Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Renjian Xie | + 2111 word(s) | 2111 | 2021-10-18 02:42:10 | | | |
| 2 | Conner Chen | Meta information modification | 2111 | 2021-11-03 08:48:51 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Xie, R. Cartilage Lubrication in Osteoarthritis. Encyclopedia. Available online: https://encyclopedia.pub/entry/15608 (accessed on 21 May 2026).
Xie R. Cartilage Lubrication in Osteoarthritis. Encyclopedia. Available at: https://encyclopedia.pub/entry/15608. Accessed May 21, 2026.
Xie, Renjian. "Cartilage Lubrication in Osteoarthritis" Encyclopedia, https://encyclopedia.pub/entry/15608 (accessed May 21, 2026).
Xie, R. (2021, November 02). Cartilage Lubrication in Osteoarthritis. In Encyclopedia. https://encyclopedia.pub/entry/15608
Xie, Renjian. "Cartilage Lubrication in Osteoarthritis." Encyclopedia. Web. 02 November, 2021.
Copy Citation
The remarkable lubrication properties of normal articular cartilage play an essential role in daily life, providing almost frictionless movements of joints. Alterations of cartilage surface or degradation of biomacromolecules within synovial fluid increase the wear and tear of the cartilage and hence determining the onset of the most common joint disease, osteoarthritis (OA).
articular cartilage
osteoarthritis
boundary lubrication
chondrocytes
1. Introduction
Articular cartilage is an avascular, aneural and alymphatic connective tissue (which determines its very poor self-recovery ability) lining the bone ends of diarthrodial joints [1]. Combined with excellent load-bearing capacities, this cushion of articular cartilage, especially the outer surface of cartilage, provides extremely low friction with a friction coefficient as low as 10−3 under a wide range of physiological pressures (even up to 100 atm) to maintain daily movements during a person’s lifetime [2][3][4]. Recently, researchers noticed that the increase of cartilage friction plays a determining role in initiating the most common degenerative joint disability disease, that is osteoarthritis (OA), which is mainly characterized by the progressive but irreversible degradation of articular cartilage [5][6][7][8][9]. Briefly, aging-related changes or lesions usually lead to the compromisation of the outer surface of cartilage, which subsequently causes an increase of the friction coefficient. Chondrocytes, as the only cell type in cartilage, in return, up-regulate the secretion of the cartilage-degrading enzymes, such as matrix metalloproteinases (MMPs) and a disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS) to degrade the type II collagen and aggrecan (the main components of cartilage matrix), respectively. Moreover, the cartilage degradation fragments are phagocytosed cells (such as macrophage and synovial fibroblasts) to inflame the synovium, promoting the production of MMPs and ADMATS to break down cartilage and deteriorate lubrication [10][11][12][13][14][15]. In this way, a positive feedback loop is formed due to the mutually reinforcing effect of increased friction and secretion of degradation enzymes, resulting in the progressive till total degradation of articular cartilage. Early, moderate, and late stages of OA can be classified mainly based on the degree of cartilage degradation. It is thought the breakdown of the type II collagen network initiates the point where the OA is considered irreversible [16][17]. Is it possible to modify and prevent the disease at its early stage especially considering the role of cartilage lubrication?
Among the population over 60 years old, 9.6% of men and 18.0% of women have OA symptoms, which made OA a serious global disease defined by the Osteoarthritis Research Society International in 2016 [18]. Cell-based therapies or cartilage tissue engineering for cartilage repair or regeneration have made significant advances recently [19][20], such as the adhesive peptide-based 3D scaffolds for cell culture [21], however, considerable efforts are still required for the tribological properties and durability of the neocartilage before clinical translation. Currently, the main nonsurgical options for OA treatment before the end of OA include using analgesics, anti-inflammatory drugs (such as acetaminophen), inhibitors (such as cyclooxygenase), or articular injection of hyaluronan and corticosteroids [22][23]. However, these nonsurgical options usually are highly controversial due to the nonuniversal effects when compared to that in state-of-the-art placebo-controlled [24][25]. Therefore, it is highly important to shed light on the remarkable lubrication of cartilage and the correlation between lubrication and cartilage regeneration, with the aim to improve the understanding of OA and encourage the development of approaches to alleviating and even treating it.
2. Articular Cartilage
A joint is a place where two or more bones meet, allowing the skeleton to move. Usually, joints differ in shape and structures according to the required movement and load, so we focus on discussing the diarthrosis knee joints. The knee joint consists of a joint capsule, ligaments, synovium, and the articular cartilage lining the ends of the opposing bones [1]. Synovial fluid within the capsule provides lubrication and nutrition, while the synovial membrane, a sac-like structure, surrounds the joint cavity and synovial fluid [26]. In daily life, it’s essential to maintain the normal structure of the synovial joint.
2.1. Structure and Components of Articular Cartilage
Articular cartilage (also referred to as hyaline cartilage) is a highly hydrated glassy connective tissue comprised of chondrocytes (the only cell type embedded within cartilage) and the extracellular matrix (ECM) which is secreted and maintained by chondrocytes [1][27]. The ECM is predominantly composed of type II collagen bundles, negatively charged proteoglycans, non-collagenous proteins, water, and ions (primarily Na+ and Cl−). 11 types of collagens could be found in articular cartilage. Among them, type II collagen, representing 90–95% of all collagens in ECM and accounting for 60% of the dry weight of articular cartilage, forms a crosslinked core network, to enable the cartilage tensile and shear strength [28][29].
The type II collagen fibril networks interweave with proteoglycans (such as aggrecans), the second most abundant macromolecules in articular cartilage, which contribute to the lubrication and load-bearing properties of cartilage due to their strong hydration [12]. Proteoglycans are proteins covalently attached to glycosaminoglycans (long repetitive dimers of hexosamine and uronic acid). The most prevalent and largest in size of proteoglycans in articular cartilage are aggrecans, representing a bottle-brush structure with a polypeptide as backbone and chondroitin sulfate and keratan sulfate as the side chains. Usually, there are over 100 chondroitin sulfate and 20–40 keratan sulfate chains in one aggrecan molecule [30][31]. Therefore, the aggrecans are highly sulfated and negatively charged conferred by the sulfate groups in their side chains. These negative charges attract large water molecules to further strengthen the cartilage matrix. Hyaluronic acid or hyaluronan (HA), the only non-sulfated glycosaminoglycan, is built by the repeated dimers of b-D-(1,4)-N-acetylglucosamine and b-D-(1–3) glucuronic acid with a molecular weight up to 6 MDa [32]. HA and aggrecan form an extensive aggregate, comprising of a central HA to which about 100 aggrecan molecules are non-covalently attached via the link protein, thereby stabilizing this aggregation [33]. These aggregates further bind to the type II collagen fibers and have been demonstrated to play a significant role in cartilage lubrication (will be discussed later).
Microscopically, three zones of the articular cartilage can be distinguished, that is the superficial zone (also referred to as the lamina splendens), the middle or transitional zone and the deep zone, as shown in Figure 1. These three zones of cartilage exhibit heterogeneity in the composition of ECM, which is reflected in the organization of collagen, size, phenotype, and metabolic activity of chondrocytes [34]. The superficial zone lies in the outer surface of articular cartilage, constitutes 10–20% of the full thickness of adult cartilage, characterized by two aspects, one is the type II collagen fibers, with a diameter of 30–35 nm, which are densely arranged and parallel to the articular surface, the other one is the long axis of flat and ellipsoidal chondrocytes parallel to the surface of the cartilage. Normally, the lubricating molecules are within the superficial zone. The middle zone is constituted of 40–60% of the thickness of total cartilage, the chondrocytes, exhibiting round or rectangular shape, are randomly distributed with their long axis perpendicular to the cartilage surface. The fibrils of type II collagen form an oblique transitional network and appear as arcades. The deep or radial zone constitutes the last 20–30% of the thickness of the cartilage. The shape of chondrocytes is round, the fibrils of type II collagen, with the largest diameter (40–80 nm), are perpendicular to the cartilage surface. The predominant biomechanical properties of the main three different zones of articular cartilage are summarized in Table 1.
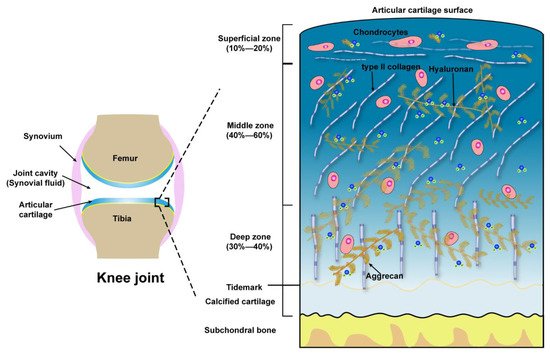
Figure 1. Schematic illustration of the knee joint and the structure, compositions of articular cartilage from the surface to subchondral bone.
Table 1. The main biomechanical properties of the superficial zone, middle zone, and deep zone of articular cartilage.
| Zones | Extracellular Molecules | Biomechanical Properties | References |
|---|---|---|---|
| Superficial zone (10 to 20%) |
Outer of Surface Lubricin, HA, Phospholipids, COMP | Boundary lubrication, chondroprotection |
[35][36][37] |
| Below outer of Surface Type II collagen aggrecans, HA |
Resist shear stress, bear ~20% load; Maintain tensile strength; As a barrier to fluid flow during loading; Subject to maximum strain; Contribute to elasticity and resiliency via interacting with collagen |
[38][39][40] | |
| Middle zone (40 to 60%) |
Upper ~1/3rd Collagen type II, other collagens, aggrecans, HA |
Transit shear and compression stresses; Exhibit high deformation during loading; Resist compression; Contribute to elasticity and resiliency to compression via interacting with collagen | [38][39][40][41] |
| Lower ~2/3rd Thick collagen type II, other collagens, aggrecan, HA, GAGs |
Compared with upper 1/3rd of the middle zone: Decrease tensile strength; Provide higher resistance to compression during loading | [42][43][44] | |
| Deep zone (30 to 40%) |
Thickest collagen type II, other collagens, aggrecans, HA, high GAGs | Relative to the middle zone: Further decreased tensile strength; Provide highest resistance to compression during loading | [42][43][44] |
2.2. Mechanotransduction of Chondrocytes
Mechanotransduction refers to the process of sensing and converting mechanical signals into biochemical signals to regulate cellular activities [45]. Located on the joint surfaces, a range of static and dynamic stresses (standing, walking, and jogging) are applied on articular cartilage. It is well documented the metabolism of chondrocyte is strongly regulated by the normal stress (compression), static loads were shown to be detrimental to the anabolic processes (biosynthesis of type II collagen and proteoglycans) while oscillatory loads with moderate frequencies and amplitudes (compression strain under 20%) have been shown to effectively promote the matrix accumulation and decrease the secretion of TNF- a and IL-6, which contribute to the degradation of matrix [46][47][48][49].
Unlike normal stress, shear stress gives rise to the shear strain of the cartilage, and then be transmitted to the chondrocytes, especially those within the superficial zone. Many previous studies suggested the shear stress activates chondrocytes and up-regulates the proinflammatory cytokines (TNF-a, and the family of interleukins) and MMPs [48][50], which elicit the degradation of cartilage, most of the underlying signaling pathways remain unclear, but we can summarize some of them in Figure 2 according to the previous reports [50][51][52][53][54][55][56]. The chondrocytes undergo a phenotypic switch to aberrantly express catabolic enzymes when the shear strain exceeds ≈1% estimated by Klein and Lin very recently under relative ideal circumstances considering the complexity of cartilage [57]. Moreover, the increased shear strain, or the shear stress or friction, induces chondrocyte apoptosis, which has been demonstrated by previous studies [58][59]. The mechanism regulating shear-mediated chondrocytes expression of IL-6 and MMPs and apoptosis is shown in Figure 2. The stimuli of high shear stress induce chondrocytes to express cyclooxygenase (Cox 2), which inhibits the activity of phosphatidylinositol 3-kinase (PI3-K), following decreases in antioxidant capacity to lead to chondrocyte apoptosis.
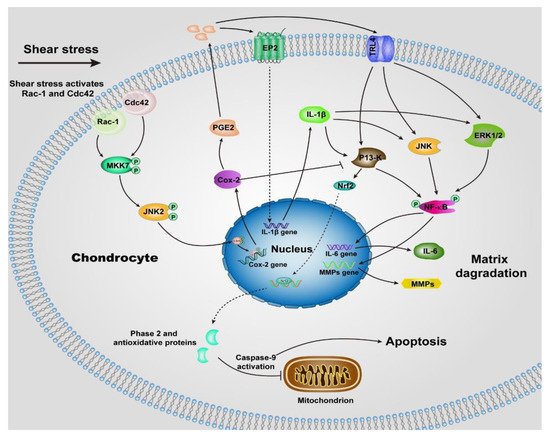
Figure 2. Schematic of shear-induced cartilage matrix degradation and apoptosis of chondrocytes. High shear stress activates the Rac-1/Cdc42, which then transactivates MKK7 to regulate JNK2 activation, and this, in turn, triggers c-Jun phosphorylation which induces the overexpression of Cox-2. Cox-2 suppresses the activity of P13-K, which represses Nrf-2 to decrease the antioxidant capacity to permit disruption of the integrity of mitochondrial, activation of caspase-9, and the apoptosis of chondrocytes. The expression of Cox-2 also triggers the expression of PGE2, as well as the concomitant downstream expression of receptor EP2, as a result, IL-1b is rapidly and sustainably synthesized. Moreover, up-regulation of TLR4 due to high shear stress activates ERK1/2, P13-K and JNK pathways, which is also activated by IL-1b, then regulates NF-kB-dependent IL-6 and MMP synthesis. Abbreviations: Cyclooxygenase-2 (Cox-2), mitogen-activated protein kinase 7 (MKK7), nuclear factor-kB (NF-kB), prostaglandin E2 (PGE2), Interleukin-1b (IL-1b), Interleukin-6 (IL-6), phosphatidylinositol 3-kinase (PI3-K), c-Jun N-terminal kinase 2 (JNK2), NF-E2 related factor 2 (Nrf2), matrix metalloproteinases (MMPs), toll-like receptor 4 (TLR4), extracellular signal-regulated kinase (ERK1/2).
Collectively, favoring cartilage longevity requires dynamic normal stress combined with quite low or even zero shear stress. To maximize the regeneration of cartilage, the main way is to decrease the shear strain arising from the shear stress by reducing the friction coefficient of cartilage. Therefore, a scenario in which treating or healing OA at its early stage by restoring the lubrication of OA-damaged cartilage can be imagined.
2.3. Lubrication Mechanism of Articular Cartilage
The friction coefficients of normal articular cartilage can be as low as ~0.002-0.02[57]. The lubrication properties of articualr cartilage have drawn attention since the 1930s and many theories have been propersed to claim the mechanisms behind the ultra-low friction of cartilage.
Recently, a new picture emerged where it is the synergy between the molecules in the synovial fluid that determines the lubrication of articular cartilage under severe joint loading [60][61]. Specifically, HA associates with aggrecan via the link protein to form a bottle-brush structure where HA serves as the backbone and aggrecan as the side chain. HA and lubricin also form a complex, which is physically trapped on the surface and contributes to effectively eliminate the wear damage of the cartilage. HA also shows a high affinity with phospholipids according to the previous report [62]. Thus, Klein et al. pointed to a scenario in which HA (alone cannot bind to cartilage surface) anchors at the outer surface of cartilage with the assistance of lubricin, then further complexes with the phospholipids to act as an effective boundary lubricant to enable the remarkable lubrication of articular cartilage at high pressure, via the hydration mechanism [63][64][65]. In this way, lubricin serves as a “carrier” between the HA, phospholipids, and the outer surface of the articular cartilage.
References
- Alice J. Sophia Fox; Asheesh Bedi; Scott A. Rodeo; The Basic Science of Articular Cartilage: Structure, Composition, and Function. Sports Health: A Multidisciplinary Approach 2009, 1, 461-468, 10.1177/1941738109350438.
- J Klein; Molecular mechanisms of synovial joint lubrication. Proceedings of the Institution of Mechanical Engineers, Part J: Journal of Engineering Tribology 2006, 220, 691-710, 10.1243/13506501jet143.
- K. C. Morrell; W. A. Hodge; D. E. Krebs; R. W. Mann; Corroboration of in vivo cartilage pressures with implications for synovial joint tribology and osteoarthritis causation. Proceedings of the National Academy of Sciences 2005, 102, 14819-14824, 10.1073/pnas.0507117102.
- John M. Marzo; Jennifer Gurske-DePerio; Effects of Medial Meniscus Posterior Horn Avulsion and Repair on Tibiofemoral Contact Area and Peak Contact Pressure with Clinical Implications. The American Journal of Sports Medicine 2008, 37, 124-129, 10.1177/0363546508323254.
- L.E. DeFrate; S.Y. Kim-Wang; Z.A. Englander; A.L. McNulty; Osteoarthritis year in review 2018: mechanics. Osteoarthritis and Cartilage 2019, 27, 392-400, 10.1016/j.joca.2018.12.011.
- Fatemeh Saberi Hosnijeh; Sita M. Bierma-Zeinstra; Anne C. Bay-Jensen; Osteoarthritis year in review 2018: biomarkers (biochemical markers). Osteoarthritis and Cartilage 2019, 27, 412-423, 10.1016/j.joca.2018.12.002.
- D. Martínez-Moreno; G. Jiménez; P. Gálvez-Martín; G. Rus; J.A. Marchal; Cartilage biomechanics: A key factor for osteoarthritis regenerative medicine. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2019, 1865, 1067-1075, 10.1016/j.bbadis.2019.03.011.
- Giulia Morgese; Edmondo M. Benetti; Marcy Zenobi-Wong; Molecularly Engineered Biolubricants for Articular Cartilage. Advanced Healthcare Materials 2018, 7, e1701463, 10.1002/adhm.201701463.
- Jane Desrochers; Matthias W. Amrein; John R. Matyas; Microscale surface friction of articular cartilage in early osteoarthritis. Journal of the Mechanical Behavior of Biomedical Materials 2013, 25, 11-22, 10.1016/j.jmbbm.2013.03.019.
- Martin K. Lotz; Beatriz Carames; Autophagy and cartilage homeostasis mechanisms in joint health, aging and OA. Nature Reviews Rheumatology 2011, 7, 579-587, 10.1038/nrrheum.2011.109.
- Johanne Martel-Pelletier; Andrew Barr; Flavia Cicuttini; Philip Conaghan; Cyrus Cooper; Mary B. Goldring; Steven R. Goldring; Graeme Jones; Andrew J. Teichtahl; Jean-Pierre Pelletier; et al. Osteoarthritis. Nature Reviews Disease Primers 2016, 2, 16072, 10.1038/nrdp.2016.72.
- Peter J Roughley; John S Mort; The role of aggrecan in normal and osteoarthritic cartilage. Journal of Experimental Orthopaedics 2014, 1, 1-11, 10.1186/s40634-014-0008-7.
- Linda Troeberg; Hideaki Nagase; Proteases involved in cartilage matrix degradation in osteoarthritis. Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics 2012, 1824, 133-145, 10.1016/j.bbapap.2011.06.020.
- Ru Liu-Bryan; Robert Terkeltaub; Emerging regulators of the inflammatory process in osteoarthritis. Nature Reviews Rheumatology 2014, 11, 35-44, 10.1038/nrrheum.2014.162.
- Jérémie Sellam; Francis Berenbaum; The role of synovitis in pathophysiology and clinical symptoms of osteoarthritis. Nature Reviews Rheumatology 2010, 6, 625-635, 10.1038/nrrheum.2010.159.
- John S Mort; Caron J Billington; Articular cartilage and changes in arthritis: Matrix degradation. Arthritis Research & Therapy 2001, 3, 337-341, 10.1186/ar325.
- Shannon Brown; Shreedevi Kumar; Blanka Sharma; Intra-articular targeting of nanomaterials for the treatment of osteoarthritis. Acta Biomaterialia 2019, 93, 239-257, 10.1016/j.actbio.2019.03.010.
- M. Kloppenburg; F. Berenbaum; Osteoarthritis year in review 2019: epidemiology and therapy. Osteoarthritis and Cartilage 2020, 28, 242-248, 10.1016/j.joca.2020.01.002.
- Heenam Kwon; Wendy E. Brown; Cassandra A. Lee; Dean Wang; Nikolaos Paschos; Jerry C. Hu; Kyriacos A. Athanasiou; Surgical and tissue engineering strategies for articular cartilage and meniscus repair. Nature Reviews Rheumatology 2019, 15, 550-570, 10.1038/s41584-019-0255-1.
- Charlotte H. Hulme; Jade Perry; Helen S. McCarthy; Karina T. Wright; Martyn Snow; Claire Mennan; Sally Roberts; Cell therapy for cartilage repair. Emerging Topics in Life Sciences 2021, 5, 575-589, 10.1042/etls20210015.
- AnnJ Zamuner; Marta Cavo; Silvia Scaglione; Grazia Maria Lucia Messina; Teresa Russo; Antonio Gloria; Giovanni Marletta; Monica Dettin; Design of Decorated Self-Assembling Peptide Hydrogels as Architecture for Mesenchymal Stem Cells. Materials 2016, 9, 727, 10.3390/ma9090727.
- Heike A. Wieland; Martin Michaelis; Bernhard J. Kirschbaum; Karl A. Rudolphi; Osteoarthritis — an untreatable disease?. Nature Reviews Drug Discovery 2005, 4, 331-344, 10.1038/nrd1693.
- Nigel K. Arden; Thomas A. Perry; Raveendhara R. Bannuru; Olivier Bruyère; Cyrus Cooper; Ida K. Haugen; Marc C. Hochberg; Timothy E. McAlindon; Ali Mobasheri; Jean-Yves Reginster; et al. Non-surgical management of knee osteoarthritis: comparison of ESCEO and OARSI 2019 guidelines. Nature Reviews Rheumatology 2020, 17, 59-66, 10.1038/s41584-020-00523-9.
- Victor M Goldberg; Richard D Coutts; Pseudoseptic Reactions to Hylan Viscosupplementation: Diagnosis and Treatment. Clinical Orthopaedics and Related Research 2004, 429, 350-351, 10.1097/00003086-200412000-00053.
- Walter van der Weegen; Jorgen A. Wullems; Ellis Bos; Hub Noten; Rogier A.M. van Drumpt; No Difference Between Intra-Articular Injection of Hyaluronic Acid and Placebo for Mild to Moderate Knee Osteoarthritis: A Randomized, Controlled, Double-Blind Trial. The Journal of Arthroplasty 2015, 30, 754-757, 10.1016/j.arth.2014.12.012.
- C Handley; D Lowther; D Mcquillan; Mini-review: The structure and synthesis of proteoglycans of articular cartilage. Cell Biology International Reports 1985, 9, 753-782, 10.1016/0309-1651(85)90095-5.
- P.M. van der Kraan; P. Buma; T. van Kuppevelt; W.B. Van Den Berg; Interaction of chondrocytes, extracellular matrix and growth factors: relevance for articular cartilage tissue engineering. Osteoarthritis and Cartilage 2002, 10, 631-637, 10.1053/joca.2002.0806.
- D R Eyre; J J Wu; P E Woods; The cartilage collagens: structural and metabolic studies.. The Journal of Rheumatology Supplement 1991, 27, 49-51.
- D.R. Eyre; The collagens of articular cartilage. Seminars in Arthritis and Rheumatism 1991, 21, 2-11, 10.1016/0049-0172(91)90035-x.
- Ambika G. Bajpayee; Alan J. Grodzinsky; Cartilage-targeting drug delivery: can electrostatic interactions help?. Nature Reviews Rheumatology 2017, 13, 183-193, 10.1038/nrrheum.2016.210.
- Chris Kiani; Liwen Chen; Yao Jiong Wu; Albert J Yee; Burton B Yang; Structure and function of aggrecan. Cell Research 2002, 12, 19-32, 10.1038/sj.cr.7290106.
- Warren Knudson; Shinya Ishizuka; Kenya Terabe; Emily B. Askew; Cheryl B. Knudson; The pericellular hyaluronan of articular chondrocytes. Matrix Biology 2018, 78-79, 32-46, 10.1016/j.matbio.2018.02.005.
- Pj Roughley; The structure and function of cartilage proteoglycans. European Cells and Materials 2006, 12, 92-101, 10.22203/ecm.v012a11.
- Van C. Mow; Anthony Ratcliffe; A. Robin Poole; Cartilage and diarthrodial joints as paradigms for hierarchical materials and structures. Biomaterials 1992, 13, 67-97, 10.1016/0142-9612(92)90001-5.
- Akanksha Raj; Min Wang; Chao Liu; Liaquat Ali; Niclas G. Karlsson; Per M. Claesson; Andra Dėdinaitė; Molecular synergy in biolubrication: The role of cartilage oligomeric matrix protein (COMP) in surface-structuring of lubricin. Journal of Colloid and Interface Science 2017, 495, 200-206, 10.1016/j.jcis.2017.02.007.
- Debby P. Chang; Farshid Guilak; Gregory D. Jay; Stefan Zauscher; Interaction of lubricin with type II collagen surfaces: Adsorption, friction, and normal forces. Journal of Biomechanics 2013, 47, 659-666, 10.1016/j.jbiomech.2013.11.048.
- Sandra Kienle; Kathrin Boettcher; Lorenz Wiegleb; Joanna Urban; Rainer Burgkart; Oliver Lieleg; Thorsten Hugel; Comparison of friction and wear of articular cartilage on different length scales. Journal of Biomechanics 2015, 48, 3052-3058, 10.1016/j.jbiomech.2015.07.027.
- J. M. Párraga Quiroga; W. Wilson; K. Ito; C. C. Van Donkelaar; Relative contribution of articular cartilage’s constitutive components to load support depending on strain rate. Biomechanics and Modeling in Mechanobiology 2016, 16, 151-158, 10.1007/s10237-016-0807-0.
- Corinne R. Henak; Keir Ross; Edward D. Bonnevie; Lisa A. Fortier; Itai Cohen; John G. Kennedy; Lawrence J. Bonassar; Human talar and femoral cartilage have distinct mechanical properties near the articular surface. Journal of Biomechanics 2016, 49, 3320-3327, 10.1016/j.jbiomech.2016.08.016.
- Max J. Kääb; Keita Ito; Berton Rahn; John M. Clark; Hubert P. Nötzli; Effect of Mechanical Load on Articular Cartilage Collagen Structure: A Scanning Electron-Microscopic Study. Cells Tissues Organs 2000, 167, 106-120, 10.1159/000016774.
- David M. Pierce; Tim Ricken; Gerhard A. Holzapfel; A hyperelastic biphasic fibre-reinforced model of articular cartilage considering distributed collagen fibre orientations: continuum basis, computational aspects and applications. Computer Methods in Biomechanics and Biomedical Engineering 2013, 16, 1344-1361, 10.1080/10255842.2012.670854.
- K S Halonen; Mika Mononen; J S Jurvelin; Juha Töyräs; Rami Korhonen; Importance of depth-wise distribution of collagen and proteoglycans in articular cartilage—A 3D finite element study of stresses and strains in human knee joint. Journal of Biomechanics 2013, 46, 1184-1192, 10.1016/j.jbiomech.2012.12.025.
- M Venn; A Maroudas; Chemical composition and swelling of normal and osteoarthrotic femoral head cartilage. I. Chemical composition.. Annals of the Rheumatic Diseases 1977, 36, 121-129, 10.1136/ard.36.2.121.
- A Maroudas; M Venn; Chemical composition and swelling of normal and osteoarthrotic femoral head cartilage. II. Swelling.. Annals of the Rheumatic Diseases 1977, 36, 399-406, 10.1136/ard.36.5.399.
- Patrizia Romani; Lorea Valcarcel-Jimenez; Christian Frezza; Sirio Dupont; Crosstalk between mechanotransduction and metabolism. Nature Reviews Molecular Cell Biology 2020, 22, 22-38, 10.1038/s41580-020-00306-w.
- Y. Li; E.H. Frank; Y. Wang; S. Chubinskaya; H.-H. Huang; A.J. Grodzinsky; Moderate dynamic compression inhibits pro-catabolic response of cartilage to mechanical injury, tumor necrosis factor-α and interleukin-6, but accentuates degradation above a strain threshold. Osteoarthritis and Cartilage 2013, 21, 1933-1941, 10.1016/j.joca.2013.08.021.
- Jonathan B. Fitzgerald; Moonsoo Jin; Alan J. Grodzinsky; Shear and Compression Differentially Regulate Clusters of Functionally Related Temporal Transcription Patterns in Cartilage Tissue. Journal of Biological Chemistry 2006, 281, 24095-24103, 10.1074/jbc.m510858200.
- Alan J. Grodzinsky; Marc E. Levenston; Moonsoo Jin; Eliot H. Frank; Cartilage Tissue Remodeling in Response to Mechanical Forces. Annual Review of Biomedical Engineering 2000, 2, 691-713, 10.1146/annurev.bioeng.2.1.691.
- Michael Buschmann; Y.A. Gluzband; A.J. Grodzinsky; E.B. Hunziker; Mechanical compression modulates matrix biosynthesis in chondrocyte/agarose culture. Journal of Cell Science 1995, 108, 1497-1508, 10.1242/jcs.108.4.1497.
- Hiroki Yokota; Mary B. Goldring; Hui Bin Sun; CITED2-mediated Regulation of MMP-1 and MMP-13 in Human Chondrocytes under Flow Shear. Journal of Biological Chemistry 2003, 278, 47275-47280, 10.1074/jbc.m304652200.
- M. Mohtai; M. K. Gupta; B. Donlon; B. Ellison; J. Cooke; G. Gibbons; D. J. Schurman; R. Lane Smith; Expression of interleukin-6 in osteoarthritic chondrocytes and effects of fluid-induced shear on this expression in normal human chondrocytesin vitro. Journal of Orthopaedic Research 1996, 14, 67-73, 10.1002/jor.1100140112.
- James P. Abulencia; Renee Gaspard; Zachary R. Healy; William A. Gaarde; John Quackenbush; Konstantinos Konstantopoulos; Shear-induced Cyclooxygenase-2 via a JNK2/c-Jun-dependent Pathway Regulates Prostaglandin Receptor Expression in Chondrocytic Cells. Journal of Biological Chemistry 2003, 278, 28388-28394, 10.1074/jbc.m301378200.
- Z. R. Healy; N. H. Lee; X. Gao; M. B. Goldring; P. Talalay; T. W. Kensler; K. Konstantopoulos; Divergent responses of chondrocytes and endothelial cells to shear stress: Cross-talk among COX-2, the phase 2 response, and apoptosis. Proceedings of the National Academy of Sciences 2005, 102, 14010-14015, 10.1073/pnas.0506620102.
- Zachary R. Healy; Fei Zhu; Joshua D. Stull; Konstantinos Konstantopoulos; Elucidation of the signaling network of COX-2 induction in sheared chondrocytes: COX-2 is induced via a Rac/MEKK1/MKK7/JNK2/c-Jun-C/EBPβ-dependent pathway. American Journal of Physiology-Cell Physiology 2008, 294, C1146-C1157, 10.1152/ajpcell.00542.2007.
- Pu Wang; Fei Zhu; Ziqiu Tong; Konstantinos Konstantopoulos; Response of chondrocytes to shear stress: antagonistic effects of the binding partners Toll‐like receptor 4 and caveolin‐1. The FASEB Journal 2011, 25, 3401-3415, 10.1096/fj.11-184861.
- Pu Wang; Pei‐Pei Guan; Chuang Guo; Fei Zhu; Konstantinos Konstantopoulos; Zhan‐You Wang; Fluid shear stress‐induced osteoarthritis: roles of cyclooxygenase‐2 and its metabolic products in inducing the expression of proinflammatory cytokines and matrix metalloproteinases. The FASEB Journal 2013, 27, 4664-4677, 10.1096/fj.13-234542.
- Weifeng Lin; Jacob Klein; Recent Progress in Cartilage Lubrication. Advanced Materials 2021, 33, 2005513, 10.1002/adma.202005513.
- Kimberly A. Waller; Ling X. Zhang; Khaled A. Elsaid; Braden C. Fleming; Matthew L. Warman; Gregory D. Jay; Role of lubricin and boundary lubrication in the prevention of chondrocyte apoptosis. Proceedings of the National Academy of Sciences 2013, 110, 5852-5857, 10.1073/pnas.1219289110.
- Edward D. Bonnevie; Michelle L. Delco; Lena R. Bartell; Naveen Jasty; Itai Cohen; Lisa A. Fortier; Lawrence J. Bonassar; Microscale frictional strains determine chondrocyte fate in loaded cartilage. Journal of Biomechanics 2018, 74, 72-78, 10.1016/j.jbiomech.2018.04.020.
- Jasmine Seror; Linyi Zhu; Robert K Goldberg; Anthony Day; Jonathan M Klein; Supramolecular synergy in the boundary lubrication of synovial joints. Nature Communications 2015, 6, 6497, 10.1038/ncomms7497.
- Linyi Zhu; Jasmine Seror; Anthony J. Day; Nir Kampf; Jacob Klein; Ultra-low friction between boundary layers of hyaluronan-phosphatidylcholine complexes. Acta Biomaterialia 2017, 59, 283-292, 10.1016/j.actbio.2017.06.043.
- Ivonne Pasquali-Ronchetti; Daniela Quaglino; Giuseppe Mori; Barbara Bacchelli; Peter Ghosh; Hyaluronan–Phospholipid Interactions. Journal of Structural Biology 1997, 120, 1-10, 10.1006/jsbi.1997.3908.
- Sabrina Jahn; Jacob Klein; Hydration Lubrication: The Macromolecular Domain. Macromolecules 2015, 48, 5059-5075, 10.1021/acs.macromol.5b00327.
- Jacob Klein; Hydration lubrication. Friction 2013, 1, 1-23, 10.1007/s40544-013-0001-7.
- Sabrina Jahn; Jasmine Seror; Jacob Klein; Lubrication of Articular Cartilage. Annual Review of Biomedical Engineering 2016, 18, 235-258, 10.1146/annurev-bioeng-081514-123305.
More
Information
Subjects:
Materials Science, Biomaterials
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.3K
Revisions:
2 times
(View History)
Update Date:
03 Nov 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





