Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ian Macreadie | + 4258 word(s) | 4258 | 2021-10-21 05:50:43 | | | |
| 2 | Catherine Yang | Meta information modification | 4258 | 2021-10-21 07:40:32 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Macreadie, I.; Dhakal, S.; Kushairi, N.; Phan, C.W.; Adhikari, B.; Sabaratnam, V. Dietary Polyphenols to Target Alzheimer’s Disease. Encyclopedia. Available online: https://encyclopedia.pub/entry/15203 (accessed on 29 May 2026).
Macreadie I, Dhakal S, Kushairi N, Phan CW, Adhikari B, Sabaratnam V. Dietary Polyphenols to Target Alzheimer’s Disease. Encyclopedia. Available at: https://encyclopedia.pub/entry/15203. Accessed May 29, 2026.
Macreadie, Ian, Sudip Dhakal, Naufal Kushairi, Chia Wei Phan, Benu Adhikari, Vikineswary Sabaratnam. "Dietary Polyphenols to Target Alzheimer’s Disease" Encyclopedia, https://encyclopedia.pub/entry/15203 (accessed May 29, 2026).
Macreadie, I., Dhakal, S., Kushairi, N., Phan, C.W., Adhikari, B., & Sabaratnam, V. (2021, October 21). Dietary Polyphenols to Target Alzheimer’s Disease. In Encyclopedia. https://encyclopedia.pub/entry/15203
Macreadie, Ian, et al. "Dietary Polyphenols to Target Alzheimer’s Disease." Encyclopedia. Web. 21 October, 2021.
Copy Citation
Dietary polyphenols may provide various levels of protection for neuronal health. This entry extensively examines this topic tabulating the in vivo and in vitro studies that have been performed, the methods used, the doses and duration of treatments, and most importantly the outcomes. The entry can be particularly useful as a reference and for those embarking on studies to further exploit dietary polyphenols for protection against neurodegenerative diseases such as Alzheimer's Disease.
Alzheimer’s Disease
nutraceuticals
pharmaceuticals
1. Introduction
Deaths due to Alzheimer’s Disease (AD) and other dementias are a major cause of mortality in the elderly worldwide, and the rate is increasing rapidly with a doubling time of 20 years [1]. AD is an age-related neurodegenerative disease that leads to cognitive impairment and death. Neuronal synapsis disruption, accumulation of amyloid plaques in brain, formation of neurofibrillary tangles in neuronal cells, loss of cellular homeostasis and accumulation of oxidative stress are major hallmarks of the disease [2]. However, mitochondrial dysfunction, loss of protein and lipid homeostasis, alterations in biometal distribution, cellular senescence, loss of nutrient sensing and accumulation of misfolded proteins are also associated with the AD [3]. Despite the efforts of more than three decades of research, the precise cause of AD has not been found. Many hypotheses have been made to address the major molecular events in the neuronal cells with AD (refer to Figure 1) [2]. Polyphenolic compounds have been reported to have multiple effects in cells including inducing antioxidant activity, induction of autophagy, restoration of lipid homeostasis, antiproliferative property, anti-proteinopathies, inhibition of choline esterases, anti-inflammatory activity, metal chelation, clearance of lipofuscin and others (refer to Table 1). This review details how polyphenols exert their neuroprotective role at the cellular level helping to prevent and possibly cure AD.
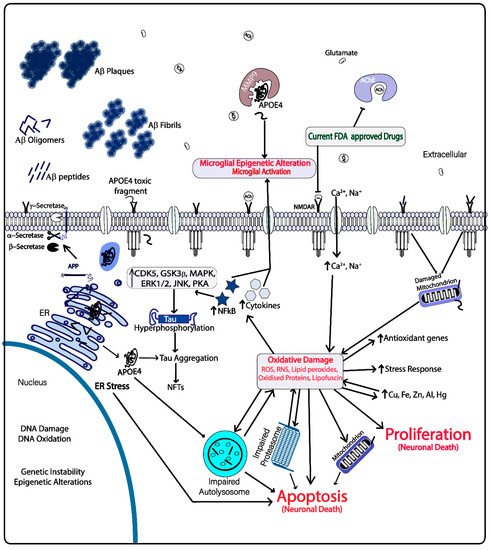
Figure 1. Current drug targets and molecular events occurring in the Alzheimer’s Disease (AD) brain microenvironment.
Table 1. Neuroprotective roles of some polyphenols for AD.
| Polyphenol | Analytical System | EPCa/ROAb | Effects of Polyphenols at Cellular Level | Effects in Relation to AD | Reference |
|---|---|---|---|---|---|
| Quercetin | In vitro | NA | mTORC inhibitor | Induces autophagy, anti-amyloidogenic, inhibits proteasomal degradation, antioxidant, restores biometal distribution, antiproliferative and enhances neuronal synapsis | [4][5][6][7][8] |
| ARPE 19 cells | 2 μM | TFEB activation | |||
| APPswe cells | 10 μM | Inhibits Aβ fibril formation | |||
| Rat neonatal cardiomyocytes | 5 μM | Inhibits all the catalytic subunits of proteasome | |||
| In vitro | NA | Chelates iron | |||
| In vitro | NA | Reduces ROS and RNS | |||
| In silico and in vitro | NA | Inhibits acetyl choline esterase | |||
| Resveratrol | Tg6799 mice | 60 mg/kg/d for 60 d/oral administration | Reduces amyloid plaque formation | Induces autophagy, increases lysosomal biogenesis, restores lipid homeostasis, increases stress resistance, regulates cell cycle, antiproliferative, anti-apoptotic, increases longevity and anti-inflammatory | [9][10][11][12][13][14] |
| Primary neuronal culture | 30 μM | SIRT1 activation and NFκB inhibition | |||
| Obese healthy men clinical trial | 150 mg/d for 30 d/oral administration | TFEB activation | |||
| Human aortic endothelial cells | 50 μM | AMPK mediated LC3II activation | |||
| Human aortic endothelial cells | 10 μM | Decreases ROS and RNS, increases SOD | |||
| LNCaP cells | 20 μM | p53 regulation, PI3K/Akt/mTOR inhibition, induces FOXO transcriptional activity including cell cycle regulation and stress resistance | |||
| Epigallocatechin gallate (EGCG) | Human bladder cancer cell line T24 | 20 μg/ml | Inhibits Beclin1 suppressors and PI3K/Akt/mTOR | Induces autophagy, restores lipid homeostasis, anti-amyloidogenic, increases antioxidant capacity, restores impaired autophagosomes and biometal distribution, increases cell survival | [15][16][17][18][19] |
| Bovine aortic endothelial cells | 10 μM | Increases LC3II formation and activates AMPK/ULK1 | |||
| HepG2 cells | 40 μM | Degrades lipid droplets through Ca2+/CAMKKB AMPK dependent mechanism | |||
| In vitro | NA | Chelates zinc and copper | |||
| PC12 cells (rat pheochromocytoma | 100 μg/mL | Interacts with Aβ40 and changes its conformation, inhibits lipofuscin formation | |||
| Anthocyanin | Sprague–Dawley rats | 100 mg/kg/d for 28 d/oral administration | Restores calcium homeostasis and activates Nrf2 subsequently activating phase II detoxifying genes | Activates autophagy, increases expression of anti-oxidant genes, reduces ROS and increases cell survival | [20][21][22][23] |
| HT22 cells and primary cultures of hippocampal neurons | 0.1 mg/mL | Induces AMPK | |||
| In vitro | 0.005 mg/mL | ROS scavenging | |||
| HCC cell lines PLC/PRF/5 and HepG2 cells | 0.2 mg/mL | Increase expression of Beclin1, LC3 II | |||
| Kaempferol | SK-HEP-1 human hepatic cancer cell | 75 μM | Increases the levels of p-AMPK, LC3-II, Atg 5, Atg 7, Atg 12 and beclin 1, inhibits PI3K/Akt/mTOR | Reduces mitochondrial dysfunction, anti-proliferative, increases autophagy, increases unfolded protein response, reduces APOE4 fragmentation and associated toxicity | [24][25][26][27] |
| BALB/c nude mice | 150 mg/kg/d for 31 d/intraperitoneal injection | Activates DNMT methyltransferase ubiquitination | |||
| SCC-4, human tongue squamous cell carcinoma cell | 50 µM | Activates IRE1-JNK-CHOP signaling, downregulates ERK1/2 signaling which reduces MMP2 | |||
| Hydroxytyrosol | Male db/db (C57BL/6J) mice | 10 mg/kg/d for 8 weeks/oral administration | Activates Nrf2 and SIRT1/AMPK/PGC-1, reduces protein oxidation, increases NMDAR1 and NGF mRNA expression | Enhances autophagy, increases stress resistance and longevity, antioxidant, anti-inflammatory, restores lipid homeostasis and improves cognition | [28][29][30][31][32][33] |
| VECs cells | 50 μM | Activates AMPK/FOXO3a | |||
| VECs cells | 10 μM | Reduces ROS | |||
| VAFs from Sprague–Dawley rats | 25 μM | Increases LC3II/LC3I, Bcl1 and SIRT1 expression | |||
| HepG2 and Huh7 cells | 100 μM | Inhibits PI3K/Akt/mTOR, expression of IL1β & IL6, and NFκB DNA binding | |||
| Rat hepatocytes | 25 μM | Inhibits Acetyl CoA carboxylase, HMG CoA reductase, diacylglycerol acyl transferase | |||
| Oleuropein aglycone | Rat ventricular myocyte | 100 μM | Increases Bcl1 and LC3II expression, TFEB nuclear localization, LAMP1 and p62 expression | Induces autophagy, increases lysosomal biogenesis and reduces oxidative damage | [33][34][35] |
| Human SH-SY5Y neuroblastoma cells and rat RIN5F insulinoma cells | 50 μM | Inhibits MAOA, induces AMPK/ULK1, inhibits mTOR | |||
| Rat hepatocytes | 25 μM | Inhibits acetyl CoA carboxylase, HMG CoA reductase and diacylglycerol acyl transferase | |||
| Curcumin | Male Sprague–Dawley rats | 15 mg/kg/d for 4 weeks/subcutaneous injection | Activates AMPK and regulates lipid metabolism | Induces autophagy, restores lipid homeostasis, antioxidant, anti-amyloidogenic, anti-inflammatory, anti-apoptotic antiproliferative, increases lysosomal biogenesis and longevity |
[36][37][38][39][40][41][42][43][44][45] |
| Adult male Wistar rats | 30 mg/kg for 30 d/oral administration | Activates Nrf2, inhibits NFκB and mTOR | |||
| Adult Swiss male albino mice | 80 mg/kg/d for 7 d/intraperitoneal injection | Inhibits MaoB and reduces ROS | |||
| APPswe Tg2576 transgenic mice (chronic 500 ppm curcumin diet) | Blood curcumin level ~2 μM for 1 h/injection in right carotid artery | Inhibits formation of Aβ, oligomers, fibrils and plaques | |||
| Tsc2+/+, Tsc2−/− MEFs and HCT116 cells | 10 μM | Activates TFEB, increases levels of LC3 and inhibits pAkt | |||
| Sprague–Dawley rats’ primary cortical neurons | 10 μM | Upregulates SIRT1 and inhibits Bax | |||
| APP/PS1 double transgenic mice | 160 ppm for 6 months/oral administration | Inhibits PI3K/Akt/mTOR signaling, increases LC3I/II and Beclin1 expression | |||
| Myricetin | HepG2 Cells | 50 μM | Inhibits mTOR and increases LC3II expression | Induces autophagy, antiproliferative, increases stress resistance, longevity, antioxidant capacity and mitochondrial regeneration | [46][47][48] |
| Adipocytes differentiated from C3H10T1/2 cells | 10 μM | Activates SIRT1/SIRT3/SIRT5 | |||
| Male ICR mice | 50 mg/kg/d for 21 d/oral administration | Increases mitochondrial mass and increases PGC1α, SIRT1, TFAM, Nrf1 & FOXO1 | |||
| Urolithin A | C2C12 myoblasts | 50 μM | Induces mitophagy, increases LC3I/LC3II and activates AMPK signaling | Increases mitophagy, and autophagy, antioxidant, increases lysosomal biogenesis, anti-inflammatory, anti-amyloidogenic, improves cognition and longevity | [49][50] |
| Female APP/PS1 transgenic mice B6C3-Tg (APPswe, PS1dE9) 85Dbo/J and age-matched wild type mice | 300 mg/kg/d for 14 d/oral administration | Activates AMPK, decreases NFκB/MAPK/BACE1 activities and APP levels | |||
| Ferulic Acid | HeLa cells and mouse primary hepatocytes | 1 mM | Increases LC3 II and inhibits mTOR | Anti-apoptotic, anti-amyloidogenic, antioxidant, anti-inflammatory and induces autophagy | [51][52][53][54] |
| In vitro | NA | Inhibits Aβ aggregation and reduces ROS | |||
| (APP)swe/presenilin 1(PS1)dE9 (APP/PS1) mouse model | 5.3 mg/kg/d for 6 months/oral administration | Reduces amyloid deposition and interleukin-1 beta (IL-1β) levels | |||
| Acacetin | Drosophila melanogaster | 100 μM | Inhibits BACE1 | Anti-amyloidogenic, antioxidant, anti-inflammatory and induces autophagy | [55][56][57][58] |
| C57BL/6J mice | ∼10 mg/kg/d for 14 d/oral administration (gavage) | Inhibits MAPK and PI3K/Akt pathways | |||
| ICR mice | 100 mg/kg for 7 h/intraperitoneal injection | Increases LC3II, Atg5 and Atg7 expression, modulates TNF-α/IL-6 expression and suppresses TLR4 signaling | |||
| Baicalein | SH-SY5Y human neuroblastoma cells | 12.5 μM | Increases ROS scavenging and activates Nrf2 | Anti-amyloidogenic, anti-apoptotic, antioxidant, anti-inflammatory, inhibits excitotoxicity, stimulates neurogenesis and neuronal differentiation | [59][60][61][62][63][64][65] |
| In vitro | NA | Chelates iron | |||
| CHO/APPwt cells | 5 μM | Induces α-secretase and inhibits Aβ formation | |||
| In vitro | 30 μM | Dissociates amyloid aggregates, Aβ oligomerization and fibrillation | |||
| HeLa cells | 100 μM | Inhibits NFκB activation | |||
| C57BL/6J APP/PS1 mice | 80 mg/kg/d for 60 d/oral administration (drinking water) | Inhibits GSK3β mediated tau phosphorylation | |||
| Sprague-Dawley male rats | 20 mg/kg 30 min before and 2/4 h after onset of ischemia/intraperitoneal injection | Induces Bcl-2/Bcl-xL associated phosphorylation | |||
| Icariin | Primary cortical neurons prepared from E16-17 mouse embryos | 1.2 μM | Activates SIRT1 | Antioxidant, anti-amyloidogenic, reduces ER stress, increases synapsis and neuronal plasticity, inhibits tau hyperphosphorylation, increases cell viability, antiapoptotic and anti-inflammatory | [66][67][68][69][70][71][72][73] |
| Wistar rats | 60 mg/kg/d for 3 months/oral administration | Increases SOD activity | |||
| Tg2576 mouse model | 60 mg/kg/d for 3 months/oral administration | Reduces expression of BACE1 and APP | |||
| Sprague-Dawley rats | 120 mg/kg/d for 28 d/oral administration | Induces PSD95, BDNF, pTrkB, pAkt, and pCREB expression | |||
| SH-SY5Y cells | 1 μM | Inhibits GSK3β activation | |||
| PC12 cells | 10 μM | Inhibits JNK/p38, MAPK and p53 activity | |||
| HT29 and HCT116 | 20 μM | Inhibits NFκB signaling | |||
| Nobiletin | Male 3XTg-AD mice | 30 mg/kg/d for 3 months/intraperitoneal injection | Reduces Aβ levels and plaque formation in brain | Anti-amyloidogenic, increases stress resistance, neuronal synapsis and plasticity, antioxidant and anti-inflammatory | [74][75][76][77] |
| Male Sprague-Dawley rats | 25 mg/kg/d for 3d/intraperitoneal injection | Increases activity of Akt, CREB, BDNF and Bcl2, increases Nrf2, HO-1, SOD1 and GSH expression, reduces NFκB, MMP-9 and MDA expression | |||
| Genistein | In silico and in vitro | NA | Inhibits chymotrypsin-like activity of proteasomes | Antioxidant, increases degradation of Aβ, increases apoptosis, enhances autophagy and inhibits proteasomal protein degradation | [78][79][80][81] |
| LNCaP cells | 100 μM | Increases Kip1 and reduces IκBα/Bax | |||
| Human dermal fibroblasts (HDFa) | 30 μM | Increases TFEB expression | |||
| Human mammary gland tumor cells (MCF-7) | 0.5 μM | Enhances antioxidant gene expression | |||
| Luteolin | HT-29 cells | 50 μM | Reduces ROS, NFκB signaling, Cox2 expression, blocks JAK/STAT signaling | Anti-inflammatory, antioxidant, modulates autophagy and apoptosis, increases survival | [82][83][84][85][86] |
| Male Sprague-Dawley rat myocytes | 8 μM | Downregulates Bax expression, upregulates PI3k/Akt signaling and Bcl-2 expression | |||
| Human HCC cell line SMMC-772 | 100 μM | Increases expression of LC3B-II, Bcl1 and caspase 8 | |||
| Mangiferin | Swiss albino male rats | 15 mg/kg/d for 14 d/intraperitoneal injection | Increases ROS scavenging, activates Nrf2, inhibits NFκB signaling, increases GSH levels, decreases lipid peroxidation | Antioxidant, anti-apoptotic, chelates metals, increases stress resistance, autophagy, longevity, neuronal synapsis and plasticity | [87][88][89][90][91] |
| In vitro | NA | Rescues mitochondrial respiration, chelates iron | |||
| Male Swiss albino mice | 40 mg/kg/d for 21 d/oral administration | Reduces lipid peroxides and ROS/RNS induced by aluminum and restores regulation of BDNF and NGF | |||
| Human astroglioma U87MG, U373MG and CRT-MG cells | 100 μM | Inhibits PI3K/Akt signaling, MAPK pathway, MMP9 gene expression |
Footnotes: NA, not applicable; a EPC, minimum concentration of the polyphenols that have significant neuroprotective effect; b ROA, route of administration of polyphenols in in vivo models.
2. Multiple Targets of Polyphenols against AD
Polyphenols are a class of compounds that are commonly found in many plants. Four major classes of polyphenols including flavonoids, stilbenes, phenolics and lignans are highly regarded as potential therapeutics for neurodegeneration, cardiovascular diseases, cancer and obesity. Many more polyphenolic compounds are yet to be studied for their potency in AD and other neurodegenerative diseases. Polyphenols are classified according to their structure (reviewed in [92]). Structures of some important polyphenols that are described in the text are depicted in Figure 2.
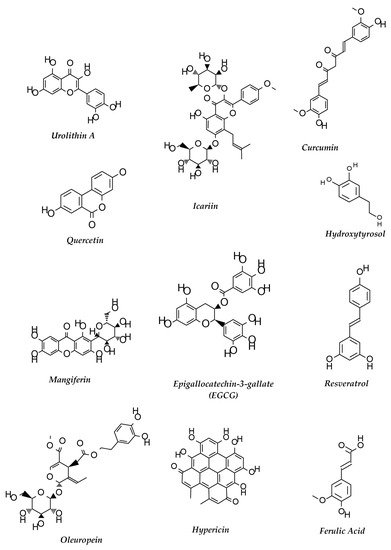
Figure 2. Structures of some polyphenols that show neuroprotective functions against AD.
Polyphenolic compounds abound in mushrooms and are one of their main antioxidants. They are mainly phenolic acids which can be divided into groups of either hydroxybenzoic acids and hydroxycinnamic acids derived from the non-phenolic molecules benzoic and cinnamic acid, respectively [93]. The most common benzoic acid derivatives present in mushrooms were reported as p-hydroxybenzoic, protocatechuic, gallic, gentisic, homogentisic, vanillic, 5-sulfosalicylic, syringic, ellagic and veratric acids as well as vanillin. Meanwhile, cinnamic acid derivatives mainly found in mushrooms were p-coumaric, o-coumaric, caffeic, ferulic, sinapic, 3-o-caffeoylquinic, 4-o-caffeoylquinic, 5-o-caffeoylquinic and tannic acids [93].
It is known that only plants synthesize flavonoids, while animals and fungi are not capable of it. However, accumulating studies indicate the presence of flavonoids in different edible mushrooms [94]. The presence of flavonoids in mushrooms could arise from absorption from the substrates where they grow or from neighboring plants by establishing symbiotic interactions via formation of mycorrhizae [95].
2.1. Polyphenols as Antioxidants
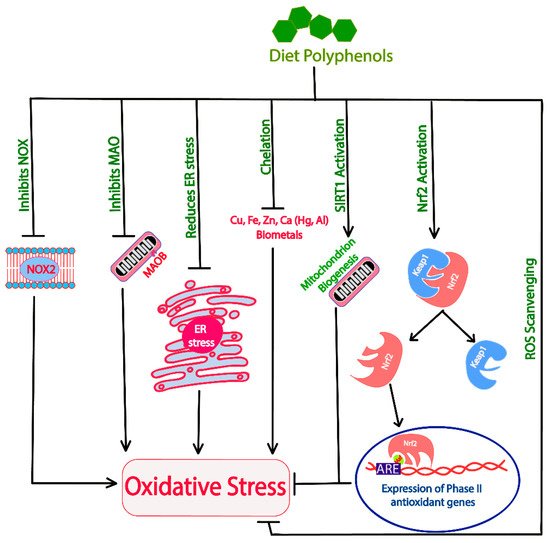
Naturally occurring polyphenols provide protection against neurodegeneration through their role as antioxidants [96]. Dietary polyphenols have direct ROS scavenging activity [97]. Several polyphenolic antioxidants identified in common edible mushrooms include protocatechuic acid, p-coumaric, and ellagic acid as well as gallic acid, pyrogallol, homogentisic acid, 5-sulfosalicylic acid, chlorogenic acid, caffeic acid, ferulic acid and quercetin [98][99]. Most of these polyphenols donate electrons to the free radicals thus neutralizing them, which ultimately reduces the levels of ROS within the cells. Polyphenols activate Nuclear factor erythroid 2-related factor 2 (Nrf2), a basic leucine zipper transcription factor. Nrf2 normally is complexed with Kelch-like ECH-associated protein 1 (Keap1) in the cellular environment inhibiting Nrf2′s nuclear translocation. Furthermore, Keap1 also facilitates ubiquitination and proteasomal degradation of Nrf2 [100]. The separation of Nrf2 from Keap1 leads to activation and nuclear translocation of Nrf2, where it complexes with musculoaponeurotic fibrosarcoma (Maf) proteins. This heteromeric Nrf2-Maf complex then binds with antioxidant response element (ARE) sequences located upstream to the phase II detoxifying genes upregulating their expression. Phase II antioxidant genes encode proteins, such as heme oxygenase 1, γ-glutamyl cysteine synthetase, peroxiredoxins, glutathione reductases, thioredoxin reductase, drug metabolizing and detoxification enzymes NAD(P)H quinone dehydrogenase 1, glutathione-S-transferase, uridine diphosphate-glucuronosyltransferase and regulators, transketolase, PPARγ-coactivator 1 β (PGC1-β), etc [101]. These proteins act in the cell as antioxidant proteins, having a major role in restoration of the redox imbalance and cellular signaling [102][103]. Additionally, polyphenols also elucidate their antioxidant property through inhibition of NADPH oxidase (NOX) activities [104]. NOX proteins are transmembrane proteins that signal the immune modulators through ROS generation [105]. Lower levels of ROS may be important for cellular signaling, however, at higher levels they can cause damage to the neuronal cells. These proteins, found to be involved in increasing Aβ-induced oxidative stress, could be potential therapeutic targets for AD [106].
Oxidative damage is more prominent when the damage is coupled with mitochondrial dysfunction. Enzymes such as monoamine oxidases (e.g., MaoB) increase the cellular stress by producing hydrogen peroxide [107]. In brains, monoamine oxidase activity of substrate neurotransmitters causes mitochondrial damage, while dietary polyphenols have been found to inhibit MaoB, thus decreasing the ROS generation and mitochondrial dysfunction [36]. Additionally, polyphenols also aid in regeneration of mitochondria in the cells through activation of the master regulator SIRT1 [108]. SIRT1 is a NAD+-dependent histone deacetylase enzyme that has multiple targets for deacetylation. SIRT1′s involvement in reducing oxidative stress comes from deacetylation of its substrate PGC-1α, which activates nuclear respiratory factors (Nrf1 and Nrf2) and peroxisome proliferator-activated receptor (PPARα) [109]. Further downstream, these molecules enhance the expression of transcription factor A, mitochondrial (TFAM) that initiates the transcription and replication of mitochondrial DNA ultimately causing the regeneration of mitochondria [109]. The activation AMPK, either directly or indirectly (through SIRT1 activation) activates PGC-1α, thus helping in mitochondrial biogenesis.
Biometals such as iron and copper are the major contributors of ROS formation in defunct mitochondria [110]. Quercetin, baicalein, curcumin, etc., are found to provide a protective antioxidant property also through biometal chelation [4][111][112]. Furthermore, alterations in biometal distribution in the neuronal cells is also an important hallmark of AD. The mechanism through which polyphenols act as antioxidants in the cellular environment is schematically presented in Figure 3. Antioxidants can also act as pro-oxidants in certain sub-optimal concentrations and cause oxidative damage to the cells. Thus, their optimum concentration needs to be considered prior to their application.
2.2. Modulation of Protein Homeostasis and Longevity with Polyphenols
Dietary polyphenols modulate the protein quality control mechanisms increasing the cellular efficiency to clear misfolded proteins. Apart from induction of autophagic clearance, the UPR and ubiquitin proteasome system are also modulated by dietary polyphenols [113][114][115]. The ability of polyphenols to activate lysosomal biogenesis and increase longevity make them an important class of neuroprotective compounds [5][34][37]. In addition, some of the polyphenols like EGCG and curcuminoids reduced the lipofuscin granules in cells, which normally are impossible to degrade or exocytose from the cell [15][116]. Reduction of lipofuscin in the cell can contribute to the restoration of the protein homeostasis by reducing the damage to autophagosomes and proteasomes.
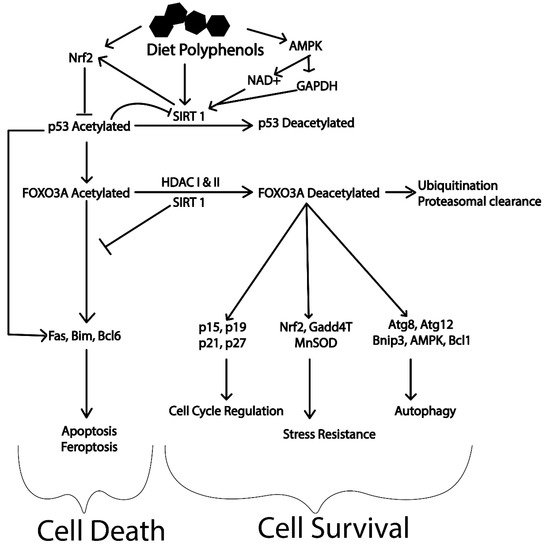
Most of the polyphenolic compounds act through upregulation of the expression of the master regulator SIRT1 [117]. The SIRT1 protein has been found to have multiple targets that play a vital role in regulating major cellular processes (refer to Figure 4) [117]. The activation of AMPK/Unc-51 like autophagy activating kinase 1 (ULK1), transcription factor EB (TFEB), Fork head box O transcription factors (FOXO), deacetylation of p53 and inhibition of PI3K/Akt/mTOR, NFkB, MAPK and the c-Jun N-terminal kinases (c-JNK) pathway are important cellular processes that will induce autophagy through SIRT1 [118][119][120]. Most of these molecular targets are deacetylation substrates of SIRT1. Activation of transcription factors like TFEB reinforces the cellular autophagy by activating lysosomal biogenesis. TFEB itself is another master regulator for the coordinated lysosomal expression and regulation (CLEAR) network. The CLEAR network has important roles in various cellular processes. Energy metabolism, DNA metabolism, steroid biosynthesis, protein clearance, haemoglobin degradation, antigen presentation, phagocytosis and signal transduction are important events regulated by TFEB [121][122].
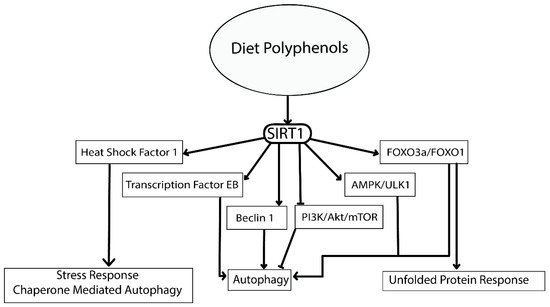
Similarly, SIRT1 has a significant role in determining cellular fate via Fork head transcription factors (FOXO1 and FOXO3). The deacetylated form of these transcription factors are major contributors of autophagy activation, cell cycle arrest, stress resistance (expression of manganese superoxide dismutase) and immune modulation. Reduction in the levels of FOXO by ubiquitination and proteasomal degradation with the help of SIRT1 reduces the levels of acetylated forms. Reduction in acetylated FOXO’s suppresses cell death caused by apoptosis driving cells towards survival and increasing longevity (refer to Figure 5) [119][123]. This is of particular interest for neurodegenerative diseases, where survival of neuronal cell after damage is crucial. It has been illustrated that polyphenols activate these master regulators of longevity (Nrf2, SIRT1 and AMPK) providing unprecedented protection against various disease [103][124][125]. However, limited bioavailability of these dietary polyphenols in human has limited their application. Polyphenols such as hydroxytyrosol, oleuropein aglycone, curcumin, resveratrol, rotenone, rutin, myricetin, urolithin A, epigallocatechin 3-gallate (EGCG), ferulic acid, genipin, etc. have been reported to induce autophagy. The olive oil polyphenol, hydroxytyrosol activates AMPK pathway and is reported to reduce Aβ levels in mouse models of AD [28][126]. Similarly, oleuropein aglycone has been reported to activate SIRT1/AMPK/mTOR and TFEB mediated autophagy [127][128].
Curcumin, one of the most studied polyphenols, has multifactorial benefits in balancing the protein homeostasis by activation of AMPK/ULK1 and inhibition of PI3K/Akt/mTOR through activation of SIRT1 [38]. EGCG, a catechin family polyphenol, inhibits the suppressors (Bcl2 and Bcl-XL) of Beclin1. However, the activity of this polyphenol is also dependent on the concentration of the compound. A higher concentration of EGCG inhibits autophagy and induces apoptosis, whereas, lower concentrations induce autophagy that also degrade lipid droplets through a Ca2+/CAMKKB/AMPK dependent mechanism. Thus, the concentration of polyphenols is a crucial factor before considering it as a therapeutic option. EGCG has also been reported to reduce the catalytic activity of 19S and 20S proteasomal proteins, deactivate NFkB pathway and enhance p53 tumour suppressor protein expression [129]. An important feature of EGCG also includes its ability to inhibit lipofuscin formation, which otherwise impairs autophagy and the proteasome during ageing [15].
Resveratrol is another important polyphenol frequently studied for its beneficial effect in increasing longevity and balances cellular protein homeostasis. The activation of SIRT1/AMPK and extracellular signal-regulated kinases (ERK1/2) is the molecular mechanism by which this polyphenol was found to be neuroprotective [9][130][131]. The metabolite of ellagitannin, urolithin A, extracted from pomegranate has been reported to activate autophagy through SIRT1 activation [132]. Furthermore, the natural compound was also found to increase mitophagy and longevity in a Caenorhabditis elegans (C. elegans) model that has provided insight on human neurodegeneration [49]. Quercetin has shown multiple benefits in human health by enhancing autophagy through SIRT1activation, inhibiting proteasomal degradation (inhibition of all the catalytic subunits), reducing proliferation and activating apoptosis [133]. Apart from autophagy inducers, hesperitin and hesperidin have also been reported to have negative effects on Aβ-induced autophagy and glucose metabolism impairment [134][135].
2.3. Polyphenols and Cellular Lipid Balance
Polyphenols are also considered as potential therapeutic agents against obesity and other life-threatening conditions [136][137][138]. This property of polyphenols is associated with the activation of AMPK, which targets lipid metabolism as well [139]. Activation of AMPK decreases the activity of acetyl CoA carboxylase, HMG-CoA reductase and diacylglycerol acyl transferase, and thus avoids hepatic accumulation of lipids [140][141]. These actions of AMPK reduce the levels of free fatty acids as well as the complex lipids. Polyphenols are also found to inhibit the adipogenesis by inhibiting proteins like PPARγ [142][143]. Additionally, as explained in previous sections, polyphenols increase autophagic clearance. Induction of autophagy is not only limited to restoring the protein balance but is also associated with the degradation of lipids to meet the energy demands of the cells. Thus, polyphenols can also reduce lipid accumulation in the intracellular environment [144]. AD is also termed as Type III diabetes due to its similarity with diabetes. High levels of cholesterol have been found to be associated with AD brains [145]. Lowering the levels of cholesterol has been an important approach for the treatment of AD, despite limited success. Furthermore, studies support increased activity of γ-secretase and β-secretase with higher levels of lipids in the membrane environment that could contribute to increased Aβ levels in the brain [146]. Considering these facts, polyphenols are hypothesized to have their neuroprotective action in part through the restoration of lipid homeostasis.
2.4. Anti-inflammatory Activity of Polyphenols
ROS act as signaling molecules for induction and release of pro-inflammatory mediators including NFκB and cytokines. NFκB exists in an inactivated form bound to an inhibitor referred to as p65/p50 dimer in normal conditions [147]. When this complex gets activated by increased ROS, the p65/p50 dimer translocates to the nucleus upregulating expression of the inflammatory markers [148]. The expression of these inflammatory mediators inside the cells triggers the downstream process of inflammation. Deacetylation of NFκB through the action of SIRT1 at specific amino acid residues renders it inactivated and reduces the inflammatory response by reducing the expression of downstream genes [147]. Since polyphenols are antioxidants capable of lowering the ROS in the cells, they can downregulate the expression of proinflammatory mediators [149]. However, the highest anti-inflammatory activity of polyphenols is attributed to their ability to activate the master regulator SIRT1 [150]. Many polyphenols have been reported to have an anti-inflammatory effect which could provide the basis for protection against diseases with chronic neuroinflammation/inflammation.
2.5. Polyphenols as Anti-amyloid Agents
Oleuropein, an olive polyphenol, is found to increase α-secretase activity. Thus, it prevents cells from producing Aβ: instead such activity results in the formation of the Aα peptide [151]. Formation of Aα instead of Aβ is anti-amyloidogenic, which may be helpful in reducing the Aβ-associated toxicity. Some polyphenols (such as rutin) reduce the β-secretase activity [6]. Similarly, other polyphenols disaggregate the amyloid aggregates in vitro [6][152]. Furthermore, the ability of polyphenols to lower the cholesterol levels in cells also favors the reduced activity of β-secretase and γ-secretase [6][145]. Apart from the anti-amyloid functions, polyphenols also possess the ability to inhibit tau aggregation [153].
Through characterization of the cell-free extracts of different bacteria, fungi and yeast, Lee et al. (2007) identified the BACE1 inhibitory effects of different mushrooms [154]. Mushroom species having anti-BACE1 effects were Flammulina velutipes, Pleurotus ostreatus, Grifola frondosa, Dictyophora echinovolvata, Fomitella fraxinea and Inonotus obliquus. Hispidin, a polyphenolic compound found in abundance in the mushroom Phellinus linteus inhibits BACE1 non-competitively and scavenges free radicals [155]. BACE1’s inhibitory effect of Auricularia polytricha has also been indicated to be hispidine mediated [156].
2.6. Polyphenols in Cognition and Synapsis
Polyphenolic compounds like α-isocubebenol, tacrine and their derivatives, 2′,4′-dihydroxy-6′methoxy-3′,5′-dimethyl-dihydrochalcone, tetrahydropyranodiquinolin-8-amines, quercetin and tiliroside have been shown to have neuroprotective properties attributed to their inhibiting activity against acetylcholine esterase [157][158][159][160]. In addition, some other polyphenols, including genistein, luteolin-7-O-rutinoside and silibinin, are reported to have a moderate effect on the butyrylcholine esterase [159]. Among the polyphenols, flavonoids are an important class of polyphenols that have anti-choline esterase activity [161]. Flavonoids extracted from Ginkgo biloba have been reported to have inhibitory effects against acetyl choline esterase [162]. Molecular docking experiments revealed the mechanism of action of quercetin was through strong hydrogen bond formation with certain amino acids of AChE, thus leading to competitive inhibition of AChE. Similarly, macluraxanthone exhibited non-competitive type interference with the activity of acetyl choline esterase [161]. The combination of numerous hydrogen bonds with several amino acids and hydrophobic interaction may be responsible for how these polyphenols inhibit acetylcholine esterase activity [163].
Polyphenols exert neuroprotective effects in experimental systems but there is a need to translate this in guidelines for neuroprotection of aging populations. For translation of animal studies to human trials, dose accuracy plays a critical role. For example, consider resveratrol levels in Table 1: an effective dose in mice is 60 mg/kg/d by oral administration. In humans this translates to ~290 mg for a 60 kg person per day [164]. Such levels are rarely reached. In the case of resveratrol, the suggested daily intake is 200 mg/day and this is unlikely to be a protective level. In addition, alterations in polyphenol administration routes may reduce the amount of polyphenol to be used on daily basis, signifying the benefits of alternative administration strategy. However, long term uptake of the polyphenol could still have beneficial effects in lower doses. On the other hand, some nutraceutical products may contain the polyphenol at more than the optimal amount, which could have negative effects in brain health [165]. This bimodal activity of polyphenols should be highly considered before translating the beneficial effects of the polyphenols for human use.
References
- Macreadie, I.G.; Arvanitis, C.; Bharadwaj, P. Finding chemopreventatives to reduce amyloid beta in yeast. Neural Regen. Res. 2016, 11, 244–245.
- Sharma, P.; Srivastava, P.; Seth, A.; Tripathi, P.N.; Banerjee, A.G.; Shrivastava, S.K. Comprehensive review of mechanisms of pathogenesis involved in Alzheimer’s disease and potential therapeutic strategies. Prog. Neurobiol. 2019, 174, 53–89.
- Kim, C.A.; Lim, S.; Kim, K.Y. Metal Ion Effects on Aβ and Tau Aggregation. Int. J. Mol. Sci. 2018, 19, 128.
- Xiao, L.; Luo, G.; Tang, Y.; Yao, P. Quercetin and iron metabolism: What we know and what we need to know. Food Chem. Toxicol. 2018, 114, 190–203.
- Huang, Y.; Chen, Y.; Shaw, A.M.; Goldfine, H.; Tian, J.; Cai, J. Enhancing TFEB-mediated cellular degradation pathways by the mTORC1 inhibitor quercetin. Oxidative Med. Cell. Longev. 2018, 2018, 5073420.
- Jimenez-Aliaga, K.; Bermejo-Bescos, P.; Benedi, J.; Martin-Aragon, S. Quercetin and rutin exhibit antiamyloidogenic and fibril-disaggregating effects in vitro and potent antioxidant activity in APPswe cells. Life Sci 2011, 89, 939–945.
- Primikyri, A.; Mazzone, G.; Lekka, C.; Tzakos, A.G.; Russo, N.; Gerothanassis, I.P. Understanding zinc(II) chelation with quercetin and luteolin: A combined NMR and theoretical study. J. Phys. Chem. B 2015, 119, 83–95.
- Dosenko, V.E.; Nagibin, V.S.; Tumanovskaya, L.V.; Zagorii, V.Y.; Moibenko, A.A. Effect of quercetin on the activity of purified 20S and 26S proteasome and proteasomal activity in isolated cardiomyocytes. Biomed. Chem. 2007, 1, 40–44.
- Chen, Y.; Shi, G.W.; Liang, Z.M.; Sheng, S.Y.; Shi, Y.S.; Peng, L.; Wang, Y.P.; Wang, F.; Zhang, X.M. Resveratrol improves cognition and decreases amyloid plaque formation in Tg6799 mice. Mol. Med. Rep. 2019, 49, 3783–3790.
- Pallàs, M.; Casadesús, G.; Smith, M.A.; Coto-Montes, A.; Pelegri, C.; Vilaplana, J.; Camins, A. Resveratrol and neurodegenerative diseases: Activation of SIRT1 as the potential pathway towards neuroprotection. Curr. Neurovas. Res. 2009, 6, 70–81.
- Suvorova, I.I.; Knyazeva, A.R.; Pospelov, V.A. Resveratrol-induced p53 activation is associated with autophagy in mouse embryonic stem cells. Biochem. Biophys. Res. Commun. 2018, 503, 2180–2185.
- Chen, J.; Zhou, Y.; Mueller-Steiner, S.; Chen, L.F.; Kwon, H.; Yi, S.; Mucke, L.; Gan, L. SIRT1 protects against microglia-dependent amyloid-β toxicity through inhibiting NF-κB signaling. J. Biol. Chem. 2005, 280, 40364–40374.
- Konings, E.; Timmers, S.; Boekschoten, M.V.; Goossens, G.H.; Jocken, J.W.; Afman, L.A.; Müller, M.; Schrauwen, P.; Mariman, E.C.; Blaak, E.E. The effects of 30 days resveratrol supplementation on adipose tissue morphology and gene expression patterns in obese men. Int. J. Obes. 2014, 38, 470–473.
- Chen, Q.; Ganapathy, S.; Singh, K.P.; Shankar, S.; Srivastava, R.K. Resveratrol induces growth arrest and apoptosis through activation of FOXO transcription factors in prostate cancer cells. PLoS ONE 2010, 5.
- Cai, S.; Yang, H.; Zeng, K.; Zhang, J.; Zhong, N.; Wang, Y.; Ye, J.; Tu, P.; Liu, Z. EGCG Inhibited Lipofuscin Formation Based on Intercepting Amyloidogenic β-Sheet-Rich Structure Conversion. PLoS ONE 2016, 11, e0152064.
- Qin, J.; Xie, L.P.; Zheng, X.Y.; Wang, Y.B.; Bai, Y.; Shen, H.F.; Li, L.C.; Dahiya, R. A component of green tea, (-)-epigallocatechin-3-gallate, promotes apoptosis in T24 human bladder cancer cells via modulation of the PI3K/Akt pathway and Bcl-2 family proteins. Biochem. Biophys. Res. Commun. 2007, 354, 852–857.
- Hyung, S.J.; Detoma, A.S.; Brender, J.R.; Lee, S.; Vivekanandan, S.; Kochi, A.; Choi, J.S.; Ramamoorthy, A.; Ruotolo, B.T.; Lim, M.H. Insights into antiamyloidogenic properties of the green tea extract (-)-epigallocatechin-3-gallate toward metal-associated amyloid-β species. Proc. Natl. Acad. Sci. USA 2013, 110, 3743–3748.
- Kim, H.S.; Montana, V.; Jang, H.J.; Parpura, V.; Kim, J.A. Epigallocatechin gallate (EGCG) stimulates autophagy in vascular endothelial cells: A potential role for reducing lipid accumulation. J. Biol. Chem. 2013, 288, 22693–22705.
- Zhou, J.; Farah, B.L.; Sinha, R.A.; Wu, Y.; Singh, B.K.; Bay, B.H.; Yang, C.S.; Yen, P.M. Epigallocatechin-3-Gallate (EGCG), a green tea polyphenol, stimulates hepatic autophagy and lipid clearance. PLoS ONE 2014, 9, e87161.
- Pacheco, S.M.; Soares, M.S.P.; Gutierres, J.M.; Gerzson, M.F.B.; Carvalho, F.B.; Azambuja, J.H.; Schetinger, M.R.C.; Stefanello, F.M.; Spanevello, R.M. Anthocyanins as a potential pharmacological agent to manage memory deficit, oxidative stress and alterations in ion pump activity induced by experimental sporadic dementia of Alzheimer’s type. J. Nutr. Biochem. 2018, 56, 193–204.
- Hwang, Y.P.; Choi, J.H.; Yun, H.J.; Han, E.H.; Kim, H.G.; Kim, J.Y.; Park, B.H.; Khanal, T.; Choi, J.M.; Chung, Y.C.; et al. Anthocyanins from purple sweet potato attenuate dimethylnitrosamine-induced liver injury in rats by inducing Nrf2-mediated antioxidant enzymes and reducing COX-2 and iNOS expression. Food Chem. Toxicol. 2011, 49, 93–99.
- Longo, L.; Platini, F.; Scardino, A.; Alabiso, O.; Vasapollo, G.; Tessitore, L. Autophagy inhibition enhances anthocyanin-induced apoptosis in hepatocellular carcinoma. Mol. Cancer Ther. 2008, 7, 2476–2485.
- Ullah, I.; Park, H.Y.; Kim, M.O. Anthocyanins Protect against Kainic Acid-induced Excitotoxicity and Apoptosis via ROS-activated AMPK Pathway in Hippocampal Neurons. Cns Neurosci. Ther. 2014, 20, 327–338.
- Kim, T.W.; Lee, S.Y.; Kim, M.; Cheon, C.; Ko, S.-G. Kaempferol induces autophagic cell death via IRE1-JNK-CHOP pathway and inhibition of G9a in gastric cancer cells. Cell Death Dis. 2018, 9, 875.
- Lin, C.-W.; Chen, P.-N.; Chen, M.-K.; Yang, W.-E.; Tang, C.-H.; Yang, S.-F.; Hsieh, Y.-S. Kaempferol Reduces Matrix Metalloproteinase-2 Expression by Down-Regulating ERK1/2 and the Activator Protein-1 Signaling Pathways in Oral Cancer Cells. PLoS ONE 2013, 8, e80883.
- Huang, W.W.; Tsai, S.C.; Peng, S.F.; Lin, M.W.; Chiang, J.H.; Chiu, Y.J.; Fushiya, S.; Tseng, M.T.; Yang, J.S. Kaempferol induces autophagy through AMPK and AKT signaling molecules and causes G 2/M arrest via downregulation of CDK1/cyclin B in SK-HEP-1 human hepatic cancer cells. Int. J. Oncol. 2013, 42, 2069–2077.
- Qiu, W.; Lin, J.; Zhu, Y.; Zhang, J.; Zeng, L.; Su, M.; Tian, Y. Kaempferol modulates DNA methylation and downregulates DNMT3B in bladder cancer. Cell. Physiol. Biochem. 2017, 41, 1325–1335.
- De Pablos, R.M.; Espinosa-Oliva, A.M.; Hornedo-Ortega, R.; Cano, M.; Arguelles, S. Hydroxytyrosol protects from aging process via AMPK and autophagy; a review of its effects on cancer, metabolic syndrome, osteoporosis, immune-mediated and neurodegenerative diseases. Pharmacol. Res. 2019, 143, 58–72.
- Zheng, A.; Li, H.; Xu, J.; Cao, K.; Li, H.; Pu, W.; Yang, Z.; Peng, Y.; Long, J.; Liu, J.; et al. Hydroxytyrosol improves mitochondrial function and reduces oxidative stress in the brain of db/db mice: Role of AMP-activated protein kinase activation. Br. J. Nutr. 2015, 113, 1667–1676.
- Zrelli, H.; Matsuoka, M.; Kitazaki, S.; Zarrouk, M.; Miyazaki, H. Hydroxytyrosol reduces intracellular reactive oxygen species levels in vascular endothelial cells by upregulating catalase expression through the AMPK-FOXO3a pathway. Eur. J. Pharmacol. 2011, 660, 275–282.
- Wang, W.; Jing, T.; Yang, X.; He, Y.; Wang, B.; Xiao, Y.; Shang, C.; Zhang, J.; Lin, R. Hydroxytyrosol regulates the autophagy of vascular adventitial fibroblasts through the SIRT1-mediated signaling pathway. Can. J. Physiol. Pharmacol. 2018, 96, 88–96.
- Zhao, B.; Ma, Y.; Xu, Z.; Wang, J.; Wang, F.; Wang, D.; Pan, S.; Wu, Y.; Pan, H.; Xu, D.; et al. Hydroxytyrosol, a natural molecule from olive oil, suppresses the growth of human hepatocellular carcinoma cells via inactivating AKT and nuclear factor-kappa B pathways. Cancer Lett. 2014, 347, 79–87.
- Priore, P.; Siculella, L.; Gnoni, G.V. Extra virgin olive oil phenols down-regulate lipid synthesis in primary-cultured rat-hepatocytes. J. Nutr. Biochem. 2014, 25, 683–691.
- Miceli, C.; Santin, Y.; Manzella, N.; Coppini, R.; Berti, A.; Stefani, M.; Parini, A.; Mialet-Perez, J.; Nediani, C. Oleuropein aglycone protects against MAO-a-induced autophagy impairment and cardiomyocyte death through activation of TFEB. Oxidative Med. Cell. Longev. 2018, 2018, 8067592.
- Rigacci, S.; Miceli, C.; Nediani, C.; Berti, A.; Cascella, R.; Pantano, D.; Nardiello, P.; Luccarini, I.; Casamenti, F.; Stefani, M. Oleuropein aglycone induces autophagy via the AMPK/mTOR signalling pathway: A mechanistic insight. Oncotarget 2015, 6, 35344–35357.
- Rajeswari, A.; Sabesan, M. Inhibition of monoamine oxidase-B by the polyphenolic compound, curcumin and its metabolite tetrahydrocurcumin, in a model of Parkinson’s disease induced by MPTP neurodegeneration in mice. Inflammopharmacology 2008, 16, 96–99.
- Zhang, J.; Wang, J.; Xu, J.; Lu, Y.; Jiang, J.; Wang, L.; Shen, H.M.; Xia, D. Curcumin targets the TFEB-lysosome pathway for induction of autophagy. Oncotarget 2016, 7, 75659–75671.
- Shakeri, A.; Cicero, A.F.G.; Panahi, Y.; Mohajeri, M.; Sahebkar, A. Curcumin: A naturally occurring autophagy modulator. J. Cell. Physiol. 2019, 234, 5643–5654.
- Saljoughian, M. Curcumin: A promising antiamyloidogenic agent. U.S. Pharm. 2011, 36, 27–32.
- Zheng, J.; Cheng, J.; Zheng, S.; Feng, Q.; Xiao, X. Curcumin, a polyphenolic curcuminoid with its protective effects and molecular mechanisms in diabetes and diabetic cardiomyopathy. Front. Pharmacol. 2018, 9, 472.
- Yang, F.; Lim, G.P.; Begum, A.N.; Ubeda, O.J.; Simmons, M.R.; Ambegaokar, S.S.; Chen, P.; Kayed, R.; Glabe, C.G.; Frautschy, S.A.; et al. Curcumin inhibits formation of amyloid β oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J. Biol. Chem. 2005, 280, 5892–5901.
- Mishra, P.; Paital, B.; Jena, S.; Swain, S.S.; Kumar, S.; Yadav, M.K.; Chainy, G.B.N.; Samanta, L. Possible activation of NRF2 by Vitamin E/Curcumin against altered thyroid hormone induced oxidative stress via NFĸB/AKT/mTOR/KEAP1 signalling in rat heart. Sci. Rep. 2019, 9, 7408.
- Sun, Q.; Jia, N.; Wang, W.; Jin, H.; Xu, J.; Hu, H. Activation of SIRT1 by curcumin blocks the neurotoxicity of amyloid-β25–35 in rat cortical neurons. Biochem. Biophys. Res. Commun. 2014, 448, 89–94.
- Wang, C.; Zhang, X.; Teng, Z.; Zhang, T.; Li, Y. Downregulation of PI3K/Akt/mTOR signaling pathway in curcumin-induced autophagy in APP/PS1 double transgenic mice. Eur. J. Pharmacol. 2014, 740, 312–320.
- Liu, Z.; Cui, C.; Xu, P.; Dang, R.; Cai, H.; Liao, D.; Yang, M.; Feng, Q.; Yan, X.; Jiang, P. Curcumin Activates AMPK Pathway and Regulates Lipid Metabolism in Rats Following Prolonged Clozapine Exposure. Front. Neurosci. 2017, 11, 558.
- Cao, J.; Chen, H.; Lu, W.; Wu, Y.; Wu, X.; Xia, D.; Zhu, J. Myricetin Induces Protective Autophagy by Inhibiting the Phosphorylation of mTOR in HepG2 Cells. Anat. Rec. 2018, 301, 786–795.
- Akindehin, S.; Jung, Y.S.; Kim, S.N.; Son, Y.H.; Lee, I.; Seong, J.K.; Jeong, H.W.; Lee, Y.H. Myricetin exerts anti-obesity effects through upregulation of SIRT3 in adipose tissue. Nutrients 2018, 10, 1962.
- Jung, H.Y.; Lee, D.; Ryu, H.G.; Choi, B.H.; Go, Y.; Lee, N.; Lee, D.; Son, H.G.; Jeon, J.; Kim, S.H.; et al. Myricetin improves endurance capacity and mitochondrial density by activating SIRT1 and PGC-1α. Sci. Rep. 2017, 7, 6237.
- Ryu, D.; Mouchiroud, L.; Andreux, P.A.; Katsyuba, E.; Moullan, N.; Nicolet-Dit-Félix, A.A.; Williams, E.G.; Jha, P.; Lo Sasso, G.; Huzard, D.; et al. Urolithin A induces mitophagy and prolongs lifespan in C. elegans and increases muscle function in rodents. Nat. Med. 2016, 22, 879–888.
- Gong, Z.; Huang, J.; Xu, B.; Ou, Z.; Zhang, L.; Lin, X.; Ye, X.; Kong, X.; Long, D.; Sun, X.; et al. Urolithin A attenuates memory impairment and neuroinflammation in APP/PS1 mice. J. Neuroinflammation 2019, 16, 62.
- Ono, K.; Hirohata, M.; Yamada, M. Ferulic acid destabilizes preformed β-amyloid fibrils in vitro. Biochem. Biophys. Res. Commun. 2005, 336, 444–449.
- Maurya, D.K.; Devasagayam, T.P.A. Antioxidant and prooxidant nature of hydroxycinnamic acid derivatives ferulic and caffeic acids. Food Chem. Toxicol. 2010, 48, 3369–3373.
- Yan, J.J.; Jung, J.S.; Kim, T.K.; Hasan, M.A.; Hong, C.W.; Nam, J.S.; Song, D.K. Protective effects of ferulic acid in amyloid precursor protein plus presenilin-1 transgenic mouse model of Alzheimer disease. Biol. Pharm. Bull. 2013, 36, 140–143.
- Bian, Z.; Furuya, N.; Zheng, D.M.; Trejo, J.A.O.; Tada, N.; Ezaki, J.; Ueno, T. Ferulic acid induces mammalian target of rapamycin inactivation in cultured mammalian cells. Biol. Pharm. Bull. 2013, 36, 120–124.
- Chen, Y.; Zheng, R.; Jia, Z.; Ju, Y. Flavonoids as superoxide scavengers and antioxidants. Free Radic. Biol. Med. 1990, 9, 19–21.
- Cho, H.-I.; Park, J.-H.; Choi, H.-S.; Kwak, J.H.; Lee, D.-U.; Lee, S.K.; Lee, S.-M. Protective Mechanisms of Acacetin against d-Galactosamine and Lipopolysaccharide-Induced Fulminant Hepatic Failure in Mice. J. Nat. Prod. 2014, 77, 2497–2503.
- Wang, X.; Perumalsamy, H.; Kwon, H.W.; Na, Y.E.; Ahn, Y.J. Effects and possible mechanisms of action of acacetin on the behavior and eye morphology of Drosophila models of Alzheimer’s disease. Sci. Rep. 2015, 5, 16127.
- Chang, W.; Wu, Q.Q.; Xiao, Y.; Jiang, X.H.; Yuan, Y.; Zeng, X.F.; Tang, Q.Z. Acacetin protects against cardiac remodeling after myocardial infarction by mediating MAPK and PI3K/Akt signal pathway. J. Pharmacol. Sci. 2017, 135, 156–163.
- Li, Y.; Zhao, J.; Holscher, C. Therapeutic Potential of Baicalein in Alzheimer’s Disease and Parkinson’s Disease. CNS Drugs 2017, 31, 639–652.
- Lee, H.J.; Noh, Y.H.; Lee, D.Y.; Kim, Y.S.; Kim, K.Y.; Chung, Y.H.; Lee, W.B.; Kim, S.S. Baicalein attenuates 6-hydroxydopamine-induced neurotoxicity in SH-SY5Y cells. Eur. J. Cell Biol. 2005, 84, 897–905.
- Zhang, S.Q.; Obregon, D.; Ehrhart, J.; Deng, J.; Tian, J.; Hou, H.; Giunta, B.; Sawmiller, D.; Tan, J. Baicalein reduces β-amyloid and promotes nonamyloidogenic amyloid precursor protein processing in an Alzheimer’s disease transgenic mouse model. J. Neurosci. Res. 2013, 91, 1239–1246.
- Lu, J.H.; Ardah, M.T.; Durairajan, S.S.K.; Liu, L.F.; Xie, L.X.; Fong, W.F.D.; Hasan, M.Y.; Huang, J.D.; El-Agnaf, O.M.A.; Li, M. Baicalein Inhibits Formation of α-Synuclein Oligomers within Living Cells and Prevents Aβ Peptide Fibrillation and Oligomerisation. ChemBioChem 2011, 12, 615–624.
- Li, J.; Ma, J.; Wang, K.S.; Mi, C.; Wang, Z.; Piao, L.X.; Xu, G.H.; Li, X.; Lee, J.J.; Jin, X. Baicalein inhibits TNF-α-induced NF-κB activation and expression of NF-κB-regulated target gene products. Oncol. Rep. 2016, 36, 2771–2776.
- Liu, C.; Wu, J.; Xu, K.; Cai, F.; Gu, J.; Ma, L.; Chen, J. Neuroprotection by baicalein in ischemic brain injury involves PTEN/AKT pathway. J. Neurochem. 2010, 112, 1500–1512.
- Gu, X.H.; Xu, L.J.; Liu, Z.Q.; Wei, B.; Yang, Y.J.; Xu, G.G.; Yin, X.P.; Wang, W. The flavonoid baicalein rescues synaptic plasticity and memory deficits in a mouse model of Alzheimer’s disease. Behav. Brain Res. 2016, 311, 309–321.
- Angeloni, C.; Barbalace, M.C.; Hrelia, S. Icariin and Its Metabolites as Potential Protective Phytochemicals Against Alzheimer’s Disease. Front. Pharmacol. 2019, 10, 271.
- Luo, Y.; Nie, J.; Gong, Q.H.; Lu, Y.F.; Wu, Q.; Shi, J.S. Protective effects of icariin against learning and memory deficits induced by aluminium in rats. Clin. Exp. Pharmacol. Physiol. 2007, 34, 792–795.
- Wang, L.; Zhang, L.; Chen, Z.B.; Wu, J.Y.; Zhang, X.; Xu, Y. Icariin enhances neuronal survival after oxygen and glucose deprivation by increasing SIRT1. Eur. J. Pharmacol. 2009, 609, 40–44.
- Li, W.W.; Gao, X.M.; Wang, X.M.; Guo, H.; Zhang, B.L. Icariin inhibits hydrogen peroxide-induced toxicity through inhibition of phosphorylation of JNK/p38 MAPK and p53 activity. Mutat. Res. Fundam. Mol. Mech. Mutagenesis 2011, 708, 1–10.
- Shi, D.B.; Li, X.X.; Zheng, H.T.; Li, D.W.; Cai, G.X.; Peng, J.J.; Gu, W.L.; Guan, Z.Q.; Xu, Y.; Cai, S.J. Icariin-Mediated Inhibition of NF-κB Activity Enhances the In Vitro and In Vivo Antitumour Effect of 5-Fluorouracil in Colorectal Cancer. Cell Biochem. Biophys. 2014, 69, 523–530.
- Li, F.; Dong, H.X.; Gong, Q.H.; Wu, Q.; Jin, F.; Shi, J.S. Icariin decreases both APP and Aβ levels and increases neurogenesis in the brain of Tg2576 mice. Neuroscience 2015, 304, 29–35.
- Song, Y.X.; Miao, J.Y.; Qiang, M.; He, R.Q.; Wang, X.M.; Li, W.W. Icariin protects SH-SY5Y cells from formaldehyde-induced injury through suppression of Tau phosphorylation. Chin. J. Integr. Med. 2016, 22, 430–437.
- Sheng, C.; Xu, P.; Zhou, K.; Deng, D.; Zhang, C.; Wang, Z. Icariin Attenuates Synaptic and Cognitive Deficits in an Aβ1–42-Induced Rat Model of Alzheimer’s Disease. Biomed. Res. Int. 2017, 2017, 7464872.
- Nakajima, A.; Ohizumi, Y. Potential benefits of nobiletin, a citrus flavonoid, against Alzheimer’s disease and Parkinson’s disease. Int. J. Mol. Sci. 2019, 20, 3380.
- Zhang, L.; Zhao, H.; Zhang, X.; Chen, L.; Zhao, X.; Bai, X.; Zhang, J. Nobiletin protects against cerebral ischemia via activating the p-Akt, p-CREB, BDNF and Bcl-2 pathway and ameliorating BBB permeability in rat. Brain Res. Bull. 2013, 96, 45–53.
- Nakajima, A.; Aoyama, Y.; Shin, E.J.; Nam, Y.; Kim, H.C.; Nagai, T.; Yokosuka, A.; Mimaki, Y.; Yokoi, T.; Ohizumi, Y.; et al. Nobiletin, a citrus flavonoid, improves cognitive impairment and reduces soluble Aβ levels in a triple transgenic mouse model of Alzheimer’s disease (3XTg-AD). Behav. Brain Res. 2015, 289, 69–77.
- Zhang, L.; Zhang, X.; Zhang, C.; Bai, X.; Zhang, J.; Zhao, X.; Chen, L.; Wang, L.; Zhu, C.; Cui, L.; et al. Nobiletin promotes antioxidant and anti-inflammatory responses and elicits protection against ischemic stroke in vivo. Brain Res. 2016, 1636, 130–141.
- Pierzynowska, K.; Podlacha, M.; Gaffke, L.; Majkutewicz, I.; Mantej, J.; Węgrzyn, A.; Osiadły, M.; Myślińska, D.; Węgrzyn, G. Autophagy-dependent mechanism of genistein-mediated elimination of behavioral and biochemical defects in the rat model of sporadic Alzheimer’s disease. Neuropharmacology 2019, 148, 332–346.
- Kazi, A.; Daniel, K.G.; Smith, D.M.; Kumar, N.B.; Dou, Q.P. Inhibition of the proteasome activity, a novel mechanism associated with the tumor cell apoptosis-inducing ability of genistein. Biochem. Pharmacol. 2003, 66, 965–976.
- Borrás, C.; Gambini, J.; Gómez-Cabrera, M.C.; Sastre, J.; Pallardó, F.V.; Mann, G.E.; Viña, J. Genistein, a soy isoflavone, up-regulates expression of antioxidant genes: Involvement of estrogen receptors, ERK1/2, and NFκB. FASEB J. 2006, 20, E1476–E1481.
- Moskot, M.; Montefusco, S.; Jakóbkiewicz-Banecka, J.; Mozolewski, P.; Wȩgrzyn, A.; Di Bernardo, D.; Wȩgrzyn, G.; Medina, D.L.; Ballabio, A.; Gabig-Cimińska, M. The phytoestrogen genistein modulates lysosomal metabolism and Transcription Factor EB (TFEB) activation. J. Biol. Chem. 2014, 289, 17054–17069.
- Kwon, Y. Luteolin as a potential preventive and therapeutic candidate for Alzheimer’s disease. Exp. Gerontol. 2017, 95, 39–43.
- Liao, Y.; Xu, Y.; Cao, M.; Huan, Y.; Zhu, L.; Jiang, Y.; Shen, W.; Zhu, G. Luteolin Induces Apoptosis and Autophagy in Mouse Macrophage ANA-1 Cells via the Bcl-2 Pathway. J. Immunol. Res. 2018, 2018, 4623919.
- Fang, F.; Li, D.; Pan, H.; Chen, D.; Qi, L.; Zhang, R.; Sun, H. Luteolin inhibits apoptosis and improves cardiomyocyte contractile function through the PI3K/Akt pathway in simulated ischemia/reperfusion. Pharmacology 2011, 88, 149–158.
- Nunes, C.; Almeida, L.; Barbosa, R.M.; Laranjinha, J. Luteolin suppresses the JAK/STAT pathway in a cellular model of intestinal inflammation. Food Funct. 2017, 8, 387–396.
- Cao, Z.; Zhang, H.; Cai, X.; Fang, W.; Chai, D.; Wen, Y.; Chen, H.; Chu, F.; Zhang, Y. Luteolin Promotes Cell Apoptosis by Inducing Autophagy in Hepatocellular Carcinoma. Cell. Physiol. Biochem. 2018, 43, 1803–1812.
- Feng, S.T.; Wang, Z.Z.; Yuan, Y.H.; Sun, H.M.; Chen, N.H.; Zhang, Y. Mangiferin: A multipotent natural product preventing neurodegeneration in Alzheimer’s and Parkinson’s disease models. Pharm. Res. 2019, 146, 104336.
- Andreu, G.P.; Delgado, R.; Velho, J.A.; Curti, C.; Vercesi, A.E. Iron complexing activity of mangiferin, a naturally occurring glucosylxanthone, inhibits mitochondrial lipid peroxidation induced by Fe2+-citrate. Eur. J. Pharmacol. 2005, 513, 47–55.
- Das, J.; Ghosh, J.; Roy, A.; Sil, P.C. Mangiferin exerts hepatoprotective activity against D-galactosamine induced acute toxicity and oxidative/nitrosative stress via Nrf2-NFκB pathways. Toxicol. Appl. Pharmacol. 2012, 260, 35–47.
- Kasbe, P.; Jangra, A.; Lahkar, M. Mangiferin ameliorates aluminium chloride-induced cognitive dysfunction via alleviation of hippocampal oxido-nitrosative stress, proinflammatory cytokines and acetylcholinesterase level. J. Trace Elem. Med. Biol. 2015, 31, 107–112.
- Jung, J.-S.; Jung, K.; Kim, D.-H.; Kim, H.-S. Selective inhibition of MMP-9 gene expression by mangiferin in PMA-stimulated human astroglioma cells: Involvement of PI3K/Akt and MAPK signaling pathways. Pharmacol. Res. 2012, 66, 95–103.
- Luna-Guevara, M.L.; Luna-Guevara, J.J.; Hernández-Carranza, P.; Ruíz-Espinosa, H.; Ochoa-Velasco, C.E. Chapter 3 - Phenolic Compounds: A Good Choice Against Chronic Degenerative Diseases. In Studies in Natural Products Chemistry; Attaur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; Volume 59, pp. 79–108.
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747.
- Ferreira, I.C.; Barros, L.; Abreu, R. Antioxidants in wild mushrooms. Curr. Med. Chem. 2009, 16, 1543–1560.
- Gil-Ramírez, A.; Pavo-Caballero, C.; Baeza, E.; Baenas, N.; Garcia-Viguera, C.; Marín, F.R.; Soler-Rivas, C. Mushrooms do not contain flavonoids. J. Funct. Foods 2016, 25, 1–13.
- Surguchov, A.; Emamzadeh, F.N.; Surguchev, A.A. Amyloidosis and longevity: A lesson from plants. Biology 2019, 8, 43.
- Sandoval-Acuña, C.; Ferreira, J.; Speisky, H. Polyphenols and mitochondria: An update on their increasingly emerging ROS-scavenging independent actions. Arch. Biochem. Biophys. 2014, 559, 75–90.
- Kim, M.Y.; Seguin, P.; Ahn, J.K.; Kim, J.J.; Chun, S.C.; Kim, E.H.; Seo, S.H.; Kang, E.Y.; Kim, S.L.; Park, Y.J.; et al. Phenolic compound concentration and antioxidant activities of edible and medicinal mushrooms from Korea. J. Agric. Food Chem. 2008, 56, 7265–7270.
- Heleno, S.A.; Barros, L.; Martins, A.; Queiroz, M.J.R.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Phenolic, polysaccharidic, and lipidic fractions of mushrooms from northeastern portugal: Chemical compounds with antioxidant properties. J. Agric. Food Chem. 2012, 60, 4634–4640.
- Zhang, D.D.; Hannink, M. Distinct Cysteine Residues in Keap1 Are Required for Keap1-Dependent Ubiquitination of Nrf2 and for Stabilization of Nrf2 by Chemopreventive Agents and Oxidative Stress. Mol. Cell. Biol. 2003, 23, 8137.
- Matzinger, M.; Fischhuber, K.; Heiss, E.H. Activation of Nrf2 signaling by natural products-can it alleviate diabetes? Biotechnol. Adv. 2018, 36, 1738–1767.
- Martínez-Huélamo, M.; Rodríguez-Morató, J.; Boronat, A.; de la Torre, R. Modulation of Nrf2 by Olive Oil and Wine Polyphenols and Neuroprotection. Antioxid 2017, 6, 73.
- Scapagnini, G.; Vasto, S.; Abraham, N.G.; Caruso, C.; Zella, D.; Fabio, G. Modulation of Nrf2/ARE pathway by food polyphenols: A nutritional neuroprotective strategy for cognitive and neurodegenerative disorders. Mol Neurobiol 2011, 44, 192–201.
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618.
- Nayernia, Z.; Jaquet, V.; Krause, K.-H. New insights on NOX enzymes in the central nervous system. Antioxid Redox Signal 2014, 20, 2815–2837.
- Gandhi, S.; Abramov, A.Y. Mechanism of Oxidative Stress in Neurodegeneration. Oxidative Med. Cell. Longev. 2012, 2012, 11.
- Cohen, G.; Kesler, N. Monoamine Oxidase and Mitochondrial Respiration. J. Neurochem. 1999, 73, 2310–2315.
- Dos Santos, T.W.; Pereira, Q.C.; Teixeira, L.; Gambero, A.; Villena, J.A.; Ribeiro, M.L. Effects of polyphenols on thermogenesis and mitochondrial biogenesis. Int. J. Mol. Sci. 2018, 19, 5757.
- Dong, W.; Wang, F.; Guo, W.; Zheng, X.; Chen, Y.; Zhang, W.; Shi, H. Aβ25–35 Suppresses Mitochondrial Biogenesis in Primary Hippocampal Neurons. Cell. Mol. Neurobiol. 2016, 36, 83–91.
- Xu, W.; Barrientos, T.; Andrews, N.C. Iron and copper in mitochondrial diseases. Cell Metab. 2013, 17, 319–328.
- Perez, C.A.; Wei, Y.; Guo, M. Iron-binding and anti-Fenton properties of baicalein and baicalin. J. Inorg. Biochem. 2009, 103, 326–332.
- Refat, M.S. Synthesis and characterization of ligational behavior of curcumin drug towards some transition metal ions: Chelation effect on their thermal stability and biological activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 105, 326–337.
- Valdés, A.; Sullini, G.; Ibáñez, E.; Cifuentes, A.; García-Cañas, V. Rosemary polyphenols induce unfolded protein response and changes in cholesterol metabolism in colon cancer cells. J. Funct. Foods 2015, 15, 429–439.
- Shen, M.; Chan, T.H.; Dou, Q.P. Targeting tumor ubiquitin-proteasome pathway with polyphenols for chemosensitization. Anti-Cancer Agents Med. Chem. 2012, 12, 891–901.
- Pallauf, K.; Rimbach, G. Autophagy, polyphenols and healthy ageing. Ageing Res. Rev. 2013, 12, 237–252.
- Rastogi, M.; Ojha, R.P.; Sagar, C.; Agrawal, A.; Dubey, G.P. Protective effect of curcuminoids on age-related mitochondrial impairment in female Wistar rat brain. Biogerontology 2014, 15, 21–31.
- Chung, S.; Yao, H.; Caito, S.; Hwang, J.W.; Arunachalam, G.; Rahman, I. Regulation of SIRT1 in cellular functions: Role of polyphenols. Arch. Biochem. Biophys. 2010, 501, 79–90.
- Horio, Y. Elucidation of the roles of protein deacetylase SIRT1 in health and diseases. Sapporo Med. J. 2018, 87, 1–8.
- Ren, Z.; He, H.; Zuo, Z.; Xu, Z.; Wei, Z.; Deng, J. The role of different SIRT1-mediated signaling pathways in toxic injury. Cell. Mol. Biol. Lett. 2019, 24, 36.
- Maiese, K. The mechanistic target of rapamycin (mTOR) and the silent mating-type information regulation 2 homolog 1 (SIRT1): Oversight for neurodegenerative disorders. Biochem. Soc. Trans. 2018, 46, 351–360.
- Bao, J.; Zheng, L.; Zhang, Q.; Li, X.; Zhang, X.; Li, Z.; Bai, X.; Zhang, Z.; Huo, W.; Zhao, X.; et al. Deacetylation of TFEB promotes fibrillar Aβ degradation by upregulating lysosomal biogenesis in microglia. Protein Cell 2016, 7, 417–433.
- Palmieri, M.; Impey, S.; Kang, H.; di Ronza, A.; Pelz, C.; Sardiello, M.; Ballabio, A. Characterization of the CLEAR network reveals an integrated control of cellular clearance pathways. Hum. Mol. Genet. 2011, 20, 3852–3866.
- Brunet, A.; Sweeney, L.B.; Sturgill, J.F.; Chua, K.F.; Greer, P.L.; Lin, Y.; Tran, H.; Ross, S.E.; Mostoslavsky, R.; Cohen, H.Y.; et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 2004, 303, 2011–2015.
- Pillarisetti, S. A Review of Sirt1 and Sirt1 Modulators in Cardiovascular and Metabolic Diseases. Recent Pat. Cardiovasc. Drug Discov. 2008, 3, 156–164.
- Allard, J.S.; Perez, E.; Zou, S.; de Cabo, R. Dietary activators of Sirt1. Mol. Cell. Endocrinol. 2009, 299, 58–63.
- Nardiello, P.; Pantano, D.; Lapucci, A.; Stefani, M.; Casamenti, F. Diet Supplementation with Hydroxytyrosol Ameliorates Brain Pathology and Restores Cognitive Functions in a Mouse Model of Amyloid-β Deposition. J. Alzheimer Dis. 2018, 63, 1161–1172.
- Pantano, D.; Luccarini, I.; Nardiello, P.; Servili, M.; Stefani, M.; Casamenti, F. Oleuropein aglycone and polyphenols from olive mill waste water ameliorate cognitive deficits and neuropathology. Br. J. Clin. Pharmacol. 2017, 83, 54–62.
- Cordero, J.G.; García-Escudero, R.; Avila, J.; Gargini, R.; García-Escudero, V. Benefit of Oleuropein Aglycone for Alzheimer’s Disease by Promoting Autophagy. Oxidative Med. Cell. Longev. 2018, 2018, 5010741.
- Kim, H.S.; Quon, M.J.; Kim, J.A. New insights into the mechanisms of polyphenols beyond antioxidant properties; lessons from the green tea polyphenol, epigallocatechin 3-gallate. Redox Biol 2014, 2, 187–195.
- Ulakcsai, Z.; Bagaméry, F.; Szökő, É.; Tábi, T. The role of autophagy induction in the mechanism of cytoprotective effect of resveratrol. Eur. J. Pharm. Sci. 2018, 123, 135–142.
- Song, J.; Huang, Y.; Zheng, W.; Yan, J.; Cheng, M.; Zhao, R.; Chen, L.; Hu, C.; Jia, W. Resveratrol reduces intracellular reactive oxygen species levels by inducing autophagy through the AMPK-mTOR pathway. Front. Med. 2018, 12, 697–706.
- Velagapudi, R.; Lepiarz, I.; El-Bakoush, A.; Katola, F.O.; Bhatia, H.; Fiebich, B.L.; Olajide, O.A. Induction of Autophagy and Activation of SIRT-1 Deacetylation Mechanisms Mediate Neuroprotection by the Pomegranate Metabolite Urolithin A in BV2 Microglia and Differentiated 3D Human Neural Progenitor Cells. Mol. Nutr. Food Res. 2019, 63, 1801237.
- Sarubbo, F.; Ramis, M.R.; Kienzer, C.; Aparicio, S.; Esteban, S.; Miralles, A.; Moranta, D. Chronic Silymarin, Quercetin and Naringenin Treatments Increase Monoamines Synthesis and Hippocampal Sirt1 Levels Improving Cognition in Aged Rats. J. Neuroimmune Pharmacol. 2018, 13, 24–38.
- Putteeraj, M.; Lim, W.L.; Teoh, S.L.; Yahaya, M.F. Flavonoids and its neuroprotective effects on brain ischemia and neurodegenerative diseases. Curr. Drug Targets 2018, 19, 1710–1720.
- Huang, S.M.; Tsai, S.Y.; Lin, J.A.; Wu, C.H.; Yen, G.C. Cytoprotective effects of hesperetin and hesperidin against amyloid ss-induced impairment of glucose transport through downregulation of neuronal autophagy. Mol. Nutr. Food Res. 2012, 56, 601–609.
- Xiao, J.B.; Högger, P. Dietary polyphenols and type 2 diabetes: Current insights and future perspectives. Curr. Med. Chem. 2015, 22, 23–38.
- Rodríguez-García, C.; Sánchez-Quesada, C.; Gaforio, J.J.; Gaforio, J.J. Dietary flavonoids as cancer chemopreventive agents: An updated review of human studies. Antioxidants 2019, 8, 137.
- Carrasco-Pozo, C.; Cires, M.J.; Gotteland, M. Quercetin and Epigallocatechin Gallate in the Prevention and Treatment of Obesity: From Molecular to Clinical Studies. J. Med. Food 2019, 22, 753–770.
- Zang, M.; Xu, S.; Maitland-Toolan, K.A.; Zuccollo, A.; Hou, X.; Jiang, B.; Wierzbicki, M.; Verbeuren, T.J.; Cohen, R.A. Polyphenols Stimulate AMP-Activated Protein Kinase, Lower Lipids, and Inhibit Accelerated Atherosclerosis in Diabetic LDL Receptor–Deficient Mice. Diabetes 2006, 55, 2180.
- Huang, J.; Zhang, Y.; Zhou, Y.; Zhang, Z.; Xie, Z.; Zhang, J.; Wan, X. Green Tea Polyphenols Alleviate Obesity in Broiler Chickens through the Regulation of Lipid-Metabolism-Related Genes and Transcription Factor Expression. J. Agric. Food Chem. 2013, 61, 8565–8572.
- Bigagli, E.; Toti, S.; Lodovici, M.; Giovannelli, L.; Cinci, L.; D’Ambrosio, M.; Luceri, C. Dietary extra-virgin olive oil polyphenols do not attenuate colon inflammation in transgenic HLAB-27 rats but exert hypocholesterolemic effects through the modulation of HMGCR and PPAR-α gene expression in the liver. Lifestyle Genom. 2019, 11, 99–108.
- Barquissau, V.; Ghandour, R.A.; Ailhaud, G.; Klingenspor, M.; Langin, D.; Amri, E.Z.; Pisani, D.F. Control of adipogenesis by oxylipins, GPCRs and PPARs. Biochimie 2017, 136, 3–11.
- Wang, S.; Zhang, Q.; Zhang, Y.; Shen, C.; Wang, Z.; Wu, Q.; Zhang, Y.; Li, S.; Qiao, Y. Agrimol B suppresses adipogenesis through modulation of SIRT1-PPAR gamma signal pathway. Biochem. Biophys. Res. Commun. 2016, 477, 454–460.
- Singh, R.; Kaushik, S.; Wang, Y.; Xiang, Y.; Novak, I.; Komatsu, M.; Tanaka, K.; Cuervo, A.M.; Czaja, M.J. Autophagy regulates lipid metabolism. Nature 2009, 458, 1131.
- Simons, K.; Ehehalt, R. Cholesterol, lipid rafts, and disease. J. Clin. Investig. 2002, 110, 597–603.
- Dhakal, S.; Subhan, M.; Fraser, M.J.; Gardiner, K.; Macreadie, I. Simvastatin Efficiently Reduces Levels of Alzheimer’s Amyloid Beta in Yeast. Int. J. Mol. Sci. 2019, 20, 3531.
- Salminen, A.; Kauppinen, A.; Suuronen, T.; Kaarniranta, K. SIRT1 longevity factor suppresses NF-κB-driven immune responses: Regulation of aging via NF-κB acetylation? BioEssays 2008, 30, 939–942.
- Morgan, M.J.; Liu, Z.G. Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res. 2011, 21, 103–115.
- Scalbert, A.; Johnson, I.T.; Saltmarsh, M. Polyphenols: Antioxidants and beyond. Am. J. Clin. Nutr. 2005, 81, 215S–217S.
- Rahman, I.; Chung, S. Dietary Polyphenols, Deacetylases and Chromatin Remodeling in Inflammation. World Rev. Nutr. Diet. 2010, 101, 84–94.
- Kostomoiri, M.; Fragkouli, A.; Sagnou, M.; Skaltsounis, L.A.; Pelecanou, M.; Tsilibary, E.C.; Τzinia, A.K. Oleuropein, an Anti-oxidant Polyphenol Constituent of Olive Promotes α-Secretase Cleavage of the Amyloid Precursor Protein (AβPP). Cell. Mol. Neurobiol. 2013, 33, 147–154.
- Porzoor, A.; Alford, B.; Hügel, H.M.; Grando, D.; Caine, J.; Macreadie, I. Anti-amyloidogenic properties of some phenolic compounds. Biomolecules 2015, 5, 505–527.
- Wobst, H.J.; Sharma, A.; Diamond, M.I.; Wanker, E.E.; Bieschke, J. The green tea polyphenol (-)-epigallocatechin gallate prevents the aggregation of tau protein into toxic oligomers at substoichiometric ratios. FEBS Lett. 2015, 589, 77–83.
- Lee, D.H.; Lee, D.H.; Lee, J.S. Characterization of a new antidementia β-secretase inhibitory peptide from Saccharomyces cerevisiae. Enzym. Microb. Technol. 2007, 42, 83–88.
- Park, I.H.; Jeon, S.Y.; Lee, H.J.; Kim, S.I.; Song, K.S. A β-secretase (BACE1) inhibitor hispidin from the mycelial cultures of Phellinus linteus. Planta Med. 2004, 70, 143–146.
- Bennett, L.; Sheean, P.; Zabaras, D.; Head, R. Heat-stable components of wood ear mushroom, Auricularia polytricha (higher basidiomycetes), inhibit in vitro activity of beta secretase (BACE1). Int. J. Med. Mushrooms 2013, 15, 233–249.
- Song, S.H.; Choi, S.M.; Kim, J.E.; Sung, J.E.; Lee, H.A.; Choi, Y.H.; Bae, C.J.; Choi, Y.W.; Hwang, D.Y. α-Isocubebenol alleviates scopolamine-induced cognitive impairment by repressing acetylcholinesterase activity. Neurosci. Lett. 2017, 638, 121–128.
- Sameem, B.; Saeedi, M.; Mahdavi, M.; Shafiee, A. A review on tacrine-based scaffolds as multi-target drugs (MTDLs) for Alzheimer’s disease. Eur. J. Med. Chem. 2017, 128, 332–345.
- Orhan, I.; Kartal, M.; Tosun, F.; Şener, B. Screening of various phenolic acids and flavonoid derivatives for their anticholinesterase potential. Z. Fur Nat. Sect. C J. Biosci. 2007, 62, 829–832.
- Dgachi, Y.; Sokolov, O.; Luzet, V.; Godyń, J.; Panek, D.; Bonet, A.; Martin, H.; Iriepa, I.; Moraleda, I.; García-Iriepa, C.; et al. Tetrahydropyranodiquinolin-8-amines as new, non-hepatotoxic, antioxidant, and acetylcholinesterase inhibitors for Alzheimer’s disease therapy. Eur. J. Med. Chem. 2017, 126, 576–589.
- Khan, H.; Marya; Amin, S.; Kamal, M.A.; Patel, S. Flavonoids as acetylcholinesterase inhibitors: Current therapeutic standing and future prospects. Biomed. Pharmacother. 2018, 101, 860–870.
- Ding, X.; Ouyang, M.A.; Liu, X.; Wang, R.Z. Acetylcholinesterase inhibitory activities of flavonoids from the leaves of Ginkgo biloba against brown planthopper. J. Chem. 2013, 2013, 645086.
- Kuppusamy, A.; Arumugam, M.; George, S. Combining in silico and in vitro approaches to evaluate the acetylcholinesterase inhibitory profile of some commercially available flavonoids in the management of Alzheimer’s disease. Int. J. Biol. Macromol. 2017, 95, 199–203.
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 2016, 7, 27–31.
- Renaud, J.; Martinoli, M.-G. Considerations for the Use of Polyphenols as Therapies in Neurodegenerative Diseases. Int J Mol Sci 2019, 20, 1883.
More
Information
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.2K
Entry Collection:
Neurodegeneration
Revisions:
2 times
(View History)
Update Date:
21 Oct 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No