Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Chitra Upadhyay | + 5948 word(s) | 5948 | 2021-02-24 07:03:19 | | | |
| 2 | Rita Xu | -3752 word(s) | 2196 | 2021-11-25 05:03:55 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Upadhyay, C. HIV-1 Envelope. Encyclopedia. Available online: https://encyclopedia.pub/entry/16354 (accessed on 09 April 2026).
Upadhyay C. HIV-1 Envelope. Encyclopedia. Available at: https://encyclopedia.pub/entry/16354. Accessed April 09, 2026.
Upadhyay, Chitra. "HIV-1 Envelope" Encyclopedia, https://encyclopedia.pub/entry/16354 (accessed April 09, 2026).
Upadhyay, C. (2021, November 24). HIV-1 Envelope. In Encyclopedia. https://encyclopedia.pub/entry/16354
Upadhyay, Chitra. "HIV-1 Envelope." Encyclopedia. Web. 24 November, 2021.
Copy Citation
The RV144 trial represents the only vaccine trial to demonstrate any protective effect against HIV-1 infection. While the reason(s) for this protection are still being evaluated, it serves as justification for widespread efforts aimed at developing new, more effective HIV-1 vaccines. HIV-1 immunogens and host antibody responses to these immunogens are crucial to informing vaccine design. While the envelope (Env) protein is the only viral protein present on the surface of virions, it exists in a complex trimeric conformation and is decorated with an array of variable N-linked glycans, making it an important but difficult target for vaccine design.
HIV-1
HIV envelope
glycosylation
signal peptide
PNGS
broadly neutralizing antibodies
1. Introduction
Infection of CD4+ T cells by the human immunodeficiency virus type 1 (HIV-1) leads to a drastic decrease in their number and causes acquired immune deficiency syndrome (AIDS), which can eventually lead to death. Treatment of individuals infected with HIV-1 consists of the administration of highly effective antiretroviral medications, which allow patients to live relatively normal lives assuming that treatment regimens are adhered to [1][2][3]. However, resistance to these medications is of some concern, and therefore additional treatment approaches are of importance [4]. In the absence of a cure, prevention of new infections via prophylactic vaccination is likely to have a more widespread and lasting effect and is therefore the main focus of HIV-1 research [5].
Due to the high selective pressure induced by the immune system and the high mutation rate of HIV-1, chronic infection often consists of multiple viral quasispecies [6][7]. The immune response of an HIV-1 infected individual therefore needs to effectively prevent multiple viral quasispecies from infecting CD4+ T cells. This can be achieved via the induction of a broad milieu of antibodies (Abs) with varying specificities that act in concert to prevent infection but is ideally mediated via antibodies capable of binding epitopes that are conserved between many HIV-1 strains [8]. These antibodies are known as broadly neutralizing antibodies (bnAbs) and attempts to induce their production via vaccination have been a focal point of HIV-1 vaccine design.
The trimeric envelope (Env) that is responsible for initiating HIV-1 infection is the sole target for the induction of neutralizing antibodies (nAbs). Much effort has been expended to create recombinant Env trimers that share great structural similarity to native Env trimers (termed SOSIP, uncleaved prefusion optimized (UFO), single-chain (SC), and native-flexible-linker (NFL), among others), with the hope of inducing bnAbs. However, none have thus far succeeded in this aim. These efforts are no doubt a crucial step in immunogen design and have been reviewed elsewhere [9][10]. Complicating these efforts is the existence of a wide array of N-linked glycan structures on the Env surface that modulate its interaction with the host immune response and whose composition is dependent on a number of variable viral and host factors. One such viral factor, the Env signal peptide (SP), has recently been shown to greatly influence the composition of N-glycans on Env [11]. Notably, glycosylation of the HIV-1 Env can modulate the antibody response.
2. HIV-1 Envelope Structure and Immunogenicity
In order to understand how Env glycosylation complicates efforts to create effective Env-based immunogens, it is important to understand the structure, function, and inherent immune evasion properties of Env. HIV-1 Env exists as a trimeric spike on the viral surface, consisting of three heterodimers. These heterodimers arise from furin-mediated cleavage of glycoprotein 160 (gp160) proteins, resulting in a glycoprotein 120 (gp120) non-covalently linked to a glycoprotein 41 (gp41) (Figure 1, top). The gp120 subunit can be further subdivided into five conserved regions (C1-C5) and five hypervariable regions (V1-V5) [12][13][14][15]. Binding to host cells is facilitated by the gp120 subunit, which contains both the CD4 and co-receptor (either CCR5 or CXCR4) binding sites. Upon binding, Env undergoes conformational changes that allow gp41 to mediate fusion of the viral membrane with the host cell membrane, leading to infection [6][13][16][17]. Env is a dynamic molecule that exists in one of three conformations at any given time: a metastable closed conformation (state 1), a partially open intermediate conformation (state 2), and an open conformation (state 3) (Figure 2). Env transitions between these three conformations, but the majority of known bnAbs, isolated from HIV-1 infected individuals, target Env in state 1. Of note, the current recombinant Env trimers present Env in a stabilized version of state 2 [18]. In state 2 or 3, the variable loops V1V2 and V3 are more available for immune recognition. This functionally means that the initial antibody response is often specific to the infecting strain, and that escape mutations are common. Uncleaved gp160 monomers, as well as gp41 stumps lacking an associated gp120, also exist on the viral surface. These nonfunctional proteins display epitopes that may lead to the generation of non-neutralizing antibodies (nnAbs). Combined with the relatively low number of functional Env trimers, these mechanisms serve to effectively divert the host immune response [19][20][21][22][23][24][25].
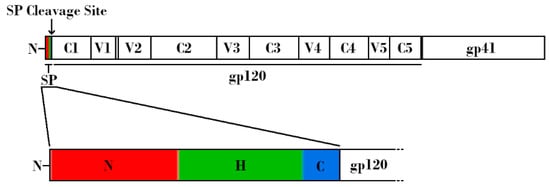
Figure 1. Schematic linear representation of the nascent HIV-1 Envelope (Env) protein attached to the HIV-1 signal peptide (SP). (Top) Regions corresponding to SP, glycoprotein 120 (gp120), and glycoprotein 41 (gp41) are indicated. Variable and constant regions of gp120 are indicated by V1-V5 and C1-C5, respectively. SP cleavage site is indicated by an arrow. (Bottom) Expanded schematic representation of the HIV-1 SP. The N-terminal hydrophilic positively charged region is shown in red, the central hydrophobic region is shown in green, and the slightly polar C-terminal region is shown in blue.
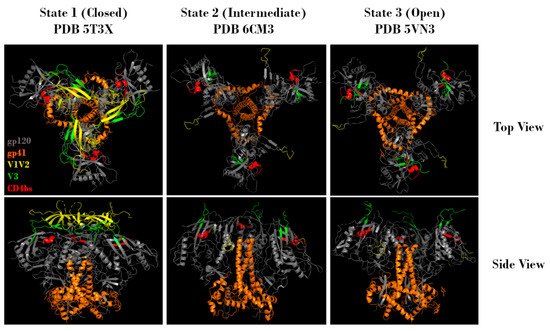
Figure 2. Representative top and side views of Env trimers in state 1 (closed), state 2 (intermediate), and state 3 (open) conformations. Regions corresponding to gp120 (grey), gp41 (orange), V1V2 (yellow), V3 (green), and the CD4 binding site (red) are indicated. Protein Data Bank identifier (PDB ID) numbers are also indicated. Structures adapted from Gristick et al., 2016 [26], Bjorkman et al., 2018 [27], and Ozorowski et al., 2017 [28].
3. HIV-1 Env N-linked Glycosylation
The existence of a wide array of N-glycans on the Env surface adds an additional layer of complexity to the already highly variable Env. Each Env of the trimer contains between 18 and 33 potential N-linked glycosylation sites (PNGSs), whose occupancy by glycans can account for up to half of the trimer’s total mass and cover up to 50–70% of the Env surface [7][29][30][31][32]. In this way, these poorly immunogenic glycans shield underlying protein residues from immune recognition. The occupancy of any particular PNGS is variable, with most being less than 90% conserved between HIV-1 strains [33][34]. Most PNGSs exist on gp120, but ~4–5 can be found on gp41 as well [33].
The mechanism by which Env is processed prior to deposition on the viral surface is a key determinant in the makeup of the Env “glycan shield”. Shortly after SP-mediated delivery of the nascent Env polypeptide to the endoplasmic reticulum (ER) for processing, PNGS occupancy is initiated via the placement of a Glc3Man9GlcNAc2 residue at each site. During subsequent transit through the ER and Golgi, this precursor glycan is modified by a milieu of host enzymes to yield its final glycoform (Figure 3A). However, the accessibility of these enzymes is greatly affected by steric constraints related to PNGS occupancy, which is in turn dependent on viral strain, host cell type, and acquired mutations [33][35][36][37][38][39]. Due to the high level of overall PNGS occupancy, these steric constraints result in a glycan landscape that consists predominantly of high-mannose (immature/unprocessed Man5-9GlcNAc2) glycans, with a smaller proportion of more mature/highly-processed complex and hybrid glycans [39][40][41][42][43][44][45][46][47]. Even with such constraints, the total number of variables involved in this process leads to incredibly diverse glycan landscapes from the outset [41][46][48].
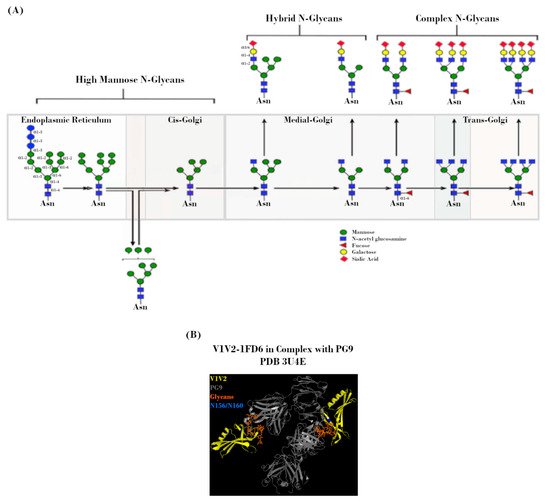
Figure 3. N-linked glycans on the HIV-1 envelope (Env). (A) Potential N-linked glycosylation site (PNGS) occupancy is initiated via the placement of a Glc3Man9GlcNAc2 residue, which is subsequently modified during transit through the endoplasmic reticulum (ER) and Golgi by a milieu of host enzymes to yield its final glycoform. Figure adapted from Jan et al., 2019 [49]. (B) Crystal structure (2.19 Å) of PG9 mAb in complex with V1V2 region from HIV-1. Regions corresponding to V1V2 (yellow), PG9 (grey), PNGS N156 and N160 (blue), and N-glycans (orange) are indicated. Protein Data Bank identifier (PDB ID) number is also indicated. Adapted from McLellan et al., 2011 [50].
In much the same way that these glycans sterically hinder host glycosidases, they also confer resistance to nAbs [30][51][52]. Interactions between antibodies and these host-derived glycans are often low affinity, due to both the nature of such interactions as well as the fact that said glycans are immunologically “self” [53][54][55]. That is not to say that glycan-targeting bnAbs cannot exist, merely that glycans are often only one component of larger, non-linear epitopes [56]. Occupancy of specific PNGSs, such as N156 and N160, has been shown to be crucial for the formation of conformational epitopes recognized by bnAbs, such as PG9 and PG16 (Figure 3B) [57]. Additionally, one of the most common bnAb targets is a high-mannose patch centered around either N332 or N334, dependent on viral strain and antibody lineage [58][59][60][61]. This so-called glycan supersite is also the target of 2G12, which binds an epitope derived solely of glycans. Due to their dependence on higher-order conformation, attempts to elicit these bnAbs in the context of vaccination with recombinant Env constructs have not been successful. There is also evidence that glycans can modulate the composition of immune responses via interactions with antigen presenting cells. Glycans of the C2V3 region of Env have been shown in a mouse model to be capable of inducing a bias for unfavorable IgG1 antibody subtype and TH2 T cell responses [62].
Over the course of disease progression, selective pressure drives mutations that further aid in immune escape. These may manifest as altered PNGS locations, such as the N332 to N334 mutation that has been shown to mutate in both directions to avoid immune recognition [63]. They may also manifest as alterations in glycoforms, due to genetic or steric determinants [33][36][48][63][64][65][66][67][68][69][70]. Furthermore, single mutations that alter the occupancy or glycoform at any particular PNGS may have cascading effects on the processing of glycans at other PNGSs. Taken together, these factors contribute to a complex and constantly moving target for the immune system [71].
Despite this variation, a number of generalized structural and glycan motifs have been identified in HIV-1 isolates from varying stages of disease progression. In the vast majority of heterosexual infection events (>80%), a single transmitted/founder (T/F) strain is responsible for the establishment of infection [72][73][74][75][76]. For this reason, T/F strains are a prime target of interest for vaccines, microbicides, and pre- and post-exposure prophylactic measures. Cases involving multiple infecting strains are often tied to alternative transmission routes (such as intravenous drug use), or breakdown of mucosal barriers [75][77][78]. These T/F strains have been shown to possess a number of characteristics that separate them from chronic isolates and may partially explain why they are effective at establishing infection. For instance, T/F viruses from clades A, C, and D have been shown to possess shorter V1V2 regions and a reduced number of occupied PNGSs compared to chronic isolates [65][77][79][80][81][82][83][84]. Additionally, some clade B isolates have been shown to possess a shorter V5 region, as well as a V3 region that contains fewer occupied PNGSs and less positively charged residues [77][81][85][86][87][88][89][90]. As these variable regions are commonly targeted by nAbs, their shortening presents fewer potential targets for the immune system [91][92][93]. Due to their important roles in infection, many conserved sites (such as the CD4 and coreceptor binding sites) within these variable regions are rarely, if ever, directly occluded from immunological access via glycans. The lower PNGS occupancy observed in T/F viruses may reflect an adaptation to divert the immune response away from these crucial sites and onto residues in the vicinity of unoccupied PNGSs. Indeed, the earliest nAbs targeting T/F viruses are often specific for non-conserved residues within the variable regions of Env.
Wagh et al. recently utilized a computational model in order to characterize the relationship between the completeness of the glycan shield at transmission and the development of nAbs [51]. They found that a more complete glycan shield at transmission correlated with more rapid development of a broad nAb response. One possibility for this observation is that an increased initial PNGS occupancy represents fewer opportunities for escape. While not all escape mutations result in increased PNGS occupancy, we know that chronic isolates have more complete glycan shielding and a higher proportion of complex/hybrid glycans, suggesting a directionality to the process [6][68][77][94][95][96][97]. Additionally, as PNGS become occupied by primarily high-mannose glycans, the virus becomes more susceptible to capture and degradation by the C-type lectin DC-SIGN on dendritic cells and macrophages [49][98][99][100][101][102]. This leads to increased antigen presentation and therefore may increase the rate at which these rounds of escape and adaptation occur. Eventual increases in the proportions of complex/hybrid glycans decrease viral capture by DC-SIGN but are also temporally associated with the development of bnAbs [49][103]. However, virus that does end up captured via this route is protected from degradation by these more mature glycans, resulting in increased levels of CD4+ T cell transinfection and thus representing an additional viral route of infection free of bnAb interference.
It is clear that the contribution of glycosylation at all stages of HIV-1 infection is great, and therefore, understanding the mechanisms governing it is going to be crucial to the development of an effective vaccine. Recently, the Env SP has been implicated as an important determining factor in this process.
References
- Harrigan, P.R.; Whaley, M.; Montaner, J.S. Rate of HIV-1 RNA rebound upon stopping antiretroviral therapy. AIDS 1999, 13, F59–F62.
- Antiretroviral Therapy Cohort Collaboration. Survival of HIV-positive patients starting antiretroviral therapy between 1996 and 2013: A collaborative analysis of cohort studies. Lancet HIV 2017, 4, e349–e356.
- Johnson, L.F.; Mossong, J.; Dorrington, R.E.; Schomaker, M.; Hoffmann, C.J.; Keiser, O.; Fox, M.P.; Wood, R.; Prozesky, H.; Giddy, J.; et al. Life expectancies of South African adults starting antiretroviral treatment: Collaborative analysis of cohort studies. PLoS Med. 2013, 10, e1001418.
- Iyidogan, P.; Anderson, K.S. Current perspectives on HIV-1 antiretroviral drug resistance. Viruses 2014, 6, 4095–4139.
- Pham, H.T.; Mesplede, T. The latest evidence for possible HIV-1 curative strategies. Drugs Context 2018, 7, 212522.
- Richman, D.D.; Wrin, T.; Little, S.J.; Petropoulos, C.J. Rapid evolution of the neutralizing antibody response to HIV type 1 infection. Proc. Natl. Acad. Sci. USA 2003, 100, 4144–4149.
- Korber, B.; Gaschen, B.; Yusim, K.; Thakallapally, R.; Kesmir, C.; Detours, V. Evolutionary and immunological implications of contemporary HIV-1 variation. Br. Med. Bull. 2001, 58, 19–42.
- Forthal, D.N.; Landucci, G.; Gorny, M.K.; Zolla-Pazner, S.; Robinson, W.E., Jr. Functional activities of 20 human immunodeficiency virus type 1 (HIV-1)-specific human monoclonal antibodies. AIDS Res. Hum. Retrovir. 1995, 11, 1095–1099.
- Seabright, G.E.; Doores, K.J.; Burton, D.R.; Crispin, M. Protein and Glycan Mimicry in HIV Vaccine Design. J. Mol. Biol. 2019, 431, 2223–2247.
- Sanders, R.W.; Moore, J.P. Native-like Env trimers as a platform for HIV-1 vaccine design. Immunol. Rev. 2017, 275, 161–182.
- Upadhyay, C.; Feyznezhad, R.; Cao, L.; Chan, K.W.; Liu, K.; Yang, W.; Zhang, H.; Yolitz, J.; Arthos, J.; Nadas, A.; et al. Signal peptide of HIV-1 envelope modulates glycosylation impacting exposure of V1V2 and other epitopes. PLoS Pathog. 2020, 16, e1009185.
- Starcich, B.R.; Hahn, B.H.; Shaw, G.M.; McNeely, P.D.; Modrow, S.; Wolf, H.; Parks, E.S.; Parks, W.P.; Josephs, S.F.; Gallo, R.C.; et al. Identification and characterization of conserved and variable regions in the envelope gene of HTLV-III/LAV, the retrovirus of AIDS. Cell 1986, 45, 637–648.
- Checkley, M.A.; Luttge, B.G.; Freed, E.O. HIV-1 envelope glycoprotein biosynthesis, trafficking, and incorporation. J. Mol. Biol. 2011, 410, 582–608.
- Moore, J.P.; Willey, R.L.; Lewis, G.K.; Robinson, J.; Sodroski, J. Immunological evidence for interactions between the first, second, and fifth conserved domains of the gp120 surface glycoprotein of human immunodeficiency virus type 1. J. Virol. 1994, 68, 6836–6847.
- Freed, E.O. HIV-1 replication. Somat. Cell Mol. Genet. 2001, 26, 13–33.
- Rong, R.; Bibollet-Ruche, F.; Mulenga, J.; Allen, S.; Blackwell, J.L.; Derdeyn, C.A. Role of V1V2 and other human immunodeficiency virus type 1 envelope domains in resistance to autologous neutralization during clade C infection. J. Virol. 2007, 81, 1350–1359.
- Chan, D.C.; Kim, P.S. HIV entry and its inhibition. Cell 1998, 93, 681–684.
- Wang, Q.; Finzi, A.; Sodroski, J. The Conformational States of the HIV-1 Envelope Glycoproteins. Trends Microbiol. 2020, 28, 655–667.
- Bachmann, M.F.; Rohrer, U.H.; Kundig, T.M.; Burki, K.; Hengartner, H.; Zinkernagel, R.M. The influence of antigen organization on B cell responsiveness. Science 1993, 262, 1448–1451.
- Zhu, P.; Liu, J.; Bess, J., Jr.; Chertova, E.; Lifson, J.D.; Grise, H.; Ofek, G.A.; Taylor, K.A.; Roux, K.H. Distribution and three-dimensional structure of AIDS virus envelope spikes. Nature 2006, 441, 847–852.
- Pancera, M.; Wyatt, R. Selective recognition of oligomeric HIV-1 primary isolate envelope glycoproteins by potently neutralizing ligands requires efficient precursor cleavage. Virology 2005, 332, 145–156.
- Parren, P.W.; Burton, D.R.; Sattentau, Q.J. HIV-1 antibody--debris or virion? Nat. Med. 1997, 3, 366–367.
- Moore, P.L.; Crooks, E.T.; Porter, L.; Zhu, P.; Cayanan, C.S.; Grise, H.; Corcoran, P.; Zwick, M.B.; Franti, M.; Morris, L.; et al. Nature of nonfunctional envelope proteins on the surface of human immunodeficiency virus type 1. J. Virol. 2006, 80, 2515–2528.
- Sattentau, Q.J.; Moore, J.P. Human immunodeficiency virus type 1 neutralization is determined by epitope exposure on the gp120 oligomer. J. Exp. Med. 1995, 182, 185–196.
- Sanders, R.W.; Derking, R.; Cupo, A.; Julien, J.P.; Yasmeen, A.; de Val, N.; Kim, H.J.; Blattner, C.; de la Pena, A.T.; Korzun, J.; et al. A next-generation cleaved, soluble HIV-1 Env trimer, BG505 SOSIP.664 gp140, expresses multiple epitopes for broadly neutralizing but not non-neutralizing antibodies. PLoS Pathog. 2013, 9, e1003618.
- Gristick, H.B.; von Boehmer, L.; West, A.P., Jr.; Schamber, M.; Gazumyan, A.; Golijanin, J.; Seaman, M.S.; Fatkenheuer, G.; Klein, F.; Nussenzweig, M.C.; et al. Natively glycosylated HIV-1 Env structure reveals new mode for antibody recognition of the CD4-binding site. Nat. Struct. Mol. Biol. 2016, 23, 906–915.
- Wang, H.; Barnes, C.O.; Yang, Z.; Nussenzweig, M.C.; Bjorkman, P.J. Partially Open HIV-1 Envelope Structures Exhibit Conformational Changes Relevant for Coreceptor Binding and Fusion. Cell Host Microbe 2018, 24, 579–592.
- Ozorowski, G.; Pallesen, J.; de Val, N.; Lyumkis, D.; Cottrell, C.A.; Torres, J.L.; Copps, J.; Stanfield, R.L.; Cupo, A.; Pugach, P.; et al. Open and closed structures reveal allostery and pliability in the HIV-1 envelope spike. Nature 2017, 547, 360–363.
- Lasky, L.A.; Groopman, J.E.; Fennie, C.W.; Benz, P.M.; Capon, D.J.; Dowbenko, D.J.; Nakamura, G.R.; Nunes, W.M.; Renz, M.E.; Berman, P.W. Neutralization of the AIDS retrovirus by antibodies to a recombinant envelope glycoprotein. Science 1986, 233, 209–212.
- Pancera, M.; Zhou, T.; Druz, A.; Georgiev, I.S.; Soto, C.; Gorman, J.; Huang, J.; Acharya, P.; Chuang, G.Y.; Ofek, G.; et al. Structure and immune recognition of trimeric pre-fusion HIV-1 Env. Nature 2014, 514, 455–461.
- Zhang, M.; Gaschen, B.; Blay, W.; Foley, B.; Haigwood, N.; Kuiken, C.; Korber, B. Tracking global patterns of N-linked glycosylation site variation in highly variable viral glycoproteins: HIV, SIV, and HCV envelopes and influenza hemagglutinin. Glycobiology 2004, 14, 1229–1246.
- Leonard, C.K.; Spellman, M.W.; Riddle, L.; Harris, R.J.; Thomas, J.N.; Gregory, T.J. Assignment of intrachain disulfide bonds and characterization of potential glycosylation sites of the type 1 recombinant human immunodeficiency virus envelope glycoprotein (gp120) expressed in Chinese hamster ovary cells. J. Biol. Chem. 1990, 265, 10373–10382.
- Go, E.P.; Ding, H.; Zhang, S.; Ringe, R.P.; Nicely, N.; Hua, D.; Steinbock, R.T.; Golabek, M.; Alin, J.; Alam, S.M.; et al. Glycosylation Benchmark Profile for HIV-1 Envelope Glycoprotein Production Based on Eleven Env Trimers. J. Virol. 2017, 91.
- Pritchard, L.K.; Spencer, D.I.; Royle, L.; Bonomelli, C.; Seabright, G.E.; Behrens, A.J.; Kulp, D.W.; Menis, S.; Krumm, S.A.; Dunlop, D.C.; et al. Glycan clustering stabilizes the mannose patch of HIV-1 and preserves vulnerability to broadly neutralizing antibodies. Nat. Commun. 2015, 6, 7479.
- Torrents de la Pena, A.; Rantalainen, K.; Cottrell, C.A.; Allen, J.D.; van Gils, M.J.; Torres, J.L.; Crispin, M.; Sanders, R.W.; Ward, A.B. Similarities and differences between native HIV-1 envelope glycoprotein trimers and stabilized soluble trimer mimetics. PLoS Pathog. 2019, 15, e1007920.
- Cao, L.; Diedrich, J.K.; Kulp, D.W.; Pauthner, M.; He, L.; Park, S.R.; Sok, D.; Su, C.Y.; Delahunty, C.M.; Menis, S.; et al. Global site-specific N-glycosylation analysis of HIV envelope glycoprotein. Nat. Commun. 2017, 8, 14954.
- Cao, L.; Pauthner, M.; Andrabi, R.; Rantalainen, K.; Berndsen, Z.; Diedrich, J.K.; Menis, S.; Sok, D.; Bastidas, R.; Park, S.R.; et al. Differential processing of HIV envelope glycans on the virus and soluble recombinant trimer. Nat. Commun. 2018, 9, 3693.
- Struwe, W.B.; Chertova, E.; Allen, J.D.; Seabright, G.E.; Watanabe, Y.; Harvey, D.J.; Medina-Ramirez, M.; Roser, J.D.; Smith, R.; Westcott, D.; et al. Site-Specific Glycosylation of Virion-Derived HIV-1 Env Is Mimicked by a Soluble Trimeric Immunogen. Cell Rep. 2018, 24, 1958–1966.
- Bonomelli, C.; Doores, K.J.; Dunlop, D.C.; Thaney, V.; Dwek, R.A.; Burton, D.R.; Crispin, M.; Scanlan, C.N. The glycan shield of HIV is predominantly oligomannose independently of production system or viral clade. PLoS ONE 2011, 6, e23521.
- Behrens, A.J.; Struwe, W.B.; Crispin, M. Glycosylation profiling to evaluate glycoprotein immunogens against HIV-1. Expert Rev. Proteom. 2017, 14, 881–890.
- Kong, L.; Sheppard, N.C.; Stewart-Jones, G.B.E.; Robson, C.L.; Chen, H.; Xu, X.; Krashias, G.; Bonomelli, C.; Scanlan, C.N.; Kwong, P.D.; et al. Expression-system-dependent modulation of HIV-1 envelope glycoprotein antigenicity and immunogenicity. J. Mol. Biol. 2010, 403, 131–147.
- Raska, M.; Takahashi, K.; Czernekova, L.; Zachova, K.; Hall, S.; Moldoveanu, Z.; Elliott, M.C.; Wilson, L.; Brown, R.; Jancova, D.; et al. Glycosylation patterns of HIV-1 gp120 depend on the type of expressing cells and affect antibody recognition. J. Biol. Chem. 2010, 285, 20860–20869.
- Doores, K.J.; Bonomelli, C.; Harvey, D.J.; Vasiljevic, S.; Dwek, R.A.; Burton, D.R.; Crispin, M.; Scanlan, C.N. Envelope glycans of immunodeficiency virions are almost entirely oligomannose antigens. Proc. Natl. Acad. Sci. USA 2010, 107, 13800–13805.
- Binley, J.M.; Ban, Y.E.; Crooks, E.T.; Eggink, D.; Osawa, K.; Schief, W.R.; Sanders, R.W. Role of complex carbohydrates in human immunodeficiency virus type 1 infection and resistance to antibody neutralization. J. Virol. 2010, 84, 5637–5655.
- Jan, M.; Upadhyay, C.; Sharma, A.; Hioe, C.E.; Arora, S.K. Short Communication: Manalpha1-2Man-Binding Anti-HIV Lectins Enhance the Exposure of V2i and V3 Crown Neutralization Epitopes on the V1/V2 and V3 Hypervariable Loops of HIV-1 Envelope. AIDS Res. Hum. Retrovir. 2017, 33, 941–945.
- Go, E.P.; Herschhorn, A.; Gu, C.; Castillo-Menendez, L.; Zhang, S.; Mao, Y.; Chen, H.; Ding, H.; Wakefield, J.K.; Hua, D.; et al. Comparative Analysis of the Glycosylation Profiles of Membrane-Anchored HIV-1 Envelope Glycoprotein Trimers and Soluble gp140. J. Virol. 2015, 89, 8245–8257.
- Go, E.P.; Liao, H.X.; Alam, S.M.; Hua, D.; Haynes, B.F.; Desaire, H. Characterization of host-cell line specific glycosylation profiles of early transmitted/founder HIV-1 gp120 envelope proteins. J. Proteome Res. 2013, 12, 1223–1234.
- Behrens, A.J.; Vasiljevic, S.; Pritchard, L.K.; Harvey, D.J.; Andev, R.S.; Krumm, S.A.; Struwe, W.B.; Cupo, A.; Kumar, A.; Zitzmann, N.; et al. Composition and Antigenic Effects of Individual Glycan Sites of a Trimeric HIV-1 Envelope Glycoprotein. Cell Rep. 2016, 14, 2695–2706.
- Jan, M.; Upadhyay, C.; Hioe, C.E. HIV-1 Envelope Glycan Composition as a Key Determinant of Efficient Virus Transmission via DC-SIGN and Resistance to Inhibitory Lectins. iScience 2019, 21, 413–427.
- McLellan, J.S.; Pancera, M.; Carrico, C.; Gorman, J.; Julien, J.P.; Khayat, R.; Louder, R.; Pejchal, R.; Sastry, M.; Dai, K.; et al. Structure of HIV-1 gp120 V1/V2 domain with broadly neutralizing antibody PG9. Nature 2011, 480, 336–343.
- Wagh, K.; Kreider, E.F.; Li, Y.; Barbian, H.J.; Learn, G.H.; Giorgi, E.; Hraber, P.T.; Decker, T.G.; Smith, A.G.; Gondim, M.V.; et al. Completeness of HIV-1 Envelope Glycan Shield at Transmission Determines Neutralization Breadth. Cell Rep. 2018, 25, 893–908.e7.
- Liang, Y.; Guttman, M.; Williams, J.A.; Verkerke, H.; Alvarado, D.; Hu, S.L.; Lee, K.K. Changes in Structure and Antigenicity of HIV-1 Env Trimers Resulting from Removal of a Conserved CD4 Binding Site-Proximal Glycan. J. Virol. 2016, 90, 9224–9236.
- Wardemann, H.; Yurasov, S.; Schaefer, A.; Young, J.W.; Meffre, E.; Nussenzweig, M.C. Predominant autoantibody production by early human B cell precursors. Science 2003, 301, 1374–1377.
- Haynes, B.F.; Verkoczy, L. AIDS/HIV. Host controls of HIV neutralizing antibodies. Science 2014, 344, 588–589.
- Cohen, M. Notable Aspects of Glycan-Protein Interactions. Biomolecules 2015, 5, 2056–2072.
- Crispin, M.; Bowden, T.A. Antibodies expose multiple weaknesses in the glycan shield of HIV. Nat. Struct. Mol. Biol. 2013, 20, 771–772.
- Doores, K.J.; Burton, D.R. Variable loop glycan dependency of the broad and potent HIV-1-neutralizing antibodies PG9 and PG16. J. Virol. 2010, 84, 10510–10521.
- Steichen, J.M.; Kulp, D.W.; Tokatlian, T.; Escolano, A.; Dosenovic, P.; Stanfield, R.L.; McCoy, L.E.; Ozorowski, G.; Hu, X.; Kalyuzhniy, O.; et al. HIV Vaccine Design to Target Germline Precursors of Glycan-Dependent Broadly Neutralizing Antibodies. Immunity 2016, 45, 483–496.
- Doores, K.J.; Kong, L.; Krumm, S.A.; Le, K.M.; Sok, D.; Laserson, U.; Garces, F.; Poignard, P.; Wilson, I.A.; Burton, D.R. Two classes of broadly neutralizing antibodies within a single lineage directed to the high-mannose patch of HIV envelope. J. Virol. 2015, 89, 1105–1118.
- Gray, E.S.; Madiga, M.C.; Hermanus, T.; Moore, P.L.; Wibmer, C.K.; Tumba, N.L.; Werner, L.; Mlisana, K.; Sibeko, S.; Williamson, C.; et al. The neutralization breadth of HIV-1 develops incrementally over four years and is associated with CD4+ T cell decline and high viral load during acute infection. J. Virol. 2011, 85, 4828–4840.
- Blazkova, J.; Refsland, E.W.; Clarridge, K.E.; Shi, V.; Justement, J.S.; Huiting, E.D.; Gittens, K.R.; Chen, X.; Schmidt, S.D.; Liu, C.; et al. Glycan-dependent HIV-specific neutralizing antibodies bind to cells of uninfected individuals. J. Clin. Investig. 2019, 129, 4832–4837.
- Hess, R.; Storcksdieck Genannt Bonsmann, M.; Lapuente, D.; Maaske, A.; Kirschning, C.; Ruland, J.; Lepenies, B.; Hannaman, D.; Tenbusch, M.; Uberla, K. Glycosylation of HIV Env Impacts IgG Subtype Responses to Vaccination. Viruses 2019, 11, 153.
- Moore, P.L.; Gray, E.S.; Wibmer, C.K.; Bhiman, J.N.; Nonyane, M.; Sheward, D.J.; Hermanus, T.; Bajimaya, S.; Tumba, N.L.; Abrahams, M.R.; et al. Evolution of an HIV glycan-dependent broadly neutralizing antibody epitope through immune escape. Nat. Med. 2012, 18, 1688–1692.
- Freund, N.T.; Wang, H.; Scharf, L.; Nogueira, L.; Horwitz, J.A.; Bar-On, Y.; Golijanin, J.; Sievers, S.A.; Sok, D.; Cai, H.; et al. Coexistence of potent HIV-1 broadly neutralizing antibodies and antibody-sensitive viruses in a viremic controller. Sci. Transl. Med. 2017, 9.
- Dacheux, L.; Moreau, A.; Ataman-Onal, Y.; Biron, F.; Verrier, B.; Barin, F. Evolutionary dynamics of the glycan shield of the human immunodeficiency virus envelope during natural infection and implications for exposure of the 2G12 epitope. J. Virol. 2004, 78, 12625–12637.
- Tian, J.; Lopez, C.A.; Derdeyn, C.A.; Jones, M.S.; Pinter, A.; Korber, B.; Gnanakaran, S. Effect of Glycosylation on an Immunodominant Region in the V1V2 Variable Domain of the HIV-1 Envelope gp120 Protein. PLoS Comput. Biol. 2016, 12, e1005094.
- Stewart-Jones, G.B.; Soto, C.; Lemmin, T.; Chuang, G.Y.; Druz, A.; Kong, R.; Thomas, P.V.; Wagh, K.; Zhou, T.; Behrens, A.J.; et al. Trimeric HIV-1-Env Structures Define Glycan Shields from Clades A, B, and G. Cell 2016, 165, 813–826.
- Wei, X.; Decker, J.M.; Wang, S.; Hui, H.; Kappes, J.C.; Wu, X.; Salazar-Gonzalez, J.F.; Salazar, M.G.; Kilby, J.M.; Saag, M.S.; et al. Antibody neutralization and escape by HIV-1. Nature 2003, 422, 307–312.
- Lemmin, T.; Soto, C.; Stuckey, J.; Kwong, P.D. Microsecond Dynamics and Network Analysis of the HIV-1 SOSIP Env Trimer Reveal Collective Behavior and Conserved Microdomains of the Glycan Shield. Structure 2017, 25, 1631–1639.
- Yang, M.; Huang, J.; Simon, R.; Wang, L.X.; MacKerell, A.D., Jr. Conformational Heterogeneity of the HIV Envelope Glycan Shield. Sci. Rep. 2017, 7, 4435.
- Rudd, P.M.; Dwek, R.A. Glycosylation: Heterogeneity and the 3D structure of proteins. Crit. Rev. Biochem. Mol. Biol. 1997, 32, 1–100.
- Keele, B.F.; Giorgi, E.E.; Salazar-Gonzalez, J.F.; Decker, J.M.; Pham, K.T.; Salazar, M.G.; Sun, C.; Grayson, T.; Wang, S.; Li, H.; et al. Identification and characterization of transmitted and early founder virus envelopes in primary HIV-1 infection. Proc. Natl. Acad. Sci. USA 2008, 105, 7552–7557.
- Shaw, G.M.; Hunter, E. HIV transmission. Cold Spring Harb Perspect. Med. 2012, 2.
- Wawer, M.J.; Gray, R.H.; Sewankambo, N.K.; Serwadda, D.; Li, X.; Laeyendecker, O.; Kiwanuka, N.; Kigozi, G.; Kiddugavu, M.; Lutalo, T.; et al. Rates of HIV-1 transmission per coital act, by stage of HIV-1 infection, in Rakai, Uganda. J. Infect. Dis. 2005, 191, 1403–1409.
- Joseph, S.B.; Swanstrom, R.; Kashuba, A.D.; Cohen, M.S. Bottlenecks in HIV-1 transmission: Insights from the study of founder viruses. Nat. Rev. Microbiol. 2015, 13, 414–425.
- Kariuki, S.M.; Selhorst, P.; Arien, K.K.; Dorfman, J.R. The HIV-1 transmission bottleneck. Retrovirology 2017, 14, 22.
- Frost, S.D.; Liu, Y.; Pond, S.L.; Chappey, C.; Wrin, T.; Petropoulos, C.J.; Little, S.J.; Richman, D.D. Characterization of human immunodeficiency virus type 1 (HIV-1) envelope variation and neutralizing antibody responses during transmission of HIV-1 subtype B. J. Virol. 2005, 79, 6523–6527.
- Bar, K.J.; Li, H.; Chamberland, A.; Tremblay, C.; Routy, J.P.; Grayson, T.; Sun, C.; Wang, S.; Learn, G.H.; Morgan, C.J.; et al. Wide variation in the multiplicity of HIV-1 infection among injection drug users. J. Virol. 2010, 84, 6241–6247.
- Chohan, B.; Lang, D.; Sagar, M.; Korber, B.; Lavreys, L.; Richardson, B.; Overbaugh, J. Selection for human immunodeficiency virus type 1 envelope glycosylation variants with shorter V1-V2 loop sequences occurs during transmission of certain genetic subtypes and may impact viral RNA levels. J. Virol. 2005, 79, 6528–6531.
- Liu, Y.; Curlin, M.E.; Diem, K.; Zhao, H.; Ghosh, A.K.; Zhu, H.; Woodward, A.S.; Maenza, J.; Stevens, C.E.; Stekler, J.; et al. Env length and N-linked glycosylation following transmission of human immunodeficiency virus Type 1 subtype B viruses. Virology 2008, 374, 229–233.
- Finzi, A.; Pacheco, B.; Xiang, S.H.; Pancera, M.; Herschhorn, A.; Wang, L.; Zeng, X.; Desormeaux, A.; Kwong, P.D.; Sodroski, J. Lineage-specific differences between human and simian immunodeficiency virus regulation of gp120 trimer association and CD4 binding. J. Virol. 2012, 86, 8974–8986.
- Gnanakaran, S.; Bhattacharya, T.; Daniels, M.; Keele, B.F.; Hraber, P.T.; Lapedes, A.S.; Shen, T.; Gaschen, B.; Krishnamoorthy, M.; Li, H.; et al. Recurrent signature patterns in HIV-1 B clade envelope glycoproteins associated with either early or chronic infections. PLoS Pathog. 2011, 7, e1002209.
- Helseth, E.; Olshevsky, U.; Furman, C.; Sodroski, J. Human immunodeficiency virus type 1 gp120 envelope glycoprotein regions important for association with the gp41 transmembrane glycoprotein. J. Virol. 1991, 65, 2119–2123.
- Derdeyn, C.A.; Decker, J.M.; Bibollet-Ruche, F.; Mokili, J.L.; Muldoon, M.; Denham, S.A.; Heil, M.L.; Kasolo, F.; Musonda, R.; Hahn, B.H.; et al. Envelope-constrained neutralization-sensitive HIV-1 after heterosexual transmission. Science 2004, 303, 2019–2022.
- Kafando, A.; Martineau, C.; El-Far, M.; Fournier, E.; Doualla-Bell, F.; Serhir, B.; Kazienga, A.; Sangare, M.N.; Sylla, M.; Chamberland, A.; et al. HIV-1 Envelope Glycoprotein Amino Acids Signatures Associated with Clade B Transmitted/Founder and Recent Viruses. Viruses 2019, 11, 1012.
- Baalwa, J.; Wang, S.; Parrish, N.F.; Decker, J.M.; Keele, B.F.; Learn, G.H.; Yue, L.; Ruzagira, E.; Ssemwanga, D.; Kamali, A.; et al. Molecular identification, cloning and characterization of transmitted/founder HIV-1 subtype A, D and A/D infectious molecular clones. Virology 2013, 436, 33–48.
- De Wolf, F.; Hogervorst, E.; Goudsmit, J.; Fenyo, E.M.; Rubsamen-Waigmann, H.; Holmes, H.; Galvao-Castro, B.; Karita, E.; Wasi, C.; Sempala, S.D.; et al. Syncytium-inducing and non-syncytium-inducing capacity of human immunodeficiency virus type 1 subtypes other than B: Phenotypic and genotypic characteristics. WHO Network for HIV Isolation and Characterization. AIDS Res. Hum. Retrovir. 1994, 10, 1387–1400.
- Wilen, C.B.; Parrish, N.F.; Pfaff, J.M.; Decker, J.M.; Henning, E.A.; Haim, H.; Petersen, J.E.; Wojcechowskyj, J.A.; Sodroski, J.; Haynes, B.F.; et al. Phenotypic and immunologic comparison of clade B transmitted/founder and chronic HIV-1 envelope glycoproteins. J. Virol. 2011, 85, 8514–8527.
- Chandramouli, B.; Chillemi, G.; Abbate, I.; Capobianchi, M.R.; Rozera, G.; Desideri, A. Importance of V3 loop flexibility and net charge in the context of co-receptor recognition. A molecular dynamics study on HIV gp120. J. Biomol. Struct. Dyn. 2012, 29, 879–891.
- Kaleebu, P.; Nankya, I.L.; Yirrell, D.L.; Shafer, L.A.; Kyosiimire-Lugemwa, J.; Lule, D.B.; Morgan, D.; Beddows, S.; Weber, J.; Whitworth, J.A. Relation between chemokine receptor use, disease stage, and HIV-1 subtypes A and D: Results from a rural Ugandan cohort. J. Acquir. Immune Defic. Syndr. 2007, 45, 28–33.
- Zolla-Pazner, S.; Cardozo, T. Structure-function relationships of HIV-1 envelope sequence-variable regions refocus vaccine design. Nat. Rev. Immunol. 2010, 10, 527–535.
- Pejchal, R.; Doores, K.J.; Walker, L.M.; Khayat, R.; Huang, P.S.; Wang, S.K.; Stanfield, R.L.; Julien, J.P.; Ramos, A.; Crispin, M.; et al. A potent and broad neutralizing antibody recognizes and penetrates the HIV glycan shield. Science 2011, 334, 1097–1103.
- Zhou, T.; Georgiev, I.; Wu, X.; Yang, Z.Y.; Dai, K.; Finzi, A.; Kwon, Y.D.; Scheid, J.F.; Shi, W.; Xu, L.; et al. Structural basis for broad and potent neutralization of HIV-1 by antibody VRC01. Science 2010, 329, 811–817.
- McNearney, T.; Hornickova, Z.; Markham, R.; Birdwell, A.; Arens, M.; Saah, A.; Ratner, L. Relationship of human immunodeficiency virus type 1 sequence heterogeneity to stage of disease. Proc. Natl. Acad. Sci. USA 1992, 89, 10247–10251.
- Bar, K.J.; Tsao, C.Y.; Iyer, S.S.; Decker, J.M.; Yang, Y.; Bonsignori, M.; Chen, X.; Hwang, K.K.; Montefiori, D.C.; Liao, H.X.; et al. Early low-titer neutralizing antibodies impede HIV-1 replication and select for virus escape. PLoS Pathog. 2012, 8, e1002721.
- Moore, P.L.; Gray, E.S.; Morris, L. Specificity of the autologous neutralizing antibody response. Curr. Opin. HIV AIDS 2009, 4, 358–363.
- Bonsignori, M.; Kreider, E.F.; Fera, D.; Meyerhoff, R.R.; Bradley, T.; Wiehe, K.; Alam, S.M.; Aussedat, B.; Walkowicz, W.E.; Hwang, K.K.; et al. Staged induction of HIV-1 glycan-dependent broadly neutralizing antibodies. Sci. Transl. Med. 2017, 9.
- van Montfort, T.; Eggink, D.; Boot, M.; Tuen, M.; Hioe, C.E.; Berkhout, B.; Sanders, R.W. HIV-1 N-glycan composition governs a balance between dendritic cell-mediated viral transmission and antigen presentation. J. Immunol. 2011, 187, 4676–4685.
- Gringhuis, S.I.; den Dunnen, J.; Litjens, M.; van der Vlist, M.; Geijtenbeek, T.B. Carbohydrate-specific signaling through the DC-SIGN signalosome tailors immunity to Mycobacterium tuberculosis, HIV-1 and Helicobacter pylori. Nat. Immunol. 2009, 10, 1081–1088.
- Jan, M.; Arora, S.K. Innate Sensing of HIV-1 by Dendritic Cell-Specific ICAM-3 Grabbing Nonintegrin on Dendritic Cells: Degradation and Presentation Versus Transmission of Virus to T Cells Is Determined by Glycan Composition of Viral Envelope. AIDS Res. Hum. Retrovir. 2017, 33, 765–767.
- van Liempt, E.; Bank, C.M.; Mehta, P.; Garcia-Vallejo, J.J.; Kawar, Z.S.; Geyer, R.; Alvarez, R.A.; Cummings, R.D.; Kooyk, Y.; van Die, I. Specificity of DC-SIGN for mannose- and fucose-containing glycans. FEBS Lett. 2006, 580, 6123–6131.
- Geijtenbeek, T.B.; Kwon, D.S.; Torensma, R.; van Vliet, S.J.; van Duijnhoven, G.C.; Middel, J.; Cornelissen, I.L.; Nottet, H.S.; KewalRamani, V.N.; Littman, D.R.; et al. DC-SIGN, a dendritic cell-specific HIV-1-binding protein that enhances trans-infection of T cells. Cell 2000, 100, 587–597.
- Yolitz, J.; Schwing, C.; Chang, J.; Van Ryk, D.; Nawaz, F.; Wei, D.; Cicala, C.; Arthos, J.; Fauci, A.S. Signal peptide of HIV envelope protein impacts glycosylation and antigenicity of gp120. Proc. Natl. Acad. Sci. USA 2018, 115, 2443–2448.
More
Information
Subjects:
Virology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
881
Revisions:
2 times
(View History)
Update Date:
25 Nov 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





