Discovery of candidate genes for maize mesocotyl elongation, and identification of the mode of inheritance of the genes, will enhance the improvement of deep-sowing tolerance in maize, and provide the possibility of developing maize varieties for sowing in areas with low water tables. Investigating the extent of genetic variability for ML in available maize germplasm and the influence of seed biochemical quality attributes on ML will provide information for determining the gene action controlling ML, and secondary traits for selection of DST maize genotypes. The objectives of this study were to determine: (i) the extent of genetic variability for mesocoty length in maize, and (ii) the influence of seed biochemical quality attributes on mesocotyl elongation in maize.
2. Results and Discussion
As shown in
Table 1, analysis of variance revealed a significant (
p ≤ 0.01) genotype mean square for mesocotyl length. Moreover, genotypic variance (8.79) was close to phenotypic variance (13.19), leading to high broad-sense heritability (0.67), while genotypic (GCV) and phenotypic coefficients of variation (PCV) were high at 0.57 and 0.70, respectively. In addition, genetic advance (as a % of mean) was extremely high (almost unity). There was also a difference between the computed genotypic and phenotypic variances, indicating the role of environmental factors in the elongation of mesocotyl in maize. The significant genotype mean square, as well as the close correspondence between the genotypic and phenotypic variances, indicated the existence of sufficient variability for mesocotyl length among the maize genotypes. Thus, there is the possibility of genetic improvement of the trait for future breeding programs. The estimate of environmental variance suggested the need to test the maize genotypes in more environments to investigate the effect of genotype × environment interaction in the elongation of mesocotyl. Similar observations have been reported for agronomic traits in maize. Sivasubramanian and Menon
[1] ranked GCVs and PCVs as low, moderate and high when the values are <0.10, 0.10–0.20, and >0.20, respectively. The high GCV (0.57) observed indicated potential for improvement of ML within the germplasm. The higher value of PCV than GCV is an indication of the role of the environment in the phenotypic expression of ML, and further suggested the need to test in more environments. According to Johnson et al.
[2], heritability values < 0.30, between 0.30 and 0.60, and above 0.60 are classified as low, moderate, and high, respectively. Thus, the observed heritability estimate of ML was high and implied that the trait was majorly controlled by the genotype and is therefore heritable. Olayiwola and Soremi
[3] have suggested that broad-sense heritability should not be solely used in determining the genetic potentials of a trait since it (broad-sense heritability) is composed of both additive and non-additive genetic variances, and as a result, high heritability is not always associated with high genetic advance
[4]. Earlier, Johnson et al.
[2] recommended that estimates of heritability and genetic advance should be jointly considered in predicting the value of selection. The high genetic advance (as a % of mean) of mesocotyl length is an indication that the trait will respond favourably to selection.
Table 1. Mean squares and genetic components of maize genotypes for mesocotyl length after 10 days in the dark.
| Source of Variation |
DF |
Mean Square |
| Genotype |
67 |
13.19 ** |
| Error |
68 |
4.40 |
| R2 |
0.75 |
| Genotypic variance |
8.79 |
| Environmental variance |
4.40 |
| Phenotypic variance |
13.19 |
| Broad-sense heritability |
0.67 |
| Genotypic coefficient of variation |
0.57 |
| Phenotypic coefficient of variation |
0.70 |
| Genetic advance (% of Mean) |
0.96 |
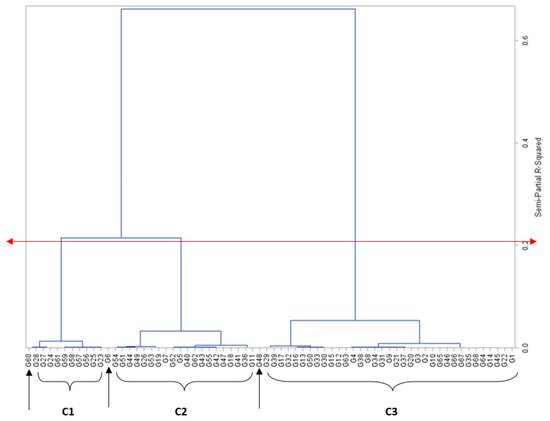
Grouping genotypes into clusters has been found effective in minimising the genotype pool and easing the process of selection
[5]. Sixty-five of the maize genotypes were clearly delineated into three clusters based on mesocotyl length, while three genotypes, PVAQEH-4, LY1919-14, and A1804-66, were ungrouped (
Table 2,
Figure 1). Cluster 1 was composed of 10 genotypes with no or short mesocotyl growth. Cluster 2 was composed of 20 genotypes with moderate mesocotyl growth ranging from 2.45 cm for †PVAEH 36 to 5.56 cm for PVAEH 29, while cluster 3 had 36 genotypes characterised by long observed mesocotyls ranging from 5.95 cm for †PVAEH 32 to 9.02 cm for LY1901-18. The observed clustering pattern implied the existence of considerable genetic diversity for ML among the maize genotypes. Several past works have classified maize genotypes into clusters based on a single trait. For instance, Oyetunde et al.
[6] and Badu-Apraku et al.
[7] classified maize genotypes into heterotic groups based on specific combining ability for grain yield. Crosses can be planned involving genotypes from clusters 1 and 2 (the most divergent groups) to exploit heterosis for ML.
Figure 1. Dendrogram of relatedness among maize genotypes (X-axis), based on genetic similarity (Y-axis) from Ward’s minimum variance cluster analysis. The red double-arrowed line delineates the genotypes into clusters at approximately 65% level of similarity; C1, C2, C3 are clusters 1, 2, and 3, respectively; single-arrowed line signifies ungrouped genotypes.
Table 2. Mean ± standard deviation (Std) of mesocotyl length (ML) and classification into clusters based on ML of maize genotypes germinated in the dark for 10 days.
| ID |
Genotype |
Mean |
Std |
Cluster |
ID |
Genotype |
Mean |
Std |
Cluster |
| G25 |
A1804-14 |
0 |
0 |
1 |
G2 |
PVAEH 30 |
5.99 |
1.57 |
3 |
| G56 |
LY1001-18 |
0 |
0 |
1 |
G37 |
LY1901-11 |
6.08 |
3.11 |
3 |
| G57 |
LY1409-14 |
0 |
0 |
1 |
G20 |
Oba super 2 |
6.12 |
1.27 |
3 |
| G58 |
LY1312-11 |
0 |
0 |
1 |
G9 |
†PVAEH 41 |
6.2 |
0.83 |
3 |
| G59 |
LY1901-15 |
0 |
0 |
1 |
G21 |
LY1312-23 |
6.2 |
0.99 |
3 |
| G61 |
A1804-67 |
0 |
0 |
1 |
G8 |
†PVAEH 43 |
6.28 |
1.22 |
3 |
| G23 |
A1312-12 |
0.58 |
0.19 |
1 |
G34 |
LY1901-23 |
6.3 |
2.12 |
3 |
| G24 |
LY1501-7 |
1.18 |
0.22 |
1 |
G31 |
LY1501-8 |
6.31 |
0.86 |
3 |
| G27 |
LY1901-14 |
1.36 |
0.55 |
1 |
G38 |
†PVAEH 40 |
6.45 |
2.81 |
3 |
| G28 |
LY1913-3 |
1.74 |
1.02 |
1 |
G4 |
PVAEH 28 |
6.47 |
1.54 |
3 |
| G60 |
A1804-66 * |
2.02 |
2.86 |
- |
G63 |
A1736-6 |
6.57 |
1.51 |
3 |
| G19 |
†PVAEH 36 |
2.45 |
3.46 |
2 |
G66 |
LY1001-23 |
6.77 |
2.73 |
3 |
| G53 |
Local check |
2.65 |
3.75 |
2 |
G67 |
Mkt-cultivar A |
6.88 |
1.27 |
3 |
| G51 |
M1124-31 |
3.06 |
3.62 |
2 |
G35 |
LY1901-20 |
6.93 |
3.15 |
3 |
| G54 |
LY1501-6 |
3.23 |
1.1 |
2 |
G68 |
Mkt-cultivar B |
6.98 |
0.76 |
3 |
| G6 |
PVAQEH-4 * |
3.28 |
1.64 |
- |
G14 |
PVAQEH-6 |
7.02 |
2.88 |
3 |
| G44 |
LY1901-12 |
3.57 |
3.01 |
2 |
G64 |
LY1901-24 |
7.02 |
0.68 |
3 |
| G49 |
A1802-4 |
3.67 |
0.61 |
2 |
G45 |
A1706-2 |
7.04 |
2.88 |
3 |
| G26 |
A1802-12 |
3.7 |
0.95 |
2 |
G22 |
LY1901-22 |
7.05 |
3.1 |
3 |
| G36 |
LY1901-17 |
4.1 |
0.11 |
2 |
G1 |
PVAEH 26 |
7.06 |
2.43 |
3 |
| G41 |
LY1409-21 |
4.11 |
2.42 |
2 |
G65 |
LY1501-9 |
7.19 |
0.24 |
3 |
| G11 |
PVAQEH-3 |
4.4 |
0.62 |
2 |
G46 |
Ife hybrid-4 |
7.29 |
4.26 |
3 |
| G18 |
†PVAEH 37 |
4.65 |
6.58 |
2 |
G48 |
LY1914-14* |
7.67 |
3.95 |
- |
| G47 |
LY1901-19 |
4.78 |
4.1 |
2 |
G29 |
LY1501-5 |
7.68 |
0.99 |
3 |
| G62 |
LY1501-1 |
4.89 |
1.99 |
2 |
G39 |
†PVAEH 33 |
7.87 |
0.52 |
3 |
| G43 |
LY1901-25 |
4.94 |
1.92 |
2 |
G17 |
†PVAEH 44 |
7.89 |
0.78 |
3 |
| G55 |
LY1901-21 |
5.05 |
1.53 |
2 |
G33 |
A1736-13 |
8.18 |
0.85 |
3 |
| G42 |
LY1901-16 |
5.11 |
1.2 |
2 |
G50 |
A1804-15 |
8.22 |
2.8 |
3 |
| G40 |
Ife hybrid-3 |
5.24 |
0.62 |
2 |
G30 |
LY1913-16 |
8.38 |
2.38 |
3 |
| G5 |
Check (RE) |
5.39 |
0.69 |
2 |
G15 |
PVAEH 27 |
8.42 |
0.28 |
3 |
| G52 |
LY1901-13 |
5.39 |
0.87 |
2 |
G12 |
Local check |
8.52 |
0.51 |
3 |
| G7 |
PVAEH 29 |
5.56 |
0.81 |
2 |
G13 |
†PVAEH 31 |
8.73 |
1.6 |
3 |
| G3 |
†PVAEH 32 |
5.95 |
2.5 |
3 |
G16 |
†PVAEH 34 |
8.94 |
0.03 |
3 |
| G10 |
PVAQEH-5 |
5.98 |
0.72 |
3 |
G32 |
LY1901-18 |
9.02 |
0.42 |
3 |
Sixty-two of the maize genotypes tested developed measurable mesocotyl. The mean mesocotyl length ranged from 0.58 cm for A1312-12 to 9.02 cm for LY1901-18 (Table 2). The differences in ML lengths of the maize genotypes further indicated the existence of variability of the trait, and genotypes with high ML values, such as LY191-01, †PVAEH 34, and †PVAEH 31 have potential to draw moisture from the subsoil and could be adaptable to drought-prone areas. The standard deviation of a set of numbers is an indication of the spread of the numbers from the mean. A low standard deviation value implies that the majority of the numbers are close to the average, while a high value implies that most numbers are far from the mean. Genotypes with low standard deviation estimates should, therefore, be more reliable for prediction. Hence, genotypes that combined high ML with low standard deviation values, such as LY1901-18 and †PVAEH 34, would be more reliable in predicting the ML of maize. In addition, planned crosses can be made involving the genotypes A1312-12, LY1501-7, and LY1901-14 to develop maize hybrids with potential for adaptation to drought conditions. Moreover, genotypes with high standard deviation values could be useful for development of inbred lines for future hybrid programs with focus on developing deep-sowing tolerant maize.
Information on seed micronutrients and biochemical composition which affects the seedling vigor and organ development may provide some insight to the understanding of the mechanism underlying mesocotyl elongation. In this study, significant (
p ≤ 0.05/0.01) associations were observed in the relationship of ML with all the measured biochemical parameters except selenium (
Table 3). Furthermore, all the significant associations were positive except for the negative association between ML and amylase content. The significant levels of association observed implied the possibility of simultaneous improvement of each of iron, zinc, crude protein, free fatty acid, oil content, linoleic acid, and amylase content with mesocotyl length of maize. The quality attributes could serve as selection indices for ML. Muhammad
[8] employed nutrient priming to reveal the impact of seed reserves on seedling development and root biomass in maize. According to Martinez-Ballesta et al.
[9], seed biofortification enhances seed vigour, and can play a role in abiotic stress tolerance.
Table 3. Pearson correlation coefficients of seed biochemical parameters with mesocotyl length (ML).
| Biochemical Content |
ML |
| Iron |
0.31 * |
| Zinc |
0.29 * |
| Selenium |
0.01 ns |
| Crude protein |
0.33 ** |
| Free fatty acid |
0.26 * |
| Oil |
0.30 * |
| Linoleic acid |
0.28 * |
| Amylase |
−0.31 * |