
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ka-Liong Tan | + 2064 word(s) | 2064 | 2021-08-31 06:20:39 | | | |
| 2 | Peter Tang | Meta information modification | 2064 | 2021-09-08 11:13:06 | | |
Video Upload Options
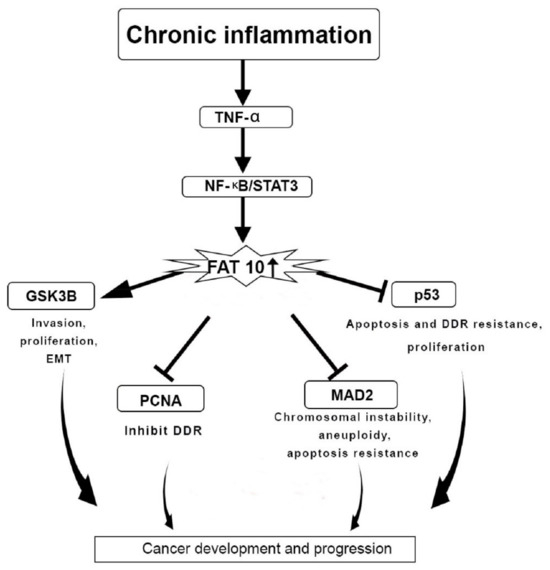
The upregulated expression of both NUB1 and FAT10 has been observed in various cancers. NUB1 protein binds to FAT10 non-covalently to promote FAT10 degradation. An overexpressed FAT10 stimulates nuclear factor-kappa β, activates the inflammatory pathways, and induces the proliferation of cancer. The FAT10 protein interacts with the mitotic arrest deficient 2 protein, causing chromosomal instability and breast tumourigenesis. FAT10 binds to the proliferating cell nuclear antigen protein and inhibits the DNA damage repair response. In addition, FAT10 involves epithelial–mesenchymal transition, invasion, apoptosis, and multiplication in hepatocellular carcinoma.
1. Introduction
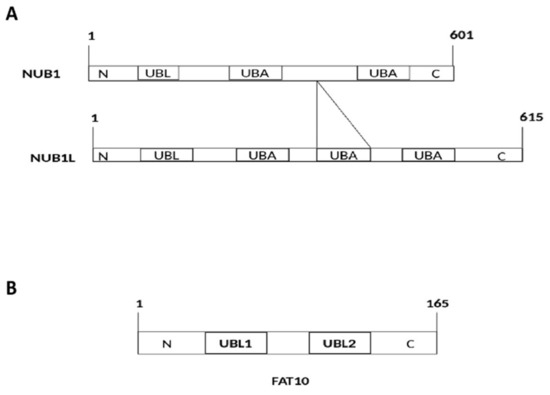
2. Interaction of FAT10 and NUB1
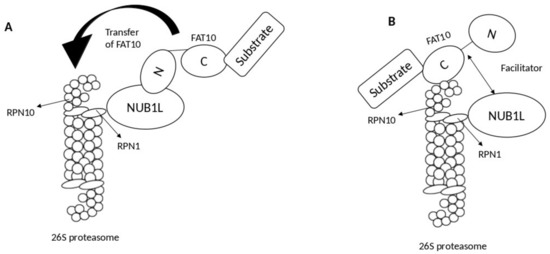
3. NUB1 Protein Actions in Cancer
4. FAT10 Protein Actions in Cancer
|
Cancer Type |
Remarks |
References |
|---|---|---|
|
Hepatocellular carcinoma (HCC) |
GRP78 protein increases FAT10 protein expression via direct activation on the NF-κB pathway. |
[17] |
|
Breast cancer |
FAT10 protein induces pro-metastasis effect with the help of ZEB2 overexpression. |
[18] |
|
Bladder cancer |
FAT10 protein non-covalently binds to Survivin protein to inhibit ubiquitin-mediated degradation. |
[19] |
|
B-cell non-Hodgkin lymphomas |
FAT10 protein non-covalently binds to MAD2 protein to maintain mitosis. |
[20] |
|
Colorectal cancer, HCC, Gastric cancer |
FAT10 protein disrupts the DNA damage repair response via modification of PCNA protein. |
|
|
NSCLC |
FAT10 causes NSCLS malignancy via interaction with NF-κB signalling pathway. |
[24] |
|
Glioma |
FAT10 protein increases phosphorylation of SMAD2 protein, which triggers FAT10 induced oncogenic activities. |
[25] |
|
Neuroblastoma |
FAT10 protein stabilises the survivin protein via non-covalent binding. |
5. Use of NUB1 and FAT10 as Biomarkers in a Clinical Setting
|
Type |
Human Sample Types |
Sample Size |
Antibody Clone and Host Species |
Method of Detection and Biomarker Type |
Findings |
References |
|---|---|---|---|---|---|---|
|
Anti-NUB1 and -NUB1L |
Gastric cancer patients |
116 |
Ab38438 (Rabbit polyclonal) |
Immunohistochemistry/ Prognostic |
Reduced NUB1 level associated to poor prognosis of gastric cancer. p < 0.05; HR: 0.33 (0.20–0.54) |
[6] |
|
Breast cancer patients |
114 |
4H2 (Mouse Monoclonal Antibody) |
Immunohistochemistry/ Prognostic |
Low cytoplasmic NUB1 protein level exerts poorer overall survival. p = 0.048, HR: 1.779 (1.006–3.346) |
[39] |
|
|
Anti-FAT10 antibody |
Bladder cancer samples |
133 |
MBS4750652 (Rabbit Polyclonal Antibody) |
Immunohistochemistry/ Prognostic |
Higher FAT10 expression in bladder cancer tissues had poorer survival than those with lower FAT10 expression. p = 0.002; HR:? |
[19] |
|
Non-small cell lung carcinoma (NSCLC) samples |
45 |
sc-133199 (mouse monoclonal antibody) |
Immunohistochemistry/ Prognostic |
High FAT10 expression confers quick chemoresistance than the lower FAT10 expression group. p = 0.001; HR:? |
[24] |
|
|
Breast cancer tissues |
120 |
ab168680 (Mouse polyclonal antibody) |
Immunohistochemistry/ Prognostic |
FAT10 overexpression leads to poor prognostic factor for poorer outcomes of patients with breast cancer. p < 0.05; HR:1.563 (1.232–2.531) |
[18] |
References
- Hipp, M.S.; Raasi, S.; Groettrup, M.; Schmidtke, G. NEDD8 ultimate buster-1L interacts with the ubiquitin-like protein FAT10 and accelerates its degradation. J. Biol. Chem. 2004, 279, 16503–16510.
- Schmidtke, G.; Aichem, A.; Groettrup, M. FAT10ylation as a signal for proteasomal degradation. Biochim. Biophys. Acta. Mol. Cell Res. 2014, 1843, 97–102.
- Rani, N.; Aichem, A.; Schmidtke, G.; Kreft, S.G.; Groettrup, M. FAT10 and NUB1L bind to the VWA domain of Rpn10 and Rpn1 to enable proteasome-mediated proteolysis. Nat. Commun. 2012, 3, 749.
- Kito, K.; Yeh, E.T.; Kamitani, T. NUB1, a NEDD8-interacting protein, is induced by interferon and down-regulates the NEDD8 expression. J. Biol. Chem. 2001, 276, 20603–20609.
- Hosono, T.; Tanaka, T.; Tanji, K.; Nakatani, T.; Kamitani, T. NUB1, an interferon-inducible protein, mediates anti-proliferative actions and apoptosis in renal cell carcinoma cells through cell-cycle regulation. Br. J. Cancer 2010, 102, 873–882.
- Zhang, D.; Wu, P.; Zhang, Z.; An, W.; Zhang, C.; Pan, S.; Tan, Y.; Xu, H. Overexpression of negative regulator of ubiquitin-like proteins 1 (NUB1) inhibits proliferation and invasion of gastric cancer cells through upregulation of p27Kip1 and inhibition of epithelial-mesenchymal transition. Pathol. Res. Pract. 2020, 216, 153002.
- Masuda, T.A.; Inoue, H.; Sonoda, H.; Mine, S.; Yoshikawa, Y.; Nakayama, K.; Nakayama, K.; Mori, M. Clinical and biological significance of S-phase kinase-associated protein 2 (Skp2) gene expression in gastric carcinoma: Modulation of malignant phenotype by Skp2 overexpression, possibly via p27 proteolysis. Cancer Res. 2002, 62, 3819–3825.
- Tanaka, T.; Nakatani, T.; Kamitani, T. Inhibition of NEDD8-conjugation pathway by novel molecules: Potential approaches to anticancer therapy. Mol. Oncol. 2012, 6, 267–275.
- Kamitani, T.; Kito, K.; Nguyen, H.P.; Yeh, E.T. Characterization of NEDD8, a developmentally down-regulated ubiquitin-like protein. J. Biol. Chem. 1997, 272, 28557–28562.
- He, S.; Cao, Y.; Xie, P.; Dong, G.; Zhang, L. The Nedd8 non-covalent binding region in the Smurf HECT domain is critical to its ubiquitn ligase function. Sci. Rep. 2017, 7, 1–12.
- Yao, Y.; Lu, B. NUB1 suppression of Huntington toxicity: Mechanistic insights. Res. Rep. Biochem. 2015, 5, 129–136.
- Aichem, A.; Groettrup, M. The ubiquitin-like modifier FAT10 in cancer development. Int. J. Biochem. Cell Biol. 2016, 79, 451–461.
- Lee, C.G.; Ren, J.; Cheong, I.S.; Ban, K.H.; Ooi, L.L.; Yong Tan, S.; Kan, A.; Nuchprayoon, I.; Jin, R.; Lee, K.H.; et al. Expression of the FAT10 gene is highly upregulated in hepatocellular carcinoma and other gastrointestinal and gynecological cancers. Oncogene 2003, 22, 2592–2603.
- Fiebiger, B.M.; Pfister, H.; Behrends, U.; Mautner, J. Polyubiquitination of lysine-48 is an essential but indirect signal for MHC class I antigen processing. Eur. J. Immunol. 2015, 45, 716–727.
- Yuan, R.; Wang, K.; Hu, J.; Yan, C.; Li, M.; Yu, X.; Liu, X.; Lei, J.; Guo, W.; Wu, L.; et al. Ubiquitin-like protein FAT10 promotes the invasion and metastasis of hepatocellular carcinoma by modifying β-catenin degradation. Cancer Res. 2014, 74, 5287–5300.
- Liu, X.; Chen, L.; Ge, J.; Yan, C.; Huang, Z.; Hu, J.; Wen, C.; Li, M.; Huang, D.; Qiu, Y.; et al. The Ubiquitin-like Protein FAT10 Stabilizes eEF1A1 Expression to Promote Tumor Proliferation in a Complex Manner. Cancer Res. 2016, 76, 4897–4907.
- Luo, B.; Lee, A.S. The critical roles of endoplasmic reticulum chaperones and unfolded protein response in tumorigenesis and anticancer therapies. Oncogene 2013, 32, 805–818.
- Zou, Y.; Ouyang, Q.; Wei, W.; Yang, S.; Zhang, Y.; Yang, W. FAT10 promotes the invasion and migration of breast cancer cell through stabilization of ZEB2. Biochem. Biophys. Res. Commun. 2018, 506, 563–570.
- Dong, D.; Jiang, W.; Lei, J.; Chen, L.; Liu, X.; Ge, J.; Che, B.; Xi, X.; Shao, J. Ubiquitin-like protein FAT10 promotes bladder cancer progression by stabilizing survivin. Oncotarget 2016, 7, 81463–81473.
- Luo, C.; Xiong, H.; Chen, L.; Liu, X.; Zou, S.; Guan, J.; Wang, K. GRP78 Promotes Hepatocellular Carcinoma proliferation by increasing FAT10 expression through the NF-κB pathway. Exp. Cell Res. 2018, 365, 1–11.
- Chen, Z.; Zhang, W.; Yun, Z.; Zhang, X.; Gong, F.; Wang, Y.; Ji, S.; Leng, L. Ubiquitin-like protein FAT10 regulates DNA damage repair via modification of proliferating cell nuclear antigen. Mol. Med. Rep. 2018, 17, 7487–7496.
- Xirodimas, D.P.; Saville, M.K.; Bourdon, J.-C.; Hay, R.T.; Lane, D.P. Mdm2-mediated NEDD8 conjugation of p53 inhibits its transcriptional activity. Cell 2004, 118, 83–97.
- Liu, G.; Xirodimas, D. NUB1 promotes cytoplasmic localization of p53 through cooperation of the NEDD8 and ubiquitin pathways. Oncogene 2010, 29, 2252–2261.
- Xue, F.; Zhu, L.; Meng, Q.W.; Wang, L.; Chen, X.S.; Zhao, Y.B.; Xing, Y.; Wang, X.Y.; Cai, L. FAT10 is associated with the malignancy and drug resistance of non-small-cell lung cancer. Onco Targets Ther. 2016, 9, 4397–4409.
- Dai, B.; Zhang, Y.; Zhang, P.; Pan, C.; Xu, C.; Wan, W.; Wu, Z.; Zhang, J.; Zhang, L. Upregulation of p-Smad2 contributes to FAT10-induced oncogenic activities in glioma. Tumor Biol. 2016, 37, 8621–8631.
- Chen, H.A.; Su, C.M.; Hsieh, H.Y.; Tung, C.L.; Hsu, C.D.; Wang, Y.H.; Shen, C.H. Clinical significance of survivin expression in patients with urothelial carcinoma. Dis. Markers 2014, 2014, 574985.
- Azuhata, T.; Scott, D.; Takamizawa, S.; Wen, J.; Davidoff, A.; Fukuzawa, M.; Sandler, A. The inhibitor of apoptosis protein survivin is associated with high-risk behavior of neuroblastoma. J. Pediatr. Surg. 2001, 36, 1785–1791.
- Ni, M.; Zhang, Y.; Lee, A.S. Beyond the endoplasmic reticulum: Atypical GRP78 in cell viability, signalling and therapeutic targeting. Biochem. J. 2011, 434, 181–188.
- Ren, J.; Wang, Y.; Gao, Y.; Mehta, S.B.; Lee, C.G. FAT10 mediates the effect of TNF-α in inducing chromosomal instability. J. Cell Sci. 2011, 124, 3665–3675.
- Choi, Y.; Kim, J.K.; Yoo, J.Y. NFκB and STAT3 synergistically activate the expression of FAT10, a gene counteracting the tumor suppressor p53. Mol. Oncol. 2014, 8, 642–655.
- Sethi, G.; Ahn, K.S.; Sung, B.; Aggarwal, B.B. Pinitol targets nuclear factor-kB activation pathway leading to inhibition of gene products associated with proliferation, apoptosis, invasion, and angiogenesis. Mol. Cancer Ther. 2008, 7, 1604–1614.
- Guo, K.; Kang, N.X.; Li, Y.; Sun, L.; Gan, L.; Cui, F.J.; Gao, M.D.; Liu, K.Y. Regulation of HSP27 on NF-kB pathway activation may be involved in metastatic hepatocellular carcinoma cells apoptosis. BMC Cancer 2009, 9, 100.
- Lukasiak, S.; Schiller, C.; Oehlschlaeger, P.; Schmidtke, G.; Krause, P.; Legler, D.F.; Autschbach, F.; Schirmacher, P.; Breuhahn, K.; Groettrup, M. Proinflammatory cytokines cause FAT10 upregulation in cancers of liver and colon. Oncogene 2008, 27, 6068–6074.
- Liu, L.; Dong, Z.; Liang, J.; Cao, C.; Sun, J.; Ding, Y.; Wu, D. As an independent prognostic factor, FAT10 promotes hepatitis B virus-related hepatocellular carcinoma progression via Akt/GSK3β pathway. Oncogene 2014, 33, 909–920.
- Singh, N.; Baby, D.; Rajguru, J.P.; Patil, P.B.; Thakkannavar, S.S.; Pujari, V.B. Inflammation and cancer. Ann. Afr. Med. 2019, 18, 121–126.
- Wang, X.; Lin, Y. Tumor necrosis factor and cancer, buddies or foes? Acta Pharmacol. Sin. 2008, 29, 1275–1288.
- Gong, P.; Canaan, A.; Wang, B.; Leventhal, J.; Snyder, A.; Nair, V.; Cohen, C.D.; Kretzler, M.; D’Agati, V.; Weissman, S.; et al. The ubiquitin-like protein FAT10 mediates NF-kB activation. J. Am. Soc. Nephrol. 2010, 21, 316–326.
- Zhang, D.W.; Jeang, K.T.; Lee, C.G. p53 negatively regulates the expression of FAT10, a gene upregulated in various cancers. Oncogene 2006, 25, 2318–2327.
- Tan, K.; Pezzella, F.; Harris, A.; Acuto, O. PO-479 NUB1 as a prognostic marker in breast cancer: A retrospective, integrated genomic, transcriptomic, and protein analysis. ESMO Open 2018, 3, A417–A418.





