Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Susanna Var | + 2702 word(s) | 2702 | 2021-12-23 09:21:53 | | | |
| 2 | Lindsay Dong | Meta information modification | 2702 | 2021-12-24 02:27:40 | | | | |
| 3 | Lindsay Dong | Meta information modification | 2702 | 2022-03-28 04:38:01 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Var, S. Microglia and Macrophages as Emerging Therapies for Stroke. Encyclopedia. Available online: https://encyclopedia.pub/entry/17528 (accessed on 08 June 2026).
Var S. Microglia and Macrophages as Emerging Therapies for Stroke. Encyclopedia. Available at: https://encyclopedia.pub/entry/17528. Accessed June 08, 2026.
Var, Susanna. "Microglia and Macrophages as Emerging Therapies for Stroke" Encyclopedia, https://encyclopedia.pub/entry/17528 (accessed June 08, 2026).
Var, S. (2021, December 23). Microglia and Macrophages as Emerging Therapies for Stroke. In Encyclopedia. https://encyclopedia.pub/entry/17528
Var, Susanna. "Microglia and Macrophages as Emerging Therapies for Stroke." Encyclopedia. Web. 23 December, 2021.
Copy Citation
The mechanisms underlying the involvement of microglia and macrophages in both neuroprotection and neurogenesis after stroke are widely acknowledged as having a role in ischemic stroke pathology Since microglia and macrophages are regarded as major players in the pathological progression of ischemic stroke, modulating inflammation and neurological recovery is a hopeful strategy for treating the long-term outcomes after ischemic injury.
stroke
macrophage
microglia
neurogenesis
neuroprotection
inflammation
transplantation
therapies
1. Dichotomous Role of Microglia and Macrophages
1.1. Microglial Activation during Stroke
After the onset of brain ischemia, microglia undergo morphological and functional changes in the penumbra, while macrophages also infiltrate the brain parenchyma and migrate toward the infarct area. Studies have shown that activated microglia can have both a beneficial and detrimental effect during all stages of ischemic stroke; however, the timing and kinetics at which specific anti- and proinflammatory events occur may influence the nature of the outcome (Figure 1). Activated microglia release cytotoxic factors that can exacerbate ischemia and enable poststroke inflammation, displaying elevated levels of reactive oxygen species and TNF [1]. Several reports maintain this detrimental role of microglia in stroke; however, these reports are not consistent with their markers, often looking broadly at Iba1+ or CD11b+ cells, and sometimes using glial fibrillary acidic protein (GFAP), a marker often used to label reactive astrocytes to identify microglia.
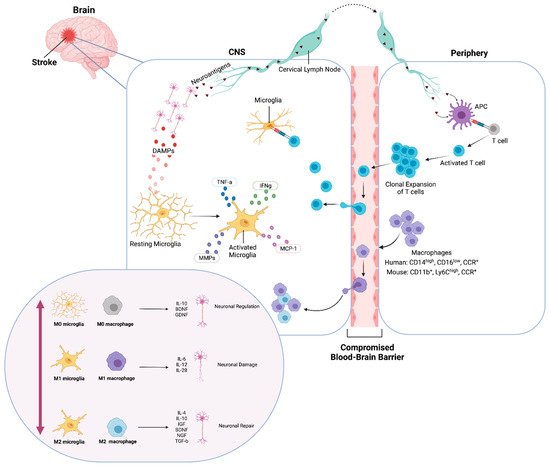
Figure 1. Ischemic inflammatory response of microglia and macrophages. Immediately after an ischemic event, DAMPs activate resting (M0) microglia and neuroantigens are released. Microglia produce cytokines and chemokines. Transendothelial migration of monocytes and macrophages occurs through the compromised BBB. Neuroantigens are processed and presented by APCs and activate CD4+ T-cells, which undergo clonal expansion, promoting inflammation and neuronal damage. Classically activated (M1) microglia and macrophages release pro-inflammatory factors and contribute to neuronal damage. Conversely, alternatively activated (M2) microglia and macrophages release anti-inflammatory factors and contribute to neuronal repair and neurogenesis. Created with BioRender.com.
1.2. Macrophage Activation Profiles
There has been evidence of infiltration of peripheral monocytes to the brain in response to DAMPs produced after ischemic stroke, with the presence of neutrophils and macrophages in the site of CNS injury [2][3][4][5] (Figure 1). Activated macrophages can be categorized on a spectrum of functional activity, starting at resting (M0), classical activation (M1), which promotes inflammatory responses, to alternate activation (M2), which promotes tissue remodeling, wound healing, and immune regulation [6][7].
This switch in macrophage phenotype has been explored in ischemic models. For example, monocyte chemoattractant protein-1 (MCP-1) possesses cytokine-like properties and plays a significant role in macrophage and Ly-6Chi (CCR2+) proinflammatory monocyte migration to injury sites. The increase in CCL2 (MCP-1) expression in the ischemic hemisphere leads to an increased infarct volume [8][9], while the inhibition of CCR2 or MCP-1 resulted in a reduced infarct size [10][11]. In contrast, Ly-6Clo monocytes that do not express CCR2, but express the CX3CR1 receptor for CXC3CL1 fractalkine, develop into M2 macrophages after recruitment to normal tissues [12]. This understanding has led to many studies to focus on skewing microglia and macrophage toward the M2 phenotype rather than M1.
It is important to note that the M0/M1/M2 trichotomy is an oversimplification of activation states as the status of macrophages may include overlapping functional phenotype as well as different ones. For example, the M2 phenotype includes subpopulations such as M2a, M2b, M2c, and Mox, each with distinct physiological functions [13]. Not all current studies in CNS injuries have characterized these subpopulations, so while the M0/M1/M2 classification is broad, it is still a meaningful concept to facilitate our understanding of the functional status of macrophages.
2. Neuroprotection after Stroke
2.1. Angiogenesis
Microglia are known to influence angiogenesis, whereas the elimination of microglia or deficiency in macrophage colony-stimulating factory causes a reduction in retinal vasculature [14][15]. During ischemic stroke, Iba1+ microglia and macrophages cluster around the vasculature and release vascular endothelial growth factor to promote the reconstruction of blood vessels following stroke [16][17][18]. Pro-angiogenic microglia and macrophages may enhance neural proliferation and differentiation following stroke, possibly contributing to neurogenesis and CNS repair.
2.2. Synaptic Remodeling
It is often reported that ischemic stroke can lead to synaptic dysfunction; however, it is known that microglia and macrophages participate in synaptic remodeling and refining neural circuitry [19]. Microglia promote spine formation and synaptic maturation through CX3CR1 and complement proteins as well as synaptic pruning in the brain during development [20][21][22]. Modulation of synaptic function and the neural circuitry is also dependent on the activation state of microglia and macrophages. For example, one of the inflammatory mediators of neurotoxicity in stroke is NADPH oxidase, which is triggered by CR3 activation on macrophages and microglia, resulting in long-term synaptic depression [23][24]. Modulation of activation states of both microglia and macrophages may thus change the inflammatory environment following ischemic injury, encouraging neuroprotection and angiogenesis.
3. Neurogenesis after Stroke
Macrophage and microglial activation have been linked to changes in neurogenesis following ischemic stroke in mammals [25][26]. It has been suggested that newly generated neuroblasts from the subgranular zone (SGZ) of the hippocampus and the subventricular zone (SVZ) of the lateral ventricles can migrate and terminally differentiate into specific cell types in order to generate new granule cell neurons in the hippocampal dentate gyrus as anatomical substrates for learning and memory [27], or replenish the loss of neurons in other regions of the adult brain [28][29], respectively. Some studies suggest that the brain is capable of self-repair after insults of extensive neuronal death through a number of compensatory neurogenesis mechanisms after stroke [30][31][32]. Additionally, enhanced SVZ neurogenesis has been observed after stroke [33][34], and newborn neurons have been found in the ischemic penumbra [35][36].
Microglia and macrophages may participate in regulating neurogenesis by supporting axonal regrowth and regeneration to allow for functional recovery after stroke. Their production of local trophic gradients helps to stimulate axonal sprouting toward the infarct area [37]. Microglia also are necessary for regulating synaptic maturation, while in the setting of microglial injury, there can be synaptic dysfunction [38]. This active regulation of functional synapses in the CNS through axon guidance, synaptic patterning, and cell migration is evidence of their role in modulating neurogenesis [39][40][41].
The increase in neurogenesis after stroke may only be transient, and similar to what is seen in traumatic brain injury models, appears to be disrupted by a secondary inflammatory response [42]. While the production of trophic factors is essential for the migration of newborn neurons [43], activation of microglia and macrophages can also mediate inflammation that is detrimental to neurogenesis [44][45]. CD4+ T cells are essential to maintain homeostatic neurogenesis [46], and they also contribute to learning and memory [47]; however, activated cells responding to the injury may inhibit neurogenesis [48]. New treatment modalities can arise from shedding light on the mechanisms that modulate neurogenesis after stroke, as currently there are none.
4. Therapeutic Perspectives
4.1. Polarization via Small Molecules
Modulating microglial and macrophage activation and polarization through use of various pharmacological and small molecules has been a popular area of study (Figure 2). Minocycline, a tetracycline antibiotic commonly used as an inhibitor of microglial activation, administered five times a week in amyotrophic lateral sclerosis (ALS) mice from 8–24 weeks of age, diminished the expression of M1 microglia and macrophages but not M2 microglia and macrophages [49]. Additionally, treatment with minocycline for one week, beginning at four days after reperfusion injury in rats, preserved adult new neurons, reduced reactive astrocytes, and improved dentate gyrus neurogenesis and neurological function [50]. Typically, ischemic stroke is characterized by the downregulation of the Wnt/beta-catenin signaling pathway; however, activation of Wnt/beta-catenin signaling through TWS119 attenuated neuroinflammation after stroke, by driving microglial anti-inflammatory activation, promotes angiogenesis [51][52].
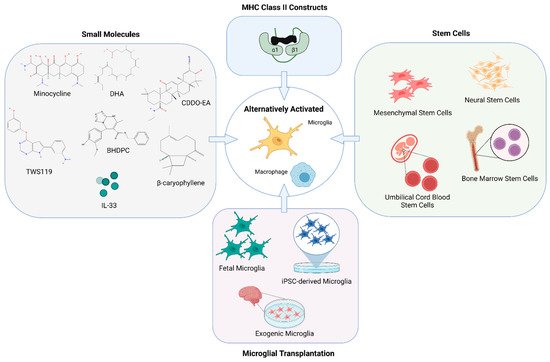
Figure 2. Modulating microglia and macrophage for therapy. Many novel therapeutic approaches have emerged to promote remodeling of the injured brain in stroke models by altering the activation phenotype of microglia and macrophages. Methods for alternative activation of microglia and macrophages include but are not limited to the use of various small molecules, partial MHC class II constructs, stem cell therapies as well as transplantation of microglia as a treatment for both the acute and chronic effects of stroke. Created with BioRender.com.
The nature of the ischemic environment is a key determinant in driving microglia and macrophage function and their activation phenotype. For example, microglia and macrophages are highly susceptible to energy deficits, leading to activation and recruitment; thus, metabolic status of the lesion environment is a major factor in determining the nature of microglial response [53][54]. A number of studies have examined the effects of small molecules on the shift in microglial polarization toward an M2 phenotype after an ischemic event.
Microglia and macrophage activation could also be modulated toward the M2 phenotype through post-stroke treatment with docosahexaenoic acid (DHA) administered immediately after reperfusion and daily for three days thereafter. Treatment with DHA also significantly inhibits infiltration of neutrophils, T, and B lymphocytes [55]. A polarization shift of microglia and macrophages toward the M2 phenotype with a reduction in neurological deficit and infarct volume was also seen when a redox transcription factor NFE2 related factor 2 activator, CDDO-EA, was administered 30 min after the end of the ischemic period in mice [56]. Modulation of microglial M2 polarization via the toll-like receptor 4 pathway, a regulator of macrophage activation and polarization after injury, through the use of β-caryophyllene, a natural bicyclic sesquiterpene, also reduced infarct volume and neurologic deficits in mice after a transient MCAO [57]. Additionally, human stroke patients have increased plasma levels of sST2, an inhibitory IL-33 receptor; while in mice, treatment with IL-33, a cytokine known to induce a shift toward M2 polarization, increased peripheral levels of IL-4 in the spleen and peri-infarct area [58].
Angiogenesis can also be stimulated by modulating microglial polarization toward M2 through AMPK signaling after stroke. The treatment of human umbilical vein endothelial cells (HUVECs) in vitro with conditioned media collected from BV-2 microglial cells in culture incubated in berberine, an isoquinoline alkaloid extract from traditional Chinese medicine, facilitated angiogenesis of HUVEC cells [59]. Moreover, oral administration of berberine in mice subjected to transient MCAO resulted in angiogenesis as revealed by PET/CT imaging.
4.2. Stem Cell Therapies
Stem cell therapies for stroke have the potential to provide neurorestorative benefits. The type of stem cell and route of administration determines the efficacy of stem cell-based therapies in mediating a therapeutic effect (Figure 2). The therapeutic of effect of neural stem cells have been attributed to the promotion of neurogenesis and regeneration.
Neural stem cell (NSC) transplants have been shown to improve behavioral outcomes and angiogenesis in rat models of ischemic stroke, leading to studies interrogating human NSCs (hNSC) [60][61]. Grafted hNSCs can differentiate into neurons, astrocytes, and oligodendrocytes in the ischemic brain, even making direct contact with stroke-damaged vasculature and participating in remyelination, respectively [62]. More recently, administration of clinical-grade hNSC line CTX0E03 by intracerebral implantation has been shown to improve upper limb function in stroke patients [63]. Success in previous clinical trials will allow researchers to utilize this cell line for other neurological diseases and disorders.
Bone marrow stem cells (BMSC) have been shown to migrate to the site of ischemia and differentiate into neural cells [64]. More recently, bone marrow-derived mesenchymal stem cells (MSC) SB632 were associated with improvement in clinical outcome in stroke patients with chronic motor deficits [65]. These cells were intracerebrally transplanted and well tolerated after 12 months. The advantages of MSC transplantation for functional recovery, angiogenesis, and endogenous neurogenesis have also been shown in animal stroke models [66][67][68][69]. However, one major limitation of the current stem cell therapies is the sparse migration of MCSs to the ischemic brain regions after transplantation.
Umbilical cord blood stem cells (UCBSC) offer many advantages over other types of stem cells. The immune properties of UCBSCs allow for increased tolerance for human leukocyte antigen mismatches and decreased incidence of graft-versus-host disease as well as ease of procurement and availability [70][71]. Non-hematopoietic umbilical cord blood stem cell (nh-UCBSC) has also been shown to ameliorate ischemic brain injury by reducing the number of macrophages and microglia and normalizing the number of B cells and T cells in the brain following stroke when administered 48 h after the ischemic injury [72][73].
5. Chronic Stroke
Antigen Presentation and Cognitive Decline
After an ischemic event, antigen-presenting cells (APCs) accumulate in the brain parenchyma and express MHC class I and II cell surface molecules [74][75][76][77]. APCs engulf and process the peptides of damaged and dying cells and then present these peptides to CD8+ T cells, in association with MHC class I, and to CD4 T+ cells, in association with MHC class II (Figure 1). The activation of the T cell population to target self-antigens in the CNS can potentially contribute to the progressive, cognitive decline seen in many patients after stroke. Macrophage populations of dendritic cells that express both Iba1 and CD11lb markers migrate in the choroid plexus in mice and behave as APCs [78][79]. A population of Iba1+CD11b+CD45int are a subset of microglial cells that can also upregulate antigen presentation and activation markers [80][81][82]. While CD4+ T-cell mediated neuroprotection is initiated by APCs, MHC class II+ microglia APCs are required for a secondary restimulation and can drive antigen-specific neuroprotection [83], demonstrating a close relationship between these two cell types.
The volume of infarction and severity of stroke is correlated with the concentration of neural antigens found in the serum of patients after stroke [84][85]. Autoantibodies against brain antigens have been found in the CSF of patients with stroke, possibly contributing to the development of post-stroke cognitive impairment [86][87][88].
6. Microglial Transplantation as a Treatment for Chronic Stroke
6.1. Transplantation of Fetal Microglia
Transplantation of fetal microglia has been demonstrated to improve ischemia-induced functional changes and apoptotic events after stroke. Ischemia was induced in rats by MCAO and human microglial cells (HMO6) from fetal telencephalon tissue were transplanted into a treated group [89]. Animals that received HMO6 transplantation showed significantly reduced infarct volume and apoptotic cells in the infarct core and penumbra when compared to the control group that did not receive the transplant. Gene expression analysis showed that HMO6 cells migrated to the ischemic area and produced neurotrophic factors such as GDNF and BDNF and anti-inflammatory cytokines IL-4 and IL-5, which reduced the endogenous glial response. The accumulation of transplanted microglia in the lesion core suggests that microglia can potentially be used in gene therapy as a vehicle for the transfer of therapeutic genes.
Fetal microglial transplantation may improve stroke outcomes by modulating inflammation and facilitating angiogenesis. Expression of IL-1β is high in HMO6 and has been shown to increase VEGF mRNA expression in HMO6 cell lines when co-cultured with a mesenchymal stem cell line B10 [90]. White matter lesions (WMLs) as a result of chronic cerebral ischemia are thought to contribute to vascular dementia. HMO6 cells were injected intravenously and WML development was assessed in a chronic cerebral hypoperfusion rat model induced by bilateral common carotid artery occlusion (BCAO) [91]. The authors found that the transplantation of HMO6 inhibited BCAO-induced WMLs and displayed an early and prolonged improvement in WMLs.
6.2. Transplantation of iPSC Derived Microglia
Microglia derived from iPSCs are an important consideration for translational neuroimmunology research and hold great potential for therapy. iPSC-microglia are able to recapitulate the inflammatory-modulating properties of brain-resident microglia by resembling the in vivo phenotypical profiles of microglia within stroke lesions and responding to IL-13 stimulation [92]. While primary microglia grown in culture tend to have major differences in morphology and gene expression when compared to resident-microglia in the brain, single-cell RNA-sequencing of transplanted iPSC-derived microglia showed similarities to healthy primary microglia in both gene expression profiles and phenotypic morphology and are able to functionally integrate in the chimeric mouse brain [93][94]. Beyond iPSC-derived microglia serving as a suitable representation of human microglial cells, the cells have also demonstrated the ability to mitigate neuronal loss after stroke in rats aged 24 months.
6.3. Prospects for Transplanting Exogenic Microglia
Exogenous microglia also have the potential to protect against neuronal damage after stroke by exhibiting an affinity for ischemic brain lesions. Injection of exogenous microglia was show to promote CA1 cell survival by migrating to the CA1 cell layer and increasing the expression of BDNF and GDNF in the ischemic hippocampus [95]. Cell therapies involving hypoxic preconditioning is becoming a popular strategy for treating ischemic stroke. Microglia subjected to oxygen-glucose deprivation before transplantation can induce anti-inflammatory microglia and result in the overexpression of remodeling factors such as MMP-9, VEGF, and TGF-β in the injured brain parenchyma [96]. Such therapeutic potential has led to hypoxic preconditioning of stem cells to facilitate the switching of microglia toward an anti-inflammatory polarization for use in alleviating ischemic injury [97].
References
- Szalay, G.; Martinecz, B.; Lénárt, N.; Környei, Z.; Orsolits, B.; Judák, L.; Császár, E.; Fekete, R.; West, B.L.; Katona, G.; et al. Microglia protect against brain injury and their selective elimination dysregulates neuronal network activity after stroke. Nat. Commun. 2016, 7, 11499.
- Amantea, D.; Nappi, G.; Bernardi, G.; Bagetta, G.; Corasaniti, M.T. Post-ischemic brain damage: Pathophysiology and role of inflammatory mediators. FEBS J. 2008, 276, 13–26.
- Stevens, S.L.; Bao, J.; Hollis, J.; Lessov, N.S.; Clark, W.M.; Stenzel-Poore, M.P. The use of flow cytometry to evaluate temporal changes in inflammatory cells following focal cerebral ischemia in mice. Brain Res. 2002, 932, 110–119.
- Schilling, M.; Strecker, J.-K.; Ringelstein, E.B.; Schäbitz, W.-R.; Kiefer, R. The role of CC chemokine receptor 2 on microglia activation and blood-borne cell recruitment after transient focal cerebral ischemia in mice. Brain Res. 2009, 1289, 79–84.
- Biswas, S.K.; Mantovani, A. Macrophage plasticity and interaction with lymphocyte subsets: Cancer as a paradigm. Nat. Immunol. 2010, 11, 889–896.
- Das, A.; Sinha, M.; Datta, S.; Abas, M.; Chaffee, S.; Sen, C.K.; Roy, S. Monocyte and Macrophage Plasticity in Tissue Repair and Regeneration. Am. J. Pathol. 2015, 185, 2596–2606.
- Zhao, Y.-L.; Tian, P.-X.; Han, F.; Zheng, J.; Xia, X.-X.; Xue, W.-J.; Ding, X.-M.; Ding, C.-G. Comparison of the characteristics of macrophages derived from murine spleen, peritoneal cavity, and bone marrow. J. Zhejiang Univ. Sci. B 2017, 18, 1055–1063.
- Chen, Y.; Hallenbeck, J.M.; Ruetzler, C.; Bol, D.; Thomas, K.; Berman, N.E.J.; Vogel, S.N. Overexpression of Monocyte Chemoattractant Protein 1 in the Brain Exacerbates Ischemic Brain Injury and is Associated with Recruitment of Inflammatory Cells. J. Cereb. Blood Flow Metab. 2003, 23, 748–755.
- Schuette-Nuetgen, K.; Strecker, J.-K.; Minnerup, J.; Ringelstein, E.B.; Schilling, M. MCP-1/CCR-2-double-deficiency severely impairs the migration of hematogenous inflammatory cells following transient cerebral ischemia in mice. Exp. Neurol. 2012, 233, 849–858.
- Hughes, P.M.; Allegrini, P.R.; Rudin, M.; Perry, V.H.; Mir, A.K.; Wiessner, C. Monocyte Chemoattractant Protein-1 Deficiency is Protective in a Murine Stroke Model. J. Cereb. Blood Flow Metab. 2002, 22, 308–317.
- Dimitrijevic, O.B.; Stamatovic, S.M.; Keep, R.; Andjelkovic, A.V. Absence of the Chemokine Receptor CCR2 Protects Against Cerebral Ischemia/Reperfusion Injury in Mice. Stroke 2007, 38, 1345–1353.
- Auffray, C.; Sieweke, M.H.; Geissmann, F. Blood Monocytes: Development, Heterogeneity, and Relationship with Dendritic Cells. Annu. Rev. Immunol. 2009, 27, 669–692.
- Hu, X.; Leak, R.K.; Shi, Y.; Suenaga, J.; Gao, Y.; Zheng, P.; Chen, J. Microglial and macrophage polarization—new prospects for brain repair. Nat. Rev. Neurol. 2015, 11, 56–64.
- Kubota, Y.; Takubo, K.; Shimizu, T.; Ohno, H.; Kishi, K.; Shibuya, M.; Saya, H.; Suda, T. M-CSF inhibition selectively targets pathological angiogenesis and lymphangiogenesis. J. Cell Biol. 2009, 185, i6.
- Zhao, X.; Eyo, U.B.; Murugan, M.; Wu, L.-J.; Murguan, M. Microglial interactions with the neurovascular system in physiology and pathology. Dev. Neurobiol. 2018, 78, 604–617.
- Xie, L.; Mao, X.; Jin, K.; Greenberg, D.A. Vascular endothelial growth factor-B expression in postischemic rat brain. Vasc. Cell 2013, 5, 8.
- Zhang, Z.G.; Zhang, L.; Jiang, Q.; Zhang, R.; Davies, K.; Powers, C.; van Bruggen, N.; Chopp, M. VEGF enhances angiogenesis and promotes blood-brain barrier leakage in the ischemic brain. J. Clin. Investig. 2000, 106, 829–838.
- Jolivel, V.; Bicker, F.; Binamé, F.; Ploen, R.; Keller, S.; Gollan, R.; Jurek, B.; Birkenstock, J.; Poisa-Beiro, L.; Bruttger, J.; et al. Perivascular microglia promote blood vessel disintegration in the ischemic penumbra. Acta Neuropathol. 2014, 129, 279–295.
- Nie, J.; Yang, X. Modulation of Synaptic Plasticity by Exercise Training as a Basis for Ischemic Stroke Rehabilitation. Cell. Mol. Neurobiol. 2016, 37, 5–16.
- Wu, Y.; Dissing-Olesen, L.; MacVicar, B.; Stevens, B. Microglia: Dynamic Mediators of Synapse Development and Plasticity. Trends Immunol. 2015, 36, 605–613.
- Paolicelli, R.C.; Bolasco, G.; Pagani, F.; Maggi, L.; Scianni, M.; Panzanelli, P.; Giustetto, M.; Ferreira, T.A.; Guiducci, E.; Dumas, L.; et al. Synaptic Pruning by Microglia Is Necessary for Normal Brain Development. Science 2011, 333, 1456–1458.
- Miyamoto, A.; Wake, H.; Ishikawa, A.W.; Eto, K.; Shibata, K.; Murakoshi, H.W.H.; Koizumi, K.S.S.; Moorhouse, A.; Yoshimura, A.W.I.Y.; Nabekura, A.M. Microglia contact induces synapse formation in developing somatosensory cortex. Nat. Commun. 2016, 7, 12540.
- Zhang, L.; Wu, J.; Duan, X.; Tian, X.; Shen, H.; Sun, Q.; Chen, G. NADPH Oxidase: A Potential Target for Treatment of Stroke. Oxid. Med. Cell. Longev. 2016, 2016, 5026984.
- Zhang, J.; Malik, A.; Choi, H.B.; Ko, R.W.; Dissing-Olesen, L.; MacVicar, B.A. Microglial CR3 Activation Triggers Long-Term Synaptic Depression in the Hippocampus via NADPH Oxidase. Neuron 2014, 82, 195–207.
- Xiong, X.-Y.; Liu, L.; Yang, Q.-W. Functions and mechanisms of microglia/macrophages in neuroinflammation and neurogenesis after stroke. Prog. Neurobiol. 2016, 142, 23–44.
- Qin, C.; Zhou, L.-Q.; Ma, X.-T.; Hu, Z.-W.; Yang, S.; Chen, M.; Bosco, D.; Wu, L.-J.; Tian, D.S. Dual Functions of Microglia in Ischemic Stroke. Neurosci. Bull. 2019, 35, 921–933.
- Glover, L.R.; Schoenfeld, T.J.; Karlsson, R.-M.; Bannerman, D.M.; Cameron, H.A. Ongoing neurogenesis in the adult dentate gyrus mediates behavioral responses to ambiguous threat cues. PLoS Biol. 2017, 15, e2001154.
- Kerschensteiner, M.; Meinl, E.; Hohlfeld, R. Neuro-Immune Crosstalk in CNS Diseases. Results Probl. Cell Differ. 2010, 51, 197–216.
- Zhao, C.; Deng, W.; Gage, F.H. Mechanisms and Functional Implications of Adult Neurogenesis. Cell 2008, 132, 645–660.
- Lin, R.; Iacovitti, L. Classic and novel stem cell niches in brain homeostasis and repair. Brain Res. 2015, 1628, 327–342.
- Otero, L.; Zurita, M.; Bonilla, C.; Rico, M.A.; Aguayo, C.; Rodriguez, A.; Vaquero, J. Endogenous neurogenesis after intracerebral hemorrhage. Histol. Histopathol. 2012, 27, 303–315.
- Tobin, M.K.; Bonds, J.A.; Minshall, R.D.; Pelligrino, D.A.; Testai, F.D.; Lazarov, O. Neurogenesis and Inflammation after Ischemic Stroke: What is Known and Where We Go from Here. J. Cereb. Blood Flow Metab. 2014, 34, 1573–1584.
- Kreuzberg, M.; Kanov, E.; Timofeev, O.; Schwaninger, M.; Monyer, H.; Khodosevich, K. Increased subventricular zone-derived cortical neurogenesis after ischemic lesion. Exp. Neurol. 2010, 226, 90–99.
- Lindvall, O.; Kokaia, Z. Neurogenesis following Stroke Affecting the Adult Brain. Cold Spring Harb. Perspect. Biol. 2015, 7, a019034.
- Tang, Y.; Wang, J.; Lin, X.; Wang, L.; Shao, B.; Jin, K.; Wang, Y.; Yang, G.-Y. Neural Stem Cell Protects Aged Rat Brain from Ischemia–Reperfusion Injury through Neurogenesis and Angiogenesis. J. Cereb. Blood Flow Metab. 2014, 34, 1138–1147.
- Nada, S.E.; Tulsulkar, J.; Shah, Z.A. Heme Oxygenase 1-Mediated Neurogenesis Is Enhanced by Ginkgo biloba (EGb 761®) After Permanent Ischemic Stroke in Mice. Mol. Neurobiol. 2013, 49, 945–956.
- Batchelor, P.E.; Porritt, M.J.; Martinello, P.; Parish, C.L.; Liberatore, G.T.; Donnan, G.A.; Howells, D.W. Macrophages and Microglia Produce Local Trophic Gradients That Stimulate Axonal Sprouting Toward but Not beyond the Wound Edge. Mol. Cell. Neurosci. 2002, 21, 436–453.
- Paolicelli, R.C.; Ferretti, M.T. Function and Dysfunction of Microglia during Brain Development: Consequences for Synapses and Neural Circuits. Front. Synaptic Neurosci. 2017, 9, 9.
- Ji, K.; Akgul, G.; Wollmuth, L.P.; Tsirka, S.E. Microglia Actively Regulate the Number of Functional Synapses. PLoS ONE 2013, 8, e56293.
- Lenz, K.M.; Nelson, L. Microglia and Beyond: Innate Immune Cells as Regulators of Brain Development and Behavioral Function. Front. Immunol. 2018, 9, 698.
- Yin, J.; Valin, K.L.; Dixon, M.L.; Leavenworth, J.W. The Role of Microglia and Macrophages in CNS Homeostasis, Autoimmunity, and Cancer. J. Immunol. Res. 2017, 2017, 5150678.
- Acosta, S.A.; Tajiri, N.; Shinozuka, K.; Ishikawa, H.; Grimmig, B.; Diamond, D.; Sanberg, P.R.; Bickford, P.; Kaneko, Y.; Borlongan, C.V. Long-Term Upregulation of Inflammation and Suppression of Cell Proliferation in the Brain of Adult Rats Exposed to Traumatic Brain Injury Using the Controlled Cortical Impact Model. PLoS ONE 2013, 8, e53376.
- Rotschafer, J.H.; Hu, S.; Little, M.; Erickson, M.; Low, W.C.; Cheeran, M.C. Modulation of neural stem/progenitor cell proliferation during experimental Herpes Simplex encephalitis is mediated by differential FGF-2 expression in the adult brain. Neurobiol. Dis. 2013, 58, 144–155.
- Ma, Y.; Wang, J.; Wang, Y.; Yang, G.-Y. The biphasic function of microglia in ischemic stroke. Prog. Neurobiol. 2016, 157, 247–272.
- Fan, W.; Dai, Y.; Xu, H.; Zhu, X.; Cai, P.; Wang, L.; Sun, C.; Hu, C.; Zheng, P.; Zhao, B. Caspase-3 Modulates Regenerative Response After Stroke. Stem Cells 2013, 32, 473–486.
- Kempermann, G.; Gage, F.H.; Aigner, L.; Song, H.; Curtis, M.A.; Thuret, S.; Kuhn, H.-G.; Jessberger, S.; Frankland, P.W.; Cameron, H.A.; et al. Human Adult Neurogenesis: Evidence and Remaining Questions. Cell Stem Cell 2018, 23, 25–30.
- Baruch, K.; Ron-Harel, N.; Gal, H.; Deczkowska, A.; Shifrut, E.; Ndifon, W.; Mirlas-Neisberg, N.; Cardon, M.; Vaknin, I.; Cahalon, L.; et al. CNS-specific immunity at the choroid plexus shifts toward destructive Th2 inflammation in brain aging. Proc. Natl. Acad. Sci. USA 2013, 110, 2264–2269.
- Hu, S.; Rotschafer, J.H.; Lokensgard, J.R.; Cheeran, M.C.-J. Activated CD8+ T Lymphocytes Inhibit Neural Stem/Progenitor Cell Proliferation: Role of Interferon-Gamma. PLoS ONE 2014, 9, e105219.
- Kobayashi, K.; Imagama, S.; Ohgomori, T.; Hirano, K.; Uchimura, K.; Sakamoto, K.; Hirakawa, A.; Takeuchi, H.; Suzumura, A.; Ishiguro, N.; et al. Minocycline selectively inhibits M1 polarization of microglia. Cell Death Dis. 2013, 4, e525.
- Liu, Z.; Fan, Y.; Won, S.J.; Neumann, M.; Hu, D.; Zhou, L.; Weinstein, P.R.; Liu, J. Chronic Treatment with Minocycline Preserves Adult New Neurons and Reduces Functional Impairment After Focal Cerebral Ischemia. Stroke 2007, 38, 146–152.
- Song, D.; Zhang, X.; Chen, J.; Liu, X.; Xue, J.; Zhang, L.; Lan, X. Wnt canonical pathway activator TWS119 drives microglial anti-inflammatory activation and facilitates neurological recovery following experimental stroke. J. Neuroinflamm. 2019, 16, 1–17.
- Laksitorini, M.; Yathindranath, V.; Xiong, W.; Hombach-Klonisch, S.; Miller, D.W. Modulation of Wnt/β-catenin signaling promotes blood-brain barrier phenotype in cultured brain endothelial cells. Sci. Rep. 2019, 9, 19718.
- Fumagalli, S.; Perego, C.; Pischiutta, F.; Zanier, E.; de Simoni, M.G. The Ischemic Environment Drives Microglia and Macrophage Function. Front. Neurol. 2015, 6, 81.
- Gimeno-Bayón, J.; López-López, A.; Rodríguez, M.; Mahy, N. Glucose pathways adaptation supports acquisition of activated microglia phenotype. J. Neurosci. Res. 2014, 92, 723–731.
- Cai, W.; Liu, S.; Hu, M.; Sun, X.; Qiu, W.; Zheng, S.; Hu, X.; Lu, Z. Post-stroke DHA Treatment Protects Against Acute Ischemic Brain Injury by Skewing Macrophage Polarity Toward the M2 Phenotype. Transl. Stroke Res. 2018, 9, 669–680.
- Lei, X.; Li, H.; Li, M.; Dong, Q.; Zhao, H.; Zhang, Z.; Sun, B.; Mao, L. The novel Nrf2 activator CDDO-EA attenuates cerebral ischemic injury by promoting microglia/macrophage polarization toward M2 phenotype in mice. CNS Neurosci. Ther. 2020, 27, 82–91.
- Tian, X.; Liu, H.; Xiang, F.; Xu, L.; Dong, Z. β-Caryophyllene protects against ischemic stroke by promoting polarization of microglia toward M2 phenotype via the TLR4 pathway. Life Sci. 2019, 237, 116915.
- Korhonen, P.; Kanninen, K.M.; Lehtonen, S.; Lemarchant, S.; Puttonen, K.; Oksanen, M.; Dhungana, H.; Loppi, S.; Pollari, E.; Wojciechowski, S.; et al. Immunomodulation by interleukin-33 is protective in stroke through modulation of inflammation. Brain Behav. Immun. 2015, 49, 322–336.
- Zhu, J.; Cao, D.; Guo, C.; Liu, M.; Tao, Y.; Zhou, J.; Wang, F.; Zhao, Y.; Wei, J.; Zhang, Y.; et al. Berberine Facilitates Angiogenesis Against Ischemic Stroke Through Modulating Microglial Polarization via AMPK Signaling. Cell. Mol. Neurobiol. 2019, 39, 751–768.
- Ishibashi, S.; Sakaguchi, M.; Kuroiwa, T.; Yamasaki, M.; Kanemura, Y.; Shizuko, I.; Shimazaki, T.; Onodera, M.; Okano, H.; Mizusawa, H. Human neural stem/progenitor cells, expanded in long-term neurosphere culture, promote functional recovery after focal ischemia in Mongolian gerbils. J. Neurosci. Res. 2004, 78, 215–223.
- Fukunaga, A.; Uchida, K.; Hara, K.; Kuroshima, Y.; Kawase, T. Differentiation and angiogenesis of central nervous system stem cells implanted with mesenchyme into ischemic rat brain. Cell Transplant. 1999, 8, 435–441.
- Daadi, M.M.; Li, Z.; Arac, A.; Grueter, B.; Sofilos, M.; Malenka, R.C.; Wu, J.C.; Steinberg, G.K. Molecular and Magnetic Resonance Imaging of Human Embryonic Stem Cell–Derived Neural Stem Cell Grafts in Ischemic Rat Brain. Mol. Ther. 2009, 17, 1282–1291.
- Muir, K.W.; Bulters, D.; Willmot, M.; Sprigg, N.; Dixit, A.; Ward, N.; Tyrrell, P.; Majid, A.; Dunn, L.; Bath, P.; et al. Intracerebral implantation of human neural stem cells and motor recovery after stroke: Multicentre prospective single-arm study (PISCES-2). J. Neurol. Neurosurg. Psychiatry 2020, 91, 396–401.
- Chen, J.; Li, Y.; Wang, L.; Lu, M.; Zhang, X.; Chopp, M. Therapeutic benefit of intracerebral transplantation of bone marrow stromal cells after cerebral ischemia in rats. J. Neurol. Sci. 2001, 189, 49–57.
- Steinberg, G.K.; Kondziolka, D.; Wechsler, L.R.; Lunsford, L.D.; Coburn, M.L.; Billigen, J.B.; Kim, A.S.; Bates, D.; King, B.; Case, C. Clinical outcomes of transplanted modified bone marrow-derived mesenchymal stem cells in stroke: A phase 1/2a study. Stroke 2016, 47, 1817–1824.
- Luan, X.; Qiu, H.; Hong, X.; Wu, C.; Zhao, K.; Chen, H.; Zhu, Z.; Li, X.; Shen, H.; He, J. High serum nerve growth factor concentrations are associated with good functional outcome at 3 months following acute ischemic stroke. Clin. Chim. Acta 2018, 488, 20–24.
- Li, Y.; Li, Y.-J.; Li, Z.; Qiu, J.-Y.; Zheng, X.-X.; Bian, T.-T.; Gao, F.-L.; Yu, Y.-Y.; Yang, D.-Z.; Tang, D.-Q. Screening for Potential Bioactive Components in Ginkgo biloba Extract by the Rat Renal Tubular Epithelial Cell Extraction and LC-MS/MS. Comb. Chem. High Throughput Screen. 2015, 18, 514–523.
- Tang, W.; Lv, X.; Huang, J.; Wang, B.; Lin, L.; Shen, Y.; Yao, Y. Neuroprotective Effect of Stroke Pretreated Mesenchymal Stem Cells Against Cerebral Ischemia/Reperfusion Injury in Rats. World Neurosurg. 2021.
- Maacha, S.; Sidahmed, H.; Jacob, S.; Gentilcore, G.; Calzone, R.; Grivel, J.-C.; Cugno, C. Paracrine Mechanisms of Mesenchymal Stromal Cells in Angiogenesis. Stem Cells Int. 2020, 2020, 1–12.
- Metheny, L.; Caimi, P.; De Lima, M. Cord Blood Transplantation: Can We Make it Better? Front. Oncol. 2013, 3, 238.
- Gupta, A.O.; Wagner, J.E. Umbilical Cord Blood Transplants: Current Status and Evolving Therapies. Front. Pediatr. 2020, 8, 570282.
- Shiao, M.L.; Yuan, C.; Crane, A.T.; Voth, J.; Juliano, M.; Stone, L.L.H.; Nan, Z.; Zhang, Y.; Kuzmin-Nichols, N.; Sanberg, P.R.; et al. Immunomodulation with Human Umbilical Cord Blood Stem Cells Ameliorates Ischemic Brain Injury—A Brain Transcriptome Profiling Analysis. Cell Transplant. 2019, 28, 864–873.
- Stone, L.L.H.; Xiao, F.; Rotschafer, J.; Nan, Z.; Juliano, M.; Sanberg, C.D.; Sanberg, P.R.; Kuzmin-Nichols, N.; Grande, A.; Cheeran, M.; et al. Amelioration of Ischemic Brain Injury in Rats with Human Umbilical Cord Blood Stem Cells: Mechanisms of Action. Cell Transplant. 2016, 25, 1473–1488.
- Pösel, C.; Uri, A.; Schulz, I.; Boltze, J.; Weise, G.; Wagner, D.-C. Flow cytometric characterization of brain dendritic cell subsets after murine stroke. Exp. Transl. Stroke Med. 2014, 6, 11.
- Felger, J.C.; Abe, T.; Kaunzner, U.W.; Gottfried-Blackmore, A.; Gal-Toth, J.; McEwen, B.S.; Iadecola, C.; Bulloch, K. Brain dendritic cells in ischemic stroke: Time course, activation state, and origin. Brain Behav. Immun. 2010, 24, 724–737.
- Schwab, J.; Nguyen, T.; Meyermann, R.; Schluesener, H. Human focal cerebral infarctions induce differential lesional interleukin-16 (IL-16) expression confined to infiltrating granulocytes, CD8+ T-lymphocytes and activated microglia/macrophages. J. Neuroimmunol. 2001, 114, 232–241.
- Miro-Mur, F.A.; Urra, X.; Gallizioli, M.; Chamorro, A.; Planas, A.M. Antigen Presentation after Stroke. Neurotherapeutics 2016, 13, 719–728.
- Gottfried-Blackmore, A.; Kaunzner, U.W.; Idoyaga, J.; Felger, J.C.; McEwen, B.S.; Bulloch, K. Acute in vivo exposure to interferon- enables resident brain dendritic cells to become effective antigen presenting cells. Proc. Natl. Acad. Sci. USA 2009, 106, 20918–20923.
- Reboldi, A.; Coisne, C.; Baumjohann, D.; Benvenuto, F.; Bottinelli, D.; Lira, S.A.; Uccelli, A.; Lanzavecchia, A.; Engelhardt, B.; Sallusto, F. C-C chemokine receptor 6-regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nat. Immunol. 2009, 10, 514–523.
- Dando, S.J.; Naranjo Golborne, C.; Chinnery, H.R.; Ruitenberg, M.J.; McMenamin, P.G. A case of mistaken identity: CD11c-eYFP+ cells in the normal mouse brain parenchyma and neural retina display the phenotype of microglia, not dendritic cells. Glia 2016, 64, 1331–1349.
- Gregerson, D.S.; Sam, T.N.; McPherson, S.W. The antigen-presenting activity of fresh, adult parenchymal microglia and perivascular cells from retina. J. Immunol. 2004, 172, 6587–6597.
- Wlodarczyk, A.; Løbner, M.; Cédile, O.; Owens, T. Comparison of microglia and infiltrating CD11c+ cells as antigen presenting cells for T cell proliferation and cytokine response. J. Neuroinflamm. 2014, 11, 57.
- Schetters, S.T.T.; Gomez-Nicola, D.; Garcia-Vallejo, J.J.; Van Kooyk, Y. Neuroinflammation: Microglia and T Cells Get Ready to Tango. Front. Immunol. 2018, 8, 1905.
- Planas, A.M.; Gómez-Choco, M.; Urra, X.; Gorina, R.; Caballero, M.; Chamorro, Á. Brain-Derived Antigens in Lymphoid Tissue of Patients with Acute Stroke. J. Immunol. 2012, 188, 2156–2163.
- Jauch, E.C.; Lindsell, C.; Broderick, J.; Fagan, S.C.; Tilley, B.C.; Levine, S.R. Association of serial biochemical markers with acute ischemic stroke: The National Institute of Neurological Disorders and Stroke recombinant tissue plasminogen activator Stroke Study. Stroke 2006, 37, 2508–2513.
- Doyle, K.; Quach, L.N.; Solé, M.; Axtell, R.C.; Nguyen, T.-V.V.; Soler-Llavina, G.J.; Jurado, S.; Han, J.; Steinman, L.; Longo, F.M.; et al. B-Lymphocyte-Mediated Delayed Cognitive Impairment following Stroke. J. Neurosci. 2015, 35, 2133–2145.
- Bornstein, N.; Aronovich, B.; Korczyn, A.; Shavit, S.; Michaelson, D.; Chapman, J. Antibodies to brain antigens following stroke. Neurology 2001, 56, 529–530.
- Prüss, H.; Iggena, D.; Baldinger, T.; Prinz, V.; Meisel, A.; Endres, M.; Dirnagl, U.; Schwab, J. Evidence of intrathecal immunoglobulin synthesis in stroke: A cohort study. Arch. Neurol. 2012, 69, 714–717.
- Narantuya, D.; Nagai, A.; Sheikh, A.M.; Masuda, J.; Kobayashi, S.; Yamaguchi, S.; Kim, S.U. Human Microglia Transplanted in Rat Focal Ischemia Brain Induce Neuroprotection and Behavioral Improvement. PLoS ONE 2010, 5, e11746.
- Sheikh, A.M.; Yano, S.; Mitaki, S.; Haque, M.A.; Yamaguchi, S.; Nagai, A. A Mesenchymal stem cell line (B10) increases angiogenesis in a rat MCAO model. Exp. Neurol. 2018, 311, 182–193.
- Narantuya, D.; Nagai, A.; Sheikh, A.M.; Wakabayashi, K.; Shiota, Y.; Watanabe, T.; Masuda, J.; Kobayashi, S.; Kim, S.U.; Yamaguchi, S. Microglia transplantation attenuates white matter injury in rat chronic ischemia model via matrix metalloproteinase-2 inhibition. Brain Res. 2010, 1316, 145–152.
- Quarta, A.; Le Blon, D.; D’Aes, T.; Pieters, Z.; Taj, S.H.; Miro-Mur, F.A.; Luyckx, E.; Van Breedam, E.; Daans, J.; Goossens, H.; et al. Murine iPSC-derived microglia and macrophage cell culture models recapitulate distinct phenotypical and functional properties of classical and alternative neuro-immune polarisation. Brain Behav. Immun. 2019, 82, 406–421.
- Svoboda, D.S.; Barrasa, M.I.; Shu, J.; Rietjens, R.; Zhang, S.; Mitalipova, M.; Berube, P.; Fu, D.; Shultz, L.D.; Bell, G.W.; et al. Human iPSC-derived microglia assume a primary microglia-like state after transplantation into the neonatal mouse brain. Proc. Natl. Acad. Sci. USA 2019, 116, 25293–25303.
- Xu, R.; Li, X.; Boreland, A.; Posyton, A.; Kwan, K.; Hart, R.P.; Jiang, P. Human iPSC-derived mature microglia retain their identity and functionally integrate in the chimeric mouse brain. Nat. Commun. 2020, 11, 1–16.
- Imai, F.; Suzuki, H.; Oda, J.; Ninomiya, T.; Ono, K.; Sano, H.; Sawada, M. Neuroprotective Effect of Exogenous Microglia in Global Brain Ischemia. Br. J. Pharmacol. 2006, 27, 488–500.
- Kanazawa, M.; Miura, M.; Toriyabe, M.; Koyama, M.; Hatakeyama, M.; Ishikawa, M.; Nakajima, T.; Onodera, O.; Takahashi, T.; Nishizawa, M.; et al. Microglia preconditioned by oxygen-glucose deprivation promote functional recovery in ischemic rats. Sci. Rep. 2017, 7, 42582.
- Yu, H.; Xu, Z.; Qu, G.; Wang, H.; Lin, L.; Li, X.; Xie, X.; Lei, Y.; He, X.; Chen, Y.; et al. Hypoxic Preconditioning Enhances the Efficacy of Mesenchymal Stem Cells-Derived Conditioned Medium in Switching Microglia toward Anti-inflammatory Polarization in Ischemia/Reperfusion. Cell. Mol. Neurobiol. 2020, 41, 505–524.
More
Information
Subjects:
Neurosciences
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.5K
Revisions:
3 times
(View History)
Update Date:
28 Mar 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





