Diallyl disulfide (DADS) is a major allelochemical of the volatile organic compounds in garlic.
1. Introduction
Garlic (
Allium sativum L.), an important economic vegetable, contains various sulphureous volatile organic compounds (VOCs), including sulfides, disulfides, and trisulfides, all of which are formed by the decomposition of allicin and are released upon crushing garlic
[1]. These sulphureous VOCs give garlic strong allelopathic effects, which makes garlic a popular targeted crop in rotation and intercropping systems. Until now, garlic has been rotated or intercropped with many other vegetable crops such as eggplant
[2], pepper
[3], tomato
[4], and cucumber
[5][6]. When garlic is used in the intercropping systems, several beneficial effects have been reported
[7], such as improving the nutritional status of soil, reducing plant diseases and pests, alleviating continuous cropping obstacles, and increasing land use efficiency and net return. Wang et al.
[2] reported that eggplant intercropped with garlic could be an ideal farming system to effectively improve soil nutrients and to partially alleviate soil-borne diseases. In the pepper–garlic intercropping system, garlic root exudates with lower concentrations can stimulate the protective enzyme systems and then promote the growth of pepper, while those with higher concentrations will show deleterious effects
[3]. For the tomato–garlic intercropping system, the increased microorganism populations and enzyme activities in the substrates were observed, which further improved the fruit quality of tomato and produced higher economic benefits than the mono-cultivated tomatoes
[4]. In addition, compared with the monoculture system, both the cucumber–garlic and cucumber–green garlic intercropping systems showed improving effects on the populations of microorganisms and the activities of soil enzymes, as well as the nutritional status of both soils and plants
[5][6]. Our previous studies indicated that intercropping green garlic or garlic bulbs with cucumbers can significantly reduce the incidence and disease index of downy mildew in cucumber at the early stage of onset, and intercropping with green garlic had more significant effects. In the cultivation system of green garlic intercropped with cucumber, the VOCs released from green garlic might play important roles in reducing the incidence of downy mildew in cucumber; especially for the period of harvesting green garlics, more intense VOCs will be released. However, the regulating mechanism of these green garlic released VOCs in inducing cucumber downy mildew resistance is still unknown.
Diallyl disulfide (DADS) is a major allelochemical of the VOCs in garlic
[1][8]. Previous studies found that DADS can promote root growth in tomato by affecting cell division, changing endogenous phytohormones, increasing expressions of expansion genes
[8]. Additionally, the pathways of sulphate assimilation and glutathione (GSH) metabolism are also involved in the regulation of DADS-induced tomato root growth
[9]. Recently, Cheng et al. reported that DADS might increase resistance and promote plant growth in tomato by increasing its root activity and photosynthetic capacity and development to reduce the autotoxicity of tomato
[10]. In addition, lower concentrations of DADS can promote root growth and induce the elongation of main roots in cucumber
[11]. Our previous work indicated that both the green garlic VOCs and DADS play vital roles in the reactive oxygen species (ROS) production and regulation of antioxidant enzymes in cucumber seedlings
[1]. Moreover, DADS was also reported to have antifungal activity against several fungi (e.g.,
C. albicans,
C. tropicalis, and
Blastoschizomyces capitatus)
[12]. Therefore, the DADS might also play an important role in the inducing of disease resistance in cucumber.
Downy mildew, caused by the obligate oomycete pathogen
P. cubensis, is one of the most devastating diseases in cucumber
[13]. Although some resistant germplasms have been identified, the regulating genes and the pathways involved in mediating downy mildew resistance in cucumber are still unclear
[14]. Several quantitative trait loci associated with downy mildew resistance have been mapped onto five of the seven cucumber chromosomes, suggesting that downy mildew resistance in cucumber is likely due to the associative effects of multiple genes
[15]. Recently,
CsSGR was reported as a major-effect resistant gene for cucumber downy mildew. Loss-of-function of
CsSGR gives cucumber durable and broad-spectrum disease resistance to several diseases including the downy mildew. This probably is due to the fact that
CsSGR functions in the inhibitions of ROS over-accumulation and phytotoxic catabolite over-buildup in the chlorophyll degradation pathway
[16]. Additionally, overexpression of
CsWRKY50 also enhanced cucumber resistance to
P. cubensis, probably, through upregulating the transcript levels of several phytohormone-related defense genes that include salicylic acid (SA)-responsive genes, jasmonic acid (JA) responsive genes, and SA biosynthesis genes
[17]. Furthermore, transcriptome profiling in the context of
P. cubensis infection also gives some insight into understanding the molecular mechanisms of host responses to downy mildew infection. In cucumber, transcriptome data revealed that resistance to downy mildew is associated with an earlier response to the pathogen, hormone signaling, and regulation of nutrient supply
[14]. Additionally, the pathways of pathogen-associated molecular pattern (PAMP) recognition, signal transduction, ROS and lignin accumulation, and some transcription regulators are all involved in the resistance of cucumber to downy mildew
[15]. Although some research on germplasm identification, regulatory genes, and pathways of cucumber resistance to downy mildew were performed, studies on the use of allelopathic substances to induce cucumber resistance to downy mildew are rarely reported, and its potential regulatory mechanism is also unknown.
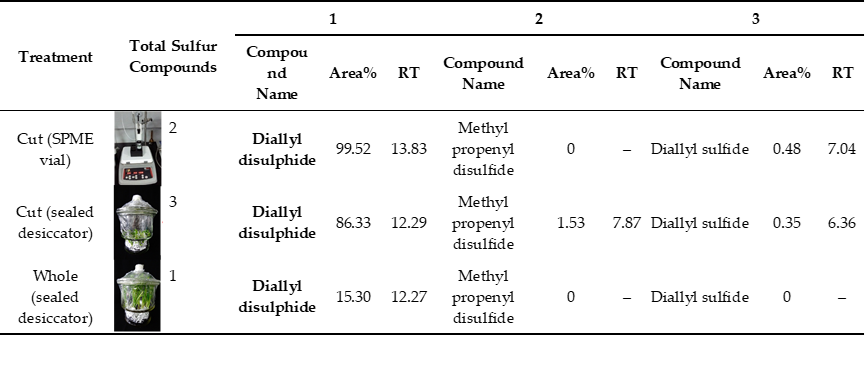
2. Identification of Volatile Allelochemicals in Green Garlic
Identification of the effective components of VOCs collected from green garlic is the basis to reveal its mechanism in scavenging ROS. For the VOCs collected from the green garlic segments that were contained in the SPME vial, only two compounds including DADS and diallyl sulfide (DAS) were identified (Figure 1A and Table 1). The retention time and relative peak area of DADS were 13.83 min and 99.52%, respectively, while, DAS had a retention time of 7.04 min and a relative peak area of 0.48%. Among the VOCs collected using green garlic segments that were put in a sealed desiccator, 17 compounds were detected, in which there were three sulfur compounds including DADS, methyl propenyl disulfide and DAS, with relative peak areas of 86.33%, 1.53% and 0.35%, respectively (Figure 1B and Table 1). All the other compounds were heterocyclic compounds and long chain hydrocarbon compounds, which are impurities in air. There were 42 compounds identified in the VOCs obtained from the whole green garlics that were placed in a sealed desiccator. Among the 42 detected compounds there was only one sulfur compound, which was DADS with a relative peak area of 15.3% (Figure 1C and Table 1), while the others were also heterocyclic compounds and long chain hydrocarbon compounds. The qualitative identification result showed that DADS is the main volatile compound of green garlic. Compared with the whole green garlic, the cut green garlic segments produced 5-fold more DADS.
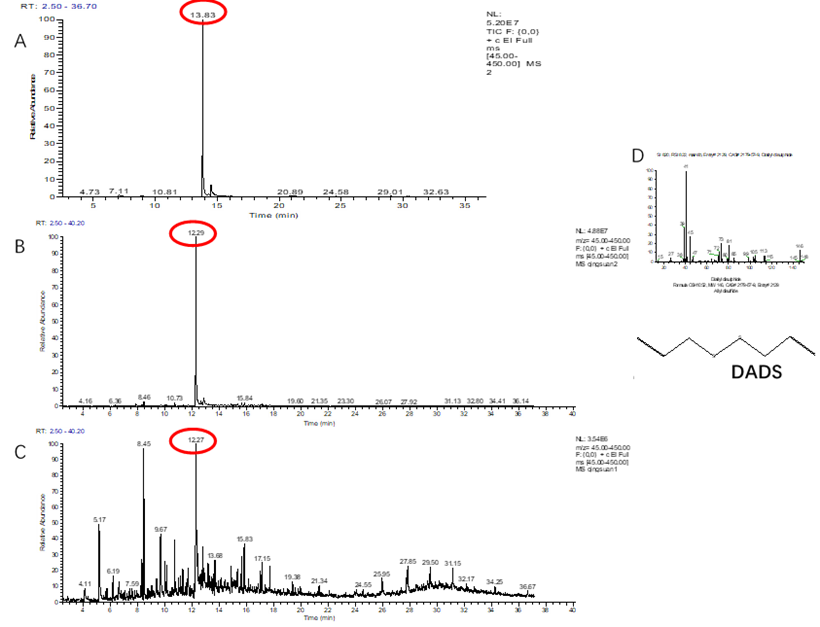
Figure 1. Identification of the collected volatiles from green garlic volatiles by GC-MS. A, Cut (SPME vial); B, Cut (sealed desiccator); C, Whole (sealed desiccator); D, MS spectrum and compound structure of DADS. n = 3, three biological replicates.
The result of an external standard method for quantitative analysis of volatiles showed that the linear relation between DADS concentration (mmol/L) (y) and peak area (x) fits a simple linear regression equation (Equation (1):
|
y = 5E + 09x + 3E + 07, R2 = 0.9903
|
(1)
|
Based on this equation, the DADS that released by green garlics has a concentration of about 0.08 mg/g.
Table 1. Total sulfur compounds of green garlic volatile compounds among different collecting methods.
Our biological tests showed that the green garlic treatment had a consistent effect with the DADS treatment (Figure 2). When the concentration of DADS was higher than 1 mmol/L, cucumber leaves turned yellowish (Figure 2). Inversely, when the green garlic treatment was less than 2 g or the concentration of DADS was lower than 1 mmol/L, the cucumber leaves had no morphology changes (Figure 2). The amount of DADS volatilized by 2 g of green garlic approached 1 mmol/L DADS treatment based on Equation (1). In addition, higher concentrations of green garlic volatiles and DADS can cause cucumber leaves turn to soft and rot (Figure 2). These results confirmed that DADS is the main allelochemical of green garlic volatiles.

Figure 2. The biological test and its results of green garlic volatiles and DADS on cucumber leaf. A, effect of green garlic (0.0, 1.0, 2.0, 3.0, 5.0 g) volatiles on cucumber leaf color and structure. B, effect of DADS (0, 0.5, 1, 2, 3, 5, and 10 mmol/L) on cucumber leaf color and structure. Five leaves from 5 plants were used for each treatment.
3. DADS-Induced Cucumber Resistance to Downy Mildew
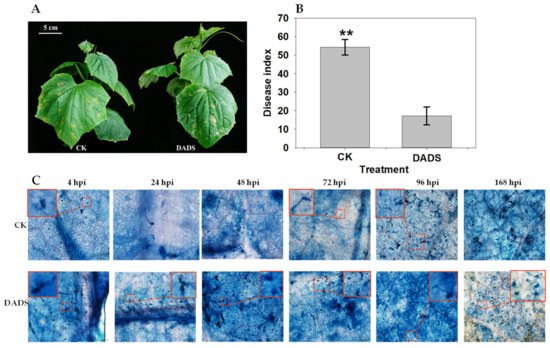
To investigate the effect of DADS on P. cubensis infection, the phenotypic changes of cucumber seedlings under the inoculation of pathogens were observed and recorded. For the seedling of CK, the pathogen-inoculated area firstly showed a water-soaking phenotype and then developed chlorotic lesions (Figure 3A). However, the DADS-treated seedlings showed smaller, less chlorotic lesions (Figure 3A). Statistically, the disease index of DADS treatment (DI = 17.2) was significantly lower than that of CK (DI = 54.3) (p < 0.01) (Figure 3B). Briefly, compared with the CK seedlings, the DADS-treated cucumber seedlings showed better resistance to downy mildew based on the occurred symptoms.
Figure 3. Comparison of leaf morphology (A), disease index (B), and process of P. cubensis infection (C) between DADS treated and CK cucumber seedlings. The inserted box in the top corner is a magnified image of the selected area. Data are means ± standard errors (n = 3, three biological replicates). Asterisks indicate significant differences between DADS treatment and CK (** p < 0.01) based on a one-way ANOVA followed by a t-test. Scale bars: 5 cm (A), 100 μm, 20 μm (C).
Microscopic examination of the growth process of P. cubensis among the infected lesions also showed consistent results with the observed phenotypic changes. The sporangiophore occurred as early as 24 hpi (hours of post inoculation) in CK seedlings, while the DADS-treated seedlings had not shown sporangiophore at this time point (Figure 3C). At 96 hpi, many sporangiophores grew in CK seedlings, but it was not observed in DADS-treated seedlings. Sporangiophore development occurred with a lot of sporangium in CK seedlings at 168 hpi, but these phenomena had not occurred in DADS-treated seedlings. In general, the growth of P. cubensis in the DADS treated seedling were relatively slower and abnormal (less sporangiophore and rare sporangium) compared with that in the seedlings of CK, which might further result in the DADS-treated cucumber seedlings with resistance features to downy mildew.
4. DADS Induced More ROS and Lignin Accumulations and Defense Enzymes Divergences after P. cubensis Inoculation
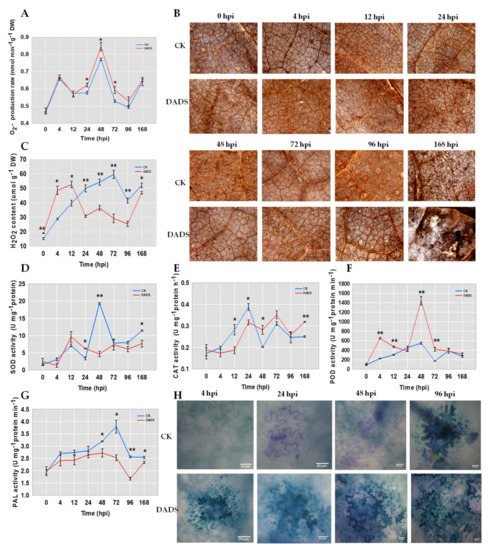
The production of cellular ROS, including H2O2 and superoxide anion, can be induced by pathogen invasion . To verify whether ROS accumulation was associated with the DADS induced cucumber resistance to P. cubensis, the contents of H2O2 and superoxide anion in the leaves of DADS-treated seedlings were checked. DADS-treated leaves showed a similar changing trend with CK for the content of superoxide anion, but the contents in DADS-treated leaves were higher than that in CK, especially for the time points of 24, 48, and 72 hpi (Figure 4A). Either inoculated or not, H2O2 was found in both DADS treatment and CK, as manifested by quantitative measurement and histochemical staining with DAB (Figure 4B, C). DADS-treated leaves accumulated significantly higher levels of H2O2 at 0, 4, and 12 hpi than those of CK leaves (Figure 4B, C). However, DADS-treated leaves accumulated significantly lower levels of H2O2 than those of CK leaves after 24 hpi (Figure 4C). In DADS-treated leaves, a dramatic increase in H2O2 content was observed at 4 hpi, while a second slight increase was detected between 24 hpi and 48 hpi (Figure 4C). In CK leaves, only a single increase was observed, peaking at 72 hpi (Figure 4C). At the final monitored time point of 168 hpi, H2O2 content in the DADS treated leaves was lower than that in CK (Figure 4C).
Figure 4. Comparison of ROS (A–C), defense enzyme (D–G), and lignin (H) in DADS-treated and CK cucumber leaves with P. cubensis infection. (B) Histological observation of H2O2 by DAB dyed. (H) Histological observation of lignin by TB dyed. Data are means ± standard errors (n = 3, three biological replicates). Asterisks indicate significant differences between the DADS treatment and CK (* p < 0.05; ** p < 0.01) based on a one-way ANOVA followed by a t−test. Scale bars: 200 μm (B), 100 μm (H).
Since the biosynthesis of H2O2 was mainly controlled by SOD, and its degradation was regulated by POD and CAT, enzymatic assays were performed to firstly check the activities of these three enzymes (SOD, CAT, and POD). Additionally, the activities of PAL, the first enzyme in the phenylpropanoid metabolism pathway , were also examined. The results showed that the activity of SOD in DADS-treated leaves was significantly higher than that in CK leaves at 24 hpi, while significantly lower activities were shown in DADS treated leaves at both 48 and 168 hpi (Figure 4D). The activities of CAT in DADS-treated leaves were significantly higher and lower than those in CK leaves at both 48 and 168 hpi and both 12 and 24 hpi, respectively (Figure 4E). DADS-treated leaves showed continuously higher POD activities than those in CK leaves at 4, 12, 48, and 72 hpi (Figure 4F). The activities of PAL in DADS-treated leaves were lower than those in CK leaves, especially for the time points of 48, 72, 96, and 168 hpi (Figure 4G). Collectively, considering the enzyme corresponding functions, the enzyme activity changes of SOD, POD, and CAT were consistent with the changes of H2O2 content, and the DADS-treated leaves showed stronger ROS scavenging capabilities than the CK. This might suggest that the different accumulation of H2O2 may be a reason for the defense resistance difference between DADS treated and CK cucumber seedlings.
Previous studies reported that lignin protects cell wall polysaccharides from microbial degradation, and its biosynthesis was induced upon pathogen infection. Therefore, we also check the lignin accumulations in the leaves of DADS-treated and CK cucumber seedlings after the infection of P. cubensis. The accumulations of lignin on the cell walls of cucumber leaves were truly induced when inoculated with downy mildew pathogens (Figure 4H), in which the leaf cells showed extension of lignification stained with toluidine blue (Figure 4H). During the infection process, the cell walls were gradually thickened, and their intensities were simultaneously deepened, especially at 4 hpi, and remained nearly unchanging since the 48 hpi (Figure 4H). However, the stained cell walls were not shown in CK leaves at 4 hpi (Figure 4H), and until the 96 hpi, the cell walls of CK leaves had similar performance with that of DADS-treated leaves at 48 hpi (Figure 4H). Therefore, DADS-treated leaves could accumulate lignin quicker than CK after the pathogen infection. Taken together, the activations of ROS and lignin accumulation pathways might take important roles in the DADS-induced cucumber resistance to P. cubensis.
5. DADS Induced Phytohormone Changes under the Infection of Downy Mildew
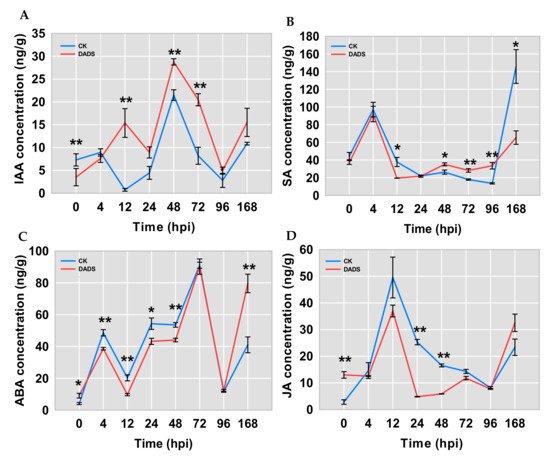
To determine whether phytohormone pathways were involved in the infection of P. cubensis, the contents of IAA, ABA, SA, and JA in cucumber leaves were measured by HPLC-ESI-MS/MS. The results showed that IAA levels in DADS-treated leaves were significantly increased and higher than those in CK leaves at 12, 48, and 72 hpi, while lower IAA content in DADS-treated leaves was detected at 0 hpi (Figure 5A). The contents of SA in DADS-treated leaves were significantly higher than those in CK leaves at 48, 72, and 96 hpi, and were significantly lower at both 12 and 168 hpi (Figure 5B). The SA content had a peak at 4 hpi for both the DADS-treated and CK leaves and then decreased and kept relatively steady until 96 hpi. Compared with the CK, the ABA content in DADS-treated leaves showed a significant fluctuation of higher to lower and then to higher levels (Figure 5C). The content of JA in DADS-treated leaves was significantly higher than that in CK leaves at 0 hpi, while, inversely, it showed significantly lower levels at both 24 and 48 hpi (Figure 5D). Similarly, when compared with the CK, the JA content in DADS-treated leaves also showed higher to lower and then higher levels from 0 to 168 hpi (Figure 3D).
Figure 5. Comparisons of the IAA (A), ABA (B), SA (C), and JA (D) content in DADS-treated and CK leaves with P. cubensis infection. Data are means ± standard errors (n = 3, three biological replicates). Asterisks indicate significant differences between the DADS treatment and CK (* p < 0.05; ** p < 0.01) based on a one-way ANOVA followed by a t−test.