
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | J. FERNANDO AYALA-ZAVALA | + 2376 word(s) | 2376 | 2021-11-16 09:05:40 | | | |
| 2 | Amina Yu | Meta information modification | 2376 | 2021-11-26 04:09:10 | | |
Video Upload Options
Plant extracts are the current trend in the market, but it is also essential to consider their antibacterial, antioxidant, and sensory characteristics. Plant essential oils are constituted by secondary metabolites, such as terpenes, phenols, and aldehydes with antibacterial, antioxidant, and flavoring properties. Other industries take advantage of the essential oil properties; some examples of their applications are found in the food, pharmaceutical, and cosmetic industries.
1. Introduction
Some synthetic substances, such as chlorhexidine and fluorides, among other compounds, have antibacterial activity against S. mutans; therefore, they have been incorporated into different dental products, such as pastes and mouthwashes [1]. However, these compounds can cause adverse effects, such as dental pigmentation, changes in taste perception, and burning sensation that cause their rejection by consumers [2][3]. Plant extracts are the current trend in the market, but it is also essential to consider their antibacterial, antioxidant, and sensory characteristics. Plant essential oils are constituted by secondary metabolites, such as terpenes, phenols, and aldehydes with antibacterial, antioxidant, and flavoring properties [4]. Other industries take advantage of the essential oil properties; some examples of their applications are found in the food, pharmaceutical, and cosmetic industries. In addition, these plant extracts have been useful for the in vitro inactivation of Escherichia coli, Staphylococcus aureus, Listeria monocytogenes, and S. mutans. Therefore, the incorporation of essential oils into oral products can be found in mouthwashes and toothpaste.
Essential oils have antioxidant and anticarcinogenic properties that could be useful to preserve oral health. For example, essential oils of Thymus serpyllum, Mentha piperita, Juniperus communis, Rosmarinus officinalis, Melissa officinalis, Achillea millefolium, Zingiber officinale, and Helichrysum arenarium inhibited the proliferation of oral squamous cell carcinoma culture and SCC-25 cell line [5]. Besides, Menta piperita extracts at 1.5 mg/mL showed a cytotoxic effect on tumor cell lines. At the same time, rosmarinic and carnosic acids from Melissa officinalis and rosemary essential oils can inhibit growth and stimulate apoptosis in cancer cells at the same dose [5]. This evidence highlights that plant essential oils are a source of antioxidant agents with potential anticarcinogenic activity.
The search for substances to preserve oral health showed that synthetic compounds, such as chlorhexidine cause irritation, dryness, and alteration in flavors’ perception [6]. As a counterpart, essential oils can provide pleasant sensory attributes; for example, Cymbopogon citratus or lemongrass essential oil has a fresh citrus smell and a sweet-spicy flavor [4]. On the other hand, essential oils obtained from Melaleuca bracteata and Ocimum basilicum plants contain gallic acid and quercetin that produce sweet and bitter flavors, respectively [7]. Therefore, the substance election is important for formulating dental products with antibacterial activity, antioxidant capacity, and sensory properties. Thus, the analyzed evidence indicates that the use of plant extracts shows a broad profile of advantages (antibacterial, antioxidant, and flavoring properties) to formulate oral health care products compared to conventionally used synthetic compounds.
2. Eubiosis vs. Dysbiosis of Oral Microbiota: Health Impact
The oral cavity favors the development of many microorganisms; this area includes epithelial surfaces of mucous membranes and dental pieces prone to be invaded mainly by bacteria [8]. Facultative anaerobes are colonizers of oral surfaces, mainly of the Streptococcal genus, found in the subgingival and dental areas forming biofilms and plaque [8]. It is currently estimated that more than 700 bacterial species co-exist in oral tissues; some of them can present benefits to the host, while others can be harmful [9].
The oral microbiota comprises many microorganisms, including viruses, bacteria, Archae, fungi, and protozoa; these participate in the homeostasis and maintenance of oral health. Microbiota must co-exist in harmony among them and with the host to contribute to their well-being; this balance and harmony is called eubiosis, while the opposite state is named dysbiosis. Oral eubiosis can prevent the colonization of exogenous microorganisms because they are better adapted to these surfaces [10]. However, the increment in certain pathogens can be detected when oral dysbiosis occurs. Among the most indicated oral pathogenic bacterial species are S. mutans [11]; while other streptococci can be considered benefic bacteria, e.g., S. sanguinis, S. oralis, S. gordonii, and S. mitis produced hydrogen peroxide and extracellular proteases that inhibited the growth of pathogenic bacteria in the oral environment [12]; however, hydrogen peroxide also causes oxidation [13]. It has to be considered that many other non-cultivable bacteria could be involved in the symptomatology of dental caries.
Using S. mutans as a bacterial model to explain its pathogenicity caused during dysbiosis, Figure 1 shows how this bacterium activates quorum sensing to control biofilm production and other virulence factors [14][15]. ComCDE secretes a competent stimulating peptide (CSP), acting also as a signal molecule; ComC synthesizes this peptide, and it is recognized and phosphorylated by the transmembrane histidine kinase (ComD) and its response regulator (ComE) [15]. The phosphorylated peptide activates the signaling cascade, expressing different genes involved to synthesize proteins and enzymes that S. mutans use to adhere to dental surfaces and form biofilms.
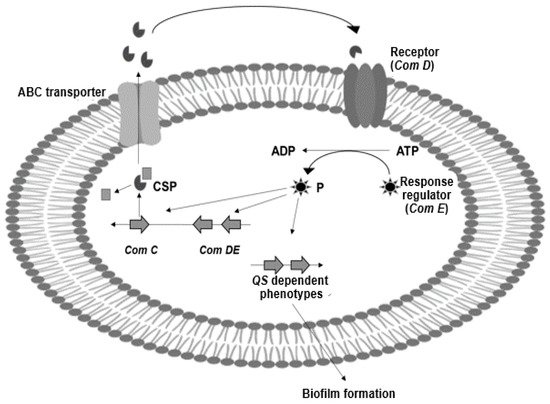
During dental caries development, degradation of the teeth’s surface occurs by demineralization of the enamel, mainly caused by acids (lactic, propionic, and formic) produced by S. mutans; these are introduced through the pores of the enamel and begin its degradation [16]. Caries can also cause pain, phonation problems, dental aesthetics, and even be an economic problem due to expenses in treatment and absence from work [17]. Dental caries is also a risk factor for developing endocarditis, atherosclerosis, and cerebrovascular diseases [17][18]. The relationship between periodontitis and cerebrovascular health has been evidenced by the damage of endothelial cells caused by oral inflammations and injuries to the blood-brain barrier [19][20]. On the other hand, some oral pathogenic species, such as S. mutans, can spread through the blood and lodge in vascular lesions, causing endocarditis [19]. Therefore, the negative health impact of pathogenic bacteria during dysbiosis stimulates the development of oral health care products with eubiotic properties.
3. Oral Cancer and Its Relationship with Reactive Oxygen Species
Oral cancer can be caused by spontaneous mutations and external environmental factors that induce the inadequate functioning of processes, such as cell division and the expression of defective genes [21]. As a result, this disease causes abnormal cell proliferation, tumor mass formation and decreases nutrient availability for healthy tissues [21]. These cells can also travel through the lymphatic chain and lodge in other organs and tissues, such as the mouth, causing malignant neoplasms that damage the oral cavity [22]. The oral squamous cell carcinoma is the primary tumor responsible for developing oral cancer, capable of producing 90% of all malignant neoplasms [23]. This type of cancer is the sixth most common worldwide, affecting 354,864 people and causing 177,384 deaths in 2018 [24][25]. Therefore, it is important to understand the development of this pathology since it can cause irreversible damage to oral tissues and death.
Dysbiosis of the oral microbiota can also cause cancer through different mechanisms of action. The first mechanism involves stimulating chronic inflammation caused by microorganisms, such as Fusobacterium nucleatum and Porphiromonas gingivalis [26]. These bacteria generate periodontal diseases that stimulate the production of inflammatory mediators, damaging fibroblasts, epithelial, and endothelial cells [27]. Some examples of these mediators are NF-kB, STAT3, and H1F-α that trigger genetic mutations, leading to cancer development [26]. On the other hand, it has been reported that the increase in IL-1β favors the production of angiogenic factors, such as TNF-α (tumor necrosis factor), which is responsible for increasing the content of malignant tumors in oral tissues [27].
Microbial metabolism in the oral cavity can include the synthesis of carcinogenic substances. The oral microbiota can synthesize substances related to cancer development, such as free radicals and reactive oxygen species (ROS) [27][28]. Specifically, hydrogen peroxide is a type of ROS produced by some bacteria, such as Lactobacillus acidophilus, Bifidobacterium adolescentis, and Streptococcus species; it increases the expression and accumulation of NADPH oxidase [27][28]. This compound can enter and damage the mitochondria, and it causes the cells’ inadequate functioning ( Figure 2 ) [27]. Furthermore, it has been reported that ROS generate genetic damage by reducing the expression of pro-tumor genes, allowing for the presence of this type of abnormal agglomeration. Besides, hydrogen peroxide can react with Fe 2+ producing substances that stimulate DNA mutations and cause cell cycle disorders [28].
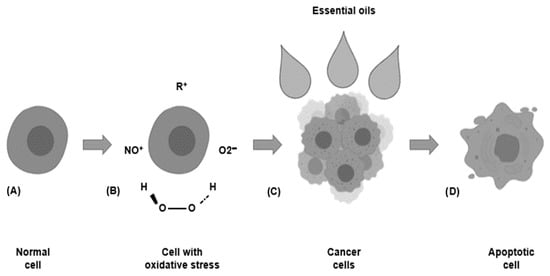
The oxidative stress in the mouth is also attributed to free radicals synthesized by oral bacteria; e.g., nitric oxide (NO) is produced from nitrate in human saliva [29]. Furthermore, it has been reported that the increase of free radicals in the environment causes damage to cellular structures and promotes mutations related to cardiovascular disease and cancer [30]. Also, there is a connection between periodontal diseases produced by oral bacteria and the content of carcinogenic substances. This effect was attributed to the high amounts of free radicals produced by the host’s immune system in the presence of oral bacterial infection [27][30]. Therefore, oral microbiota and ROS control can be considered a target to prevent oral cancer and caries.
4. Conventional and Alternative Ingredients with Antibacterial, Antioxidants, and Flavoring Properties Used in Dental Health Care Products
Thymol, menthol, eucalyptol, and methyl salicylate are the constituents of several plant essential oils with antibacterial activity against cariogenic strains of S. mutans ( Table 1 ). They have been used to formulate Listerine ® due to their ability to inactivate S. mutans biofilms at low concentrations [31]. Plant compounds found in this mouthwash are menthol 0.042%, eucalyptol 0.092%, methyl salicylate 0.06%, and thymol 0.064% [32]. Other extracts with this property are C. citratus and Cinnamon zeylanicum, constituted mainly by terpenes and effective against planktonic S. mutans. Similarly, Acacia nilotica, peel extract of Citrus aurantifolia, and the fruit extract from Berberis vulgaris were also effective against this bacterium. In addition, evidence is emerging about the eubiotic effect of using plant essential oils in animal systems, e.g., the administration of thymol, eugenol, and piperine was added as an additive in chickens’ diets, resulting in the maintenance of lactobacilli and other beneficial species and reducing pathogenic bacteria in broiler chicks [33]. Similarly, the inclusion of thymol, eugenol, and piperine in the diet of nursery pigs increased the Bacteroides and decreased Prevotella, creating a eubiotic effect [34]. However, it must be considered that even when synthetic substances like chlorhexidine derivatives showed higher effectivity than natural plant compounds, the synthetic agents did not create a eubiotic system. Therefore, it will be interesting to test the effect of plant substances added to dental care products in the eubiosys/dysbiosis of oral tissues.
| Component | MIC (mg/mL) | MBC (mg/mL) | Reference |
|---|---|---|---|
| Menthol | 0.5 | 1.0 | [35] |
| Eugenol | 0.1 | 0.2 | [36] |
| Thymol | 0.312 | 0.312 | [37] |
| Eucalyptol | 0.250 | 0.5 | [35] |
| Methyl salicylate | 1.0 | 1.0 | [35] |
| Cymbopogon citratus | 0.125–0.250 | 0.250–0.5 | [36] |
| Cinnamon zeylanicum | 0.250–0.5 | 0.5–1 | [36] |
| Fruit extract of Berberis vulgaris | 0.064 | 0.128 | [38] |
| Peel extract of Citrus aurantifolia | 0.02 | - | [39] |
| Bark extract of Acacia nilotica | 0.3 | 4 | [40] |
| Tea of Camellia sinensis | 6.25 | 12.5 | [41] |
| Tea of Hibiscus sabdariffa | 25 | 50 | [41] |
| Listerine® | 1.25 | - | [42] |
| Chlorhexidine digluconate | 0.0094 | 0.0094 | [42] |
| Chlorhexidine dihydrochloride | 0.00092 | - | [39] |
- Not determined.
The combination of different plant compounds could increase their effectiveness in inactivating pathogenic bacteria. The interaction of essential oil components can present various effects; an example is the synergy shown in binary combinations of C. citratus (0.44 mg/mL) and Allium cepa (5.26 mg/mL) essential oils that at decreased doses (0.22 mg/mL + 1.28 mg/mL) inhibited the growth of S. aureus [43]. In addition, ternary combinations of protocatechuic acid (12.98 mM), vanillic acid (11.8 mM), and catechin (13.78 mM), phenolic compounds found in several plants, reduced their effective doses (1.62 mM + 0.74 mM + 0.05 mM), inhibiting the growth of E. coli [44]. Therefore, binary or tertiary mixtures of plant compounds can be used to combat pathogenic bacteria; their combination can increase effectiveness, making them an attractive option to replace synthetic compounds. However, more studies testing the effects of these combinations are needed, specifically against cariogenic bacteria. Also, plant essential oils can present antioxidant capacity and anticarcinogenic potential, in contrast to other synthetic antibacterial agents commonly used in oral care products.
Derivatives of natural plants possess antioxidant and anticarcinogenic properties, which allows them to be used as a preventive measure of oral cancer (Table 2). An example of these compounds is turmeric acid from Curcuma longa, polyphenolics from green tea, apigenin from moringa plants, cyanidin from berries, gingerol from ginger roots, eugenol and thymol from essential oils that inhibited several biochemical parameters of cancerous cells, arresting tumor development and causing apoptosis [45][46][47][48][49][50]. Thus, studying natural plant derivatives facilitates understanding how they act to prevent this deadly disease.
| Component | Proposed Mechanisms | Reference |
|---|---|---|
| Turmeric acid from Curcuma longa | Inhibits the transcription of NF-kB, Cox-2, TNF-α, cyclin D1, ICAM-1, c-myc, Bcl-2, MMP-9, iNOS, IL-6, IL-8, causes cell cycle arrest, promotes apoptosis, and acts as ROS scavenger. | [45][46] |
| Epicatechin, epigallocatechin, epicatechin-3-gallate, and epigallocatechin-3-gallate from Camelia sinensis | ROS scavengers, inhibit tumor proliferation, induce apoptosis, arrest cells in G0 and G1 phase, downregulate cyclin D1, increase p14 and p16 proteins levels, blocks angiogenesis. | [47][51] |
| Apigening from Moringa oleifera | Induces apoptosis | [48][52] |
| Cyanidin from fruits and vegetables | Inhibits tumor cell growth, COX-2 gen, MMP, and EFGR expression. | [53][54] |
| Gingerol from Zingiber officinale | Decreases INOs and TNF-α expression and induces apoptosis. | [49][55] |
| Eugenol and trans-cinnamaldehyde from Cinnamomum zeylanicum | ROS scavenger, inhibits the growth of cancerous cells, decreases Bcl-2, Ki67, VEGF, and CD24 expressions and MDA levels. | [56][57] |
| Thymol from Thymus vulgaris | Induces apoptosis and mitochondrial dysfunction in cancerous cells and inhibits the activity of COX-2 and 5LOX. | [50] |
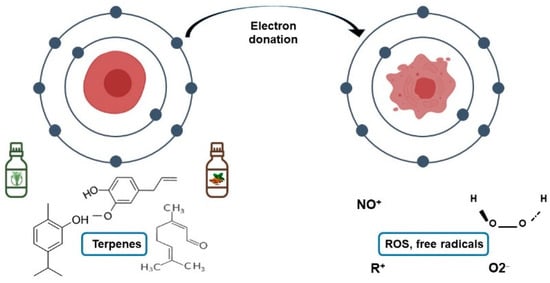
The anticarcinogenic capacity of natural plant derivatives favors their introduction into dental products that could prevent oral cancer. Mentha piperita extract is an example of compounds that have been introduced to dental products, which has a cytotoxic effect on tumor cell lines. On the other hand, thymol is a derivative of thyme; this terpenoid is considered one of the main antibacterial agents in mouthwashes and an antioxidant compound since it reduces the damage caused by hydrogen peroxide and nitrous oxide, among others ROS ( Figure 3 ) [50]. Current knowledge indicates that bioactive compounds from natural plants can prevent cancer by protecting DNA, stabilizing ROS, and regulating the cell cycle [58]. Therefore, incorporating plant compounds into dental products can prevent oral cancer by propitiating a balanced oxidative status.
References
- Elkerbout, T.; Slot, D.; Van Loveren, C.; Van Der Weijden, G. Will a chlorhexidine-fluoride mouthwash reduce plaque and gingivitis? Int. J. Dent. Hyg. 2018, 17, 3–15.
- Pretty, I.A. High Fluoride Concentration Toothpastes for Children and Adolescents. Caries Res. 2016, 50, 9–14.
- Su, C.-Y.; Chen, C.-C.; Chen, H.-Y.; Lin, C.-P.; Lin, F.-H.; Fang, H.-W. Characteristics of an alternative antibacterial biomaterial for mouthwash in the absence of alcohol. J. Dent. Sci. 2019, 14, 192–197.
- Ortega-Ramirez, L.A.; Rodriguez-Garcia, I.; Leyva, J.M.; Valenzuela, M.R.C.; Silva-Espinoza, B.A.; Gonzalez-Aguilar, G.A.; Siddiqui, M.W.; Ayala-Zavala, J.F. Potential of Medicinal Plants as Antimicrobial and Antioxidant Agents in Food Industry: A Hypothesis. J. Food Sci. 2014, 79, R129–R137.
- Lazarević, M.; Milošević, M.; Petrović, N.; Petrović, S.; Damante, G.; Milašin, J.; Milovanović, B. Cytotoxic Effects of Different Aromatic Plants Essential Oils on Oral Squamous Cell Carcinoma- an in vitro Study. Balk. J. Dent. Med. 2019, 23, 73–79.
- Bhat, N.; Mitra, R.; Oza, S.; Mantu, V.K.; Bishnoi, S.; Gohil, M.; Gupta, R. The antiplaque effect of herbal mouthwash in comparison to chlorhexidine in human gingival disease: A randomized placebo controlled clinical trial. J. Complement. Integr. Med. 2014, 11, 129–137.
- Cardoso-Ugarte, G.A.; López-Malo, A.; Sosa-Morales, M.E. Cinnamon (Cinnamomum zeylanicum) Essential Oils. In Essential Oils in Food Preservation, Flavor and Safety; Elsevier: Amsterdam, The Netherlands, 2016; pp. 339–347.
- Lamont, R.J.; Koo, H.; Hajishengallis, G. The oral microbiota: Dynamic communities and host interactions. Nat. Rev. Genet. 2018, 16, 745–759.
- Sarduy Bermúdez, L.; González Díaz, M.E. La biopelícula: Una nueva concepción de la placa dentobacteriana. Medicentro 2016, 20, 167–175.
- Marsh, P.D.; Head, D.A.; Devine, D.A. Dental plaque as a biofilm and a microbial community—Implications for treatment. J. Oral Biosci. 2015, 57, 185–191.
- Aas, J.A.; Griffen, A.L.; Dardis, S.R.; Lee, A.M.; Olsen, I.; Dewhirst, F.E.; Leys, E.J.; Paster, B.J. Bacteria of Dental Caries in Primary and Permanent Teeth in Children and Young Adults. J. Clin. Microbiol. 2008, 46, 1407–1417.
- Graves, D.; Corrêa, J.; Silva, T. The Oral Microbiota Is Modified by Systemic Diseases. J. Dent. Res. 2019, 98, 148–156.
- Jakubovics, N.S.; Yassin, S.A.; Rickard, A.H. Community Interactions of Oral Streptococci. In Advances in Applied Microbiology; Elsevier: Amsterdam, The Netherlands, 2014; Volume 87, pp. 43–110.
- Matsumoto-Nakano, M. Role of Streptococcus mutans surface proteins for biofilm formation. Jpn. Dent. Sci. Rev. 2017, 54, 22–29.
- Kaur, G.; Rajesh, S.; Princy, S.A. Plausible Drug Targets in the Streptococcus mutans Quorum Sensing Pathways to Combat Dental Biofilms and Associated Risks. Indian J. Microbiol. 2015, 55, 349–356.
- Ojeda-Garcés, J.C.; Oviedo-García, E.; Salas, L.A. Streptococcus mutans and dental caries. Ces Odontología 2013, 26, 44–56.
- Tofiño-Rivera, A.; Ortega-Cuadros, M.; Galvis-Pareja, D.; Ríos, H.J.; Merini, L.; Martínez-Pabón, M. Effect of Lippia alba and Cymbopogon citratus essential oils on biofilms of Streptococcus mutans and cytotoxicity in CHO cells. J. Ethnopharmacol. 2016, 194, 749–754.
- Lemos, J.; Palmer, S.; Zeng, L.; Wen, Z.; Kajfasz, J.; Freires, I.; Abranches, J.; Brady, L. The Biology of Streptococcus mutans. Microbiol. Spectr. 2019, 7.
- Aarabi, G.; Thomalla, G.; Heydecke, G.; Seedorf, U. Chronic oral infection: An emerging risk factor of cerebral small vessel disease. Oral Dis. 2019, 25, 710–719.
- Leira, Y.; Seoane, J.; Blanco, M.; Rodríguez-Yáñez, M.; Takkouche, B.; Blanco, J.; Castillo, J. Association between periodontitis and ischemic stroke: A systematic review and meta-analysis. Eur. J. Epidemiol. 2017, 32, 43–53.
- Ghizoni, J.S.; Nichele, R.; De Oliveira, M.T.; Pamato, S.; Pereira, J.R. The utilization of saliva as an early diagnostic tool for oral cancer: microRNA as a biomarker. Clin. Transl. Oncol. 2019, 22, 804–812.
- Tahmasebi, E.; Alikhani, M.; Yazdanian, A.; Yazdanian, M.; Tebyanian, H.; Seifalian, A. The current markers of cancer stem cell in oral cancers. Life Sci. 2020, 249, 117483.
- Zhang, Z.; Gao, Z.; Rajthala, S.; Sapkota, D.; Dongre, H.; Parajuli, H.; Suliman, S.; Das, R.; Li, L.; Bindoff, L.A.; et al. Metabolic reprogramming of normal oral fibroblasts correlated with increased glycolytic metabolism of oral squamous cell carcinoma and precedes their activation into carcinoma associated fibroblasts. Experientia 2019, 77, 1115–1133.
- Nagao, T.; Warnakulasuriya, S. Screening for oral cancer: Future prospects, research and policy development for Asia. Oral Oncol. 2020, 105, 104632.
- Yang, G.; Yin, X.; Ma, D.; Su, Z. Anticancer activity of Phloretin against the human oral cancer cells is due to G0/G1 cell cycle arrest and ROS mediated cell death. J. BUON 2020, 25, 344–349.
- Li, Y.; Tan, X.; Zhao, X.; Xu, Z.; Dai, W.; Duan, W.; Huang, S.; Zhang, E.; Liu, J.; Zhang, S.; et al. Composition and function of oral microbiota between gingival squamous cell carcinoma and periodontitis. Oral Oncol. 2020, 107, 104710.
- Karpiński, T.M. Role of Oral Microbiota in Cancer Development. Microorganisms 2019, 7, 20.
- La Rosa, G.R.M.; Gattuso, G.; Pedullà, E.; Rapisarda, E.; Nicolosi, D.; Salmeri, M. Association of oral dysbiosis with oral cancer development (Review). Oncol. Lett. 2020, 19, 3045–3058.
- Ambe, K.; Watanabe, H.; Takahashi, S.; Nakagawa, T.; Sasaki, J. Production and physiological role of NO in the oral cavity. Jpn. Dent. Sci. Rev. 2016, 52, 14–21.
- Sardaro, N.; della Vella, F.; Incalza, M.A.; Di Stasio, D.; Lucchese, A.; Contaldo, M.; Laudadio, C.; Petruzzi, M. Oxidative Stress and Oral Mucosal Diseases: An Overview. In Vivo 2019, 33, 289–296.
- Wińska, K.; Mączka, W.; Łyczko, J.; Grabarczyk, M.; Czubaszek, A.; Szumny, A. Essential Oils as Antimicrobial Agents—Myth or Real Alternative? Molecules 2019, 24, 2130.
- Vlachojannis, C.; Chrubasik-Hausmann, S.; Hellwig, E.; Al-Ahmad, A. A Preliminary Investigation on the Antimicrobial Activity of Listerine®, Its Components, and of Mixtures Thereof. Phytother. Res. 2015, 29, 1590–1594.
- Weber, G.M.; Michalczuk, M.; Huyghebaert, G.; Juin, H.; Kwakernaak, C.; Gracia, M.I. Effects of a blend of essential oil compounds and benzoic acid on performance of broiler chickens as revealed by a meta-analysis of 4 growth trials in various locations. Poult. Sci. 2012, 91, 2820–2828.
- Zhai, H.; Luo, Y.; Ren, W.; Schyns, G.; Guggenbuhl, P. The effects of benzoic acid and essential oils on growth performance, nutrient digestibility, and colonic microbiota in nursery pigs. Anim. Feed. Sci. Technol. 2020, 262, 114426.
- Chung, J.; Choo, J.; Lee, M.; Hwang, J. Anticariogenic activity of macelignan isolated from Myristica fragrans (nutmeg) against Streptococcus mutans. Phytomedicine 2006, 13, 261–266.
- Freires, I.A.; Denny, C.; Benso, B.; De Alencar, S.M.; Rosalen, P.L. Antibacterial Activity of Essential Oils and Their Isolated Constituents against Cariogenic Bacteria: A Systematic Review. Molecules 2015, 20, 7329–7358.
- Ferreira, G.L.S.; Bezerra, L.M.D.; Ribeiro, I.L.A.; Júnior, R.C.D.M.; Castro, R.D. Susceptibility of cariogenic microorganisms to phytoconstituents. Braz. J. Biol. 2018, 78, 691–696.
- Kazemipoor, M.; Tehrani, P.F.; Zandi, H.; Yazdi, R.G. Chemical composition and antibacterial activity of Berberis vulgaris (barberry) against bacteria associated with caries. Clin. Exp. Dent. Res. 2020, 7, 601–608.
- Lemes, R.S.; Alves, C.C.; Estevam, E.B.; Santiago, M.B.; Martins, C.H.; Dos Santos, T.C.; Crotti, A.E.; Miranda, M.L. Chemical composition and antibacterial activity of essential oils from Citrus aurantifolia leaves and fruit peel against oral pathogenic bacteria. Anais da Academia Brasileira de Ciências 2018, 90, 1285–1292.
- Muddathir, A.M.; Mohieldin, E.A.M.; Mitsunaga, T. In vitro activities of Acacia nilotica (L.) Delile bark fractions against Oral Bacteria, Glucosyltransferase and as antioxidant. BMC Complement. Med. Ther. 2020, 20, 1–9.
- Golmohammadi, A. A comparative study on antimicrobial effect of iranian green tea and hibiscus tea on growth of oral cariogenic bacteria Streptococcus mutans ptcc 1683. J. Res. Med Dent. Sci. 2018, 6, 361–364.
- Filoche, S.; Soma, K.; Sissons, C.H. Antimicrobial effects of essential oils in combination with chlorhexidine digluconate. Oral Microbiol. Immunol. 2005, 20, 221–225.
- Ortega-Ramirez, A.L.; Silva-Espinoza, A.B.; Vargas-Arispuro, I.; Gonzalez-Aguilar, A.G.; Cruz-Valenzuela, M.R.; Nazzaro, F.; Ayala-Zavala, J.F. Combination of Cymbopogon citratus and Allium cepa essential oils increased antibacterial activity in leafy vegetables. J. Sci. Food Agric. 2016, 97, 2166–2173.
- Bernal-Mercado, A.T.; Vazquez-Armenta, F.J.; Tapia-Rodriguez, M.R.; Islas-Osuna, M.A.; Mata-Haro, V.; Gonzalez-Aguilar, G.A.; Lopez-Zavala, A.A.; Ayala-Zavala, J.F. Comparison of single and combined use of catechin, protocatechuic, and vanillic acids as antioxidant and antibacterial agents against uropathogenic Escherichia coli at planktonic and biofilm levels. Molecules 2018, 23, 2813.
- Rai, A.; Kumar, N.; Sharma, S.; Parveen, S.; Rasheed, A. Turmeric in the management of oral submucous fibrosis: A systematic review and meta-analysis. J. Cancer Res. Ther. 2021, 17, 327–335.
- SSoni, T.P.; Gupta, A.K.; Sharma, L.M.; Singhal, H.; Sharma, S.; Gothwal, R.S. A Randomized, Placebo-Controlled Study to Evaluate the Effect of Bio-Enhanced Turmeric Formulation on Radiation-Induced Oral Mucositis. Orl-J. Oto-Rhino-Laryngol. Head Neck Surg. 2021, 1–11.
- Wang, W.; Yang, Y.; Zhang, W.; Wu, W. Association of tea consumption and the risk of oral cancer: A meta-analysis. Oral Oncol. 2014, 50, 276–281.
- Jung, I.L.; Lee, J.H.; Kang, S.C. A potential oral anticancer drug candidate, Moringa oleifera leaf extract, induces the apoptosis of human hepatocellular carcinoma cells. Oncol. Lett. 2015, 10, 1597–1604.
- Hitomi, S.; Ono, K.; Terawaki, K.; Matsumoto, C.; Mizuno, K.; Yamaguchi, K.; Imai, R.; Omiya, Y.; Hattori, T.; Kase, Y.; et al. -gingerol and -shogaol, active ingredients of the traditional Japanese medicine hangeshashinto, relief oral ulcerative mucositis-induced pain via action on Na+ channels. Pharmacol. Res. 2017, 117, 288–302.
- De La Chapa, J.J.; Singha, P.K.; Lee, D.R.; Gonzales, C.B. Thymol inhibits oral squamous cell carcinoma growth via mitochondria-mediated apoptosis. J. Oral Pathol. Med. 2018, 47, 674–682.
- Fu, J.-Y.; Gao, J.; Zhang, Z.-Y.; Zheng, J.-W.; Luo, J.-F.; Zhong, L.-P.; Xiang, Y.-B. Tea consumption and the risk of oral cancer incidence: A case-control study from China. Oral Oncol. 2013, 49, 918–922.
- Sharma, V.; Paliwal, R.; Janmeda, P.; Sharma, S. Chemopreventive Efficacy of Moringa oleifera Pods Against 7, 12-Dimethylbenzanthracene Induced Hepatic Carcinogenesis in Mice. Asian Pac. J. Cancer Prev. 2012, 13, 2563–2569.
- Knobloch, T.J.; Uhrig, L.K.; Pearl, D.K.; Casto, B.C.; Warner, B.M.; Clinton, S.K.; Sardo-Molmenti, C.L.; Ferguson, J.M.; Daly, B.T.; Riedl, K.; et al. Suppression of Proinflammatory and Prosurvival Biomarkers in Oral Cancer Patients Consuming a Black Raspberry Phytochemical-Rich Troche. Cancer Prev. Res. 2016, 9, 159–171.
- Mallery, S.R.; Budendorf, D.E.; Larsen, M.P.; Pei, P.; Tong, M.; Holpuch, A.S.; Larsen, P.E.; Stoner, G.D.; Fields, H.W.; Chan, K.K.; et al. Effects of Human Oral Mucosal Tissue, Saliva, and Oral Microflora on Intraoral Metabolism and Bioactivation of Black Raspberry Anthocyanins. Cancer Prev. Res. 2011, 4, 1209–1221.
- Wang, Q.; Wei, Q.; Yang, Q.; Cao, X.; Li, Q.; Shi, F.; Tong, S.S.; Feng, C.; Yu, Q.; Yu, J.; et al. A novel formulation of -gingerol: Proliposomes with enhanced oral bioavailability and antitumor effect. Int. J. Pharm. 2018, 535, 308–315.
- Kubatka, P.; Kello, M.; Kajo, K.; Samec, M.; Jasek, K.; Vybohova, D.; Uramova, S.; Liskova, A.; Sadlonova, V.; Koklesova, L.; et al. Chemopreventive and Therapeutic Efficacy of Cinnamomum zeylanicum L. Bark in Experimental Breast Carcinoma: Mechanistic In Vivo and In Vitro Analyses. Molecules 2020, 25, 1399.
- Dongmo, P.M.J.; Tatsadjieu, L.N.; Tchoumbougnang, F.; Sameza, M.L.; Dongmo, B.N.; Zollo, P.H.A.; Menut, C. Chemical Composition, Antiradical and Antifungal Activities of Essential Oil of the Leaves of Cinnamomum zeylanicum Blume from Cameroon. Nat. Prod. Commun. 2007, 2.
- Gezici, S. Current Perspectives in the Application of Medicinal Plants Against Cancer: Novel Therapeutic Agents. Anti-Cancer Agents Med. Chem. 2019, 19, 101–111.





