
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | David Julian McClements | + 1499 word(s) | 1499 | 2021-11-26 09:26:37 | | | |
| 2 | Amina Yu | -5 word(s) | 1494 | 2021-11-30 04:41:32 | | |
Video Upload Options
Colloidal delivery systems can be used to alter the gastrointestinal fate of oil-soluble vitamins, such as their chemical stability and bioaccessibility. The effectiveness of these delivery systems depends on their composition and structure. For instance, the bioaccessibility of oil-soluble vitamins (A, D, and E) has been shown to increase with decreasing droplet size in nanoemulsions, which is attributed to the faster release and solubilization of the vitamins when lipid digestion occurs more rapidly.
1. Introduction
Recently, many sectors of the food industry are focusing on the creation of high-quality plant-based foods that are designed to accurately mimic the sensory attributes of animal-based foods, such as meat, seafood, eggs, and milk [1]. These plant-based foods are being created in response to consumer demand for products that are better for the environment, human health, and animal welfare. Indeed, this is one of the fastest-growing segments of the modern food industry, with rapid advances being made in all food categories [2]. Plant-based foods do not have the same nutritional profiles as the animal-based ones they are designed to replace. They are often deficient in some key nutrients, such as vitamin B 12, vitamin D, omega-3 fatty acids, iron, and zinc. Consequently, there is interest in fortifying them with these nutrients. In addition, the healthiness of these products may be further enhanced by fortifying them with nutraceuticals, such as carotenoids, curcuminoids, and polyphenols. This entry focus on the fortification of plant-based foods with oil-soluble bioactive agents since these are difficult to incorporate into many food and beverage products and often have a low bioavailability after consumption. In particular, focus on the utilization of nanoenabled delivery systems that are entirely assembled from plant-based ingredients to improve the functional performance of these hydrophobic bioactives.
2. Delivery System Types
Microgel beads have also been used to inhibit the digestion of encapsulated lipids by retarding the movement of lipase molecules through the biopolymer network and to the oil droplet surfaces ( Figure 3 ). As an example, encapsulation of lipid droplets within alginate beads significantly reduced the rate and extent of lipid digestion under simulated gastrointestinal conditions [3][4]. The lipid digestion profiles could be modulated by altering the external dimensions and internal pore size of the microgels. For instance, lipid digestion is slower for microgels with smaller internal pores because this inhibits the diffusion of lipase through the biopolymer network [5][6]. In addition, it is slower for larger microgels than smaller ones because the lipase molecules have further to diffuse to reach the encapsulated lipid droplets [6][7]. However, these protective microgels must be designed to retain their physical integrity under the environmental conditions under which they are used; otherwise, they will just release the encapsulated substances.
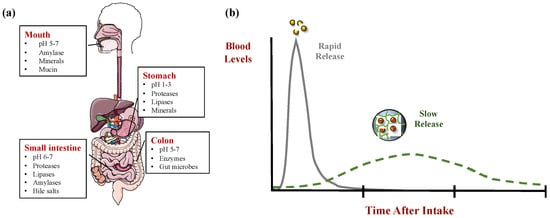
The gastrointestinal fate of nanoemulsions can also be controlled by manipulating the interfacial layers. For instance, the interfacial layer can be designed to inhibit the attachment of bile salts or lipase to the oil droplet surfaces, which can be achieved by using strongly surface-active emulsifiers that are difficult to displace [8] or by forming a mechanically robust coating that is difficult to disrupt [9]. In addition, the emulsifier might form droplets that are unstable to aggregation under gastrointestinal conditions, which leads to flocculation or coalescence, thereby reducing the surface area of the lipase to adsorb and inhibiting lipid digestion [10]. The properties of the interfacial layer can also be manipulated by controlling the aggregation state of the proteins. For instance, some plant proteins (such as soy protein) can form a protective gel layer around oil droplets by controlled thermal treatment and/or salt addition [11][12].
Various kinds of additives can be incorporated into nanoemulsions to improve their performance, including antioxidants to inhibit chemical degradation, weighting agents to retard creaming, ripening inhibitors to prevent Ostwald ripening, thickening, or gelling agents to modify texture and stability, and flavors and colors to enhance the sensory attributes.
The physical state of the lipid nanoparticles influences lipid digestion and bioactive bioavailability. Lipid digestion is typically slower for solid fats than liquid ones since it is more difficult for lipase to hydrolyze crystalline fats [13]. A reduction in lipid digestion usually leads to a reduction in the release of the encapsulated bioactive molecules from the lipid nanoparticles. Consequently, SLNs or NLCs can be used to prolong the release of bioactives within the gastrointestinal tract [14]. It should be noted that solid lipid nanoparticles are still digested within the small intestine (albeit at a slower rate), and so they can still be used as delivery systems for hydrophobic bioactives [15]. For instance, β-carotene encapsulated in SLNs prepared from cocoa butter was shown to have a good bioaccessibility after lipid digestion under simulated small intestine conditions [14].
3. Applications
Colloidal delivery systems can also be used to alter the gastrointestinal fate of oil-soluble vitamins, such as their chemical stability and bioaccessibility [16]. The effectiveness of these delivery systems depends on their composition and structure. For instance, the bioaccessibility of oil-soluble vitamins (A, D, and E) has been shown to increase with decreasing droplet size in nanoemulsions, which is attributed to the faster release and solubilization of the vitamins when lipid digestion occurs more rapidly [17]. Other critical factors for vitamin bioaccessibility are oil phase composition, interfacial properties, and additives [16]. In particular, the lipid phase used should form mixed micelles that are large enough to solubilize the vitamins released from the colloidal particles during digestion.
The water-dispersibility of omega-3 oils can be improved by trapping them inside colloidal particles that have hydrophobic interiors and hydrophilic exteriors, like nanoemulsions, SLNs, or NLCs. The tendency for omega-3 oils to oxidize can be reduced using several approaches. First, it is critical to avoid conditions that could promote oxidation when preparing omega-3 oil-loaded delivery systems, such as oxygen, light, heat, and transition metals. Second, the delivery systems themselves can be designed to inhibit oxidation, i.e., by adding antioxidants or chelating agents [18], by engineering the interfacial layers [19], or by encapsulating within microgels [20][21]. For instance, plant proteins such as those from lentils, peas, and fava beans, are natural antioxidants, which means they can be used to improve the oxidative stability of encapsulated ω-3 oils [22]. Interfacial engineering has been used to improve the oxidative stability of algae oil nanoemulsions by forming pea protein-flaxseed gum coatings around the oil droplets [23]. Encapsulation of ω-3 oil droplets inside alginate microgels has also been shown to improve their oxidative stability [20][21]. The bioavailability of omega-3 oils can also be increased by encapsulating them within colloidal delivery systems. For instance, the encapsulation of these oils within nanoemulsions has been shown to increase their bioaccessibility and bioavailability using in vitro and in vivo studies [24][25], which was mainly attributed to the faster and more extensive digestion of the lipids when they were present within small droplets.
Nanoemulsions are commonly used as delivery systems for carotenoids because of their versatility and ease of production. Like oil-soluble vitamins, the bioavailability of carotenoids depends on oil phase-type, oil concentration, droplet size, emulsifier type, additives, and various other factors. Typically, the bioaccessibility of β-carotene increases as the droplet size decreases because faster lipid digestion leads to faster carotenoid release and solubility [26][27]. The chemical stability of β-carotene in plant-based nanoemulsions has been enhanced by adding a plant-derived natural antioxidant (tannic acid) without adversely affecting the bioaccessibility [28].
Hydrophobic polyphenols need to be encapsulated in delivery systems containing colloidal particles with hydrophobic interiors and hydrophilic exteriors, like nanoliposomes, nanoemulsions, SLNs, NLCs, or some biopolymer nanoparticles. As an example, the water dispersibility, chemical stability, and bioavailability of hydrophobic polyphenols have been improved by encapsulating them within nanoliposomes, nanoemulsions, and zein nanoparticles [29]. Plant-based nanoemulsions have also been used to increase the stability and bioaccessibility of curcumin, resveratrol, and quercetin [30]. It should be noted that polyphenols can also be added to colloidal delivery systems as antioxidants to protect other nutraceuticals [31].
4. Conclusions and Future Studies
One of the fastest-growing segments of the modern food industry is the plant-based foods market. Meat, fish, egg, and dairy analogs are being developed to address consumer concerns about the negative environmental, health, and animal welfare impacts of traditional animal-based foods. However, plant-based foods often do not have the same nutritional profiles as animal-based ones and may therefore benefit from being fortified with micronutrients or nutraceuticals. This entry have shown that colloidal delivery systems that are suitable for the encapsulation, protection, and delivery of vitamins and nutraceuticals can be assembled entirely from plant-based ingredients. Several types of delivery systems are available for this purpose, and the most appropriate one must be selected for a particular application. Moreover, the composition and structure should be controlled to enhance the water dispersibility, stability, and bioavailability of the encapsulated bioactive agents. These plant-based delivery systems may be particularly useful for the development of the next generation of healthier plant-based food products. For instance, they could be used to incorporate vitamins and nutraceuticals into these products, such as vitamins A, B, C, D, E, carotenoids, curcuminoids, and polyphenols, as well as natural pigments, flavors, and preservatives.
References
- McClements, D.J.; Grossmann, L. The science of plant-based foods: Constructing next-generation meat, fish, milk and egg analogs. Compr. Rev. Food Sci. Food Saf. 2021, 20, 4049–4100.
- GFI. State of the Industry Report: Plant-Based Meat, Eggs, and Dairy; Good Food Institute: Washington, DC, USA, 2021; pp. 1–85.
- Chen, F.; Deng, Z.; Zhang, Z.; Zhang, R.; Xu, Q.; Fan, G.; Luo, T.; McClements, D.J. Controlling lipid digestion profiles using mixtures of different types of microgel: Alginate beads and carrageenan beads. J. Food Eng. 2018, 238, 156–163.
- Zhang, Z.; Zhang, R.; McClements, D.J. Encapsulation of β-carotene in alginate-based hydrogel beads: Impact on physicochemical stability and bioaccessibility. Food Hydrocoll. 2016, 61, 1–10.
- Corstens, M.N.; Berton-Carabin, C.C.; Elichiry-Ortiz, P.T.; Hol, K.; Troost, F.J.; Masclee, A.A.M.; Schroën, K. Emulsion-alginate beads designed to control in vitro intestinal lipolysis: Towards appetite control. J. Funct. Foods 2017, 34, 319–328.
- Li, Y.; Hu, M.; Du, Y.M.; Xiao, H.; McClements, D.J. Control of lipase digestibility of emulsified lipids by encapsulation within calcium alginate beads. Food Hydrocoll. 2011, 25, 122–130.
- van Leusden, P.; den Hartog, G.J.M.; Bast, A.; Postema, M.; van der Linden, E.; Sagis, L.M.C. Lipase diffusion in oil-filled, alginate micro- and macrobeads. Food Hydrocoll. 2018, 85, 242–247.
- Salvia-Trujillo, L.; Verkempinck, S.H.E.; Zhang, X.; Van Loey, A.M.; Grauwet, T.; Hendrickx, M.E. Comparative study on lipid digestion and carotenoid bioaccessibility of emulsions, nanoemulsions and vegetable-based in situ emulsions. Food Hydrocoll. 2019, 87, 119–128.
- Scheuble, N.; Schaffner, J.; Schumacher, M.; Windhab, E.J.; Liu, D.; Parker, H.; Steingoetter, A.; Fischer, P. Tailoring Emulsions for Controlled Lipid Release: Establishing in vitro-in Vivo Correlation for Digestion of Lipids. ACS Appl. Mater. Interfaces 2018, 10, 17571–17581.
- Golding, M.; Wooster, T.J. The influence of emulsion structure and stability on lipid digestion. Curr. Opin. Colloid Interface Sci. 2010, 15, 90–101.
- Matsumiya, K.; Murray, B.S. Soybean protein isolate gel particles as foaming and emulsifying agents. Food Hydrocoll. 2016, 60, 206–215.
- Xu, H.N.; Liu, Y.; Zhang, L. Salting-out and salting-in: Competitive effects of salt on the aggregation behavior of soy protein particles and their emulsifying properties. Soft Matter 2015, 11, 5926–5932.
- Guo, Q.; Bellissimo, N.; Rousseau, D. The Physical State of Emulsified Edible Oil Modulates Its in Vitro Digestion. J. Agric. Food Chem. 2017, 65, 9120–9127.
- Salvia-Trujillo, L.; Verkempinck, S.; Rijal, S.K.; Van Loey, A.; Grauwet, T.; Hendrickx, M. Lipid nanoparticles with fats or oils containing beta-carotene: Storage stability and in vitro digestibility kinetics. Food Chem. 2019, 278, 396–405.
- Jannin, V.; Dellera, E.; Chevrier, S.; Chavant, Y.; Voutsinas, C.; Bonferoni, C.; Demarne, F. In vitro lipolysis tests on lipid nanoparticles: Comparison between lipase/co-lipase and pancreatic extract. Drug Dev. Ind. Pharm. 2015, 41, 1582–1588.
- Tan, Y.; McClements, D.J. Improving the bioavailability of oil-soluble vitamins by optimizing food matrix effects: A review. Food Chem. 2021, 348, 129148.
- Tan, Y.; Zhou, H.; Zhang, Z.; McClements, D.J. Bioaccessibility of oil-soluble vitamins (A, D, E) in plant-based emulsions: Impact of oil droplet size. Food Func. 2021, 12, 3883–3897.
- Walker, R.; Decker, E.A.; McClements, D.J. Development of food-grade nanoemulsions and emulsions for delivery of omega-3 fatty acids: Opportunities and obstacles in the food industry. Food Funct. 2015, 6, 42–55.
- McClements, D.J.; Decker, E. Interfacial Antioxidants: A Review of Natural and Synthetic Emulsifiers and Coemulsifiers That Can Inhibit Lipid Oxidation. J. Agric. Food Chem. 2018, 66, 20–35.
- Chen, F.; Liang, L.; Zhang, Z.; Deng, Z.; Decker, E.A.; McClements, D.J. Inhibition of lipid oxidation in nanoemulsions and filled microgels fortified with omega-3 fatty acids using casein as a natural antioxidant. Food Hydrocoll. 2017, 63, 240–248.
- Rahiminezhad, Z.; Hashemi Gahruie, H.; Esteghlal, S.; Mesbahi, G.R.; Golmakani, M.-T.; Hosseini, S.M.H. Oxidative stability of linseed oil nano-emulsions filled in calcium alginate hydrogels. Lwt 2020, 127, 109392.
- Gumus, C.E.; Decker, E.A.; McClements, D.J. Impact of legume protein type and location on lipid oxidation in fish oil-in-water emulsions: Lentil, pea, and faba bean proteins. Food Res. Int. 2017, 100, 175–185.
- Sun, M.; Li, X.; McClements, D.J.; Xiao, M.; Chen, H.; Zhou, Q.; Xu, S.; Chen, Y.; Deng, Q. Reducing off-flavors in plant-based omega-3 oil emulsions using interfacial engineering: Coating algae oil droplets with pea protein/flaxseed gum. Food Hydrocol. 2022, 122, 107069.
- Hu, M.; Xie, F.; Zhang, S.; Qi, B.; Li, Y. Effect of nanoemulsion particle size on the bioavailability and bioactivity of perilla oil in rats. J. Food Sci. 2021, 86, 206–214.
- Inapurapu, S.P.; Ibrahim, A.; Kona, S.R.; Pawar, S.C.; Bodiga, S.; Bodiga, V.L. Development and characterization of ω-3 fatty acid nanoemulsions with improved physicochemical stability and bioaccessibility. Colloids Surf. A Physicochem. Eng. Asp. 2020, 606, 125515.
- Tan, Y.; Zhang, Z.; Muriel Mundo, J.; McClements, D.J. Factors impacting lipid digestion and nutraceutical bioaccessibility assessed by standardized gastrointestinal model (INFOGEST): Emulsifier type. Food Res. Int. 2020, 137, 109739.
- Tan, Y.; Zhang, Z.; Liu, J.; Xiao, H.; McClements, D.J. Factors impacting lipid digestion and nutraceutical bioaccessibility assessed by standardized gastrointestinal model (INFOGEST): Oil droplet size. Food Funct. 2020, 11, 9936–9946.
- Li, R.; Tan, Y.; Dai, T.; Zhang, R.; Fu, G.; Wan, Y.; Liu, C.; McClements, D.J. Bioaccessibility and stability of β-carotene encapsulated in plant-based emulsions: Impact of emulsifier type and tannic acid. Food Funct. 2019, 10, 7239–7252.
- Fang, Z.; Bhandari, B. Encapsulation of polyphenols—A review. Trends Food Sci. Technol. 2010, 21, 510–523.
- Zhou, H.; Zheng, B.; McClements, D.J. Encapsulation of lipophilic polyphenols in plant-based nanoemulsions: Impact of carrier oil on lipid digestion and curcumin, resveratrol and quercetin bioaccessibility. Food Funct. 2021, 12, 3420–3432.
- Liu, F.; Ma, C.; McClements, D.J.; Gao, Y. Development of polyphenol-protein-polysaccharide ternary complexes as emulsifiers for nutraceutical emulsions: Impact on formation, stability, and bioaccessibility of β-carotene emulsions. Food Hydrocoll. 2016, 61, 578–588.





