
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Patricia Luis | + 1823 word(s) | 1823 | 2021-10-28 09:09:24 | | | |
| 2 | Peter Tang | Meta information modification | 1823 | 2021-10-29 06:02:38 | | |
Video Upload Options
Modern definition of polyelectrolytes (PEs) according to various sources describes them as polymer chains with charged monomer units that can dissolve into a charged macroion and small counterions upon the PE dissolution in a polar solvent. The PEs properties can be ascribed to three major categories: origin, matrix, and charge. The PEs origin associates with the source of the raw polymer precursor, where the molecules such as proteins, cellulose, and deoxyribonucleic acid (DNA) represent the natural PEs. Opposed, with the development of chemical synthesis and especially polymeric chemistry a large field of synthetic polyelectrolytes have emerged to accommodate the needs of petrochemical, pharmaceutical, water recovery, and other industries. These synthetic routes may roughly be distinguished as ‘bottom-up’ by monomer polymerisation and ‘top-down’ by post-synthetic modification of neutral polymers.
1. Introduction

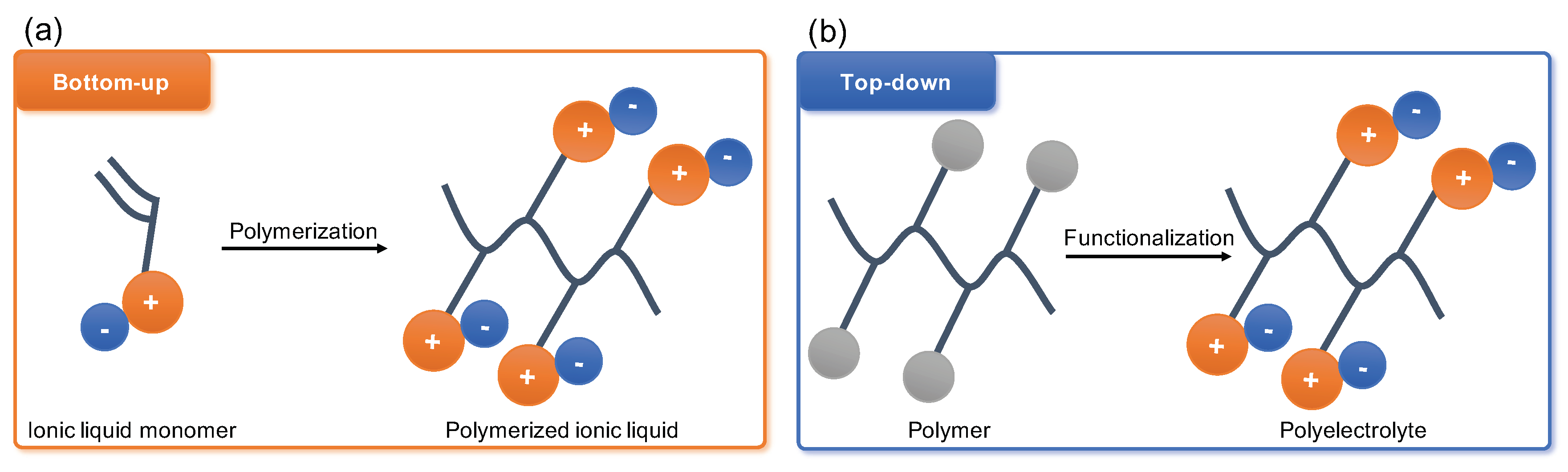
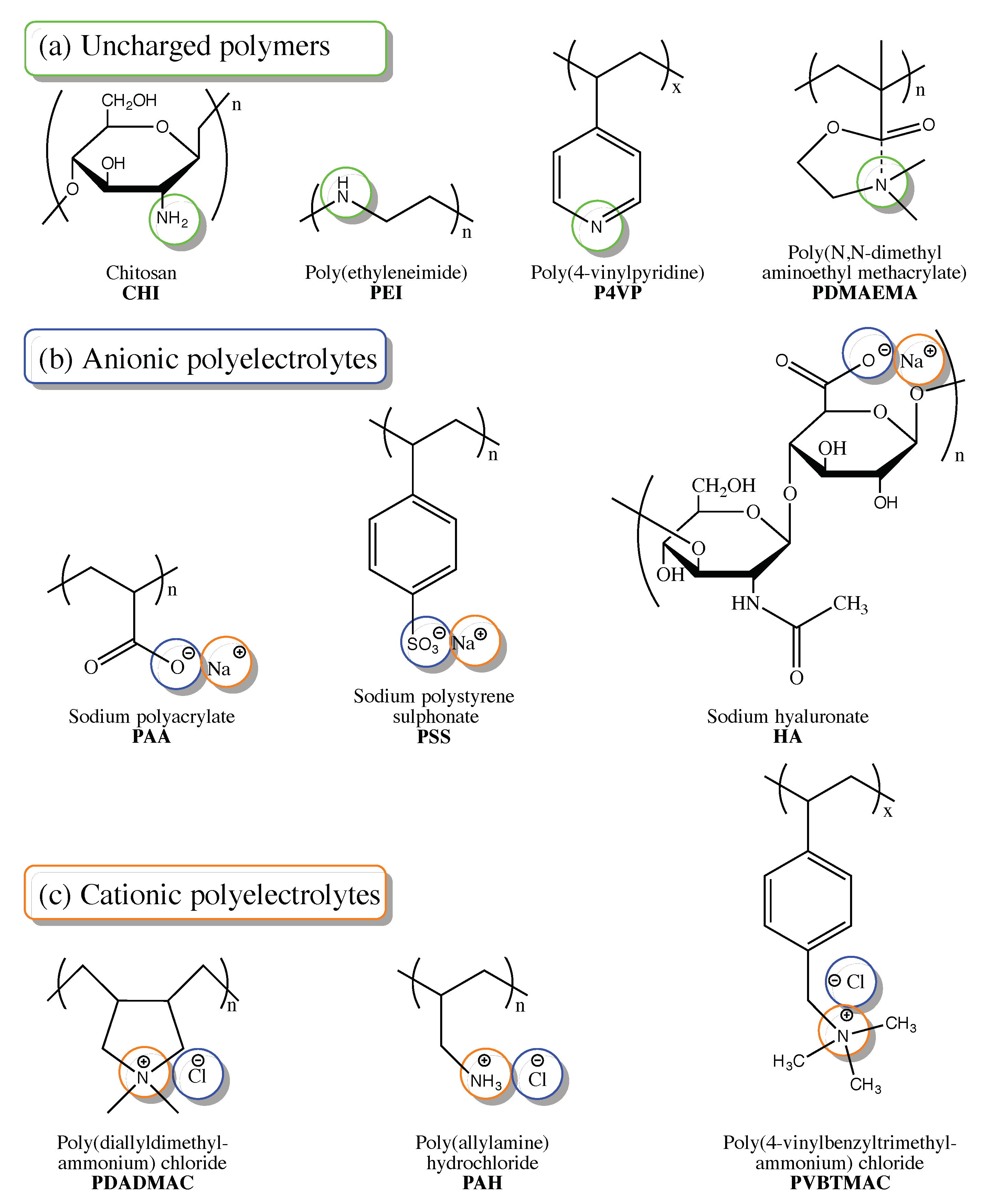
2. Post-Synthetic Modification of Polyelectrolytes
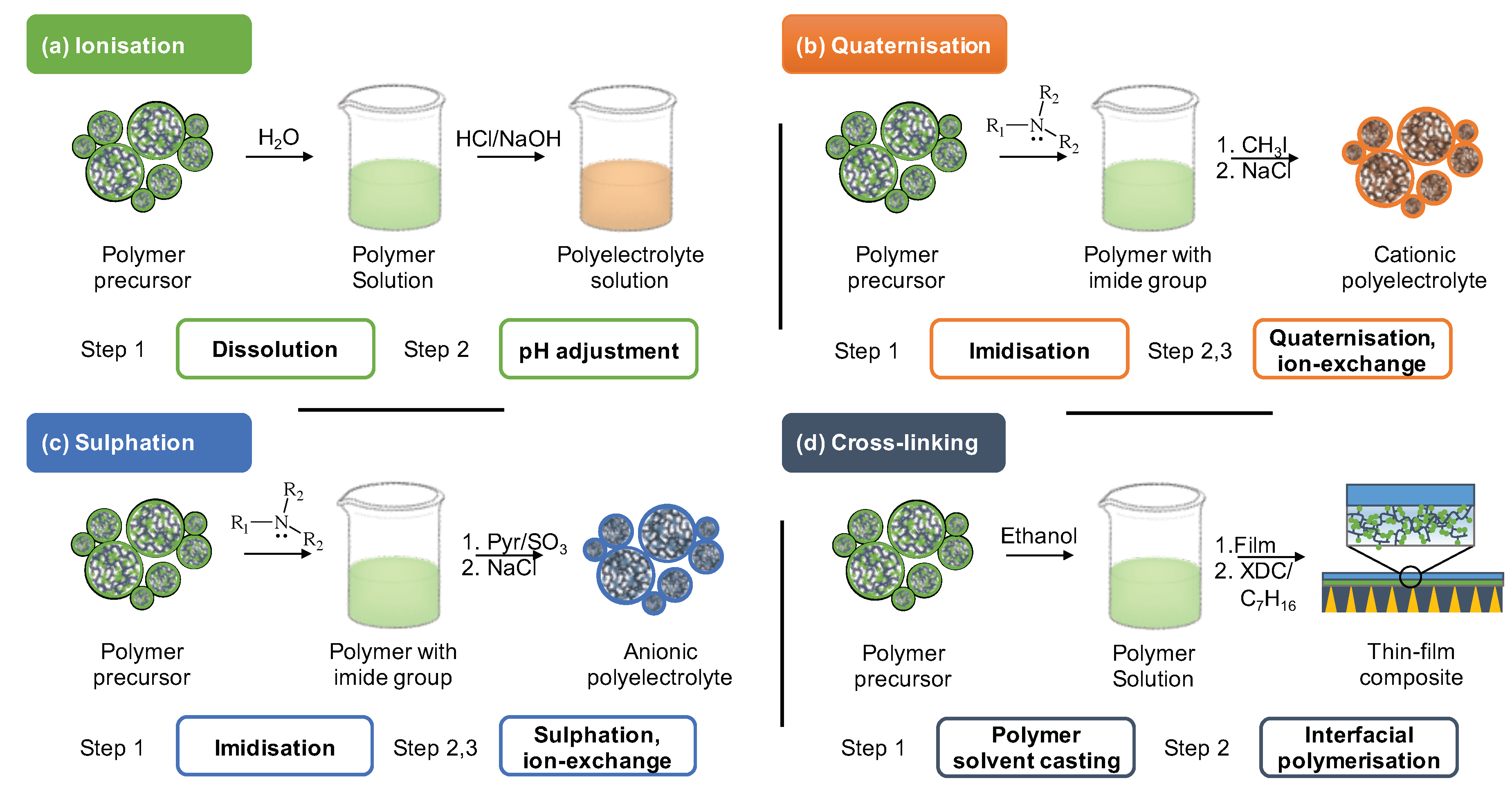
3. Membrane Preparation
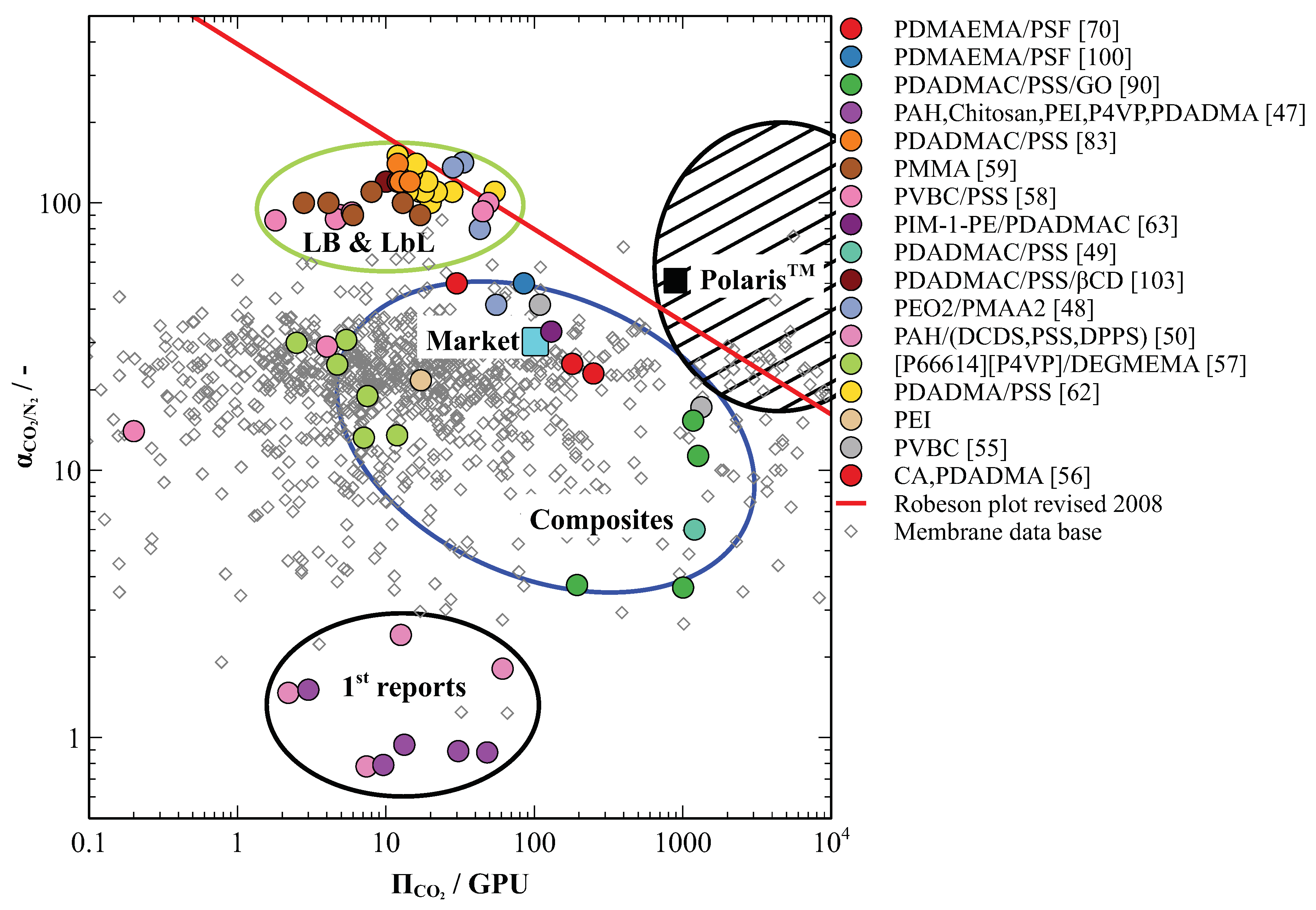
4. PE-Based Membranes Show High Selectivities for CO2 in Flue Gas Separation
References
- Climate Change 2014: Mitigation of Climate Change. Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Edenhofer, O.; Pichs-Madruga, R.; Sokona, Y.; Farahani, E.; Kadner, S.; Seyboth, K.; Adler, A.; Baum, I.; Brunner, S.; Eickemeier, P.; et al. (Eds.) Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014; pp. 418–510.
- Figueroa, J.D.; Fout, T.; Plasynski, S.; McIlvried, H.; Srivastava, R.D. Advances in CO2 capture technology—The U.S. Department of Energy’s Carbon Sequestration Program. Int. J. Greenh. Gas Control 2008, 2, 9–20.
- Luis, P.; Van Gerven, T.; Van der Bruggen, B. Recent developments in membrane-based technologies for CO2 capture. Prog. Energy Combust. Sci. 2012, 38, 419–448.
- Audus, H. Greenhouse gas mitigation technology: An overview of the CO2 capture and sequestration studies and further activities of the IEA greenhouse gas R and D programme. Energy 1997, 22, 217–221.
- Göttlicher, G.; Pruschek, R. Comparison of CO2 removal systems for fossil-fuelled power plant processes. Energy Convers. Manag. 1997, 38, 173–178.
- Wong, S.; Bioletti, R. Carbon Dioxide Separation Technologies. Carb. Energy Manag. 2002, 1–14.
- Brunetti, A.; Drioli, E.; Lee, Y.M.; Barbieri, G. Engineering evaluation of CO2 separation by membrane gas separation systems. J. Memb. Sci. 2014, 454, 305–315.
- Gibson, J.A.A.; Mangano, E.; Shiko, E.; Greenaway, A.G.; Gromov, A.V.; Lozinska, M.M.; Friedrich, D.; Campbell, E.E.B.; Wright, P.A.; Brandani, S. Adsorption Materials and Processes for Carbon Capture from Gas-Fired Power Plants: AMPGas. Ind. Eng. Chem. Res. 2016, 55, 3840–3851.
- Hart, A.; Gnanendran, N. Cryogenic CO2 capture in natural gas. Energy Procedia 2009, 1, 697–706.
- Aaron, D.; Tsouris, C. Separation of CO2 from flue gas: A review. Sep. Sci. Technol. 2005, 40, 321–348.
- Leung, D.Y.C.; Caramanna, G.; Maroto-Valer, M.M. An overview of current status of carbon dioxide capture and storage technologies. Renew. Sustain. Energy Rev. 2014, 39, 426–443.
- Powell, C.E.; Qiao, G.G. Polymeric CO2/N2 gas separation membranes for the capture of carbon dioxide from power plant flue gases. J. Memb. Sci. 2006, 279, 1–49.
- Wind, J.D.; Paul, D.R.; Koros, W.J. Natural gas permeation in polyimide membranes. J. Memb. Sci. 2004, 228, 227–236.
- Kentish, S.E.; Scholes, C.A.; Stevens, G.W. Carbon Dioxide Separation through Polymeric Membrane Systems for Flue Gas Applications. Recent Patents Chem. Eng. 2010, 1, 52–66.
- Sanders, D.F.; Smith, Z.P.; Guo, R.; Robeson, L.M.; McGrath, J.E.; Paul, D.R.; Freeman, B.D. Energy-efficient polymeric gas separation membranes for a sustainable future: A review. Polymer 2013, 54, 4729–4761.
- Budd, P.M.; Ghanem, B.S.; Makhseed, S.; McKeown, N.B.; Msayib, K.J.; Tattershall, C.E. Polymers of intrinsic microporosity (PIMs): Robust, solution-processable, organic nanoporous materials. Chem. Commun. 2004, 4, 230–231.
- McKeown, N.B.; Budd, P.M. Polymers of intrinsic microporosity (PIMs): Organic materials for membrane separations, heterogeneous catalysis and hydrogen storage. Chem. Soc. Rev. 2006, 35, 675–683.
- Lasseuguette, E.; Carta, M.; Brandani, S.; Ferrari, M.C. Effect of humidity and flue gas impurities on CO2 permeation of a polymer of intrinsic microporosity for post-combustion capture. Int. J. Greenh. Gas Control 2016, 50, 93–99.
- Park, H.B.; Jung, C.H.; Lee, Y.M.; Hill, A.J.; Pas, S.J.; Mudie, S.T.; Van Wagner, E.; Freeman, B.D.; Cookson, D.J. Polymers with cavities tuned for fast selective transport of small molecules and ions. Science 2007, 318, 254–258.
- Shamsipur, H.; Dawood, B.A.; Budd, P.M.; Bernardo, P.; Clarizia, G.; Jansen, J.C. Thermally rearrangeable PIM-polyimides for gas separation membranes. Macromolecules 2014, 47, 5595–5606.
- Cersosimo, M.; Brunetti, A.; Drioli, E.; Fiorino, F.; Dong, G.; Woo, K.T.; Lee, J.; Lee, Y.M.; Barbieri, G. Separation of CO2 from humidified ternary gas mixtures using thermally rearranged polymeric membranes. J. Memb. Sci. 2015, 492, 257–262.
- Woo, K.T.; Dong, G.; Lee, J.; Kim, J.S.; Do, Y.S.; Lee, W.H.; Lee, H.S.; Lee, Y.M. Ternary mixed-gas separation for flue gas CO2 capture using high performance thermally rearranged (TR) hollow fiber membranes. J. Memb. Sci. 2016, 510, 472–480.
- Dai, Y.; Johnson, J.R.; Karvan, O.; Sholl, D.S.; Koros, W.J. Ultem®/ZIF-8 mixed matrix hollow fiber membranes for CO2/N2 separations. J. Memb. Sci. 2012, 401–402, 76–82.
- Burmann, P.; Zornoza, B.; Téllez, C.; Coronas, J. Mixed matrix membranes comprising MOFs and porous silicate fillers prepared via spin coating for gas separation. Chem. Eng. Sci. 2014, 107, 66–75.
- Li, Y.; Xin, Q.; Wu, H.; Guo, R.; Tian, Z.; Liu, Y.; Wang, S.; He, G.; Pan, F.; Jiang, Z. Efficient CO2 capture by humidified polymer electrolyte membranes with tunable water state. Energy Environ. Sci. 2014, 7, 1489–1499.
- Fernández-Barquín, A.; Casado-Coterillo, C.; Palomino, M.; Valencia, S.; Irabien, A. Permselectivity improvement in membranes for CO2/N2 separation. Sep. Purif. Technol. 2016, 157, 102–111.
- Ward, W.J.; Robb, W.L. Carbon dioxide-oxygen separation: Facilitated transport of carbon dioxide across a liquid film. Science 1967, 156, 1481–1484.
- Way, J.D.; Noble, R.D. Facilitated transport. In Membrane Handbook; Springer: New York, NY, USA, 1992; pp. 833–866.
- Deng, L.; Kim, T.J.; Hägg, M.B. Facilitated transport of CO2 in novel PVAm/PVA blend membrane. J. Memb. Sci. 2009, 340, 154–163.
- Zhang, H.; Tian, H.; Zhang, J.; Guo, R.; Li, X. Facilitated transport membranes with an amino acid salt for highly efficient CO2 separation. Int. J. Greenh. Gas Control 2018, 78, 85–93.
- Qu, Z.; Wu, H.; Zhou, Y.; Yang, L.; Wu, X.; Wu, Y.; Ren, Y.; Zhang, N.; Liu, Y.; Jiang, Z. Constructing interconnected ionic cluster network in polyelectrolyte membranes for enhanced CO2 permeation. Chem. Eng. Sci. 2019, 199, 275–284.
- Lilleby Helberg, R.M.; Dai, Z.; Ansaloni, L.; Deng, L. PVA/PVP blend polymer matrix for hosting carriers in facilitated transport membranes: Synergistic enhancement of CO2 separation performance. Green Energy Environ. 2019.
- Morozova, S.M.; Shaplov, A.S.; Lozinskaya, E.I.; Vlasov, P.S.; Sardon, H.; Mecerreyes, D.; Vygodskii, Y.S. Poly(ionic liquid)-based polyurethanes having imidazolium, ammonium, morpholinium or pyrrolidinium cations. High Perform. Polym. 2017, 29, 691–703.
- Teodoro, R.M.; Tomé, L.C.; Mantione, D.; Mecerreyes, D.; Marrucho, I.M. Mixing poly(ionic liquid)s and ionic liquids with different cyano anions: Membrane forming ability and CO2/N2 separation properties. J. Memb. Sci. 2018, 552, 341–348.
- Whitley, J.W.; Jeffrey Horne, W.; Shannon, M.S.; Andrews, M.A.; Terrell, K.L.; Hayward, S.S.; Yue, S.; Mittenthal, M.S.; O’Harra, K.E.; Bara, J.E. Systematic Investigation of the Photopolymerization of Imidazolium-Based Ionic Liquid Styrene and Vinyl Monomers. J. Polym. Sci. Part A Polym. Chem. 2018, 56, 2364–2375.
- Nellepalli, P.; Tomé, L.C.; Vijayakrishna, K.; Marrucho, I.M. Imidazolium-Based Copoly(Ionic Liquid) Membranes for CO2/N2 Separation. Ind. Eng. Chem. Res. 2019, 58, 2017–2026.
- Zulfiqar, S.; Mantione, D.; El Tall, O.; Ruipérez, F.; Sarwar, M.I.; Rothenberger, A.; Mecerreyes, D. Pyridinium Containing Amide Based Polymeric Ionic Liquids for CO2/CH4 Separation. ACS Sustain. Chem. Eng. 2019, 7, 10241–10247.
- Yuan, J.; Mecerreyes, D.; Antonietti, M. Poly(ionic liquid)s: An update. Prog. Polym. Sci. 2013, 38, 1009–1036.
- Nishimura, N.; Ohno, H. 15Th Anniversary of Polymerised Ionic Liquids. Polymer 2014, 3289–3297.
- Xu, W.; Ledin, P.A.; Shevchenko, V.V.; Tsukruk, V.V. Architecture, Assembly, and Emerging Applications of Branched Functional Polyelectrolytes and Poly(ionic liquid)s. ACS Appl. Mater. Interfaces 2015, 7, 12570–12596.
- Shaplov, A.S.; Ponkratov, D.O.; Vygodskii, Y.S. Poly(ionic liquid)s: Synthesis, properties, and application. Polym. Sci. Ser. B 2016, 58, 73–142.
- Tomé, L.C.; Marrucho, I.M. Ionic liquid-based materials: A platform to design engineered CO2 separation membranes. Chem. Soc. Rev. 2016, 45, 2785–2824.
- Mortimer, D.A. Synthetic polyelectrolytes—A review. Polym. Int. 1991, 25, 29–41.
- Laschewsky, A. Recent trends in the synthesis of polyelectrolytes. Curr. Opin. Colloid Interface Sci. 2012, 17, 56–63.
- Loeb, S.; Sourirajan, S.; Weaver, D.E. High Flow Porous Membranes for Separating Water From Saline Solutions. US 3133137, 12 May 1964.
- Paul, D.R. Gas transport in homogeneous multicomponent polymers. J. Memb. Sci. 1984, 18, 75–86.
- Robeson, L.M. The upper bound revisited. J. Memb. Sci. 2008, 320, 390–400.
- Baker, R.W.; Low, B.T. Gas separation membrane materials: A perspective. Macromolecules 2014, 47, 6999–7013.
- Robeson, L.M. Correlation of separation factor versus permeability for polymeric membranes. J. Memb. Sci. 1991, 62, 165–185.
- Merkel, T.C.; Lin, H.; Wei, X.; Baker, R. Power plant post-combustion carbon dioxide capture: An opportunity for membranes. J. Memb. Sci. 2010, 359, 126–139.
- Krasemann, L.; Tieke, B. Composite membranes with ultrathin separation layer prepared by self-assembly of polyelectrolytes. Mater. Sci. Eng. C 1999, 8–9, 513–518.
- Van Ackern, F.; Krasemann, L.; Tieke, B. Ultrathin membranes for gas separation and pervaporation prepared upon electrostatic self-assembly of polyelectrolytes. Thin Solid Films 1998, 327–329, 762–766.





