
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Gomaa A. M. Ali | + 3343 word(s) | 3343 | 2021-11-18 08:59:30 | | | |
| 2 | Gomaa A. M. Ali | + 28 word(s) | 3371 | 2021-11-18 23:13:31 | | | | |
| 3 | Jessie Wu | -22 word(s) | 3349 | 2021-11-19 02:32:49 | | | | |
| 4 | Jessie Wu | -22 word(s) | 3349 | 2021-11-19 02:34:03 | | | | |
| 5 | ABDERRHMANE BOUAFIA | Meta information modification | 3349 | 2021-11-20 20:19:07 | | |
Video Upload Options
Nanomaterials are highly effective, environmentally friendly, and applicable for various applications. Recently, silver nanoparticles (Ag NPs) are increasingly being synthesized due to their physical, chemical, and biomedical properties. Silver nanoparticles can be synthesized using physical, chemical, and biological methods. Ag NPs are widely applied in electronic and sensing applications.
1. Background
Nanoscience enables researchers to develop new and cost-effective nanomaterials for energy, healthcare, and medical applications. Silver nanoparticles (Ag NPs) are currently increasingly synthesized for their superior physicochemical and electronic properties. Good knowledge of these characteristics allows the development of applications in all sensitive and essential fields in the service of humans and the environment. This review aims to summarize the Ag NPs synthesis methods, properties, applications, and future challenges. Generally, Ag NPs can be synthesized using physical, chemical, and biological routes. Due to the great and increasing demand for metal and metal oxide nanoparticles, researchers have invented a new, environmentally friendly, inexpensive synthetic method that replaces other methods with many defects. Studies of Ag NPs have increased after clear and substantial support from governments to develop nanotechnology. Ag NPs are the most widely due to their various potent properties.
2. Silver Nanoparticles
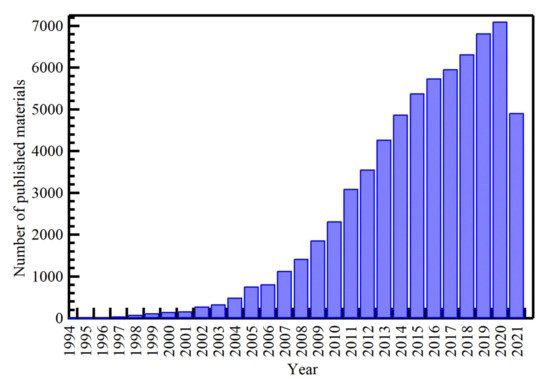
2.1. History of Silver Nanoparticles
2.2. Synthesis of Silver Nanoparticles
2.2.1. Top-Down and Bottom-Up Approaches
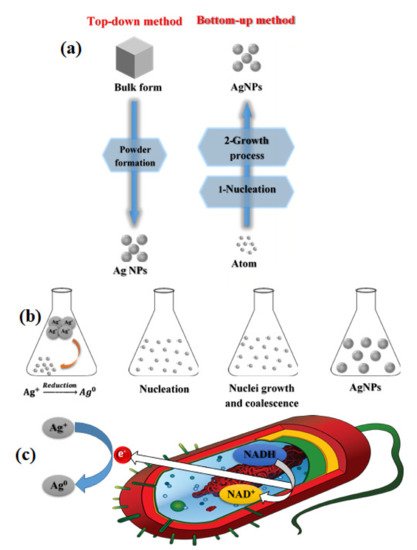
2.2.2. Physical Methods
| Physical Method | Applications | Shape | Size (nm) | Reference |
|---|---|---|---|---|
| Laser ablation | Antibacterial efficiency | Semi-spherical | 14 | [26] |
| Spherical | 13–32 | [27] | ||
| Antibacterial activity | 5–30 | [28] | ||
| Catalytic degradation activity | Spherical-like | 17 | [29] | |
| Spherical | 8–10 | [30] | ||
| Thermal decomposition |
40–50 | [31] | ||
| 5–15 | [32] | |||
| Cubic/hexagonal | 3.0–4.5 | [33] | ||
| Small Ceramic Heater | Inhalation toxicity studies | Spherical | 14 | [34] |
2.2.3. Chemical Methods
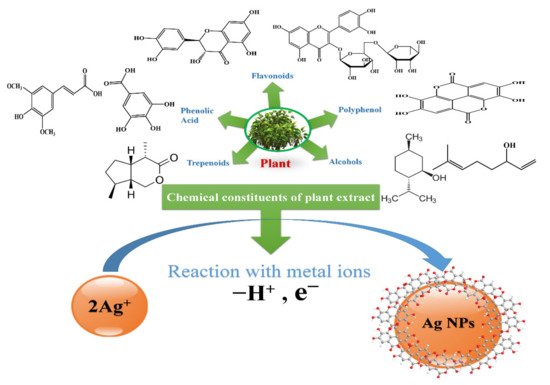
2.2.4. Green Methods
| Source | Bioreducing Agent of Silver Nitrate |
The Mechanism for the Synthesis |
|---|---|---|
| Plants | Alkaloids, Terpenes, Steroids and Saponins, Flavonoids and Tannins, Alcohol, Phenolic Acids. | Electrostatic interaction between the functional groups of a respective constituent of plant extract and Ag+ |
| Bacteria | Bacillus Cereus, Bacillus licheniformis, Staphylococcus aureus, Enterobacteriaceae, Pantoeaananatis, Proteus mirabilis |
In the extra-cellular synthesis of Ag NPs by Bacillus subtilis, the synthesis of Ag NPs was observed in the reaction mixture after 6 h contact time at room temperature. |
| Fungi | Proteins, peptides enzymes, napthoquinones, NADH, NADPH, peptides, nitrogenous biomacromolecules, | Intracellular and extracellular synthesis of Ag NPs |
| Biopolymers | Cellulose, Chitosan, polypeptides, alginate, lignin, protein | Electrostatic interaction between Ag+ ion and polar groups attached to the polymer |
2.5. Green Synthesis by Plant Extract
| Plant Sources | Part | Applications | Operating Conditions | Size/Shape | Reference |
|---|---|---|---|---|---|
| Onion, tomato, Acacia catechu |
Pieces | Photocatalytic application |
16.987 g of AgNO3 in 1 L of distilled water, 10 mL extract, stirred for 10 min. and kept for 24 h at room temperature | 32.1, 22.6, 14.5 nm, Spherical | [56] |
| Curcuma longa L. | Leaf | Antibacterial activity | 10 mL of Curcuma longa L. leaf extrac, 90 mL of 1 mM AgNO3. | 15–40 nm, spherical | [57] |
| Phaseolus vulgaris L. | Seeds | Photocatalytic activity, antimicrobial activity | Extract (1, 2, 3, and 4 mL), AgNO3 (0.01 M, 50 mL), (300 rpm) for 30 min | 10–20 nm, spherical | [58] |
| Solanum nigrum L. | Leaves | Ecotoxicity Studies | 1 mL leaf extract, AgNO3 (10−3 M, 50 mL), stirred continuously at room temperature | 10–50 nm, spherical | [59] |
| f Citrus reticulata |
Peels | Biocide and anticorrosion properties |
1 mM AgNO3, ratio 1:1 with tangerine peels extract | 39.6–56.1 nm, round | [60] |
| Apple | Fruits | Antibacterial activity |
AgNO3 (1 mM, 90 mL), 10 mL of extract, at 70 °C. | 45–110 nm, spherical shape |
[61] |
| Clove | Buds | Antibacterial and antidiatom activity | AgNO3 (1 mM, 400 mL), 80 mL of extract, darkness for 24 h | 9.42 nm | [62] |
| Delonix regia | Leaf | In vitro cytotoxicity and interaction studies with bovine serum albumin | Leaf extract and an aqueous solution of 1 mM AgNO3 (20:80, V/V). | 72.77 nm, non-uniform |
[63] |
References
- Arora, S.; Jain, J.; Rajwade, M.; Paknikar, K.M. Cellular responses induced by silver nanoparticles: In vitro studies. Toxicol. Lett. 2008, 179, 93–100.
- Ma, W.; Yang, H.; Wang, W.; Gao, P.; Yao, J. Ethanol vapor sensing properties of triangular silver nanostructures based on localized surface plasmon resonance. Sensors 2011, 11, 8643–8653.
- Chen, X.; Schluesener, H.J. Nanosilver: A nanoproduct in medical application. Toxicol. Lett. 2008, 176, 1–12.
- Farré, M.; Gajda-Schrantz, K.; Kantiani, L.; Barceló, D. Ecotoxicity and analysis of nanomaterials in the aquatic environment. Anal. Bioanal. Chem. 2009, 393, 81–95.
- Scown, T.M.; Santos, E.M.; Johnston, B.D.; Gaiser, B.; Baalousha, M.; Mitov, S.; Lead, J.R.; Stone, V.; Fernandes, T.F.; Jepson, M.; et al. Effects of Aqueous Exposure to Silver Nanoparticles of Different Sizes in Rainbow Trout. Toxicol. Sci. 2010, 115, 521–534.
- Scopus. Available online: https://www.scopus.com/ (accessed on 20 August 2021).
- Lee, P.C.; Meisel, D. Adsorption and Surface-Enhanced Raman of Dyes on Silver and Gold Sols. J. Phys. Chem. 1982, 86, 3391–3395.
- Van Hyning, D.L.; Zukoski, C.F. Formation Mechanisms and Aggregation Behavior of Borohydride Reduced Silver Particles. Langmuir 1998, 14, 7034–7046.
- Zielinska, A.; Skwarek, E.; Zaleska, A.; Gazda, M.; Hupka, J. Preparation of silver nanoparticles with controlled particle size. Procedia Chem. 2009, 1, 1560–1566.
- Suzanne, E.; Howson, A.R.; Scott, P.; Bolhuis, A.; Brabec, V.; Clarkson, G.J.; Malina, J. Optically pure, water-stable metallo-helical flexicate’ assemblies with antibiotic activity. Nat. Chem. 2012, 4, 31–36.
- Hsu, S.L.-C.; Wu, R.-T. Preparation of Silver Nanoparticle with Different Particle Sizes for Low-Temperature Sintering. Int. Conf. Nanotechnol. Biosens. IPCBEE 2010, 2, 55–58.
- Rodríguez-Sánchez, L.; Blanco, M.C.; López-Quintela, M.A. Electrochemical Synthesis of Silver Nanoparticles. J. Phys. Chem. B 2000, 104, 9683–9688.
- Khaydarov, R.A.; Khaydarov, R.R.; Gapurova, O.; Estrin, Y.; Scheper, T. Electrochemical method for the synthesis of silver nanoparticles. J Nanoparticle Res 2009, 11, 1193–1200.
- Dikovska, A.O.; Alexandrov, M.T.; Atanasova, G.B.; Tsankov, N.T.; Stefanov, P.K. Silver nanoparticles produced by PLD in vacuum: Role of the laser wavelength used. Appl. Phys. A 2013, 113, 83–88.
- Wei, L.; Lu, J.; Xu, H.; Patel, A.; Chen, Z.; Chen, G. Silver nanoparticles: Synthesis, properties, and therapeutic applications. Drug Discov. Today 2015, 20, 595–601.
- Burdușel, A.-C.; Gherasim, O.; Mogoantă, L.; Ficai, A.; Andronescu, E.; Grumezescu, A.M. Biomedical Applications of Silver Nanoparticles: An Up-to-Date Overview. Nanomaterials 2018, 8, 681.
- Chugh, H.; Sood, D.; Chandra, I.; Tomar, V.; Dhawan, G.; Chandra, R. Role of gold and silver nanoparticles in cancer nano-medicine. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1210–1220.
- Tran, Q.H.; Nguyen, V.Q.; Le, A.-T. Silver nanoparticles: Synthesis, properties, toxicology, applications and perspectives. Adv. Nat. Sci. Nanosci. Nanotechnol. 2013, 4, 033001.
- Lee, S.H.; Jun, B.H. Silver Nanoparticles: Synthesis and Application for Nanomedicine. Int. J. Mol. Sci. 2019, 20, 865.
- Tsuji, T.; Iryo, K.; Ohta, H.; Nishimura, Y. Preparation of Metal Colloids by a Laser Ablation Technique in Solution: Influence of Laser Wavelength on the Efficiencies of Colloid Formation. Jpn. J. Appl. Phys. 2000, 39, 981–983.
- Amendola, V.; Meneghettia, M. Laser ablation synthesis in solution and size manipulation of noble metal nanoparticles. Phys. Chem. Chem. Phys. 2009, 11, 3805–3821.
- Martin, H.; Magnusson, K.D.; Malm, J.; Bovin, J.; Samuelson, L. Gold nanoparticles: Production, reshaping, and thermal charging. J. Nanoparticle Res. 1999, 1, 243–251.
- Jung, J.H.; Oh, H.; Noh, H.S.; Ji, J.H.; Kim, S.S. Metal nanoparticle generation using a small ceramic heater with a local heating area. Aerosol Sci. 2006, 37, 1662–1670.
- Tsuji, T.; Kakita, T.; Tsuji, M. Preparation of nano-size particles of silver with femtosecond laser ablation in water. Appl. Surf. Sci. 2003, 206, 314–320.
- Menazea, A.A. Femtosecond laser ablation-assisted synthesis of silver nanoparticles in organic and inorganic liquids medium and their antibacterial efficiency. Radiat. Phys. Chem. 2020, 168, 108616.
- Elmira Solati, M.M.; Dorranian, D. Effects of laser pulse wavelength and laser fluence on the characteristics of silver nanoparticle generated by laser ablation. Appl Phys. A 2013, 112, 689–694.
- Menazeaa, A.A.; Ahmed, M.K. Silver and copper oxide nanoparticles-decorated graphene oxide via pulsed laser ablation technique: Preparation, characterization, and photoactivated antibacterial activity. Nano-Struct. Nano-Objects 2020, 22, 100464.
- Ayman, M.; Mostafa, A.A.M. Polyvinyl Alcohol/Silver nanoparticles film prepared via pulsed laser ablation: An eco-friendly nano-catalyst for 4-nitrophenol degradation. J. Mol. Struct. 2020, 1212, 128125.
- Wagener, P.; Ibrahimkutty, S.; Menzel, A.; Plech, A.; Barcikowski, S. Dynamics of silver nanoparticle formation and agglomeration inside the cavitation bubble after pulsed laser ablation in liquid. Phys. Chem. Chem. Phys. 2013, 15, 3068–3074.
- Hosseinpour-Mashkani, S.M.; Ramezani, M. Silver and silver oxide nanoparticles: Synthesis and characterizationby thermal decomposition. Mater. Lett. 2014, 130, 259–262.
- Goudarzi, M.; Mir, N.; Mousavi-Kamazani, M.; Bagheri, S.; Salavati-Niasari, M. Biosynthesis and characterization of silver nanoparticles prepared from two novel natural precursors by facile thermal decomposition methods. Sci. Rep. 2016, 6, 32539.
- Jeevanandam, P.; Srikanth, C.K.; Dixit, S. Synthesis of monodisperse silver nanoparticles and their self-assembly through simple thermal decomposition approach. Mater. Chem. Phys. 2010, 122, 402–407.
- Ji, J.H.; Jung, J.H.; Yu, J.; Kim, S.S. Long-Term Stability Characteristics of Metal Nanoparticle Generator Using Small Ceramic Heater for Inhalation Toxicity Studies. Inhal. Toxicol. 2007, 19, 745–751.
- Merga, G.; Wilson, R.; Lynn, G.; Milosavljevic, B.H.; Meisel, D. Redox Catalysis on “Naked” Silver Nanoparticles. J. Phys. Chem. C 2007, 111, 12220–12226.
- Oliveira, M.M.; Ugarte, D.; Zanchet, D.; Zarbin, A.J.G. Influence of synthetic parameters on the size, structure, and stability of dodecanethiol-stabilized silver nanoparticles. J. Colloid Interface Sci. 2005, 292, 429–435.
- Bai, J.; Li, Y.; Du, J.; Wang, S.; Zheng, J.; Yang, Q.; Chen, X. One-pot synthesis of polyacrylamide-gold nanocomposite. Mater. Chem. Phys. 2007, 106, 412–415.
- Pillai, Z.S.; Kamat, P.V. What Factors Control the Size and Shape of Silver Nanoparticles in the Citrate Ion Reduction Method? J. Phys. Chem. B 2004, 108, 945–951.
- Bouafia, A.; Laouini, S.E. Green synthesis of iron oxide nanoparticles by aqueous leaves extract of Mentha Pulegium, L.: Effect of ferric chloride concentration on the type of product. Mater. Lett. 2020, 265, 127364.
- Gudimalla, A.; Jose, J.; Varghese, R.J.; Thomas, S. Green Synthesis of Silver Nanoparticles Using Nymphae odorata Extract Incorporated Films and Antimicrobial Activity. J. Polym. Environ. 2021, 29, 1412–1423.
- Jung, W.K.; Kim, S.H.; Koo, H.C.; Shin, S.; Kim, J.M.; Park, Y.K.; Hwang, S.Y.; Yang, H.; Park, Y.H. Antifungal activity of the silver ion against contaminated fabric. Mycoses 2007, 50, 265–269.
- Morales-Lozoya, V.; Espinoza-Gómez, H.; Flores-López, Z.L.; Sotelo-Barrera, E.L.; Núñez-Rivera, A.; Cadena-Nava, R.D.; Alonso-Nuñez, G.; Rivero, I.A. Study of the effect of the different parts of Morinda citrifolia L. (noni) on the green synthesis of silver nanoparticles and their antibacterial activity. Appl. Surf. Sci. 2021, 537, 147855.
- Kiani, M.; Rabiee, N.; Bagherzadeh, M.; Ghadiri, A.M.; Fatahi, Y.; Dinarvand, R.; Webster, T.J. Improved Green Biosynthesis of Chitosan Decorated Ag- and Co3O4-Nanoparticles: A Relationship Between Surface Morphology, Photocatalytic and Biomedical Applications. Nanomed. Nanotechnol. Biol. Med. 2021, 32, 102331.
- Vanlalveni, C.; Lallianrawna, S.; Biswas, A.; Selvaraj, M.; Changmai, B.; Rokhum, S.L. Green synthesis of silver nanoparticles using plant extracts and their antimicrobial activities: A review of recent literature. RSC Adv. 2021, 11, 2804–2837.
- He, Y.; Li, X.; Zheng, Y.; Wang, Z.; Ma, Z.; Yang, Q.; Yao, B.; Zhao, Y.; Zhang, H. A green approach for synthesizing silver nanoparticles, and their antibacterial and cytotoxic activities. New J. Chem. 2018, 42, 2882–2888.
- Laid, T.M.; Abdelhamid, K.; Eddine, L.S.; Bouafia, A. Optimizing the biosynthesis parameters of iron oxide nanoparticles using central composite design. J. Mol. Struct. 2021, 1229, 129497.
- Abdullah, J.A.A.; Salah Eddine, L.; Bouafia, A.; Alonso-González, M.; Guerrero, A.; Romero, A. Green synthesis and characterization of iron oxide nanoparticles by pheonix dactylifera leaf extract and evaluation of their antioxidant activity. Sustain. Chem. Pharm. 2020, 17, 100280.
- Pirtarighat, S.; Ghannadnia, M.; Baghshahi, S. Green synthesis of silver nanoparticles using the plant extract of Salvia spinosa grown in vitro and their antibacterial activity assessment. J. Nanostructure Chem. 2019, 9, 1–9.
- Gardea-Torresdey, J.L.; Gomez, E.; Peralta-Videa, J.R.; Parsons, J.G.; Troiani, H.; Jose-Yacaman, M. Alfalfa Sprouts: A Natural Source for the Synthesis of Silver Nanoparticles. Langmuir 2003, 19, 1357–1361.
- Laouini, S.E.; Bouafia, A.; Soldatov, A.V.; Algarni, H.; Tedjani, M.L.; Ali, G.A.M.; Barhoum, A. Green Synthesized of Ag/Ag2O Nanoparticles Using Aqueous Leaves Extracts of Phoenix dactylifera L. and Their Azo Dye Photodegradation. Membranes 2021, 11, 468.
- Krithiga, N.; Rajalakshmi, A.; Jayachitra, A. Green Synthesis of Silver Nanoparticles Using Leaf Extracts of Clitoria ternatea and Solanum nigrum and Study of Its Antibacterial Effect against Common Nosocomial Pathogens. J. Nanosci. 2015, 2015, 1–8.
- Yadav, S.; Khurana, J.M. Cinnamomum tamala leaf extract-mediated green synthesis of Ag nanoparticles and their use in pyranopyrazles synthesis. Chin. J. Catal. 2015, 36, 1042–1046.
- Shankar, S.S.; Ahmad, A.; Sastry, M. Geranium Leaf Assisted Biosynthesis of Silver Nanoparticles. Biotechnol. Prog. 2003, 19, 1627–1631.
- Mittal, A.K.; Chisti, Y.; Banerjee, U.C. Synthesis of metallic nanoparticles using plant extracts. Biotechnol. Adv. 2013, 31, 346–356.
- Tarannum, N.; Gautam, Y.K. Facile green synthesis and applications of silver nanoparticles: A state-of-the-art review. RSC Adv. 2019, 9, 34926–34948.
- Chand, K.; Cao, D.; Eldin Fouad, D.; Hussain Shah, A.; Qadeer Dayo, A.; Zhu, K.; Nazim Lakhan, M.; Mehdi, G.; Dong, S. Green synthesis, characterization and photocatalytic application of silver nanoparticles synthesized by various plant extracts. Arab. J. Chem. 2020, 13, 8248–8261.
- Maghimaaa, M.; Alharbi, S.A. Green synthesis of silver nanoparticles from Curcuma longa L. and coating on the cotton fabrics for antimicrobial applications and wound healing activity. J. Photochem. Photobiol. B Biol. 2020, 204, 111806.
- Rani, P.; Kumar, V.; Singh, P.P.; Matharu, A.S.; Zhang, W.; Kim, K.-H.; Singh, J.; Rawat, M. Highly stable AgNPs prepared via a novel green approach for catalytic and photocatalytic removal of biological and non-biological pollutants. Environ. Int. 2020, 143, 105924.
- Jenifer, A.A.; Anjugam, M.; Malaikozhundan, B.; Iswarya, A. Green Synthesis and Characterization of Silver Nanoparticles (AgNPs) Using Leaf Extract of Solanum nigrum and Assessment of Toxicity in Vertebrate and Invertebrate Aquatic Animals. J. Clust. Sci. 2020, 31, 989–1002.
- Ituen, E.; Ekemini, E.; Yuanhua, L.; Singh, A. Green synthesis of Citrus reticulata peels extract silver nanoparticles and characterization of structural, biocide and anticorrosion properties. J. Mol. Struct. 2020, 1207, 127819.
- Kazlagić, A.; Abud, O.A.; Ćibo, M.; Hamidović, S.; Borovac, B.; Omanović-Mikličanin, E. Green synthesis of silver nanoparticles using apple extract and its antimicrobial properties. Health Technol. 2020, 10, 147–150.
- Lakhana, M.N.; Chena, R.; Shara, A.H.; Chanda, K.; Shahb, A.H.; Ahmeda, M.; Alic, I.; Ahmedd, R.; Liua, J.; Takahashia, K.; et al. Eco-friendly green synthesis of clove buds extract functionalized silver nanoparticles and evaluation of antibacterial and antidiatom activity. J. Microbiol. Methods 2020, 173, 105934.
- Siddiquee, M.A.; Parray, M.u.d.; Mehdi, S.H.; Alzahrani, K.A.; Alshehri, A.A.; Malik, M.A.; Patel, R. Green synthesis of silver nanoparticles from Delonix regia leaf extracts: In-vitro cytotoxicity and interaction studies with bovine serum albumin. Mater. Chem. Phys. 2020, 242, 122493.
- Kanmani, P.; Lim, S.T. Synthesis and structural characterization of silver nanoparticles using bacterial exopolysaccharide and its antimicrobial activity against food and multidrug resistant pathogens. Process Biochem. 2013, 48, 1099–1106.





