
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ahmad Ilyas Rushdan | + 2698 word(s) | 2698 | 2021-10-25 05:37:11 | | | |
| 2 | Abdoulhdi A. Borhana Omran | -5 word(s) | 2693 | 2022-10-20 08:04:46 | | |
Video Upload Options
Wheat is a grass plant of the Poaceae plants family; the scientific name of wheat plant is Triticum. Due to its mechanical and physical properties, wheat starch, gluten, and fiber are vital in the biopolymer industry. Glycerol as a plasticizer considerably increased the elongation and water vapor permeability of wheat films. Wheat fiber developed mechanical and thermal properties as a result of various matrices; wheat gluten is water insoluble, elastic, non-toxic, and biodegradable, making it useful in biocomposite materials.
1. Introduction
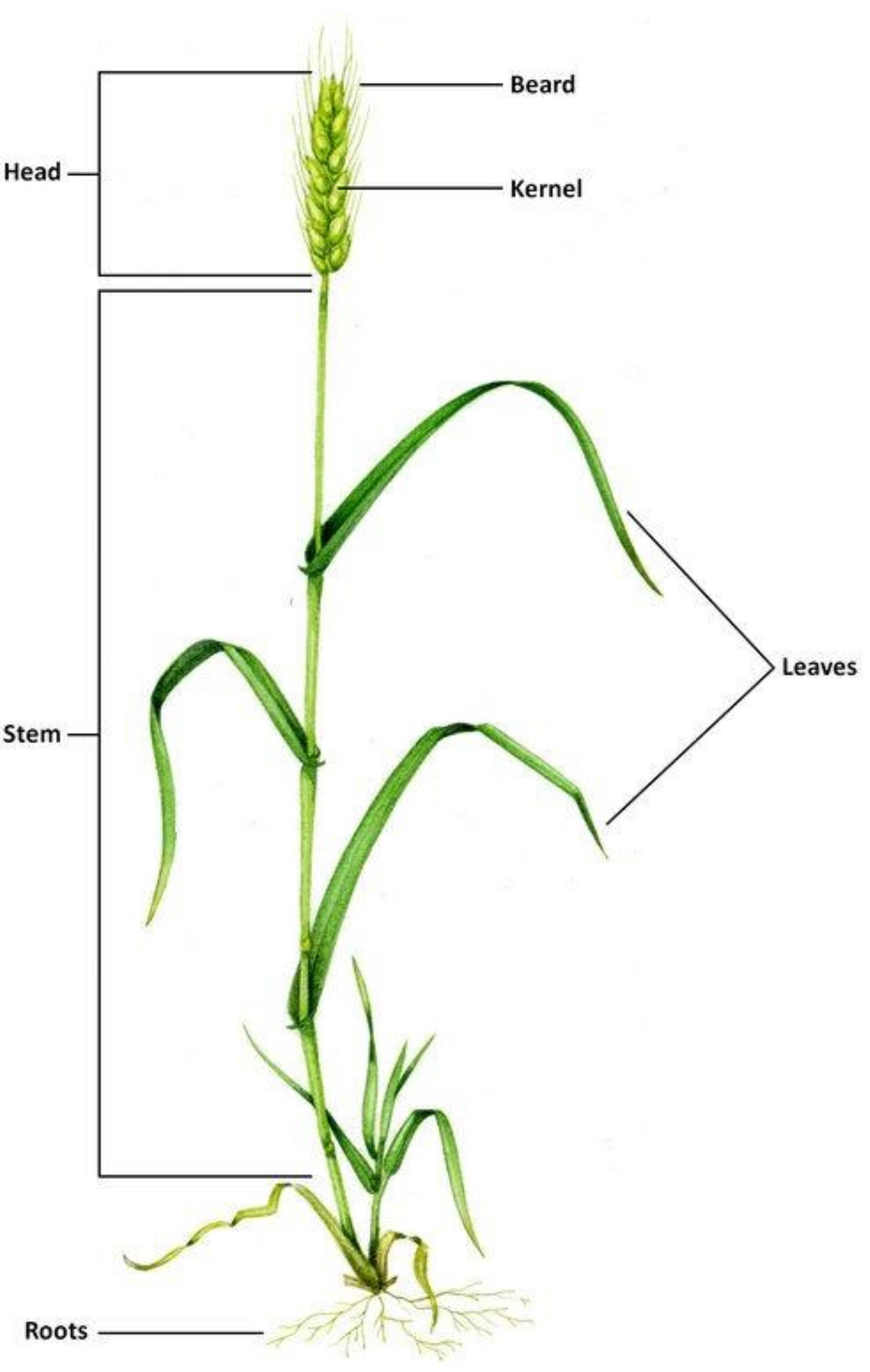
2. Wheat Plant
3. Film Preparation and Properties Characterization of Wheat Starch Based Films
3.1. Physical and Chemical Properties of Wheat Starch
|
Type of Starch |
||||
|---|---|---|---|---|
|
Parameter |
Wheat Starch |
Corn Starch |
Rice Starch |
Potato Starch |
|
Amylose (%) |
16.0–31.5 |
20.0–28 |
20–28 |
25–31 |
|
Amylopectin (%) |
68.5–75 |
75–83 |
65–85 |
76–83 |
|
Ash (%) |
0.20–0.29 |
0.32–0.62 |
0.17–0.19 |
15.95–16.05 |
|
Proteins (%) |
0.40–0.46 |
0.38–7.7 |
0.33–0.38 |
4.26–4.82 |
|
Density (g/cm3) |
1.5 |
1.356–1.4029 |
1.282 |
0.763 |
|
Moisture content (%) |
10.65–13.3 |
10.45–10.82 |
3.60 |
15.98 ± 0.36 |
3.2. Production of Wheat Starch Based Films
4. Wheat Gluten-Based Film; Preparation and Characterization
4.1. Production of Wheat Gluten-Based Film
4.1.1. Wet Method
4.1.2. Dry Method
4.2. Properties Characterization of Films Based Wheat Gluten
5. Antioxidant Properties of Wheat Based Film
6. Antimicrobial Properties of Wheat Based Film
References
- Omran, A.A.B.; Mohammed, A.A.B.A.; Sapuan, S.M.; Ilyas, R.A.; Asyraf, M.R.M.; Koloor, S.S.R.; Petrů, M. Micro- and Nanocellulose in Polymer Composite Materials: A Review. Polymers 2021, 13, 231.
- Nurazzi, N.M.; Asyraf, M.R.M.; Khalina, A.; Abdullah, N.; Aisyah, H.A.; Rafiqah, S.A.; Sabaruddin, F.A.; Kamarudin, S.H.; Norrrahim, M.N.F.; Ilyas, R.A.; et al. A Review on Natural Fiber Reinforced Polymer Composite for Bullet Proof and Ballistic Applications. Polymers 2021, 13, 646.
- Alsubari, S.; Zuhri, M.Y.M.; Sapuan, S.M.; Ishak, M.R.; Ilyas, R.A.; Asyraf, M.R.M. Potential of natural fiber reinforced polymer composites in sandwich structures: A review on its mechanical properties. Polymers 2021, 13, 423.
- Diyana, Z.N.; Jumaidin, R.; Selamat, M.Z.; Ghazali, I.; Julmohammad, N.; Huda, N.; Ilyas, R.A. Physical Properties of Thermoplastic Starch Derived from Natural Resources and Its Blends: A Review. Polymers 2021, 13, 1396.
- Mohd Nurazzi, N.; Asyraf, M.R.M.; Khalina, A.; Abdullah, N.; Sabaruddin, F.A.; Kamarudin, S.H.; Ahmad, S.; Mahat, A.M.; Lee, C.L.; Aisyah, H.A.; et al. Fabrication, Functionalization, and Application of Carbon Nanotube-Reinforced Polymer Composite: An Overview. Polymers 2021, 13, 1047.
- Ilyas, R.A.; Sapuan, S.M.; Harussani, M.M.; Hakimi, M.Y.A.Y.; Haziq, M.Z.M.; Atikah, M.S.N.; Asyraf, M.R.M.; Ishak, M.R.; Razman, M.R.; Nurazzi, N.M.; et al. Polylactic Acid (PLA) Biocomposite: Processing, Additive Manufacturing and Advanced Applications. Polymers 2021, 13, 1326.
- Supian, A.B.M.; Sapuan, S.M.; Jawaid, M.; Zuhri, M.Y.M.; Ilyas, R.A.; Syamsir, A. Crashworthiness Response of Filament Wound Kenaf/Glass Fibre-reinforced Epoxy Composite Tubes with Influence of Stacking Sequence under Intermediate-velocity Impact Load. Fibers Polym. 2021, 1–12.
- Simon, F.; Loussert-Ajaka, I.; Damond, F.; Saragosti, S.; Barin, F.; Brun-Vézinet, F. A REVIEW ON NATURAL FIBERS. AIDS Res. Hum. Retrovir. 1996, 12, 1427–1433.
- Nam, S.; Netravali, A.N. Green composites. I. physical properties of ramie fibers for environment-friendly green composites. Fibers Polym. 2006, 7, 372–379.
- Ramesh, M.; Palanikumar, K.; Reddy, K.H. Plant fibre based bio-composites: Sustainable and renewable green materials. Renew. Sustain. Energy Rev. 2017, 79, 558–584.
- Shevkani, K.; Singh, N.; Bajaj, R.; Kaur, A. Wheat starch production, structure, functionality and applications—a review. Int. J. Food Sci. Technol. 2017, 52, 38–58.
- Liu, W.; Mohanty, A.K.; Askeland, P.; Drzal, L.T.; Misra, M. Influence of fiber surface treatment on properties of Indian grass fiber reinforced soy protein based biocomposites. Polymer 2004, 45, 7589–7596.
- Bledzki, A.K.; Gassan, J. Composites reinforced with cellulose based fibers. Prog. Polym. Sci. 1999, 24, 221–274.
- Norrrahim, M.N.F.; Huzaifah, M.R.M.; Farid, M.A.A.; Shazleen, S.S.; Misenan, M.S.M.; Yasim-Anuar, T.A.T.; Naveen, J.; Nurazzi, N.M.; Rani, M.S.A.; Hakimi, M.I.; et al. Greener Pretreatment Approaches for the Valorisation of Natural Fibre Biomass into Bioproducts. Polymers 2021, 13, 2971.
- Nurazzi, N.M.; Sabaruddin, F.A.; Harussani, M.M.; Kamarudin, S.H.; Rayung, M.; Asyraf, M.R.M.; Aisyah, H.A.; Norrrahim, M.N.F.; Ilyas, R.A.; Abdullah, N.; et al. Mechanical Performance and Applications of CNTs Reinforced Polymer Composites—A Review. Nanomaterials 2021, 11, 2186.
- Chan, J.X.; Wong, J.F.; Petrů, M.; Hassan, A.; Nirmal, U.; Othman, N.; Ilyas, R.A. Effect of Nanofillers on Tribological Properties of Polymer Nanocomposites: A Review on Recent Development. Polymers 2021, 13, 2867.
- Sreenivasan, S.; Iyer, P.B.; Iyer, K.R.K. Influence of delignification and alkali treatment on the fine structure of coir fibres (Cocos Nucifera). J. Mater. Sci. 1996, 31, 721–726.
- Gassan, J.; Bledzki, A.K. Alkali Treatment of Jute Fibers: Relationship Between. J. Appl. Polym. Sci. 1998, 71, 623–629.
- Janker-Obermeier, I.; Sieber, V.; Faulstich, M.; Schieder, D. Solubilization of hemicellulose and lignin from wheat straw through microwave-assisted alkali treatment. Ind. Crops Prod. 2012, 39, 198–203.
- Ilyas, R.A.; Sapuan, S.M.; Atikah, M.S.N.; Asyraf, M.R.M.; Rafiqah, S.A.; Aisyah, H.A.; Nurazzi, N.M.; Norrrahim, M.N.F. Effect of hydrolysis time on the morphological, physical, chemical, and thermal behavior of sugar palm nanocrystalline cellulose ( Arenga pinnata (Wurmb.) Merr ). Text. Res. J. 2021, 91, 152–167.
- Ilyas, R.A.; Sapuan, S.M.; Ishak, M.R.; Zainudin, E.S. Sugar palm nanofibrillated cellulose (Arenga pinnata (Wurmb.) Merr): Effect of cycles on their yield, physic-chemical, morphological and thermal behavior. Int. J. Biol. Macromol. 2019, 123, 379–388.
- Ahmad Ilyas, R.; Mohd Sapuan, S.; Ibrahim, R.; Abral, H.; Ishak, M.R.; Zainudin, E.S.; Asrofi, M.; Siti Nur Atikah, M.; Muhammad Huzaifah, M.R.; Radzi, M.A.; et al. Sugar palm (Arenga pinnata (Wurmb.) Merr)cellulosic fibre hierarchy: A comprehensiveapproach from macro to nano scale. J. Mater. Res. Technol. 2019, 8, 2753–2766.
- Zhao, L.; Xia, W.; Tarverdi, K.; Song, J. Biocomposite boards from wheat straw without addition of bonding agent. Mater. Sci. Technol. 2014, 30, 603–610.
- USDA. World agricultural production. Ekon. APK 2021. Available online: https://www.fas.usda.gov/data/world-agricultural-production (accessed on 14 August 2021).
- Dicharry, R.M.; Ye, P.; Saha, G.; Waxman, E.; Asandei, A.D.; Parnas, R.S. Wheat gluten-thiolated poly(vinyl alcohol) blends with improved mechanical properties. Biomacromolecules 2006, 7, 2837–2844.
- Yang, Z.; Peng, H.; Wang, W.; Liu, T. Crystallization behavior of poly(ε-caprolactone)/layered double hydroxide nanocomposites. J. Appl. Polym. Sci. 2010, 116, 2658–2667.
- Gällstedt, M.; Mattozzi, A.; Johansson, E.; Hedenqvist, M.S. Transport and Tensile Properties of Compression-Molded Wheat Gluten Films. Biomacromolecules 2004, 5, 2020–2028.
- Kunanopparat, T.; Menut, P.; Morel, M.-H.; Guilbert, S. Reinforcement of plasticized wheat gluten with natural fibers: From mechanical improvement to deplasticizing effect. Compos. Part A Appl. Sci. Manuf. 2008, 39, 777–785.
- Zárate-Ramírez, L.S.; Martínez, I.; Romero, A.; Partal, P.; Guerrero, A. Wheat gluten-based materials plasticised with glycerol and water by thermoplastic mixing and thermomoulding. J. Sci. Food Agric. 2011, 91, 625–633.
- Domenek, S.; Feuilloley, P.; Gratraud, J.; Morel, M.-H.; Guilbert, S. Biodegradability of wheat gluten based bioplastics. Chemosphere 2004, 54, 551–559.
- Edwin, A.; Habeych, N. Development of Starch-Based Materials; Wageningen University: Wageningen, The Netherlands, 2009; ISBN 9789085854333.
- Woerdeman, D.L.; Veraverbeke, W.S.; Parnas, R.S.; Johnson, D.; Delcour, J.A.; Verpoest, I.; Plummer, C.J.G. Designing New Materials from Wheat Protein. Biomacromolecules 2004, 5, 1262–1269.
- Lagrain, B.; Goderis, B.; Brijs, K.; Delcour, J.A. Molecular Basis of Processing Wheat Gluten toward Biobased Materials. Biomacromolecules 2010, 11, 533–541.
- Jansens, K.J.A.; Vo, N.; Telen, L.; Brijs, K.; Lagrain, B.; Willem, A.; Vuure, V.; Van Acker, K.; Verpoest, I.; Van Puyvelde, P.; et al. Effect of molding conditions and moisture content on the mechanical properties of compression molded glassy, wheat gluten bioplastics. Ind. Crops Prod. 2013, 44, 480–487.
- Thakur, R.; Pristijono, P.; Scarlett, C.J.; Bowyer, M.; Singh, S.P.; Vuong, Q.V. Starch-based films: Major factors affecting their properties. Int. J. Biol. Macromol. 2019, 132, 1079–1089.
- Punia Bangar, S.; Nehra, M.; Siroha, A.K.; Petrů, M.; Ilyas, R.A.; Devi, U.; Devi, P. Development and Characterization of Physical Modified Pearl Millet Starch-Based Films. Foods 2021, 10, 1609.
- Kumari, N.; Bangar, S.P.; Petrů, M.; Ilyas, R.A.; Singh, A.; Kumar, P. Development and Characterization of Fenugreek Protein-Based Edible Film. Foods 2021, 10, 1976.
- Isotton, F.S.; Bernardo, G.L.; Baldasso, C.; Rosa, L.M.; Zeni, M. The plasticizer effect on preparation and properties of etherified corn starchs films. Ind. Crops Prod. 2015, 76, 717–724.
- Ibrahim, M.I.J.; Sapuan, S.M.; Zainudin, E.S.; Zuhri, M.Y.M. Physical, thermal, morphological, and tensile properties of cornstarch-based films as affected by different plasticizers. Int. J. Food Prop. 2019, 22, 925–941.
- Ibrahim, M.I.J.; Sapuan, S.M.; Zainudin, E.S.; Zuhri, M.Y.M. Preparation and characterization of cornhusk/sugar palm fiber reinforced Cornstarch-based hybrid composites. J. Mater. Res. Technol. 2020, 9, 200–211.
- Sanyang, M.; Sapuan, S.; Jawaid, M.; Ishak, M.; Sahari, J. Effect of Plasticizer Type and Concentration on Tensile, Thermal and Barrier Properties of Biodegradable Films Based on Sugar Palm (Arenga pinnata) Starch. Polymers 2015, 7, 1106.
- Edhirej, A.; Sapuan, S.M.; Jawaid, M.; Zahari, N.I. Effect of various plasticizers and concentration on the physical, thermal, mechanical, and structural properties of cassava-starch-based films. Starch/Staerke 2017, 69, 1–11.
- Laohakunjit, N.; Noomhorm, A. Effect of Plasticizers on Mechanical and Barrier Properties of Rice Starch Film. Starch 2004, 56, 348–356.
- Hong-rui, C.; Hai-tao, C.; Shuang, L.; Guo-qiang, D.; Ying, Z. ScienceDirect Effect of Plasticizers on Properties of Rice Straw Fiber Film. J. Northeast Agric. Univ. 2014, 21, 67–72.
- Wheat | Production, Types, Nutrition, Uses, & Facts | Britannica. Available online: https://www.britannica.com/plant/wheat (accessed on 14 August 2021).
- A Kernel of Wheat | National Festival of Breads. Available online: https://nationalfestivalofbreads.com/nutrition-education/a-kernel-of-wheat (accessed on 14 August 2021).
- The Parts of a Wheat Plant. Available online: https://sciencing.com/the-parts-of-a-wheat-plant-12211988.html (accessed on 14 August 2021).
- Wheat — Louisiana Ag in the Classroom, Reproduced with the permission of Louisiana Agriculture in the Classroom. Available online: https://aitcla.org/wheat (accessed on 14 August 2021).
- Zhang, W.; Gu, J.; Wang, Z.; Wei, C.; Yang, J.; Zhang, J. Comparison of Structural and Functional Properties of Wheat Starch under Different Soil Drought Conditions. Sci. Rep. 2017, 7, 1–18.
- Jha, P.; Dharmalingam, K.; Nishizu, T.; Katsuno, N.; Anandalakshmi, R. Effect of Amylose–Amylopectin Ratios on Physical, Mechanical, and Thermal Properties of Starch-Based Bionanocomposite Films Incorporated with CMC and Nanoclay. Starch/Staerke 2020, 72, 1–9.
- Jiang, T.; Duan, Q.; Zhu, J.; Liu, H.; Yu, L. Starch-based biodegradable materials: Challenges and opportunities. Adv. Ind. Eng. Polym. Res. 2020, 3, 8–18.
- Liu, H.; Yu, L.; Simon, G.; Zhang, X.; Dean, K.; Chen, L. Effect of annealing and pressure on microstructure of cornstarches with different amylose/amylopectin ratios. Carbohydr. Res. 2009, 344, 350–354.
- Yu, L.; Dean, K.; Li, L. Polymer blends and composites from renewable resources. Prog. Polym. Sci. 2006, 31, 576–602.
- Zhu, T.; Jackson, D.S.; Wehling, R.L.; Geera, B. Comparison of amylose determination methods and the development of a dual wavelength iodine binding technique. Cereal Chem. 2008, 85, 51–58.
- Avaro, M.R.A.; Pan, Z.; Yoshida, T.; Wada, Y. Two Alternative Methods to Predict Amylose Content of Rice Grain by Using Tristimulus CIE Lab Values and Developing a Specific Color Board Of Starch-Iodine Complex Solution. Plant Prod. Sci. 2011, 14, 164–168.
- Jian, Y.; Xiaorong, Y.; Zhaoci, W.; Xiarong, Y.; Zhaoci, W. Research on method for determination of amylose content in rice. Proc. 7th Int. Work. Conf. Stored-Product Prot. 1998, 2, 1710–1714.
- Boonpo, S.; Kungwankunakorn, S. Study on Amylose Iodine Complex from Cassava Starch by Colorimetric Method. J. Adv. Agric. Technol. 2017, 4, 345–349.
- Landers, P.S.; Gbur, E.E.; Sharp, R.N. Comparison of Two Models to Predict Amylose Concentration in Rice Flours as Determined by Spectrophotometric Assay. Cereal Chem. 1991, 68, 545–548.
- Cauvain, S.P. Bread Making: Improving Quality; CRC Press: Boca Raton, FL, USA, 2003; ISBN 9781855735538.
- Chen, X.; He, X.; Fu, X.; Huang, Q. In vitro digestion and physicochemical properties of wheat starch/flour modified by heat-moisture treatment. J. Cereal Sci. 2015, 63, 109–115.
- Chen, G.X.; Zhou, J.W.; Liu, Y.L.; Lu, X.B.; Han, C.X.; Zhang, W.Y.; Xu, Y.H.; Yan, Y.M. Biosynthesis and Regulation of Wheat Amylose and Amylopectin from Proteomic and Phosphoproteomic Characterization of Granule-binding Proteins. Sci. Rep. 2016, 6, 33111.
- Qiu, S.; Yadav, M.P.; Liu, Y.; Chen, H.; Tatsumi, E.; Yin, L. Effects of corn fiber gum with different molecular weights on the gelatinization behaviors of corn and wheat starch. Food Hydrocoll. 2016, 53, 180–186.
- De Pilli, T.; Legrand, J.; Derossi, A.; Severini, C. Effect of proteins on the formation of starch-lipid complexes during extrusion cooking of wheat flour with the addition of oleic acid. Int. J. Food Sci. Technol. 2015, 50, 515–521.
- Wang, S.; Luo, H.; Zhang, J.; Zhang, Y.; He, Z.; Wang, S. Alkali-induced changes in functional properties and in vitro digestibility of wheat starch: The role of surface proteins and lipids. J. Agric. Food Chem. 2014, 62, 3636–3643.
- Dengate, H.N.; Baruch, D.W.; Meredith, P. The Density of Wheat Starch Granules: A Tracer Dilution Procedure for Determining the Density of an Immiscible Dispersed Phase. Starch-Stärke 1978, 30, 80–84.
- Jang, J.K.; Pyun, Y.R. Effect of moisture content on the melting of wheat starch. Starch/Staerke 1996, 48, 48–51.
- Bertoft, E. Understanding starch structure: Recent progress. Agronomy 2017, 7, 56.
- Zakaria, N.H.; Muhammad, N.; Abdullah, M.M.A.B. Potential of Starch Nanocomposites for Biomedical Applications. IOP Conf. Ser. Mater. Sci. Eng. 2017, 209, 012087.
- Yoo, S.H.; Chang, Y.H. Effect of tara gum addition on steady and dynamic shear rheological properties of rice starch isolated from the Korean rice variety “Boramchan”. Prev. Nutr. Food Sci. 2018, 23, 254–259.
- Puncha-arnon, S.; Uttapap, D. Rice starch vs. rice flour: Differences in their properties when modified by heat-moisture treatment. Carbohydr. Polym. 2013, 91, 85–91.
- Tharise, N.; Julianti, E.; Nurminah, M. Evaluation of physico-chemical and functional properties of composite flour from cassava, rice, potato, soybean and xanthan gum as alternative of wheat flour. Int. Food Res. J. 2014, 21, 1641–1649.
- Marichelvam, M.K.; Jawaid, M.; Asim, M. Corn and Rice Starch-Based Bio-Plastics as Alternative Packaging Materials. Fibers 2019, 7, 32.
- Gifuni, I.; Olivieri, G.; Krauss, I.R.; D’Errico, G.; Pollio, A.; Marzocchella, A. Microalgae as new sources of starch: Isolation and characterization of microalgal starch granules. Chem. Eng. Trans. 2017, 57, 1423–1428.
- Pouplin, M.; Redl, A.; Gontard, N. Glass transition of wheat gluten plasticized with water, glycerol, or sorbitol. J. Agric. Food Chem. 1999, 47, 538–543.
- Patni, N.; Yadava, P.; Agarwal, A.; Maroo, V. Study on Wheat Gluten Biopolymer: A Novel Way to Eradicate Plastic Waste. Indian J. Appl. Res. 2011, 3, 253–255.
- ROY, S.B.; SHIT, D.S.C.; SEN GUPTA, D.R.A.; SHUKLA, D.P.R. A Review on Bio-Composites: Fabrication, Properties and Applications. Int. J. Innov. Res. Sci. Eng. Technol. 2014, 03, 16814–16824.
- Vo Hong, N.; Van Puyvelde, P.; Van Vuure, A.W.; Verpoest, I. Preparation of biocomposites based on gluten resin and unidirectional flax fibers. In Proceedings of the 15th European Conference on Composite Materials (ECCM 2012), Venice, Italy, 24–28 June 2012.
- Hou, T.H.; Su, C.H.; Liu, W.L. Parameters optimization of a nano-particle wet milling process using the Taguchi method, response surface method and genetic algorithm. Powder Technol. 2007, 173, 153–162.
- Krieger, K.M.; Duvick, S.A.; Pollak, L.M.; White, P.J. Thermal properties of corn starch extracted with different blending methods: Microblender and homogenizer. Cereal Chem. 1997, 74, 553–555.
- Hong, N.V.; Pyka, G.; Wevers, M.; Goderis, B.; Van Puyvelde, P.; Verpoest, I.; Van Vuure, A.W. Processing rigid wheat gluten biocomposites for high mechanical performance. Compos. Part A 2015, 79, 74–81.
- Kim, J.T.; Netravali, A.N. Mechanical, thermal, and interfacial properties of green composites with ramie fiber and soy resins. J. Agric. Food Chem. 2010, 58, 5400–5407.
- Zelaziński, T.; Słoma, J.; Skudlarski, J.; Ekielski, A. The rape pomace and microcrystalline cellulose composites made by press processing. Sustainability 2020, 12, 1311.
- Hemsri, S.; Thongpin, C.; Supatti, N.; Manomai, P.; Socharoentham, A. Bio-based Blends of Wheat Gluten and Maleated Natural Rubber: Morphology, Mechanical Properties and Water Absorption. Energy Procedia 2016, 89, 264–273.
- Edhirej, A.; Sapuan, S.M.; Jawaid, M.; Zahari, N.I. Cassava/sugar palm fiber reinforced cassava starch hybrid composites: Physical, thermal and structural properties. Int. J. Biol. Macromol. 2017, 101, 75–83.
- Lee, J.; Cousineau, A. Production and Characterization of Wheat Gluten Films. Master’s Thesis, University of Waterloo, Waterloo, ON, Canada, 2012.
- Mojumdar, S.C.; Moresoli, C.; Simon, L.C.; Legge, R.L. Edible wheat gluten (WG) protein films. J. Therm. Anal. Calorim. J. Therm. Anal. Calorim. 2011, 104, 929–936.
- Tarique, J.; Sapuan, S.M.; Khalina, A. Effect of glycerol plasticizer loading on the physical, mechanical, thermal, and barrier properties of arrowroot (Maranta arundinacea) starch biopolymers. Sci. Rep. 2021, 11, 1–17.
- Dou, Y.; Zhang, L.; Zhang, B.; He, M.; Shi, W.; Yang, S.; Cui, Y.; Yin, G. Preparation and characterization of edible dialdehyde carboxymethyl cellulose crosslinked feather keratin films for food packaging. Polymers 2020, 12, 158.
- Ye, P.; Reitz, L.; Horan, C.; Parnas, R. Manufacture and biodegradation of wheat gluten/basalt composite material. J. Polym. Environ. 2006, 14, 1–7.
- Reddy, N.; Yang, Y. Biocomposites developed using water-plasticized wheat gluten as matrix and jute fibers as reinforcement. Polym. Int. 2011, 60, 711–716.
- Muneer, F.; Johansson, E.; Hedenqvist, M.S.; Gällstedt, M.; Newson, W.R. Preparation, properties, protein cross-linking and biodegradability of plasticizer-solvent free hemp fibre reinforced wheat gluten, glutenin, and gliadin composites. BioResources 2014, 9, 5246–5261.
- Kunanopparat, T.; Menut, P.; Morel, M.H.; Guilbert, S. Plasticized wheat gluten reinforcement with natural fibers: Effect of thermal treatment on the fiber/matrix adhesion. Compos. Part A Appl. Sci. Manuf. 2008, 39, 1787–1792.
- Hemsri, S.; Grieco, K.; Asandei, A.D.; Parnas, R.S. Wheat gluten composites reinforced with coconut fiber. Compos. Part A Appl. Sci. Manuf. 2012, 43, 1160–1168.
- Nguyen, H.D.; Thuy Mai, T.T.; Nguyen, N.B.; Dang, T.D.; Phung Le, M.L.; Dang, T.T. A novel method for preparing microfibrillated cellulose from bamboo fibers. Adv. Nat. Sci. Nanosci. Nanotechnol. 2013, 4, 015016.
- Tan, M.Y.; Nicholas Kuan, H.T.; Lee, M.C. Characterization of Alkaline Treatment and Fiber Content on the Physical, Thermal, and Mechanical Properties of Ground Coffee Waste/Oxobiodegradable HDPE Biocomposites. Int. J. Polym. Sci. 2017, 2017, 6258151.
- Harwalkar, C.Y.M. Thermal analysis of food carbohydrates FCA. Elsevier Applied Science: London, UK, 1990; pp. 168–222.
- Rouilly, A.; Rigal, L. Agro-materials: A bibliographic review. J. Macromol. Sci.-Polym. Rev. 2002, 42, 441–479.
- Song, Y.; Zheng, Q. Improved tensile strength of glycerol-plasticized gluten bioplastic containing hydrophobic liquids. Bioresour. Technol. 2008, 99, 7665–7671.
- Dong, J.; Dicharry, R.; Waxman, E.; Parnas, R.S.; Asandei, A.D. Imaging and thermal studies of wheat gluten/poly(vinyl alcohol) and wheat gluten/thiolated poly(vinyl alcohol) blends. Biomacromolecules 2008, 9, 568–573.
- Mangavel, C.; Rossignol, N.; Perronnet, A.; Barbot, J.; Popineau, Y.; Guéguen, J. Properties and microstructure of thermo-pressed wheat gluten films: A comparison with cast films. Biomacromolecules 2004, 5, 1596–1601.
- Kayserilioǧlu, B.Ş.; Bakir, U.; Yilmaz, L.; Akkaş, N. Drying temperature and relative humidity effects on wheat gluten film properties. J. Agric. Food Chem. 2003, 51, 964–968.
- Muensri, P.; Kunanopparat, T.; Menut, P.; Siriwattanayotin, S. Composites: Part A Effect of lignin removal on the properties of coconut coir fiber / wheat gluten biocomposite. Compos. Part A 2011, 42, 173–179.
- Chen, L.; Reddy, N.; Wu, X.; Yang, Y. Thermoplastic films from wheat proteins. Ind. Crops Prod. 2012, 35, 70–76.
- Zubeldía, F.; Ansorena, M.R.; Marcovich, N.E. Wheat gluten films obtained by compression molding. Polym. Test. 2015, 43, 68–77.
- Gianibelli, M.C.; Larroque, O.R.; MacRitchie, F.; Wrigley, C.W. Biochemical, genetic, and molecular characterization of wheat glutenin and its component subunits. Cereal Chem. 2001, 78, 635–646.
- Nagarajan, S.; Nagarajan, R.; Kumar, J.; Salemme, A.; Togna, A.R.; Saso, L.; Bruno, F. Antioxidant activity of synthetic polymers of phenolic compounds. Polymers 2020, 12, 1646.
- Eça, K.S.; Sartori, T.; Menegalli, F.C. Films and edible coatings containing antioxidants—A review. Braz. J. Food Technol. 2014, 17, 98–112.
- Abd El-Ghaffar, M.A.; Shaffei, K.A.; Abdelwahab, N. Evaluation of some conducting polymers as novel antioxidants for rubber vulcanizates. Int. J. Polym. Sci. 2014, 2014, 893542.
- Jacob, J.; Thomas, S.; Loganathan, S.; Valapa, R.B. Chapter 10—Antioxidant incorporated biopolymer composites for active packaging. In Processing and Development of Polysaccharide-Based Biopolymers for Packaging Applications; Zhang, Y., Ed.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 239–260. ISBN 978-0-12-818795-1.
- Cheremisinoff, N.P. Condensed Encyclopedia of polymer Engineering Terms. Choice Rev. Online 2001, 39, 1288.
- Bonilla, J.; Talón, E.; Atarés, L.; Vargas, M.; Chiralt, A. Effect of the incorporation of antioxidants on physicochemical and antioxidant properties of wheat starch-chitosan films. J. Food Eng. 2013, 118, 271–278.
- Yilmaz-Turan, S.; Jiménez-Quero, A.; Menzel, C.; de Carvalho, D.M.; Lindström, M.E.; Sevastyanova, O.; Moriana, R.; Vilaplana, F. Bio-based films from wheat bran feruloylated arabinoxylan: Effect of extraction technique, acetylation and feruloylation. Carbohydr. Polym. 2020, 250, 116916.
- Díez-Pascual, A.M. Antimicrobial polymer-based materials for food packaging applications. Polymers 2020, 12, 731.
- Park, S.-K.; Bae, D.H. Antimicrobial properties of wheat gluten-chitosan composite film in intermediate-moisture food systems. Food Sci. Biotechnol. 2006, 15, 133–137.
- Güçbilmez, Ç.M.; Yemenicioǧlu, A.; Arslanoǧlu, A. Antimicrobial and antioxidant activity of edible zein films incorporated with lysozyme, albumin proteins and disodium EDTA. Food Res. Int. 2007, 40, 80–91.
- Pintado, C.; Ferreira, S.; Sousa, I. Properties of Whey Protein-Based Films Containing Organic Acids and Nisin To Control Listeria monocytogenes. J. Food Prot. 2009, 72, 1891–1896.
- Türe, H.; Eroglu, E.; Soyer, F.; Özen, B. Antifungal activity of biopolymers containing natamycin and rosemary extract against Aspergillus niger and Penicillium roquefortii. Int. J. Food Sci. Technol. 2008, 43, 2026–2032.
- Pranoto, Y.; Rakshit, S.K.; Salokhe, V.M. Enhancing antimicrobial activity of chitosan films by incorporating garlic oil, potassium sorbate and nisin. LWT-Food Sci. Technol. 2005, 38, 859–865.
- Seydim, A.C.; Sarikus, G. Antimicrobial activity of whey protein based edible films incorporated with oregano, rosemary and garlic essential oils. Food Res. Int. 2006, 39, 639–644.
- Sivarooban, T.; Hettiarachchy, N.S.; Johnson, M.G. Physical and antimicrobial properties of grape seed extract, nisin, and EDTA incorporated soy protein edible films. Food Res. Int. 2008, 41, 781–785.
- Iamareerat, B.; Singh, M.; Sadiq, M.B.; Anal, A.K. Reinforced cassava starch based edible film incorporated with essential oil and sodium bentonite nanoclay as food packaging material. J. Food Sci. Technol. 2018, 55, 1953–1959.
- Acevedo-Fani, A.; Salvia-Trujillo, L.; Rojas-Graü, M.A.; Martín-Belloso, O. Edible films from essential-oil-loaded nanoemulsions: Physicochemical characterization and antimicrobial properties. Food Hydrocoll. 2015, 47, 168–177.
- Ribeiro-Santos, R.; Andrade, M.; Sanches-Silva, A. Application of encapsulated essential oils as antimicrobial agents in food packaging. Curr. Opin. Food Sci. 2017, 14, 78–84.
- Kan, Y.; Chen, T.; Wu, Y.; Wu, J.; Wu, J. Antioxidant activity of polysaccharide extracted from Ganoderma lucidum using response surface methodology. Int. J. Biol. Macromol. 2015, 72, 151–157.
- Atarés, L.; Chiralt, A. Essential oils as additives in biodegradable films and coatings for active food packaging. Trends Food Sci. Technol. 2016, 48, 51–62.
- Viuda-Martos, M.; El Gendy, A.E.N.G.S.; Sendra, E.; Fernández-López, J.; El Razik, K.A.A.; Omer, E.A.; Pérez-Alvarezj, J.A. Chemical composition and antioxidant and anti-Listeria activities of essential oils obtained from some Egyptian plants. J. Agric. Food Chem. 2010, 58, 9063–9070.
- Syafiq, R.; Sapuan, S.M.; Zuhri, M.Y.M.; Ilyas, R.A.; Nazrin, A.; Sherwani, S.F.K.; Khalina, A. Antimicrobial activities of starch-based biopolymers and biocomposites incorporated with plant essential oils: A review. Polymers 2020, 12, 2403.
- Song, X.; Zuo, G.; Chen, F. Effect of essential oil and surfactant on the physical and antimicrobial properties of corn and wheat starch films. Int. J. Biol. Macromol. 2018, 107, 1302–1309.
- Jamróz, E.; Juszczak, L.; Kucharek, M. Investigation of the physical properties, antioxidant and antimicrobial activity of ternary potato starch-furcellaran-gelatin films incorporated with lavender essential oil. Int. J. Biol. Macromol. 2018, 114, 1094–1101.
- Türe, H.; Gällstedt, M.; Hedenqvist, M.S. Antimicrobial compression-moulded wheat gluten films containing potassium sorbate. Food Res. Int. 2012, 45, 109–115.
- Khairuddin, N.; Muhamad, I.I.; Abd Rahman, W.A.W.; Siddique, B.M. Physicochemical and thermal characterization of hydroxyethyl cellulose—Wheat starch based films incorporated thymol intended for active packaging. Sains Malays. 2020, 49, 323–333.





