
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Rahaba Marima | + 1790 word(s) | 1790 | 2021-09-23 04:57:27 | | | |
| 2 | Nora Tang | + 156 word(s) | 1946 | 2021-10-08 06:05:43 | | |
Video Upload Options
Histologically, cervical cancer can be divided into two main types, squamous cell carcinoma (SCC) and adenocarcinoma (AC). SCC is the most common type accounting for over 70% of all cases. Cervical cancer can be attributed to various risk factors, and these include smoking, alcohol consumption, multiple sexual partners, unprotected sex, socioeconomic status, a family history of cervical cancer, extended use of oral contraceptives, human immunodeficiency virus (HIV) infection and persistent human papillomavirus (HPV) infection.
1. Introduction
Histologically, cervical cancer can be divided into two main types, squamous cell carcinoma (SCC) and adenocarcinoma (AC). SCC is the most common type accounting for over 70% of all cases [1][2]. Cervical cancer can be attributed to various risk factors, and these include smoking, alcohol consumption, multiple sexual partners, unprotected sex, socioeconomic status, a family history of cervical cancer, extended use of oral contraceptives, human immunodeficiency virus (HIV) infection and persistent human papillomavirus (HPV) infection [3][4][5][6]. However, persistent HPV infection is the key contributor to the development of cervical cancer [1][7]. HPV infections can be classified as either low-risk or high-risk, with over 200 genotypes documented [8]. HPV 16 and 18 are the most dominant high-risk genotypes responsible for the development of invasive cervical cancer [9][10][11]. Persistent high-risk HPV infection is responsible for over 90% of cervical cancers while prophylactic vaccination against HPV infections including high-risk genotypes can reduce cervical cancers [10].
In sub-Saharan Africa (SSA), HIV positive women have increased HPV prevalence and cervical cancer incidence compared to HIV negative women. This may be partly attributed to HIV’s altering effect on HPV pathogenesis. Furthermore, HIV positive women have increased risk of HPV infection [12]. Globally, and in SSA, cervical cancer is the leading cause of mortality. SSA has elevated the dual burden of both HPV and HIV infection [13][14]. HIV infection is linked to higher HPV infection rates, reduced HPV clearance, more severe cervical precancerous lesions and higher risk of progression to invasive cervical cancer [15][16]. HIV positive women have been reported to have almost two times higher cervical cancer mortality compared to HIV negative women [17][18]. The advent of highly active antiretroviral therapy (HAART) has improved the life expectancy of HIV positive people, and HIV positive women. As an AIDS defining cancer (ADC), there is still a growing need for cervical cancer prevention, particularly in those countries with a high HIV/AIDS infection rate as well as in low-to-middle-income countries (LTMICs) [19]. In the USA, cervical cancer incidence and prevalence have been reduced by highly effective cervical cancer screening programs and HPV vaccination. A comprehensive background knowledge of the molecular mechanisms involved in the dual HPV/HIV infection promoting cervical cancer pathogenesis can aid in the development of informed cervical cancer management, policy and decision-making. This is especially true for HIV positive women. To date, various studies have evaluated the HPV/HIV interplay pathogenesis, while the gap in the understanding of this dual viral molecular pathogenesis is still extensive. While HAART reduces the incidence of other ADCs, the relationship between HAART and cervical cancer is still poorly understood [20].
Several extrinsic dynamics pose a threat in the fight against cervical cancer in LTMICs. Such factors include gender inequality, inequitable laws and inappropriate traditional practices and violence between intimate partners. These factors are unfavourable to women, limit women’s access to information, education, employment, opportunities and access to social and health services. Increased HPV and HIV co-infection has been frequently documented. Unfortunately, African women are mostly affected by this catastrophic dual viral infection. Globally, young women <35 years in the sub-Saharan African (SSA) region make up ~60% of HIV infection prevalence and almost 70% of newly acquired infections in Africa. With no proper interventions in place, the World Health Organization (WHO) estimates that cervical cancer deaths will double to over 400,000 by 2030, and the SSA region will make up 90% of the cases resulting in mortality [17][19].
Cancers associated with viral infections are on the rise globally, and in SSA [18][21] ( Figure 1 ). Approximately a third of all cancers in Africa are linked to a viral etiology [22]. Viruses such as HPV are generally dependent on the cellular machinery of the host for replication [18]. As a result of this, the splicing machinery of the host is also compromised upon HPV infection. As an oncovirus, HPV infection will result in the synthesis of splice variants of viral and cellular mRNA that favour cell survival mechanisms such as excessive cell proliferation, immune response evasion and inhibition of tumour suppressor proteins [3][23]. This review focuses on the link between cellular splicing machinery, HPV/HIV infection and the aberrant alternative splicing events that take place in HIV/HPV-associated cervical cancer. Finally, we investigate how these aberrant splicing events can be targeted for the development of new therapeutic strategies against HPV/HIV-associated cervical cancer.
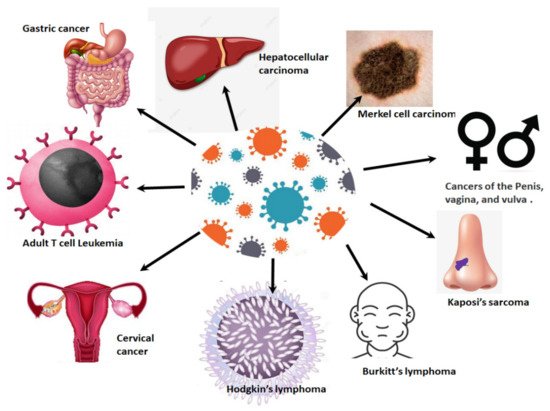
2. Cervical Cancer Epidemiology
With a worldwide age standardised incidence rate (ASR) of 15.6 per 100,000 per year, cervical cancer is the fourth most diagnosed cancer in women worldwide. The three cancers most common in women are breast, colorectal, and lung cancer. Based on the 2020 GLOBOCAN stats, the Southern African region has the highest incidence rates with an ASR of 36 per 100,000 ( Figure 2 A). This region also has the highest mortality rate of 20 per 100,000 as a result of cervical cancer ( Figure 2 B). All the countries in this region have incidence rates above 35 cases per 100,000 ( Figure 2 C). The country with the highest incidence rate is Eswatini with an ASR for incidence rates of 84.5, followed by Malawi (ASR 67.9), Zambia (ASR 65.5), Zimbabwe (ASR 61.7), Lesotho (56.8), Mozambique (ASR 50.2), Angola (ASR 37.5), Namibia (ASR 37.4), South Africa (ASR 35.3) and the country in the region with the lowest incidence, Botswana with an ASR of 34.4 ( Figure 2 B). The geographical regions with the second highest incidence rate for cervical cancer are Melanesia and the Eastern African region. Melanesia includes countries such as Fiji, Vanuatu, the Solomon Islands, and Papua New Guinea. The Eastern African region includes countries such as Burundi, Kenya, Rwanda, Tanzania, and Uganda. These two regions have an ASR of 24.3–24.4. Eastern Africa has the higher mortality rate with an ASR of 16.3 compared to that of Melanesia (ASR 15) ( Figure 2 A,B). The area with the third highest incidence rate for cervical cancer is Central and Eastern Europe (ASR 20.8), followed closely by Southeast Asia (ASR 20.4). The country with the highest incidence rate in Eastern and Central region is Romania (ASR 22.6) while Indonesia has the highest incidence rate in Southeast Asia with an ASR of 24.4 [24].
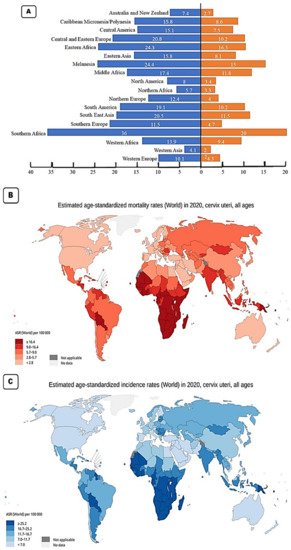
The region with the next highest incidence rate is South America with an ASR of 19.1 per 100,000, followed by Middle Africa (ASR 17.4), Eastern Asia and the Caribbean (both with an ASR of 15.8), Central America (ASR 15.1), Western Africa (ASR 13.9), Northern Europe (ASR 12.1), Southern Europe (ASR 11.5) and Western Europe (ASR 10.1). The Western Europe region contains countries such as the UK with an incidence rate of 9.9 and a mortality rate of 1.9 ( Figure 2 C). It also includes France with an incidence rate of 7 and a mortality rate of 2.2. These low incidence and low mortality rates are typical in developed nations. The regions with the lowest incidence of cervical cancer include North America (ASR 8), Australia and New Zealand (ASR 7.4), North Africa (ASR 5.7) and the region with the lowest incidence rate is West Asia with an incidence rate of 4.1 and a mortality rate of 2 per 100,000. In the North American region, the USA has an incidence rate of 6.2 per 100,000 and a mortality rate of 2.1 per 100,000 [24].
3. HPV Related Cancers and Pathogenesis
More than 200 genotypes of HPV based on their sequence can be divided into α, β, γ, δ, µ genera [4][6]. The HPV E1 and E2 proteins are early viral proteins required for the viral DNA replication and protein synthesis. In addition, E2 also regulates the expression of E6 and E7, while E4 and E5 help in viral assembly and stimulation of cellular proliferation. The late proteins, L1 and L2 form the minor and major capsid proteins [25].
HPV infects the squamous epithelial cells, which have the ability to proliferate and gain access to basal cells during abrasion or trauma. HPV then induces the viral genes’ expression, which is critical for viral replication in the basal cells. The HPV interaction with the host cells occurs via heparin sulfate proteoglycans and host cell surface receptors such as α-6 integrins. The E2 protein acts as a transcriptional repressor of E6 and E7. HPV replicates by the rolling circle mechanism, during which it may integrate into the human genome. This integration perturbs the expression of E2, thereby favouring the upregulation of the expression of the E6 and E7 oncoproteins, leading ultimately to cell transformation. Following viral replication, L1 and L2 proteins form the virus capsid and the subsequent mature virus production. The mature virus is then released with the help of the E4 protein and desquamation of cells [5][6][7].
4. HPV/HIV, HAART and Cervical Cancer
Cancers caused by HPV arise in tissues and organs where HPV infects epithelial cells. These tissues contain squamous cells that line the inside of these organs and are present within these tissues. It is these cells that are the target of HPV infection. The sites of HPV infection include the oropharynx, anus, penis, vagina, vulva and most importantly for this review, the cervix ( Figure 3 ). With some geographical variation, HPV infection causes 70% of cancers of the vulva, 75% of all vaginal cancers, 60% of all penile cancers, 90% of all anal cancers and 70% of all oropharyngeal cancers. In terms of cervical cancer, it is thought that nearly all cervical cancers are caused by persistent HPV infection [3][26].
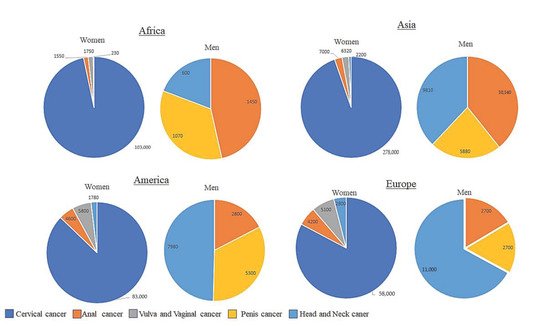
The association between HPV, HAART components and cervical cancer in HIV positive women remains poorly understood. It has been reported that initiating HAART early and attaining sustained adherence is more likely to reduce SIL (squamous intraepithelial lesion) and cervical intraepithelial neoplasia (CIN), and thus reduce the incidence of invasive cervical cancer [15]. Furthermore, the long-term effects of HAART on cervical cancer progression in HIV positive women is also unknown. Kelly et al. (2017) reported that HIV positive women on HAART showed reduced prevalence of invasive cervical cancer [27]. This study is in concordance with Clifford et al. (2016) [15]. However, conflicting reports stated that increasing incidence of invasive cervical cancer occurred in patients receiving HAART, probably because longer survival provides the time window for cervical cancer to develop [28]. The lack of adequate studies from African populations in particular, poses a challenge in attempting to decipher the effects of HAART on invasive cervical cancer. There have been a few African studies indicating that there is an inverse relationship pattern between invasive cervical cancer incidence and the length of time the patient has been undergoing HAART [20][27][29][30]. Conversely, the few studies performed in Latin American and Asian studies reveal a paradoxical relationship between HAART use and incidence of invasive cervical cancer [31][32][33].
HPV has been classified as a direct carcinogen while HIV has been classified as an indirect carcinogen through immunosuppression. The carcinogenic classification of these viruses was done by the International Agency for Research on Cancer [1][34].
References
- Hull, R.; Mbele, M.; Makhafola, T.; Hicks, C.; Wang, S.M.; Reis, R.M.; Mehrotra, R.; Mkhize-Kwitshana, Z.; Kibiki, G.; Bates, D.O.; et al. Cervical cancer in low and middle-income countries. Oncol. Lett. 2020, 20, 2058–2074.
- Herrero, R.; González, P.; Markowitz, L.E. Present status of human papillomavirus vaccine development and implementation. Lancet Oncol. 2015, 16, e206–e216.
- Francies, F.; Bassa, S.; Chatziioannou, A.; Kaufmann, A.; Dlamini, Z. Splicing Genomics Events in Cervical Cancer: Insights for Phenotypic Stratification and Biomarker Potency. Genes 2021, 12, 130.
- Momenimovahed, Z.; Salehiniya, H. Cervical cancer in Iran: Integrative insights of epidemiological analysis. BioMedicine 2018, 8, 18.
- Kabekkodu, S.P.; Chakrabarty, S.; Ghosh, S.; Brand, A.; Satyamoorthy, K. Epigenomics, Pharmacoepigenomics, and Personalized Medicine in Cervical Cancer. Public Health Genom. 2017, 20, 100–115.
- Akinlotan, M.; Bolin, J.N.; Helduser, J.; Ojinnaka, C.; Lichorad, A.; McClellan, D. Cervical Cancer Screening Barriers and Risk Factor Knowledge among Uninsured Women. J. Community Health 2017, 42, 770–778.
- Chan, C.K.; Aimagambetova, G.; Ukybassova, T.; Kongrtay, K.; Azizan, A. Human Papillomavirus Infection and Cervical Cancer: Epidemiology, Screening, and Vaccination—Review of Current Perspectives. J. Oncol. 2019, 2019, 3257939.
- Burd, E.M. Human papillomavirus and cervical cancer. Clin. Microbiol. Rev. 2003, 16, 1–17.
- Sankaranarayanan, R. HPV vaccination: The most pragmatic cervical cancer primary prevention strategy. Int. J. Gynaecol. Obstet. 2015, 131, S33–S35.
- Joura, E.A.; Giuliano, A.R.; Iversen, O.E.; Bouchard, C.; Mao, C.; Mehlsen, J.; Moreira, E.D., Jr.; Ngan, Y.; Petersen, L.K.; Lazcano-Ponce, E.; et al. A 9-valent HPV vaccine against infection and intraepithelial neoplasia in women. N. Engl. J. Med. 2015, 372, 711–723.
- Liu, F.; Dai, M.; Xu, Q.; Zhu, X.; Zhou, Y.; Jiang, S.; Wang, Y.; Ai, Z.; Ma, L.; Zhang, Y.; et al. SRSF10-mediated IL1RAP alternative splicing regulates cervical cancer oncogenesis via mIL1RAP-NF-κB-CD47 axis. Oncogene 2018, 37, 2394–2409.
- Liu, G.; Sharma, M.; Tan, N.; Barnabas, R.V. HIV-positive women have higher risk of human papilloma virus infection, precancerous lesions, and cervical cancer. AIDS 2018, 32, 795–808.
- Bruni, L.; Albero, G.; Serrano, B.; Mena, M.; Gómez, D.; Muñoz, J. Human Papillomavirus and Related Diseases in the World—Summary Report. ICO/IARC Information Center in HPV and Cancer (HPV Information Centre). 2019. Available online: https://hpvcentre.net/statistics/reports/XWX.pdf (accessed on 30 June 2021).
- De Vuyst, H.; Alemany, L.; Lacey, C.; Chibwesha, C.J.; Sahasrabuddhe, V.; Banura, C.; Denny, L.; Parham, G.P. The Burden of Human Papillomavirus Infections and Related Diseases in Sub-Saharan Africa. Vaccine 2013, 31, F32–F46.
- Clifford, G.M.; Franceschi, S.; Keiser, O.; Schöni-Affolter, F.; Lise, M.; Dehler, S.; Levi, F.; Mousavi, M.; Bouchardy, C.; Wolfensberger, A.; et al. Immunodeficiency and the risk of cervical intraepithelial neoplasia 2/3 and cervical cancer: A nested case-control study in the Swiss HIV cohort study. Int. J. Cancer 2016, 138, 1732–1740.
- Strickler, H.D.; Burk, R.D.; Fazzari, M.; Anastos, K.; Minkoff, H.; Massad, L.S.; Hall, C.; Bacon, M.; Levine, A.M.; Watts, D.H.; et al. Natural history and possible reactivation of human papillomavirus in human immunodeficiency virus-positive women. J. Natl. Cancer Inst. 2005, 97, 577–586.
- Coghill, A.E.; Shiels, M.S.; Suneja, G.; Engels, E.A. Elevated Cancer-Specific Mortality among HIV-Infected Patients in the United States. J. Clin. Oncol. 2015, 33, 2376–2383.
- Dryden-Peterson, S.; Bvochora-Nsingo, M.; Suneja, G.; Efstathiou, J.A.; Grover, S.; Chiyapo, S.; Ramogola-Masire, D.; Kebabonye-Pusoentsi, M.; Clayman, R.; Mapes, A.C.; et al. HIV Infection and Survival among Women with Cervical Cancer. J. Clin. Oncol. 2016, 34, 3749–3757.
- UNAIDS. Global AIDS Update 2017. Available online: http://www.unaids.org/sites/default/files/media_asset/global-AIDS-update-2016_en.pdf (accessed on 30 June 2021).
- Adler, D.H.; Kakinami, L.; Modisenyane, T.; Tshabangu, N.; Mohapi, L.; De Bruyn, G.; Martinson, N.A.; Omar, T. Increased regression and decreased incidence of human papillomavirus-related cervical lesions among HIV-infected women on HAART. AIDS 2012, 26, 1645–1652.
- Schiller, J.T.; Lowy, D.R. Virus Infection and Human Cancer: An Overview. Methods Mol. Biol. 2014, 193, 1–10.
- Moukassa, D.; Boumba, A.M.; Ngatali, C.F.; Ebatetou, A.; Mbon, J.B.N.; Ibara, J.-R. Virus-Induced Cancers in Africa: Epidemiology and Carcinogenesis Mechanisms. Open J. Pathol. 2018, 8, 1–14.
- Habbema, D.; Weinmann, S.; Arbyn, M.; Kamineni, A.; Williams, A.E.; de Kok, I.M.C.M.; van Kemenade, F.; Field, T.S.; van Rosmalen, J.; Brown, M.L. Harms of cervical cancer screening in the United States and the Netherlands. Int. J. Cancer 2017, 140, 1215–1222.
- GLOBOCAN. Global Cervical Cancer Fact She. Available online: https://gco.iarc.fr/to¬day/data/factsheets/cancers/27-Cervix-uteri-fact-sheet.pdf (accessed on 27 May 2021).
- Bansal, A.; Singh, M.P.; Rai, B. Human papillomavirus-associated cancers: A growing global problem. Int. J. Appl. Basic Med Res. 2016, 6, 84–89.
- Francies, F.; Dlamini, Z. Aberrant Splicing Events and Epigenetics in Viral Oncogenomics: Current Therapeutic Strategies. Cells 2021, 10, 239.
- Kelly, H.A.; Sawadogo, B.; Chikandiwa, A.; Segondy, M.; Gilham, C.; Lompo, O.; Omar, T.; Didelot, M.N.; Nagot, N.; Meda, N.; et al. Epidemiology of high-risk human papillomavirus and cervical lesions in African women living with HIV/AIDS: Effect of anti-retroviral therapy. AIDS 2017, 31, 273–285.
- Cobucci, R.N.; Lima, P.H.; de Souza, P.C.; Costa, V.V.; Cornetta Mda, C.; Fernandes, J.V.; Gonçalves, A.K. Assessing the impact of HAART on the incidence of defining and non-defining AIDS cancers among patients with HIV/AIDS: A systematic review. J. Infect. Public Health 2015, 8, 1–10.
- Firnhaber, C.; Westreich, D.; Schulze, D.; Williams, S.; Siminya, M.; Michelow, P.; Levin, S.; Faesen, M.; Smith, J.S. Highly active antiretroviral therapy and cervical dysplasia in HIV-positive women in South Africa. J. Int. AIDS Soc. 2012, 15, 17382.
- Omar, T.; Schwartz, S.; Hanrahan, C.; Modisenyane, T.; Tshabangu, N.; Golub, J.E.; McIntyre, J.A.; Gray, G.E.; Mohapi, L.; Martinson, N.A. Progression and regression of premalignant cervical lesions in HIV-infected women from Soweto: A prospective cohort. AIDS 2011, 25, 87–94.
- De Andrade, A.C.V.; Luz, P.M.; Velasque, L.; Veloso, V.G.; Moreira, R.I.; Russomano, F.; Chicarino-Coelho, J.; Pires, E.; Levi, J.E.; Grinsztejn, B.; et al. Factors Associated with Colposcopy-Histopathology Confirmed Cervical Intraepithelial Neoplasia among HIV-Infected Women from Rio De Janeiro, Brazil. PLoS ONE 2011, 6, e18297.
- Rocha-Brischiliari, S.C.; Gimenes, F.; de Abreu, A.L.; Irie, M.M.T.; Souza, R.P.; Santana, R.G.; Gravena, A.A.F.; Carvalho, M.D.d.B.; Consolaro, M.E.; Pelloso, S.M. Risk factors for cervical HPV infection and genotypes distribution in HIV-infected South Brazilian women. Infect. Agents Cancer 2014, 9, 6.
- Zhang, H.-Y.; Fei, M.-D.; Jiang, Y.; Fei, Q.-Y.; Qian, H.; Xu, L.; Jin, Y.-N.; Jiang, C.-Q.; Li, H.-X.; Tiggelaar, S.M.; et al. The diversity of human papillomavirus infection among human immunodeficiency virus-infected women in Yunnan, China. Virol. J. 2014, 11, 202.
- Papillomaviruses, H. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2011.





