Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Lukasz Piszczyk | + 916 word(s) | 916 | 2021-08-20 08:45:55 | | | |
| 2 | Camila Xu | Meta information modification | 916 | 2021-09-14 09:45:58 | | | | |
| 3 | Camila Xu | Meta information modification | 916 | 2021-09-14 09:48:30 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Piszczyk, L. Chitosan-Based Polyurethane Flexible Foams. Encyclopedia. Available online: https://encyclopedia.pub/entry/14166 (accessed on 23 May 2026).
Piszczyk L. Chitosan-Based Polyurethane Flexible Foams. Encyclopedia. Available at: https://encyclopedia.pub/entry/14166. Accessed May 23, 2026.
Piszczyk, Lukasz. "Chitosan-Based Polyurethane Flexible Foams" Encyclopedia, https://encyclopedia.pub/entry/14166 (accessed May 23, 2026).
Piszczyk, L. (2021, September 14). Chitosan-Based Polyurethane Flexible Foams. In Encyclopedia. https://encyclopedia.pub/entry/14166
Piszczyk, Lukasz. "Chitosan-Based Polyurethane Flexible Foams." Encyclopedia. Web. 14 September, 2021.
Copy Citation
Polyurethane (PUR) foam is a synthetic polymer.
chitosan
polyurethane
synthesis
flexible foams
physicochemical properties
1. Introduction
Polyurethane (PUR) foam is a synthetic polymer [1]. PUR can provide protection and insulation of materials against temperature, moisture, abrasion, or impact. In 2016, PUR foams constituted 67% of global PUR consumption [2]. PUR foams are commonly formed by the reaction of diisocyanate or polyisocyanate with polyol. These foams may occur in several forms, e.g., flexible or rigid foams and in a spray. The global PUR market was characterized by moderate growth during 2015–2020 [3]. According to the Global Polyurethane Foam Market Report, published on January 2020 by BCC Publishing, the global PUR foam market may increase by USD 9.8 billion between 2019 and 2024 [4].
PUR foams for oil sorption are known in the literature [5][6][7]. These foams have also been modified by natural components, for example, acerola and bagasse malt residues [5], Australian palm residues (PR) [6], or chitosan (Ch) from northern prawn shrimp waste [7].
2. Chemical Structure
The chemical structure of Ch, PUR and PUR+Ch foams were evaluated on the basis of the ATR-FTIR analysis. The spectrum of Ch was consistent with the literature [7][8][9]. It was possible to observe a broad band in the range of 3600–3100 cm−1 wavenumbers, which is characteristic of the stretching vibrations of the N–H and O–H bonds. Moreover, the bands visible at the wavenumbers 1652.6, 1582.2, and 1315.8 cm−1, were assigned to the amide I, amide II, and amide III bands, respectively. This confirmed that Ch was the only product of partial deacetylation of chitin [10].
Since the bands characteristic for Ch were overlapping bands of urethane groups, it cannot be determined whether the chitosan was present on the foam surface or was stuck to the polyurethane chains. The FTIR spectra of all tested polyurethanes are almost identical (Figure 1). No bands were found to develop or disappear after the addition of Ch, nor were large differences found in the intensity of the bands and their wavelengths.
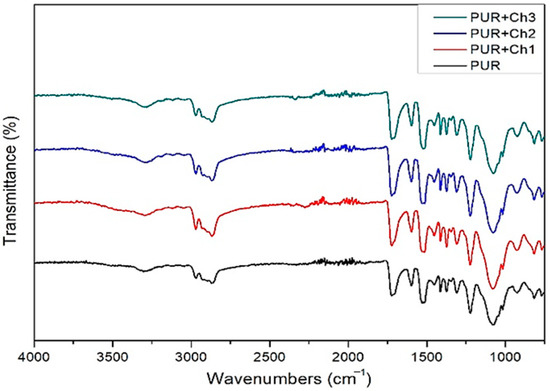
Figure 1. ATR-FTIR spectra.
The absence of the band corresponding to the -NCO groups (2237 cm−1) in the polyurethane’s spectrum indicated the complete reaction (Figure 1). The presence of bands confirming the formation of the urethane group (-NHCOO-) was found in the ATR-FTIR spectra of all tested materials. The additional oxygen in the structure of the urethane group compared to amides causes different wavenumber values of the amide bands compared to those of the chitosan-containing acetamide groups [11]. For PUR foams, the bands of amide II (δ NH, ν CN) and amide III (ν CN, δ CO, δ NH) were 1526 cm−1 and 1223 cm−1, respectively, whereas the stretching vibration of the C=O group (amide I) was at 1722.3 cm−1, and the N-H group was at 3303.3 cm−1. The spectra show that the characteristics of the chemical structure of PUR foams were preserved after the addition of Ch particles. Only a discrete shift of -N-H bands of PUR+Ch foams towards lower wavenumber values in comparison to neat PUR (up to 3289.4 cm−1) may indicate engaging of the -OH groups of Ch in the formation of hydrogen bonds with the urethane group of the hard segments of PUR+Ch foams. The bands corresponding to the urethane carbonyl groups practically did not shift after the addition of chitosan. It is known that amide band II shifts towards higher frequencies when a hydrogen bond is formed between the urethane and ether oxygen (from the soft segments) [12]. Thus, the reduction in the wavenumber of this band after the introduction of Ch into the PUR structure (from 1526 to 1514 cm−1 for PUR+Ch1 and PUR+Ch2 or to 1511 cm−1 for PUR+Ch3) indicated that the Ch particles influenced the interaction of PUR chains.
3. Structure Morphology and Characterization of Pore Structure
SEM images in Figure 2 show the structure of PUR and the exemplary PUR+Ch foam. Pore size in the neat PUR foam varied widely, and their diameters ranged from 0.09 to 0.76 mm. The introduction of Ch resulted in a decrease in the uniformity of the pore diameter (ranged from 0.01 to 0.92 mm). On the contrary, the shape of the pores was slightly more regular. The shape of PUR+Ch2 foam was the most homogenous. The PUR+Ch3 foam was characterized by a greater pore size dispersion. The presence of a larger amount of Ch particles resulted in a less uniform pore formation in the foaming process and a more compact structure of the foam structure.

Figure 2. SEM images of PUR ((a,b), where (b) shows the fragment from the red selection of image (a)) and PUR+Ch2 foam ((c,d), where (d) shows the fragment from the red selection of image (c)).
The observation of the images shows that there were small holes in the pore walls of the PUR+Ch. Thus, the surface of interaction of the foam was increased, which might improve its sorption capacity.
The tested foams had high open cell content, which is known in the literature [13][14][15]. Table 1 presents values of the open cell content in the tested materials. Incorporation of Ch into the PUR matrix influenced the content of open cells in the synthesized foams. Incorporation of Ch (1–3 wt%) into the PUR resulted in a noticeable increase in open cell content (from 84.3% to 93.4–95.8%), which was associated with differences in density between the matrix of PUR and Ch as filler. Furthermore, Husainie et al. stated that the adding of Ch filler resulted in an increase in the number of open pores [16]. The slight decrease in the open cell content after increasing the amount of filler was probably due to some agglomeration of the particles.
Table 1. Open cell content (with standard deviation SD) of PUR and PUR+Ch foams.

References
- Gama, N.V.; Ferreira, A.; Barros-Timmons, A. Polyurethane foams: Past, present, and future. Materials 2018, 11, 1841.
- Plastics Insight—Polyurethane Production, Pricing and Market Demand. Available online: https://www.plasticsinsight.com/resin-intelligence/resin-prices/polyurethane/ (accessed on 26 May 2021).
- Research and Markets—Polyurethane (PU) Foam Market: Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2021–2026. Available online: https://www.researchandmarkets.com (accessed on 26 May 2021).
- BCC Publishing—Global Polyurethane Foam Market. Available online: https://www.bccresearch.com/market-research/plastics/polyurethane-foam-market-report.html#toc (accessed on 26 May 2021).
- Amorim, F.V.; Padilha, R.J.R.; Vinhas, G.M.; Luiz, M.R.; de Souza, N.C.; de Almeida, Y.M.B. Development of hydrophobic polyurethane/castor oil biocomposites with agroindustrial residues for sorption of oils and organic solvents. J. Colloid Interface Sci. 2021, 581, 442–454.
- Martins, L.S.; Zanini, N.C.; Maia, L.S.; Souza, A.G.; Barbosa, R.F.S.; Rosa, D.S.; Mulinari, D.R. Crude oil and S500 diesel removal from seawater by polyurethane composites reinforced with palm fiber residues. Chemosphere 2021, 267, 129288.
- Piotrowska-Kirschling, A.; Szelągowska-Rudzka, K.; Karczewski, J.; Brzeska, J. Application of shrimp waste for the synthesis of polyurethane–chitosan materials with potential use in sorption of oil micro-spills in water treatment. Sustainability 2021, 13, 5098.
- Kwon, O.J.; Oh, S.T.; Lee, S.D.; Lee, N.R.; Shin, C.H.; Park, J.S. Hydrophilic and flexible polyurethane foams using sodium alginate as polyol: Effects of PEG molecular weight and cross-linking agent content on water absorbency. Fiber. Polym. 2007, 8, 347–355.
- Queiroz, M.F.; Melo, K.R.T.; Sabry, D.A.; Sassaki, G.L.; Rocha, H.A.O. Does the use of chitosan contribute to oxalate kidney stone formation? Mar. Drugs 2015, 13, 141–158.
- Kumirska, J.; Czerwicka, M.; Kaczyński, Z.; Bychowska, A.; Brzozowski, K.; Thöming, J.; Stepnowski, P. Application of spectroscopic methods for structural analysis of chitin and chitosan. Mar. Drugs 2010, 8, 1567–1636.
- Suchkova, G.G.; Maklakov, L.I. Amide bands in the IR spectra of urethanes. Vib. Spectrosc. 2009, 51, 333–339.
- Zhang, C.; Ren, Z.; Yin, Z.; Qian, H.; Ma, D. Amide II and amide III bands in polyurethane model soft and hard segments. Polym. Bull. 2008, 60, 97–101.
- Elliott, J.A.; Windle, A.H.; Hobdell, J.R.; Eeckhaut, G.; Oldman, R.J.; Ludwig, W.; Boller, E.; Cloetens, P.; Baruchel, J. In-situ deformation of an open-cell flexible polyurethane foam characterised by 3D computed microtomography. J. Mater. Sci. 2002, 37, 1547–1555.
- Koumlis, S.; Lamberson, L. Strain rate dependent compressive response of open cell polyurethane foam. Exp. Mech. 2019, 59, 1087–1103.
- Wu, J.W.; Sung, W.F.; Chu, H.S. Thermal conductivity of polyurethane foams. Int. J. Heat Mass Transf. 1999, 42, 2211–2217.
- Husainie, S.M.; Khattak, S.U.; Robinson, J.; Naguib, H.E. A comparative study on the mechanical properties of different natural fiber reinforced free-rise polyurethane foam composites. Ind. Eng. Chem. Res. 2020, 59, 21745–21755.
More
Information
Subjects:
Polymer Science
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.5K
Revisions:
3 times
(View History)
Update Date:
14 Sep 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





