
Video Upload Options
SAPCs provide a new pathway for the construction of high performance cocatalyst/photocatalyst composites. For the past few years, the preparation method, characterization technology, theoretical modeling, and mechanism investigation of SAPCs have been developed rapidly, promoting the rational design and fabrication of more efficient SAPCs. As discussed above, benefitting from their low-coordination status, unique electric structures, and metal-support interactions, SAPCs can bring about unique advantages in comparison with the nanocluster-based, nanoparticle-based, and bulk catalysts.
1. Introduction
Since the pioneering discovery of water photolysis on TiO 2 electrodes in 1972, solar-driven water splitting has been recognized as one of the most promising ways to transform solar energy into renewable and clean hydrogen energy [1]. The photocatalysts are the key component of photocatalytic processes [2]. Over the past few decades, researchers have developed various photocatalysts toward the application of water splitting [3][4][5]. A typical photocatalytic water splitting reaction on semiconductor photocatalysts mainly involves three primary steps: (1) absorption of light to generate electron-hole pairs; (2) separation of the photogenerated electron-hole pairs and their migration to semiconductor surfaces; and (3) consumption of the photogenerated electrons and holes via surface redox reactions. Specifically, only the photogenerated electrons and holes that successfully reach the surface of the catalyst have the possibility to react with water. Therein, the water molecules can be reduced by photogenerated electrons to generate H 2 (hydrogen evolution reaction, HER), and at the same time oxidized by photogenerated holes to generate oxygen (Oxygen evolution reaction, OER), hydrogen peroxide, superoxide radicals, etc [6]. The problem is that the single-component photocatalyst always suffers from poor photocatalytic activities because of its narrow light harvesting range, serious electron-hole recombination, and weak adsorption and activation abilities for reactant molecules. To overcome these bottlenecks, strategies aiming to expand the light absorption range, construct interface heterojunctions, and engineer surface reaction sites by rational design of multicomponent photocatalysts have been developed [7][8]. Among these, the loading of a cocatalyst is an effective method to improve the functions of a photocatalyst [9]. For example, noble metals, such as Pt and Pd nanoparticles, are considered as efficient cocatalysts for promoting photocatalytic H 2 evolution [10]. However, the high cost of these noble metal particles strongly hinders their wide utilization.
Single-atom photocatalysts (SAPCs) with maximized atomic utilization can effectively improve the number of exposed reaction sites on surface while reducing the loading amount of the noble metal cocatalyst [11][12]. For SAPCs, the atomically dispersed metal atoms could be anchored on the support materials by forming coordination with the host atoms, which helps to prevent agglomeration of the monodispersed metal atoms with high surface energy [13]. Compared with metal nanoparticles, the coordination interaction with the support atoms makes the single metal atoms possess a unique local electronic and geometric structure [14]. In addition, the electron transfer and charge redistribution between the single metal atoms and supports may reduce the reaction barriers to enhancing catalytic performance [15]. Nevertheless, due to the high surface free energy of isolated atoms, their aggregation is always unavoidable, and this is significantly detrimental to photocatalytic activity and stability. Therefore, how to develop SAPCs with high activity, selectivity, and stability, and how to unravel the structure-activity relationship of SAPCs still remain as the research focus in this area. In this review, the recent advances in exploiting SAPCs as the new prototype of cocatalyst/photocatalyst composites toward water splitting are introduced, especially from the aspect of their atomic structure, synthetic strategy, and applications. Then, the challenges and future research directions of single-atom photocatalysts are discussed.
2. Structure of SAPCs
Metal oxides, metal sulfides, and other inorganic semiconductor materials with appropriate bandgaps such as TiO 2, ZnO, WO 3, ZrO 2, CdS, GaS, MoS 2, BiVO 3, BiOX (x= Cl, Br), SrTiO 3, etc. have been frequently used as photocatalysts [16][17][18][19][20]. The abundant coordination atoms of O, S in the lattice and surface of these materials can provide a strong anchoring effect for stabilizing the single metal atoms.
In recent years, organic polymer semiconductors, especially g-C 3N 4, have attracted extensive attention in photocatalysis owing to their fascinating advantages such as appropriate energy band structure, good stability, nontoxicity, low cost, etc. [21][22][23]. It is worth noting that the bandgap of pristine g-C 3N 4 is about 2.7 eV, with the lowest unoccupied molecular orbital (LUMO) and highest occupied molecular orbital (HOMO) well fitted for driving water splitting from the aspect of thermodynamics [24]. At the same time, g-C 3N 4 possesses abundant N atoms with lone-pair electrons, which are ideal coordinating sites for anchoring single metal atoms [25]. It has been found that the isolated metal atoms anchored on g-C 3N 4 can serve as active centers for boosting photocatalytic HER and ameliorate the low separation efficiency of photogenerated charge carriers for pristine g-C 3N 4 [26][27]. Hence, g-C 3N 4 has been intensively explored as the support materials for hosting single atoms, giving birth to various SAPCs.
Carbon dots (CDs) are another attractive support material for hosting single atoms. Their bandgaps can be tuned by nitrogen doping to function as a good light-harvesting antenna [28]. Run Li et al. synthesized the CDs anchored with single Co atoms [29], in which the single Co atoms with a high loading content of 3.27% were stabilized through the Co−N 4 coordination structure. The authors unraveled that CDs played double functions in photocatalysis: the support for single-atom Co and the light-harvesting antenna. Meanwhile, the isolated Co atoms can boost the separation and directed migration of photogenerated charge carriers, and, more significantly, can gather the photogenerated holes for promoting oxidation reactions. As a result, the obtained photocatalyst exhibited excellent activity and stability in photocatalytic water oxidation (245 umol/g, 4h), photocatalytic oxidative coupling of aromatic amines, and photodegradation of organic dye.
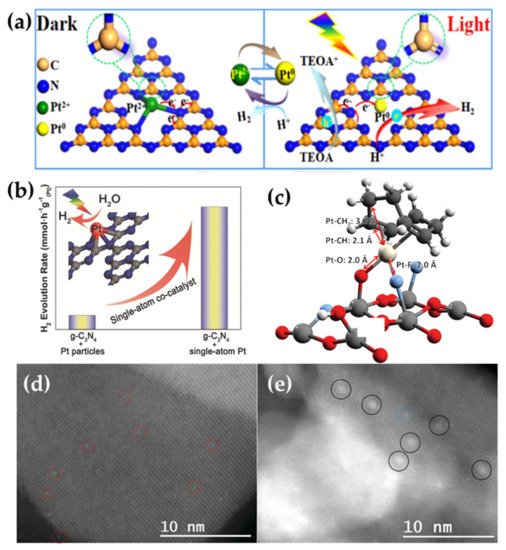
Due to their suitable Fermi level for trapping electrons and a low potential for H 2 evolution, noble metal Pt is an efficient cocatalyst for H 2 evolution and has been intensively studied in SAPCs [30][31][32][33]. Wu et al. reported a photocatalyst with significantly enhanced photocatalytic activity and maximized utilization of Pt [34], in which the isolated Pt atoms were incorporate into the sub-nanoporosity of 2D g-C 3N 4 (referred as Pt-C 3N 4, Figure 1 b) and acted as co-catalyst for photocatalytic HER. The intrinsic change of the surface trap states induced by single-atom Pt contributed to a longer lifetime of photogenerated electrons, which was the primary factor for the prominently improved photocatalytic performance. Bi et al. further investigated the in situ charge transfer and chemical bond evolution between the single-atom Pt and g-C 3N 4 during photocatalytic HER ( Figure 1 a) [35]. The results showed that different from the metallic Pt decorated-g-C 3N 4 catalyst, within single-atom Pt/C 3N 4 , the Pt-N bond broke to form Pt 0 species and the C-N bond transformed into C=N bond of g-C 3N 4 under visible light irradiation. Such dynamic changes designated Pt 0 and g-C 3N 4 as the reaction sites for reduction and oxidation reactions respectively, and this contributed to the remarkably enhanced H 2 evolution activity. Furthermore, it has been demonstrated that a higher loaded content of Pt single atoms can give much higher photocatalytic activity. For example, Zhou et al. developed a surfactant-stabilized coordination strategy to realize ultrahigh single-atom Pt loading of 12% within MOFs [36]. The resulting SAPCs exhibited an exceptionally high activity of 11320 umol g −1 h −1 for photocatalytic HER under visible light.
3. Synthesis of SAPCs
A simple and effective synthesis method is one critical factor for the practical application of SAPCs [38]. It is essential to construct strong coordination interactions between single metal atoms and support materials to avoid the aggregation of these isolated atoms with high surface free energy during synthesis and reaction processes [39]. This can be achieved through various strategies including defect engineering, coordinated design, and spatial confinement [40]. In the following section, the commonly used synthesis methods for SAPCs are summarized.
The impregnation method is a typical and flexible method for the synthesis of single-atom catalysts. The support materials are immersed in a solution containing appropriate metal precursors. In this condition, the metal ions could be anchored or absorbed on the support surface through coordination, electrostatic adsorption, or spatial confinement. This is followed by drying and activation treatment, which can make the single metal atoms stably anchored on the supports. Here, the bonding with coordination atoms in supports such as, O, N, P, S, etc. is the key to keep the metal atoms spatially isolated with restricted migration.
As for the co-precipitation method, single atoms are immobilized on the supports during their formation process. This method is suitable for support materials such as g-C 3N 4, MOF, metal sulfides, and metal [41][42][43]. For example, Lu et al. prepared a series of single-atom Mo catalysts by calcining low-cost primary material of urea with various amounts of Mo(VI) salts [44]. The isolated Mo atoms were incorporated into g-C 3N 4 through MoN 2 species in the polymerization process. The coordinatively unsaturated Mo sites acted as reaction centers for N 2 chemisorption and activation during photocatalysis. With regard to the metal supports, the atomic ratio of metal precursor to support is a key factor that should be precisely controlled for preparation of single atoms in metal supports. For example, Yang et al. synthesized a single-atom Pt 1Ag 24 alloy cocatalyst on g-C 3N 4, and the resulting composite (referred as Pt 1Ag 24/ g-C 3N 4) was applied in photocatalytic HER [45]. The H 2 evolution rate of Pt 1Ag 24/g-C 3N 4 reached 39.7 umol/h, which was four times higer than that of Ag 25/g-C 3N 4. The precise composition of single-atom Pt 1Ag 24 alloy cocatalyst was tuned by carefully tuning the molar ratio of Ag and Pt precursors during the nucleation and growth process.
( a ) Formation of the hollow nanotube MOF and the single atom-immobilized hollow nanotube MOF, which are denoted as HNTM and HNTM-M, respectively [46]; ( b ) Schematic illustration of the icing-assisted in-situ photocatalytic reduction method for preparing the supra-high-density PtSAs-loaded g-C 3N 4. Scale bar: 2 nm [47]; ( c ) Schematic illustration of the ALD mechanism for creating Pt single atoms on NGNs [48]; and ( d ) Illustration of the synthetic process of single-atom Pt photocatalysts [49].
4. Applications of SAPCs for Photocatalytic Water Splitting
H 2 produced from photocatalytic water splitting is an ideal clean energy to replace fossil fuels. However, great challenges remain for developing high-activity, durable, and economical photocatalysts that can realize practical applications toward photocatalytic H 2 generation. The creation of SAPCs provides a new clue for the development of water splitting photocatalysts with excellent activity, selectivity, and stability, as well as high atomic utilization of noble metals to lower the cost of the catalyst. Table 1 summarizes the recent progress in this research area. As discussed above, the single metal sites in SAPCs have been intensively used as efficient cocatalysts for promoting photocatalytic H 2 evolution.
| Supports | Single Atoms | Coordination | Loaded Content | Synthesis Methods | Products | Production Rate | AQE | |
|---|---|---|---|---|---|---|---|---|
| 1 [50] | TiO2 nanosheet | Rh | Rh-O | 2.6 at.% | co-precipitation | H2 | 51 umol h−1 | / |
| 2 [34] | g-C3N4 | Pt | Pt-N | 0.16 wt.% | impregnation | H2 | 318 umol h−1 | / |
| 3 [51] | g-C3N4 | Pt2+ | Pt-N | 0.18 wt.% | co-precipitation | H2 | 605 umol g−1 h−1 | / |
| 4 [52] | g-C3N4 | Co | Co1-N4 | 1 wt.% | ALD | H2 | 10.8 umol h−1 | 10.3%, 420 nm |
| 5 [45] | Ag cluster | Pt | Pt-Ag | 4 at.% | co-precipitation | H2 | 39.7 umol h−1 | / |
| 6 [53] | P doped g-C3N4 | Co | Co1-P4 | 0.40.% | impregnation | H2. O2 | 126.8 umol g−1 h−1 | 0.16%, AM 1.5 illumination |
| 7 [32] | g-C3N4 | Pt | Pt-N (maybe) | / | impregnation | H2 | 12.7 umol g−1, 4h | / |
| 8 [16] | TiO2 | Pt | Pt-O | 0.6 wt.% | photo-deposition | H2 | 85.4 umol h−1 | / |
| 9 [54] | NG | Co | / | 0.25 wt.% (Co-NG) | impregnation | H2 | 1382 umol h−1 | 50.5%, 420 nm |
| 10 [55] | g-C3N4 | Pd | surface:Pt-N;interlayer: Pt-N, Pt-C | 0.33 wt.% | photo-deposition | H2 | 6688 umol g−1 h−1 | 4%, 420 nm |
| 11 [56] | g-C3N4 | Pt | Pt1-N4 | 0.11 wt.% | photo-deposition | H2 | 42.1 umol h−1 | / |
| 12 [31] | Al- porphyrinic MOF | Pt | Pt-N | 0.29 wt.% | impregnation | H2 | 129 umol g−1 h−1 | / |
| 13 [46] | Zr–porphyrinic MOF | Ir, Pt, Ru, Au, Pd | Pt-N4Cl2 | 1.41 (Ir), 2.74 (Pt), 1.92 (Ru), 1.18 (Au), 3.68 (Pd), wt.% | impregnation | H2 | 201.9 umol g−1 h−1 | / |
| 14 [57] | g-C3N4 | PtⅡ | Pt-N | / | impregnation | H2 | 140 umol g−1 h−1 | 1.5%, 420 nm |
| 15 [58] | mesoporous TiO2 | Cu | Cu-O2.5 | <0.3 wt.% | impregnation | H2 | / | / |
| 16 [59] | CdS nanowires | Pt | Pt-S4 | 0.27–0.98 wt.% | impregnation | H2 | 47.41 mmol g−1 h−1 | / |
| 17 [60] | g-C3N4 | Pt | Pt-C/Pt-N | 0.17–1.7% | impregnation | H2 | 34.2 mmol h−1 | / |
| 18 [61] | MOF | Co | Co-N2Cl2O | 3.3 wt.% | co-precipitation | H2 | 27.853 mmol g−1, 40h | / |
| 19 [62] | NG | Co | Co-N2C | 3.5 wt.% (Co-NG) | impregnation | H2 | 677.44 umol g−1 h−1 | / |
| 20 [63] | NG | Ni | Ni-N | 0.26 wt.% | impregnation | H2 | 1351.1 umol h−1 | 48.2%, 420 nm |
| 21 [37] | TiO2 | (CH3)2Pt(COD) | O-Pt-O | / | impregnation | H2 | / | 12%, 225–387 nm |
| 22 [64] | TiO2 | Cu, Co, Fe, Ni, Rh | Cu-O | 0.75 wt.% | impregnation | H2 | 16.6 mmol g−1 h−1 | 45.5%, 340 nm |
| 23 [65] | GaS nanosheet | Ru(IV) | Ru1-S6 | 2.4 wt.% | impregnation | H2, O2 | 340 umol g−1 h−1 | / |
| 24 [33] | MOF−808-EDTA | Pt | Pt-N2O2 | 0.98 wt.% | impregnation | H2 | 68.33 mmol g−1 h−1 | 67.6%, 420 nm |
| 25 [66] | g-C3N4 | Pd | Pd-N | 0.1 wt.% | impregnation | H2 | 728 umol g−1 h−1 | / |
| 26 [67] | CdS@CDs | Pt | Pt-S | 1.15% | impregnation | H2 | 45.5 mmol g−1 h−1 | 29.8%, 400 nm |
| 27 [68] | zeolitic imidazole framework | Co | / | 0.57% | co-precipitation | H2 | 6420 umol g−1 h−1 | / |
| 28 [69] | g-C3N4 | Pt, Au | Pt-N, Au-N | 0.6 Pt, 0.8 (Au), wt.% | impregnation | H2, O2 | H2: 285 umol g−1 h−1, H2:O2 = 2:1 | / |
| 29 [28] | N doped CDs | Co | Co-N4 | 3.27 wt.% | co-precipitation | O2 | 245 umol g−1, 4h | / |
| 30 [70] | g-C3N4 | Au | Au-O | 0.18 wt.% | impregnation | H2 | 789.1 nmol h−1 | / |
| 31 [71] | g-C3N4 | Fe | Pt-N (DFT) | 0.5 at.% | impregnation | H2 | 3390 umol g−1 h−1 | 6.89%, 420 nm |
| 32 [47] | g-C3N4 | Pt | Pt-N/O/Cl | 0.35 mg m−2 | photo-deposition | H2 | 174.5 mmol g−1 h−1 | 0.544%, 420 nm |
| 33 [36] | MOF | Pt | Pt-N | 12 wt.% | co-precipitation | H2 | 11320 umol g−1 h−1 | / |
| 34 [72] | TiO2 | Pt | Pt-O | 0.36 wt.% | photo-deposition | H2 | 1077 umol h−1 | 21.7%, 365 nm |
| 35 [30] | defective TiO2 | Pt | Pt-O | 0.02wt% | impregnation | H2 | 4458 umol g−1 h−1 | / |
| 36 [73] | g-C3N4 | Rh | Rh-P | / | impregnation | H2 | 166.28 umol, 4h | / |
| 37 [74] | g-C3N4 | Co | Co-P | 0.13 wt.% | impregnation | O2, H2O2 | O2: 15.5 umol, 4h; | / |
| 38 [75] | TiO2 | Pt | / | 0.03–0.47 at.%. | impregnation | H2 | / | / |
| 39 [76] | g-C3N4 | Ag | Ag-C2N2 | 3.7 wt.% | co-precipitation | H2 | 1.8 mmol g−1 h−1 | / |
| 40 [77] | g-C3N4 | Ni | Ni-C/N | 0.5 at% | impregnation | H2 | 354.9 umol g−1 h−1 | / |
| 41 [78] | N-doped CDs | Pt | Pt-C | 0.2 wt.% | photo-deposition | H2 | 175.3 umol h−1 cm−1 | / |
| 42 [42] | Zn0.25Cd0.75S QDs | Ni | Ni-S | 0.15-0.125 at% | co-precipitation | H2 | 18.87 mmol g−1 h−1 | / |
| 43 [41] | coordination polymers | Cu | Cu-N | / | co-precipitation | H2 | 57.64 mmol g−1 h−1 | / |
| 44 [49] | g-C3N4 | Pt | Pt-N | 8.7 wt.% | ion exchange | H2 | 22650 umol g−1 h−1 | 22.5%, 420 nm |
| 45 [79] | CdS | Ni | Ni-O | 2.85 wt.% | photo-deposition | H2 | 630.1 mmol g−1 h−1 | / |
| 46 [80] | TiO2 spheres | Ru | Ru-O | 0.93 wt.% | impregnation | H2 | 7.2 mmol g−1 h−1 | / |
| 47 [35] | g-C3N4 | Pt | Pt-N | / | impregnation | H2 | 14.7 mmol g−1 h−1 | 38.8%,435 nm |
| 48 [81] | CdS | Pt | Pt-S | 0.50 wt.% | impregnation | H2 | 24.17 mmol g−1 h−1 | 46%, 420 nm |
| 49 [82] | TiO2 nanosheet | Co | Co-O | 1.11 wt.% | impregnation | H2 | 1.682 mmol g−1 h−1 | / |
| 50 [83] | g-C3N4 | Pt | Pt-C4 | 1 wt.% | Photo-deposition | H2 | 25.4 umol h−1 | 0.5%, AM 1.5 illumination |
| 51 [84] | Cs2SnI6 | Pt | Pt-I3 | 0.12 wt.% | impregnation | H2 | 430 umol g−1 h−1 | / |
| 52 [85] | unimolecular micelles | Pt | Pt-N, Pt-C-N | 4.1 wt.% | co-precipitation | H2 | 49465 umol gPt−1 h−1 | / |
In particular, benefitting from the high redox potential and work function, Pt in the form of atomically dispersed single atoms on TiO 2, g-C 3N 4, and MOF have manifested excellent performance in photocatalytic H 2 evolution [56][57]. Li et al. reported an efficient photocatalyst (referred as Pt 1/def-TiO 2) by dispersing single Pt atoms on defective TiO 2 [30]. Due to the hydrogen spillover effect, the atomically dispersed Pt atoms caused the formation of surface oxygen vacancies in neighboring TiO 2 units, giving birth to the Pt-O-Ti 3+ atomic interface. According to the experimental results and DFT calculations, the Pt-O-Ti 3+ atomic interface effectively facilitated the transfer of photogenerated electrons from Ti 3+ defective sites to single Pt atoms, thereby enhancing the separation of electron-hole pairs. This unique structure allowed Pt 1/def-TiO 2 to exhibit an exceptionally high turnover frequency of 51,423 h −1 , which was 591 times higher than that of the Pt nanoparticles-supported TiO 2 photocatalyst.
SAPCs with non-noble metals such as Co, Ni, etc. have also been developed as cocatalysts to give excellent performance in photocatalytic water splitting. Importantly, apart from the benefits for H 2 evolution reaction, it has been reported that the cocatalyst of single-atom Co could facilitate the oxidation of water for O 2 evolution half reaction or even overall water splitting [86]. For example, Wei et al. developed a single-atom, Co-based photocatalyst (referred as Co/g-C 3N 4) with single Co atoms confined within phosphide doped-g-C 3N 4 nanosheets. [53] The Co 1-phosphide sites with coordination unsaturated Co sites exhibited oxygen evolution activity by effectively promoting water molecular adsorption and activation, and also helped to suppress electron-hole recombination. Meanwhile the phosphide dopants accelerated the electric conductivity of Co/g-C 3N 4. As a result, in photocatalytic overall water splitting reaction, Co/g-C 3N 4 afforded a H 2 evolution rate as high as 410.3 umol h −1 , with the carrier lifetime over 20 times prolonged compared to the pristine g-C 3N 4.
Summary of SAPCs for photocatalytic water splitting. The order is arranged by the time of publication.
References
- Fujishima, A.; Honda, K. Electrochemical Photolysis of Water at a Semiconductor Electrode. Nature 1972, 238, 37–38.
- Fu, C.F.; Wu, X.; Yang, J. Material Design for Photocatalytic Water Splitting from a Theoretical Perspective. Adv. Mater. 2018, 30, e1802106.
- Jafari, T.; Moharreri, E.; Amin, A.S.; Miao, R.; Song, W.; Suib, S.L. Photocatalytic Water Splitting-The Untamed Dream: A Review of Recent Advances. Molecules 2016, 21, 900.
- Tong, H.; Ouyang, S.; Bi, Y.; Umezawa, N.; Oshikiri, M.; Ye, J. Nano-Photocatalytic Materials: Possibilities and Challenges. Adv. Mater. 2012, 24, 229–251.
- Chen, X.; Shen, S.; Guo, L.; Mao, S.S. Semiconductor-Based Photocatalytic Hydrogen Generation. Chem. Rev. 2010, 110, 6503–6570.
- Maeda, K.; Domen, K. Photocatalytic Water Splitting: Recent Progress and Future Challenges. J. Phys. Chem. Lett. 2010, 1, 2655–2661.
- Wang, Q.; Domen, K. Particulate Photocatalysts for Light-Driven Water Splitting: Mechanisms, Challenges, and Design Strategies. Chem. Rev. 2020, 120, 919–985.
- Bai, S.; Zhang, N.; Gao, C.; Xiong, Y. Defect Engineering in Photocatalytic Materials. Nano Energy 2018, 53, 296–336.
- Wachs, I.E.; Phivilay, S.P.; Roberts, C.A. Reporting of Reactivity for Heterogeneous Photocatalysis. ACS Catal. 2013, 3, 2606–2611.
- Sultan, S.; Tiwari, J.N.; Singh, A.N.; Zhumagali, S.; Ha, M.; Myung, C.W.; Thangavel, P.; Kim, K.S. Single Atoms and Clusters Based Nanomaterials for Hydrogen Evolution, Oxygen Evolution Reactions, and Full Water Splitting. Adv. Energy Mater. 2019, 9, 1900624.
- Mitchell, S.; Perez-Ramirez, J. Single Atom Catalysis: A Decade of Stunning Progress and the Promise for a Bright Future. Nat. Commun. 2020, 11, 4302.
- Wei, X.; Wang, K.-X.; Guo, X.-X.; Chen, J.-S. Single-Site Photocatalysts with a Porous Structure. Proc. R. Soc. A Math. Phys. Eng. Sci. 2012, 468, 2099–2112.
- Yang, X.F.; Wang, A.; Qiao, B.; Li, J.; Liu, J.; Zhang, T. Single-Atom Catalysts: A New Frontier in Heterogeneous Catalysis. Acc. Chem. Res. 2013, 46, 1740–1748.
- Wang, B.; Cai, H.; Shen, S. Single Metal Atom Photocatalysis. Small Methods 2019, 3, 1800447.
- Zhu, Y.; Sokolowski, J.; Song, X.; He, Y.; Mei, Y.; Wu, G. Engineering Local Coordination Environments of Atomically Dispersed and Heteroatom-Coordinated Single Metal Site Electrocatalysts for Clean Energy-Conversion. Adv. Energy Mater. 2019, 10, 1902844.
- Sui, Y.; Liu, S.; Li, T.; Liu, Q.; Jiang, T.; Guo, Y.; Luo, J.-L. Atomically Dispersed Pt on Specific TiO2 Facets for Photocatalytic H2 Evolution. J. Catal. 2017, 353, 250–255.
- Wang, F.; Di Valentin, C.; Pacchioni, G. Doping of WO3 for Photocatalytic Water Splitting: Hints from Density Functional Theory. J. Phys. Chem. C 2012, 116, 8901–8909.
- Wang, J.; Xia, T.; Wang, L.; Zheng, X.; Qi, Z.; Gao, C.; Zhu, J.; Li, Z.; Xu, H.; Xiong, Y. Enabling Visible-Light-Driven Selective CO2 Reduction by Doping Quantum Dots: Trapping Electrons and Suppressing H2 Evolution. Angew. Chem. Int. Ed. Engl. 2018, 57, 16447–16451.
- Xiong, X.; Mao, C.; Yang, Z.; Zhang, Q.; Waterhouse, G.I.N.; Gu, L.; Zhang, T. Photocatalytic CO2 Reduction to CO over Ni Single Atoms Supported on Defect-Rich Zirconia. Adv. Energy Mater. 2020, 10, 2002928.
- Takata, T.; Jiang, J.; Sakata, Y.; Nakabayashi, M.; Shibata, N.; Nandal, V.; Seki, K.; Hisatomi, T.; Domen, K. Photocatalytic Water Splitting with a Quantum Efficiency of Almost Unity. Nature 2020, 581, 411–414.
- Zhao, Z.; Sun, Y.; Dong, F. Graphitic Carbon Nitride Based Nanocomposites: A Review. Nanoscale 2015, 7, 15–37.
- Wen, J.; Xie, J.; Chen, X.; Li, X. A Review on g-C3N4 -Based Photocatalysts. Appl. Surf. Sci. 2017, 391, 72–123.
- Fu, J.; Yu, J.; Jiang, C.; Cheng, B. g-C3N4-Based Heterostructured Photocatalysts. Adv. Energy Mater. 2018, 8, 1701503.
- Wang, X.; Maeda, K.; Thomas, A.; Takanabe, K.; Xin, G.; Carlsson, J.M.; Domen, K.; Antonietti, M. A Metal-Free Polymeric Photocatalyst for Hydrogen Production from Water Under Visible Light. Nat. Mater. 2009, 8, 76–80.
- Chen, Z.; Mitchell, S.; Vorobyeva, E.; Leary, R.K.; Hauert, R.; Furnival, T.; Ramasse, Q.M.; Thomas, J.M.; Midgley, P.A.; Dontsova, D.; et al. Stabilization of Single Metal Atoms on Graphitic Carbon Nitride. Adv. Funct. Mater. 2017, 27, 1605785.
- Li, Y.; Kong, T.; Shen, S. Artificial Photosynthesis with Polymeric Carbon Nitride: When Meeting Metal Nanoparticles, Single Atoms, and Molecular Complexes. Small 2019, 15, e1900772.
- Gao, G.; Jiao, Y.; Waclawik, E.R.; Du, A. Single Atom (Pd/Pt) Supported on Graphitic Carbon Nitride as an Efficient Photocatalyst for Visible-Light Reduction of Carbon Dioxide. J. Am. Chem. Soc. 2016, 138, 6292–6297.
- Miao, X.; Qu, D.; Yang, D.; Nie, B.; Zhao, Y.; Fan, H.; Sun, Z. Synthesis of Carbon Dots with Multiple Color Emission by Controlled Graphitization and Surface Functionalization. Adv. Mater. 2018, 30, 1704740.
- Wang, Q.; Li, J.; Tu, X.; Liu, H.; Shu, M.; Si, R.; Ferguson, C.T.J.; Zhang, K.A.I.; Li, R. Single Atomically Anchored Cobalt on Carbon Quantum Dots as Efficient Photocatalysts for Visible Light-Promoted Oxidation Reactions. Chem. Mater. 2019, 32, 734–743.
- Chen, Y.; Ji, S.; Sun, W.; Lei, Y.; Wang, Q.; Li, A.; Chen, W.; Zhou, G.; Zhang, Z.; Wang, Y.; et al. Engineering the Atomic Interface with Single Platinum Atoms for Enhanced Photocatalytic Hydrogen Production. Angew. Chem. Int. Ed. Engl. 2020, 59, 1295–1301.
- Fang, X.; Shang, Q.; Wang, Y.; Jiao, L.; Yao, T.; Li, Y.; Zhang, Q.; Luo, Y.; Jiang, H.L. Single Pt Atoms Confined into a Metal-Organic Framework for Efficient Photocatalysis. Adv. Mater. 2018, 30, 1705112.
- Ou, M.; Wan, S.; Zhong, Q.; Zhang, S.; Wang, Y. Single Pt Atoms Deposition on g-C3N4 Nanosheets for Photocatalytic H2 Evolution or NO Oxidation under Visible Light. Int. J. Hydrog. Energy 2017, 42, 27043–27054.
- Li, J.; Huang, H.; Liu, P.; Song, X.; Mei, D.; Tang, Y.; Wang, X.; Zhong, C. Metal-Organic Framework Encapsulated Single-Atom Pt Catalysts for Efficient Photocatalytic Hydrogen Evolution. J. Catal. 2019, 375, 351–360.
- Li, X.; Bi, W.; Zhang, L.; Tao, S.; Chu, W.; Zhang, Q.; Luo, Y.; Wu, C.; Xie, Y. Single-Atom Pt as Co-Catalyst for Enhanced Photocatalytic H2 Evolution. Adv. Mater. 2016, 28, 2427–2431.
- Zhang, L.; Long, R.; Zhang, Y.; Duan, D.; Xiong, Y.; Zhang, Y.; Bi, Y. Direct Observation of Dynamic Bond Evolution in Single-Atom Pt/C3N4 Catalysts. Angew. Chem. Int. Ed. Engl. 2020, 59, 6224–6229.
- Zuo, Q.; Liu, T.; Chen, C.; Ji, Y.; Gong, X.; Mai, Y.; Zhou, Y. Ultrathin Metal-Organic Framework Nanosheets with Ultrahigh Loading of Single Pt Atoms for Efficient Visible-Light-Driven Photocatalytic H2 Evolution. Angew. Chem. Int. Ed. Engl. 2019, 58, 10198–10203.
- Jeantelot, G.; Qureshi, M.; Harb, M.; Ould-Chikh, S.; Anjum, D.H.; Abou-Hamad, E.; Aguilar-Tapia, A.; Hazemann, J.L.; Takanabe, K.; Basset, J.M. TiO2-Supported Pt Single Atoms by Surface Organometallic Chemistry for Photocatalytic Hydrogen Evolution. Phys. Chem. Chem. Phys. 2019, 21, 24429–24440.
- Yang, H.; Shang, L.; Zhang, Q.; Shi, R.; Waterhouse, G.I.N.; Gu, L.; Zhang, T. A Universal Ligand Mediated Method for Large Scale Synthesis of Transition Metal Single Atom Catalysts. Nat. Commun. 2019, 10, 4585.
- Ji, S.; Chen, Y.; Wang, X.; Zhang, Z.; Wang, D.; Li, Y. Chemical Synthesis of Single Atomic Site Catalysts. Chem. Rev. 2020, 120, 11900–11955.
- Xia, T.; Long, R.; Gao, C.; Xiong, Y. Design of Atomically Dispersed Catalytic Sites for Photocatalytic CO2 Reduction. Nanoscale 2019, 11, 11064–11070.
- Zang, Y.; Zhang, J.; Wang, R.; Wang, Z.D.; Zhu, Y.; Ren, X.; Li, S.; Dong, X.Y.; Zang, S.Q. Inter-Chain Double-Site Synergistic Photocatalytic Hydrogen Evolution in Robust Cuprous Coordination Polymers. Chem. Commun. 2020, 56, 6261–6264.
- Su, D.W.; Ran, J.; Zhuang, Z.W.; Chen, C.; Qiao, S.Z.; Li, Y.D.; Wang, G.X. Atomically Dispersed Ni in Cadmium-Zinc Sulfide Quantum Dots for High-Performance Visible-Light Photocatalytic Hydrogen Production. Sci. Adv. 2020, 6, eaaz8447.
- Wang, F.; Wang, Y.; Li, Y.; Cui, X.; Zhang, Q.; Xie, Z.; Liu, H.; Feng, Y.; Lv, W.; Liu, G. The Facile Synthesis of a Single Atom-Dispersed Silver-Modified Ultrathin g-C3N4 Hybrid for the Enhanced Visible-Light Photocatalytic Degradation of Sulfamethazine With Peroxymonosulfate. Dalton Trans. 2018, 47, 6924–6933.
- Guo, X.-W.; Chen, S.-M.; Wang, H.-J.; Zhang, Z.-M.; Lin, H.; Song, L.; Lu, T.-B. Single-Atom Molybdenum Immobilized on Photoactive Carbon Nitride as Efficient Photocatalysts for Ambient Nitrogen Fixation in Pure Water. J. Mater. Chem. A 2019, 7, 19831–19837.
- Du, X.L.; Wang, X.L.; Li, Y.H.; Wang, Y.L.; Zhao, J.J.; Fang, L.J.; Zheng, L.R.; Tong, H.; Yang, H.G. Isolation of Single Pt Atoms in a Silver Cluster: Forming Highly Efficient Silver-Based Cocatalysts for Photocatalytic Hydrogen Evolution. Chem. Commun. 2017, 53, 9402–9405.
- He, T.; Chen, S.; Ni, B.; Gong, Y.; Wu, Z.; Song, L.; Gu, L.; Hu, W.; Wang, X. Zirconium-Porphyrin-Based Metal-Organic Framework Hollow Nanotubes for Immobilization of Noble-Metal Single Atoms. Angew. Chem. Int. Ed. Engl. 2018, 57, 3493–3498.
- Zhou, P.; Lv, F.; Li, N.; Zhang, Y.; Mu, Z.; Tang, Y.; Lai, J.; Chao, Y.; Luo, M.; Lin, F.; et al. Strengthening Reactive Metal-Support Interaction to Stabilize High-Density Pt Single Atoms on Electron-Deficient g-C3N4 for Boosting Photocatalytic H2 Production. Nano Energy 2019, 56, 127–137.
- Cheng, N.; Stambula, S.; Wang, D.; Banis, M.N.; Liu, J.; Riese, A.; Xiao, B.; Li, R.; Sham, T.K.; Liu, L.M.; et al. Platinum Single-Atom and Cluster Catalysis of the Hydrogen Evolution Reaction. Nat. Commun. 2016, 7, 13638.
- Zeng, Z.; Su, Y.; Quan, X.; Choi, W.; Zhang, G.; Liu, N.; Kim, B.; Chen, S.; Yu, H.; Zhang, S. Single-Atom Platinum Confined by the Interlayer Nanospace of Carbon Nitride for Efficient Photocatalytic Hydrogen Evolution. Nano Energy 2020, 69, 104409.
- Dong, S.; Li, B.; Cui, X.; Tan, S.; Wang, B. Photoresponses of Supported Au Single Atoms on TiO2(110) through the Metal-Induced Gap States. J. Phys. Chem. Lett. 2019, 10, 4683–4691.
- Li, Y.; Wang, Z.; Xia, T.; Ju, H.; Zhang, K.; Long, R.; Xu, Q.; Wang, C.; Song, L.; Zhu, J.; et al. Implementing Metal-to-Ligand Charge Transfer in Organic Semiconductor for Improved Visible-Near-Infrared Photocatalysis. Adv. Mater. 2016, 28, 6959–6965.
- Huang, P.; Huang, J.; Pantovich, S.A.; Carl, A.D.; Fenton, T.G.; Caputo, C.A.; Grimm, R.L.; Frenkel, A.I.; Li, G. Selective CO2 Reduction Catalyzed by Single Cobalt Sites on Carbon Nitride under Visible-Light Irradiation. J. Am. Chem. Soc. 2018, 140, 16042–16047.
- Liu, W.; Cao, L.; Cheng, W.; Cao, Y.; Liu, X.; Zhang, W.; Mou, X.; Jin, L.; Zheng, X.; Che, W.; et al. Single-Site Active Cobalt-Based Photocatalyst with a Long Carrier Lifetime for Spontaneous Overall Water Splitting. Angew. Chem. Int. Ed. Engl. 2017, 56, 9312–9317.
- Zhao, Q.; Yao, W.; Huang, C.; Wu, Q.; Xu, Q. Effective and Durable Co Single Atomic Cocatalysts for Photocatalytic Hydrogen Production. ACS Appl. Mater. Interfaces 2017, 9, 42734–42741.
- Cao, S.; Li, H.; Tong, T.; Chen, H.-C.; Yu, A.; Yu, J.; Chen, H.M. Single-Atom Engineering of Directional Charge Transfer Channels and Active Sites for Photocatalytic Hydrogen Evolution. Adv. Funct. Mater. 2018, 28, 1802169.
- Cao, Y.; Wang, D.; Lin, Y.; Liu, W.; Cao, L.; Liu, X.; Zhang, W.; Mou, X.; Fang, S.; Shen, X.; et al. Single Pt Atom with Highly Vacant d-Orbital for Accelerating Photocatalytic H2 Evolution. ACS Appl. Energ. Mater. 2018, 1, 6082–6088.
- Su, H.; Che, W.; Tang, F.; Cheng, W.; Zhao, X.; Zhang, H.; Liu, Q. Valence Band Engineering via PtII Single-Atom Confinement Realizing Photocatalytic Water Splitting. J. Phys. Chem. C 2018, 122, 21108–21114.
- Tao, J.; Luttrell, T.; Batzill, M. A Two-Dimensional Phase of TiO(2) with a Reduced Bandgap. Nat. Chem. 2011, 3, 296–300.
- Zhou, P.; Zhang, Q.; Chao, Y.; Wang, L.; Li, Y.; Chen, H.; Gu, L.; Guo, S. Partially Reduced Pd Single Atoms on CdS Nanorods Enable Photocatalytic Reforming of Ethanol into High Value-Added Multicarbon Compound. Chem 2021, 7, 1033–1049.
- Xue, Y.; Lei, Y.; Liu, X.; Li, Y.; Deng, W.; Wang, F.; Min, S. Highly Active Dye-Sensitized Photocatalytic H2 Evolution Catalyzed by a Single-Atom Pt Cocatalyst Anchored Onto g-C3N4 Nanosheets under Long-Wavelength Visible Light Irradiation. New, J. Chem. 2018, 42, 14083–14086.
- Cao, Y.; Chen, S.; Luo, Q.; Yan, H.; Lin, Y.; Liu, W.; Cao, L.; Lu, J.; Yang, J.; Yao, T.; et al. Atomic-Level Insight into Optimizing the Hydrogen Evolution Pathway over a Co1-N4 Single-Site Photocatalyst. Angew. Chem. Int. Ed. Engl. 2017, 56, 12191–12196.
- Yi, L.; Lan, F.; Li, J.; Zhao, C. Efficient Noble-Metal-Free Co-NG/TiO2 Photocatalyst for H2 Evolution: Synergistic Effect between Single-Atom Co and N-Doped Graphene for Enhanced Photocatalytic Activity. ACS Sustain. Chem. Eng. 2018, 6, 12766–12775.
- Zhao, Q.; Sun, J.; Li, S.; Huang, C.; Yao, W.; Chen, W.; Zeng, T.; Wu, Q.; Xu, Q. Single Nickel Atoms Anchored on Nitrogen-Doped Graphene as a Highly Active Cocatalyst for Photocatalytic H2 Evolution. ACS Catal. 2018, 8, 11863–11874.
- Lee, B.H.; Park, S.; Kim, M.; Sinha, A.K.; Lee, S.C.; Jung, E.; Chang, W.J.; Lee, K.S.; Kim, J.H.; Cho, S.P.; et al. Reversible and Cooperative Photoactivation of Single-Atom Cu/TiO2 Photocatalysts. Nat. Mater. 2019, 18, 620–626.
- Wu, X.; Zhang, H.; Dong, J.; Qiu, M.; Kong, J.; Zhang, Y.; Li, Y.; Xu, G.; Zhang, J.; Ye, J. Surface Step Decoration of Isolated Atom as Electron Pumping: Atomic-Level Insights into Visible-Light Hydrogen Evolution. Nano Energy 2018, 45, 109–117.
- Liu, L.; Wu, X.; Wang, L.; Xu, X.; Gan, L.; Si, Z.; Li, J.; Zhang, Q.; Liu, Y.; Zhao, Y.; et al. Atomic Palladium on Graphitic Carbon Nitride as a Hydrogen Evolution Catalyst under Visible Light Irradiation. Comm. Chem. 2019, 2, 1–8.
- Qiu, S.; Shen, Y.; Wei, G.; Yao, S.; Xi, W.; Shu, M.; Si, R.; Zhang, M.; Zhu, J.; An, C. Carbon Dots Decorated Ultrathin CdS Nanosheets Enabling In-Situ Anchored Pt Single Atoms: A Highly Efficient Solar-Driven Photocatalyst for Hydrogen Evolution. Appl. Catal. B 2019, 259, 118036.
- Ran, J.; Zhang, H.; Qu, J.; Xia, B.; Zhang, X.; Chen, S.; Song, L.; Jing, L.; Zheng, R.; Qiao, S.Z. Atomically Dispersed Single Co Sites in Zeolitic Imidazole Frameworks Promoting High-Efficiency Visible-Light-Driven Hydrogen Production. Chem.-Eur. J. 2019, 25, 9670–9677.
- Su, H.; Liu, M.; Cheng, W.; Zhao, X.; Hu, F.; Liu, Q. Heterogeneous Single-Site Synergetic Catalysis for Spontaneous Photocatalytic Overall Water Splitting. J. Mater. Chem. A 2019, 7, 11170–11176.
- Zeng, L.; Dai, C.; Liu, B.; Xue, C. Oxygen-Assisted Stabilization of Single-Atom Au During Photocatalytic Hydrogen Evolution. J. Mater. Chem. A 2019, 7, 24217–24221.
- Zhang, W.; Peng, Q.; Shi, L.; Yao, Q.; Wang, X.; Yu, A.; Chen, Z.; Fu, Y. Merging Single-Atom-Dispersed Iron and Graphitic Carbon Nitride to a Joint Electronic System for High-Efficiency Photocatalytic Hydrogen Evolution. Small 2019, 15, e1905166.
- Cai, S.; Wang, L.; Heng, S.; Li, H.; Bai, Y.; Dang, D.; Wang, Q.; Zhang, P.; He, C. Interaction of Single-Atom Platinum–Oxygen Vacancy Defects for the Boosted Photosplitting Water H2 Evolution and CO2 Photoreduction: Experimental and Theoretical Study. J. Phys. Chem. C 2020, 124, 24566–24579.
- Chen, Z.; Bu, Y.; Wang, L.; Wang, X.; Ao, J.-P. Single-Sites Rh-Phosphide Modified Carbon Nitride Photocatalyst for Boosting Hydrogen Evolution under Visible Light. Appl. Catal. B 2020, 274, 119117.
- Chu, C.; Zhu, Q.; Pan, Z.; Gupta, S.; Huang, D.; Du, Y.; Weon, S.; Wu, Y.; Muhich, C.; Stavitski, E.; et al. Spatially Separating Redox Centers on 2D Carbon Nitride with Cobalt Single Atom for Photocatalytic H2O2 Production. Pans 2020, 117, 6376–6382.
- Hejazi, S.; Mohajernia, S.; Osuagwu, B.; Zoppellaro, G.; Andryskova, P.; Tomanec, O.; Kment, S.; Zboril, R.; Schmuki, P. On the Controlled Loading of Single Platinum Atoms as a Co-Catalyst on TiO2 Anatase for Optimized Photocatalytic H2 Generation. Adv. Mater. 2020, 32, e1908505.
- Jiang, X.H.; Zhang, L.S.; Liu, H.Y.; Wu, D.S.; Wu, F.Y.; Tian, L.; Liu, L.L.; Zou, J.P.; Luo, S.L.; Chen, B.B. Silver Single Atom in Carbon Nitride Catalyst for Highly Efficient Photocatalytic Hydrogen Evolution. Angew. Chem. Int. Ed. Engl. 2020, 59, 23112–23116.
- Jin, X.; Wang, R.; Zhang, L.; Si, R.; Shen, M.; Wang, M.; Tian, J.; Shi, J. Electron Configuration Modulation of Nickel Single Atoms for Elevated Photocatalytic Hydrogen Evolution. Angew. Chem. Int. Ed. Engl. 2020, 59, 6827–6831.
- Luo, H.; Liu, Y.; Dimitrov, S.D.; Steier, L.; Guo, S.; Li, X.; Feng, J.; Xie, F.; Fang, Y.; Sapelkin, A.; et al. Pt Single-Atoms Supported on Nitrogen-Doped Carbon Dots for Highly Efficient Photocatalytic Hydrogen Generation. J. Mater. Chem. A 2020, 8, 14690–14696.
- Zhang, J.; Xu, X.; Yang, L.; Cheng, D.; Cao, D. Single-Atom Ru Doping Induced Phase Transition of MoS2 and S Vacancy for Hydrogen Evolution Reaction. Small Methods 2019, 3, 1900653.
- Zhang, H.; Zuo, S.; Qiu, M.; Wang, S.; Zhang, Y.; Zhang, J.; Lou, X.W.D. Direct Probing of Atomically Dispersed Ru Species Over Multi-Edged TiO2 for Highly Efficient Photocatalytic Hydrogen Evolution. Sci. Adv. 2020, 6, eabb9823.
- Feng, R.; Wan, K.; Sui, X.; Zhao, N.; Li, H.; Lei, W.; Yu, J.; Liu, X.; Shi, X.; Zhai, M.; et al. Anchoring Single Pt Atoms and Black Phosphorene Dual Co-Catalysts on CdS Nanospheres to Boost Visible-Light Photocatalytic H2 Evolution. Nano Today 2021, 37, 101080.
- Wu, X.; Zuo, S.; Qiu, M.; Li, Y.; Zhang, Y.; An, P.; Zhang, J.; Zhang, H.; Zhang, J. Atomically Defined Co on Two-Dimensional TiO2 Nanosheet for Photocatalytic Hydrogen Evolution. Chem. Eng. J 2021, 420, 127681.
- Yan, J.; Ji, Y.; Batmunkh, M.; An, P.; Zhang, J.; Fu, Y.; Jia, B.; Li, Y.; Liu, S.; Ye, J.; et al. Breaking Platinum Nanoparticles to Single-Atomic Pt-C4 Co-catalysts for Enhanced Solar-to-Hydrogen Conversion. Angew. Chem. Int. Ed. Engl. 2021, 60, 2541–2547.
- Zhou, P.; Chen, H.; Chao, Y.; Zhang, Q.; Zhang, W.; Lv, F.; Gu, L.; Zhao, Q.; Wang, N.; Wang, J.; et al. Single-Atom Pt-I3 Sites on All-Inorganic Cs2SnI6 Perovskite for Efficient Photocatalytic Hydrogen Production. Nat. Commun. 2021, 12, 4412.
- Zuo, Q.; Feng, K.; Zhong, J.; Mai, Y.; Zhou, Y. Single-Metal-Atom Polymeric Unimolecular Micelles for Switchable Photocatalytic H2 Evolution. CCS Chemistry 2021, 3, 1963–1971.
- Zhang, G.; Huang, C.; Wang, X. Dispersing Molecular Cobalt in Graphitic Carbon Nitride Frameworks for Photocatalytic Water Oxidation. Small 2015, 11, 1215–1221.





