information during tactile tasks (such as the line bisection task) as compared to the sighed (see Reference [13]). This, in addition to the cross-modal recruitment of areas in the occipital cortex, may contribute to determining the larger leftward bias reported.
Figure 1. A pictorial, simplified view of the hemispheric asymmetries subserving spatial processing in the blind. In spatial tasks, in addition to a robust activation of parietal areas in tactile spatial processing, the blind also show a cross-modal recruitment of the occipital lobe, especially in auditory and tactile localization tasks.
References
- Concha, M.L.; Bianco, I.H.; Wilson, S.W. Encoding asymmetry within neural circuits. Nat. Rev. Neurosci. 2012, 13, 832–843.
- Güntürkün, O.; Ocklenburg, S. Ontogenesis of Lateralization. Neuron 2017, 94, 249–263.
- Zozulinsky, P.; Greenbaum, L.; Brande-Eilat, N.; Braun, Y.; Shalev, I.; Tomer, R. Dopamine system genes are associated with orienting bias among healthy individuals. Neuropsychologia 2014, 62, 48–54.
- Greene, C.M.; Robertson, I.H.; Gill, M.; Bellgrove, M.A. Dopaminergic genotype influences spatial bias in healthy adults. Neuropsychologia 2010, 48, 2458–2464.
- Ocklenburg, S.; Packheiser, J.; Schmitz, J.; Rook, N.; Güntürkün, O.; Peterburs, J.; Grimshaw, G.M. Hugs and kisses—The role of motor preferences and emotional lateralization for hemispheric asymmetries in human social touch. Neurosci. Biobehav. Rev. 2018, 95, 353–360.
- Latham, A.J.; Patston, L.L.M.; Tippett, L.J. The precision of experienced action video-game players: Line bisection reveals reduced leftward response bias. Atten. Percept. Psychophys. 2014, 76, 2193–2198.
- Schmitz, J.; Kumsta, R.; Moser, D.; Güntürkün, O.; Ocklenburg, S. DNA methylation of dopamine-related gene promoters is associated with line bisection deviation in healthy adults. Sci. Rep. 2019, 9, 1–8.
- Ocklenburg, S.; Beste, C.; Arning, L.; Peterburs, J.; Güntürkün, O. The ontogenesis of language lateralization and its relation to handedness. Neurosci. Biobehav. Rev. 2014, 43, 191–198.
- Kosslyn, S.M. Image And Brain; MIT Press: Cambridge, MA, USA, 1994.
- Cattaneo, Z.; Vecchi, T.; Cornoldi, C.; Mammarella, I.; Bonino, D.; Ricciardi, E.; Pietrini, P. Imagery and spatial processes in blindness and visual impairment. Neurosci. Biobehav. Rev. 2008, 32, 1346–1360.
- Merabet, L.B.; Rizzo, J.F.; Amedi, A.; Somers, D.C.; Pascual-Leone, A. What blindness can tell us about seeing again: Merging neuroplasticity and neuroprostheses. Nat. Rev. Neurosci. 2005, 6, 71–77.
- Ricciardi, E.; Bonino, D.; Pellegrini, S.; Pietrini, P. Mind the blind brain to understand the sighted one! Is there a supramodal cortical functional architecture? Neurosci. Biobehav. Rev. 2014, 41, 67–77.
- Leo, A.; Bernardi, G.; Handjaras, G.; Bonino, D.; Ricciardi, E.; Pietrini, P. Increased BOLD variability in the parietal cortex and enhanced parieto-occipital connectivity during tactile perception in congenitally blind individuals. Neural Plast. 2012.