1000/1000
Hot
Most Recent

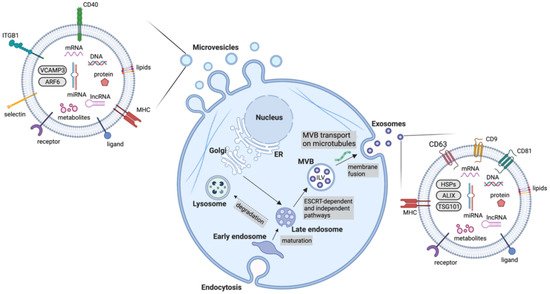
Extracellular vesicles are an important mediator of BC-TME signalling. EVs are non-replicative, lipid bilayer-delimited particles that are naturally released from cells. They have been identified in virtually every physiological fluid and are released by nearly all cell types. EV cargo consists of a number of bioactive molecules, including nucleic acids, lipids and membrane-bound and cytosolic proteins. The uptake of EVs is able to influence cell behaviour and as such, EVs are known to be important signalling particles, as well as diagnostic, predictive and prognostic biomarkers in diseases. Although EVs can be categorised in a number of ways (e.g., based on size, cargo and biological role), they are most often classified based on their biogenesis, with exosomes and microvesicles being the most commonly discussed EV subtypes. Many other subtypes of EVs have been identified, including apoptotic bodies and oncosomes, however, knowledge of their specific roles in cell–cell communication is limited. Due to the lack of consensus on biomarkers for specific subtypes of EVs, this review will use the collective term EV where the biogenesis pathway has not been demonstrated directly, in accordance with the guidelines set by the International Society for Extracellular Vesicles.
| Type of EV | Size (nm) | Biogenesis | Main Components |
|---|---|---|---|
| Exosomes | 30–150 | Early endosomes mature into late endosomes. Through the action of ESCRT machinery, MVBs containing ILVs are formed and fuse with the plasma membrane for release | Tetraspanins (CD9, CD63, CD81), HSPs, MVB biogenesis components (ALIX, TSG101) |
| Microvesicles | 100–1000 | Outward budding of the membrane followed by fission through contractile machinery | Cell adhesion molecules (integrins, selectins), Death receptors (CD40), VCAMP3, ARF6 |
| Apoptotic bodies | >1000 | Cytoplasmic fragmentation during programmed cell death | Histones, Annexin V, Caspase 3, Phosphatidylserine |
| Oncosomes | >1000 | Cleavage of large cytoplasmic extensions from cell body | Cytoskeleton components (cytokeratin 18), Tetraspanins (CD9, CD81), Cell adhesion molecules (integrins, ICAM, CD44) |