Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Luca Lo Piccolo | + 1379 word(s) | 1379 | 2021-08-23 10:28:52 | | | |
| 2 | Rita Xu | Meta information modification | 1379 | 2021-09-03 12:01:09 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Lo Piccolo, L. Osteogenic Sarcoma. Encyclopedia. Available online: https://encyclopedia.pub/entry/13737 (accessed on 29 April 2026).
Lo Piccolo L. Osteogenic Sarcoma. Encyclopedia. Available at: https://encyclopedia.pub/entry/13737. Accessed April 29, 2026.
Lo Piccolo, Luca. "Osteogenic Sarcoma" Encyclopedia, https://encyclopedia.pub/entry/13737 (accessed April 29, 2026).
Lo Piccolo, L. (2021, August 31). Osteogenic Sarcoma. In Encyclopedia. https://encyclopedia.pub/entry/13737
Lo Piccolo, Luca. "Osteogenic Sarcoma." Encyclopedia. Web. 31 August, 2021.
Copy Citation
Osteosarcoma (OS) is a highly aggressive malignant bone tumor, frequently occurring in children and adolescents with an annual incidence of over three per million worldwide.
liquid biopsy
circulating long non-coding RNA
osteosarcoma
1. Introduction
Osteosarcoma (OS) is a highly aggressive malignant bone tumor, frequently occurring in children and adolescents with an annual incidence of over three per million worldwide [1][2][3]. OS represents different pathological entities based on clinical, radiological, and histopathological features. For instance, based on histopathological features, osteosarcoma can be classified into distinct subtypes with the osteoblastic, chondroblastic, and fibroblastic OS, respectively, being the most common [4].
Nowadays, various clinical practices for OS have been notably implemented, including chemotherapy, radiotherapy, surgery, and targeted therapy; yet, the prognosis for OS still remains poor [5][6]. In fact, approximately 20% of patients showed clinical metastasis at presentation, with a 5-year survival rate less than 30% [7]. For this reason, OS strongly demands reliable, non-invasive, and clinically useful biomarkers.
In contrast to conventional biopsy, the liquid biopsy of tumor components in blood represents a simple and rapid test, easily performed, and requiring a small amount of sample (usually 10–15 mL of blood). Presently, however, the usefulness of alkaline phosphatase (ALP) and lactate dehydrogenase (LDH) as laboratory markers for OS is still considered controversial [8][9]. Likewise, studies have shown that programmed cell death 1 ligand-1 (PD-L1) and bone resorption markers, such as b-isomerized C-terminal telopeptides (b-CTx) and total procollagen type 1 amino-terminal propetide (tP1NP), still require more investigation before being able to conclude their potential value as biomarkers for OS [6][10][11][12].
Recently, circulating biomarkers, such as circulating tumor cells (CTCs) and different forms of circulating-free and extracellular vesicle/platelet-encapsulated non-coding RNA, including microRNA (miRNA) and long non-coding RNA (lncRNA), have emerged as novel promising diagnostic, prognostic, or predictive biomarkers in the clinical management of patients with OS [13][14][15][16][17][18].
Although CTCs may provide tumor-specific genomic, transcriptomic, and proteomic information, their analysis requires a large volume of fresh blood and it is laborious and expensive. On the other hands, the use of circulating ncRNAs, in spite of some obvious limitations, is more accessible, cheaper, and has shown potential as a precision medicine biomarker [19]. Early studies on circulating RNAs focused on the relevance of miRNAs. However, the current search for novel OS biomarkers has possibly shifted to lncRNAs due to their relative abundance and higher stability with respect to miRNAs [14].
Interestingly, a number of circulating lncRNAs, whose expression in liquid biopsy correlate with that of cancer tissues, have emerged as novel diagnostic or prognostic markers for several types of cancer [20][21][22][23]. However, the role of circulating lncRNAs as biomarker for OS is still elusive.
2. Long Non-Coding RNA Structures and Functions
LncRNAs are conventionally classified as transcripts longer than 200 nt with no or low coding potential [24][25][26]. Similar to protein-coding transcripts, the transcription of lncRNAs is dependent on histone-modification-mediated regulation, and lncRNA’s transcripts are processed by the canonical spliceosome machinery. Overall, lncRNA genes show fewer exons than mRNAs, and appear to be under a weaker selective pressure during evolution. Moreover, some lncRNAs are expressed at levels lower than those of mRNAs and in a more tissue- and cell-specific manner, while others are known to be fairly abundant and are expressed in diverse cell types, such as the “house-keeping” genes [27].
Of tens of thousands of metazoan lncRNAs discovered from cDNA libraries and RNAseq data by high throughput transcriptome projects, only a handful of lncRNAs have been functionally characterized. The investigations on this small cohort of lncRNAs have demonstrated that these noncoding transcripts can serve as scaffolds or guides to regulate protein–protein or protein–DNA interactions [28][29][30][31] or can modulate post-translational modification of nonhistone proteins [32]. Moreover, lncRNAs are capable of controlling microRNAs (miRNAs) [33][34][35], and function as enhancers to influence gene transcription, when transcribed from the enhancer regions (enhancer RNA) [36][37][38] or their neighboring loci (noncoding RNA activator) [39][40].
Several lines of evidence have shown that lncRNAs are capable of influencing different cellular functions that are critical to tumorigenesis, such as cell proliferation, differentiation, migration, immune response, and apoptosis [41][42][43][44][45][46][47]. Furthermore, lncRNAs have been found to act as tumor suppressors or oncogenes [48][49][50][51], and, of note, a number of lncRNAs have been reported to be significantly deregulated in tumors [52][53][54][55].
3. Origin of Circulating lncRNAs
The precise mechanism of lncRNAs release into the extracellular environment is not completely understood. Hypotheses have arisen that tumor cells, cancer-adjacent normal cells, immune cells, and other blood cells may all release lncRNAs [56][57], as shown in Figure 1. A few studies reported that lncRNAs can be encapsulated into membrane vesicles, such as exosomes or microvesicles (EV), prior to being secreted extracellularly. In such a conformation, the circulating lncRNAs have shown a higher degree stability, probably due to EVs offering protection against the nuclease-mediated degradation that may occur in the extracellular space and in body fluids [20][58][59] (Figure 1). On the other hand, other studies have suggested that the secretion pathway of lncRNAs may also occur in a similar manner to that for miRNAs. As such, lncRNAs might also be released into body fluids in an EV-independent fashion as complexes with high-density lipoproteins (HDLs) or protein Argonaute 2 (AGO2) [60] (Figure 1).
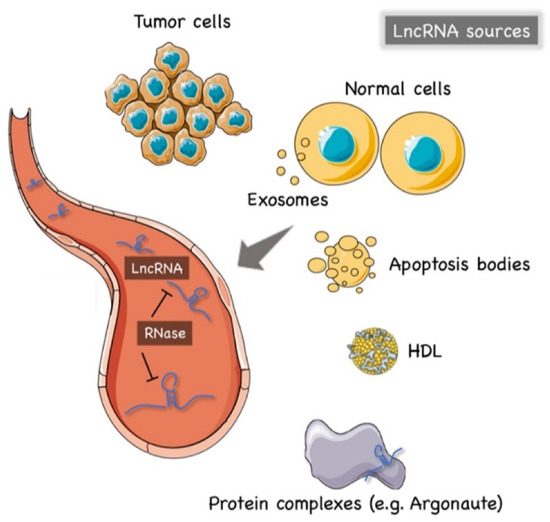
Figure 1. The origin of circulating lncRNAs. Two major sources of circulating lncRNAs have been postulated so far. LncRNAs can be encapsulated in extracellular vesicles (EV), predominantly exosomes. On the other hand, lncRNAs can also be released from live cells in an EV-independent fashion, thus, similar to circulating miRNAs, circulating lncRNAs might be detected in complexes with protein or high-density lipoproteins (HDL). The latter mechanism is likely to offer = circulating lncRNAs less protection against ribonucleases that are normally present in the extracellular space and body fluids. Created by Servier Medical Art (http://smart.servier.com/ (accessed on 15 May 2021)), licensed under Creative Commons Attribution 3.0 Unported License (https://creativecommons.org/licenses/by/3.0/ (accessed on 15 May 2021)).
The hypothesis of an EV-independent mechanism for lncRNAs secretion might seem less likely given the high abundance of ribonucleases in serum, plasma, and other bodily fluids that can dramatically affect the stability of lncRNAs in the extracellular environment. However, one can speculate that circulating lncRNAs can be capable of resisting the RNase activity through modifications such as methylation, adenylation, and uridylation [61] or via the formation of higher order structures [62].
4. Detection Methods of Circulating lncRNAs
Difference sources of liquid biopsy (i.e., whole blood, plasma, serum, urine, and gastric juice) can be used to quantify circulating lncRNAs. However, due to the possibility of blood cell RNA contamination, whole blood is the less recommended option so far [63]. In addition, EDTA-anticoagulant collecting tubes have been suggested to be more suitable for the analysis of circulating lncRNAs [57]. Of note, some studies have found that lncRNAs remained stable in plasma even under multiple cycles of freeze–thaw, incubation at 45 °C, or storage at room temperature for as long as 24 h [56].
Overall, the methods to extract circulating lncRNAs can be divided into two major groups: guanidine/phenol/chloroform-based and column-based protocols. The column-based method is currently considered more reliable, since organic and phenolic contaminants in TRIzol-based methods might invalidate results [64].
Regarding the measuring and normalization methods, some studies have suggested that the use of an equal volume of input RNA sample may be more accurate than an equal amounts of RNA measured using a NanoDrop spectrophotometer since many diseases, including cancer, may indeed release an higher degree of RNAs into body fluids than healthy control groups, leading to a significantly higher level of circulating RNA in cancer patients that causes misleading results [64].
To date, quantitative real-time PCR (qRT-PCR) is still considered the gold standard for quantitative expression analysis of lncRNAs, including circulating lncRNAs [65]. Microarrays and whole transcriptome analysis (RNA-seq) still have limited uses in this field. In fact, the high throughput potential of microarrays relies on a reference database of targets, which in the case of circulating lncRNAs, is still very limited [21]; the RNA-seq requires huge amounts of starting RNA samples. Additionally, RNA-seq is currently expensive and needs special equipment and/or expert bioinformaticians [64], whereas, the targeted-approach of qRT-PCR is still more accessible, and saves money and time. Accordingly, qRT-PCR can be divided into relative and absolute analyses. In relative quantification methods, the choice of endogenous controls is critical to properly normalize the expression levels. In this regard, it must be noted that no systematic evaluation of reference genes for serum lncRNA has yet been reported, posing some limitations for the relative qRT-PCR method in the analysis of lncRNAs from a liquid biopsy.
References
- Ottaviani, G.; Jaffe, N. The etiology of osteosarcoma. Cancer Treat Res. 2009, 152, 15–32.
- Gianferante, D.M.; Mirabello, L.; Savage, S.A. Germline and somatic genetics of osteosarcoma—Connecting aetiology, biology and therapy. Nat. Rev. Endocrinol. 2017, 13, 480–491.
- Lindsey, B.A.; Markel, J.E.; Kleinerman, E.S. Osteosarcoma Overview. Rheumatol. Ther. 2017, 4, 25–43.
- Klein, M.J.; Siegal, G.P. Osteosarcoma: Anatomic and histologic variants. Am. J. Clin. Pathol. 2006, 125, 555–581.
- Marchandet, L.; Lallier, M.; Charrier, C.; Baud’huin, M.; Ory, B.; Lamoureux, F. Mechanisms of Resistance to Conventional Therapies for Osteosarcoma. Cancers 2021, 13, 683.
- Gazouli, I.; Kyriazoglou, A.; Kotsantis, I.; Anastasiou, M.; Pantazopoulos, A.; Prevezanou, M.; Chatzidakis, I.; Kavourakis, G.; Economopoulou, P.; Kontogeorgakos, V.; et al. Systematic Review of Recurrent Osteosarcoma Systemic Therapy. Cancers 2021, 13, 1757.
- Misaghi, A.; Goldin, A.; Awad, M.; Kulidjian, A.A. Osteosarcoma: A comprehensive review. SICOT J. 2018, 4, 12.
- Zhong, J.; Hu, Y.; Si, L.; Geng, J.; Xing, Y.; Jiao, Q.; Zhang, H.; Yao, W. Clarifying prognostic factors of small cell osteosarcoma: A pooled analysis of 20 cases and the literature. J. Bone Oncol. 2020, 24, 100305.
- Kim, S.H.; Shin, K.H.; Moon, S.H.; Jang, J.; Kim, H.S.; Suh, J.S.; Yang, W.I. Reassessment of alkaline phosphatase as serum tumor marker with high specificity in osteosarcoma. Cancer Med. 2017, 6, 1311–1322.
- Cao, L.L.; Chen, Z.; Yue, Z.; Pei, L.; Jia, M.; Wang, H.; Li, T. Novel classifiers with clinical laboratory parameters for early detection of osteosarcoma. J. Clin. Lab. Anal. 2020, 34, e23189.
- Huang, X.; Zhang, W.; Zhang, Z.; Shi, D.; Wu, F.; Zhong, B.; Shao, Z. Prognostic Value of Programmed Cell Death 1 Ligand-1 (PD-L1) or PD-1 Expression in Patients with Osteosarcoma: A Meta-Analysis. J. Cancer 2018, 9, 2525–2531.
- Hu, T.; Yang, Q.; Xu, J.; Zhang, Z.; He, N.; Du, Y. Role of beta-isomerized C-terminal telopeptides (beta-CTx) and total procollagen type 1 amino-terminal propeptide (tP1NP) as osteosarcoma biomarkers. Int. J. Clin. Exp. Med. 2015, 8, 890–896.
- Zuo, Z.; Hu, H.; Xu, Q.; Luo, X.; Peng, D.; Zhu, K.; Zhao, Q.; Xie, Y.; Ren, J. BBCancer: An expression atlas of blood-based biomarkers in the early diagnosis of cancers. Nucleic Acids Res. 2020, 48, D789–D796.
- Szilagyi, M.; Pos, O.; Marton, E.; Buglyo, G.; Soltesz, B.; Keseru, J.; Penyige, A.; Szemes, T.; Nagy, B. Circulating Cell-Free Nucleic Acids: Main Characteristics and Clinical Application. Int. J. Mol. Sci. 2020, 21, 6827.
- Dudley, J.C.; Diehn, M. Detection and Diagnostic Utilization of Cellular and Cell-Free Tumor DNA. Annu. Rev. Pathol. 2021, 16, 199–222.
- Alix-Panabieres, C.; Pantel, K. Liquid Biopsy: From Discovery to Clinical Application. Cancer Discov. 2021, 11, 858–873.
- Botti, G.; Giordano, A.; Feroce, F.; De Chiara, A.R.; Cantile, M. Noncoding RNAs as circulating biomarkers in osteosarcoma patients. J. Cell Physiol. 2019, 234, 19249–19255.
- Aran, V.; Devalle, S.; Meohas, W.; Heringer, M.; Cunha Caruso, A.; Pinheiro Aguiar, D.; Leite Duarte, M.E.; Moura Neto, V. Osteosarcoma, chondrosarcoma and Ewing sarcoma: Clinical aspects, biomarker discovery and liquid biopsy. Crit. Rev. Oncol. Hematol. 2021, 162, 103340.
- Anfossi, S.; Babayan, A.; Pantel, K.; Calin, G.A. Clinical utility of circulating non-coding RNAs—An update. Nat. Rev. Clin. Oncol. 2018, 15, 541–563.
- Guo, X.; Lv, X.; Ru, Y.; Zhou, F.; Wang, N.; Xi, H.; Zhang, K.; Li, J.; Chang, R.; Xie, T.; et al. Circulating Exosomal Gastric Cancer-Associated Long Noncoding RNA1 as a Biomarker for Early Detection and Monitoring Progression of Gastric Cancer: A Multiphase Study. JAMA Surg. 2020, 155, 572–579.
- Shi, J.; Li, X.; Zhang, F.; Zhang, C.; Guan, Q.; Cao, X.; Zhu, W.; Zhang, X.; Cheng, Y.; Ou, K.; et al. Circulating lncRNAs associated with occurrence of colorectal cancer progression. Am. J. Cancer Res. 2015, 5, 2258–2265.
- Abedini, P.; Fattahi, A.; Agah, S.; Talebi, A.; Beygi, A.H.; Amini, S.M.; Mirzaei, A.; Akbari, A. Expression analysis of circulating plasma long noncoding RNAs in colorectal cancer: The relevance of lncRNAs ATB and CCAT1 as potential clinical hallmarks. J. Cell Physiol. 2019, 234, 22028–22033.
- Benoist, G.E.; Van Oort, I.M.; Boerrigter, E.; Verhaegh, G.W.; Van Hooij, O.; Groen, L.; Smit, F.; De Mol, P.; Hamberg, P.; Dezentje, V.O.; et al. Prognostic Value of Novel Liquid Biomarkers in Patients with Metastatic Castration-Resistant Prostate Cancer Treated with Enzalutamide: A Prospective Observational Study. Clin. Chem. 2020, 66, 842–851.
- Batista, P.J.; Chang, H.Y. Long noncoding RNAs: Cellular address codes in development and disease. Cell 2013, 152, 1298–1307.
- Song, Z.; Lin, J.; Li, Z.; Huang, C. The nuclear functions of long noncoding RNAs come into focus. Noncoding RNA Res. 2021, 6, 70–79.
- Uszczynska-Ratajczak, B.; Lagarde, J.; Frankish, A.; Guigo, R.; Johnson, R. Towards a complete map of the human long non-coding RNA transcriptome. Nat. Rev. Genet. 2018, 19, 535–548.
- Quinn, J.J.; Chang, H.Y. Unique features of long non-coding RNA biogenesis and function. Nat. Rev. Genet. 2016, 17, 47–62.
- Jarroux, J.; Foretek, D.; Bertrand, C.; Gabriel, M.; Szachnowski, U.; Saci, Z.; Guo, S.; Londono-Vallejo, A.; Pinskaya, M.; Morillon, A. HOTAIR lncRNA promotes epithelial-mesenchymal transition by redistributing LSD1 at regulatory chromatin regions. EMBO Rep. 2021, 22, e50193.
- Zhao, P.; Ji, M.M.; Fang, Y.; Li, X.; Yi, H.M.; Yan, Z.X.; Cheng, S.; Xu, P.P.; Janin, A.; Wang, C.F.; et al. A novel lncRNA TCLlnc1 promotes peripheral T cell lymphoma progression through acting as a modular scaffold of HNRNPD and YBX1 complexes. Cell Death Dis. 2021, 12, 321.
- Wanowska, E.; Kubiak, M.; Makalowska, I.; Szczesniak, M.W. A chromatin-associated splicing isoform of OIP5-AS1 acts in cis to regulate the OIP5 oncogene. RNA Biol. 2021, 1–12.
- Ma, P.; Pan, Y.; Yang, F.; Fang, Y.; Liu, W.; Zhao, C.; Yu, T.; Xie, M.; Jing, X.; Wu, X.; et al. KLF5-Modulated lncRNA NEAT1 Contributes to Tumorigenesis by Acting as a Scaffold for BRG1 to Silence GADD45A in Gastric Cancer. Mol. Ther. Nucleic Acids 2020, 22, 382–395.
- Jantrapirom, S.; Koonrungsesomboon, N.; Yoshida, H.; Candeias, M.M.; Pruksakorn, D.; Lo Piccolo, L. Long noncoding RNA-dependent methylation of nonhistone proteins. Wiley Interdiscip Rev. RNA 2021, e1661.
- Hansen, T.B.; Jensen, T.I.; Clausen, B.H.; Bramsen, J.B.; Finsen, B.; Damgaard, C.K.; Kjems, J. Natural RNA circles function as efficient microRNA sponges. Nature 2013, 495, 384–388.
- Munz, N.; Cascione, L.; Parmigiani, L.; Tarantelli, C.; Rinaldi, A.; Cmiljanovic, N.; Cmiljanovic, V.; Giugno, R.; Bertoni, F.; Napoli, S. Exon-Intron Differential Analysis Reveals the Role of Competing Endogenous RNAs in Post-Transcriptional Regulation of Translation. Noncoding RNA 2021, 7, 26.
- Zhao, Y.; Yu, Z.; Ma, R.; Zhang, Y.; Zhao, L.; Yan, Y.; Lv, X.; Zhang, L.; Su, P.; Bi, J.; et al. lncRNA-Xist/miR-101-3p/KLF6/C/EBPalpha axis promotes TAM polarization to regulate cancer cell proliferation and migration. Mol. Ther. Nucleic Acids 2021, 23, 536–551.
- Li, W.; Notani, D.; Ma, Q.; Tanasa, B.; Nunez, E.; Chen, A.Y.; Merkurjev, D.; Zhang, J.; Ohgi, K.; Song, X.; et al. Functional roles of enhancer RNAs for oestrogen-dependent transcriptional activation. Nature 2013, 498, 516–520.
- Huang, Z.; Yu, H.; Du, G.; Han, L.; Huang, X.; Wu, D.; Han, X.; Xia, Y.; Wang, X.; Lu, C. Enhancer RNA lnc-CES1-1 inhibits decidual cell migration by interacting with RNA-binding protein FUS and activating PPARgamma in URPL. Mol. Ther. Nucleic Acids 2021, 24, 104–112.
- Yang, M.; Lee, J.H.; Zhang, Z.; De La Rosa, R.; Bi, M.; Tan, Y.; Liao, Y.; Hong, J.; Du, B.; Wu, Y.; et al. Enhancer RNAs Mediate Estrogen-Induced Decommissioning of Selective Enhancers by Recruiting ERalpha and Its Cofactor. Cell Rep. 2020, 31, 107803.
- Lai, F.; Orom, U.A.; Cesaroni, M.; Beringer, M.; Taatjes, D.J.; Blobel, G.A.; Shiekhattar, R. Activating RNAs associate with Mediator to enhance chromatin architecture and transcription. Nature 2013, 494, 497–501.
- Nieminen, T.; Scott, T.A.; Lin, F.M.; Chen, Z.; Yla-Herttuala, S.; Morris, K.V. Long Non-Coding RNA Modulation of VEGF-A during Hypoxia. Noncoding RNA 2018, 4, 34.
- Zhao, H.; Liu, X.; Yu, L.; Lin, S.; Zhang, C.; Xu, H.; Leng, Z.; Huang, W.; Lei, J.; Li, T.; et al. Comprehensive landscape of epigenetic-dysregulated lncRNAs reveals a profound role of enhancers in carcinogenesis in BC subtypes. Mol. Ther. Nucleic Acids 2021, 23, 667–681.
- Vollmers, A.C.; Covarrubias, S.; Kuang, D.; Shulkin, A.; Iwuagwu, J.; Katzman, S.; Song, R.; Viswanathan, K.; Vollmers, C.; Wakeland, E.; et al. A conserved long noncoding RNA, GAPLINC, modulates the immune response during endotoxic shock. Proc. Natl. Acad. Sci. USA 2021, 118, e2016648118.
- Bian, X.; Sun, Y.M.; Wang, L.M.; Shang, Y.L. ELK1-induced upregulation lncRNA LINC02381 accelerates the osteosarcoma tumorigenesis through targeting CDCA4 via sponging miR-503-5p. Biochem. Biophys. Res. Commun. 2021, 548, 112–119.
- Hung, T.; Wang, Y.; Lin, M.F.; Koegel, A.K.; Kotake, Y.; Grant, G.D.; Horlings, H.M.; Shah, N.; Umbricht, C.; Wang, P.; et al. Extensive and coordinated transcription of noncoding RNAs within cell-cycle promoters. Nat. Genet. 2011, 43, 621–629.
- Zhu, G.; Luo, H.; Feng, Y.; Guryanova, O.A.; Xu, J.; Chen, S.; Lai, Q.; Sharma, A.; Xu, B.; Zhao, Z.; et al. HOXBLINC long non-coding RNA activation promotes leukemogenesis in NPM1-mutant acute myeloid leukemia. Nat. Commun. 2021, 12, 1956.
- Tripathi, V.; Shen, Z.; Chakraborty, A.; Giri, S.; Freier, S.M.; Wu, X.; Zhang, Y.; Gorospe, M.; Prasanth, S.G.; Lal, A.; et al. Long noncoding RNA MALAT1 controls cell cycle progression by regulating the expression of oncogenic transcription factor B-MYB. PLoS Genet. 2013, 9, e1003368.
- Jiang, N.; Zhang, X.; Gu, X.; Li, X.; Shang, L. Progress in understanding the role of lncRNA in programmed cell death. Cell Death Discov. 2021, 7, 30.
- Montes, M.; Lubas, M.; Arendrup, F.S.; Mentz, B.; Rohatgi, N.; Tumas, S.; Harder, L.M.; Skanderup, A.J.; Andersen, J.S.; Lund, A.H. The long non-coding RNA MIR31HG regulates the senescence associated secretory phenotype. Nat. Commun. 2021, 12, 2459.
- You, Z.; Xu, S.; Pang, D. Long noncoding RNA PVT1 acts as an oncogenic driver in human pan-cancer. J. Cell Physiol. 2020, 235, 7923–7932.
- Han, C.; Li, H.; Ma, Z.; Dong, G.; Wang, Q.; Wang, S.; Fang, P.; Li, X.; Chen, H.; Liu, T.; et al. MIR99AHG is a noncoding tumor suppressor gene in lung adenocarcinoma. Cell Death Dis. 2021, 12, 424.
- Song, J.H.; Tieu, A.H.; Cheng, Y.; Ma, K.; Akshintala, V.S.; Simsek, C.; Prasath, V.; Shin, E.J.; Ngamruengphong, S.; Khashab, M.A.; et al. Novel Long Noncoding RNA miR205HG Functions as an Esophageal Tumor-Suppressive Hedgehog Inhibitor. Cancers 2021, 13, 1707.
- Hong, W.; Liang, L.; Gu, Y.; Qi, Z.; Qiu, H.; Yang, X.; Zeng, W.; Ma, L.; Xie, J. Immune-Related lncRNA to Construct Novel Signature and Predict the Immune Landscape of Human Hepatocellular Carcinoma. Mol. Ther. Nucleic Acids 2020, 22, 937–947.
- Du, Z.; Fei, T.; Verhaak, R.G.; Su, Z.; Zhang, Y.; Brown, M.; Chen, Y.; Liu, X.S. Integrative genomic analyses reveal clinically relevant long noncoding RNAs in human cancer. Nat. Struct. Mol. Biol. 2013, 20, 908–913.
- Hua, J.T.; Chen, S.; He, H.H. Landscape of Noncoding RNA in Prostate Cancer. Trends Genet. 2019, 35, 840–851.
- Chu, K.J.; Ma, Y.S.; Jiang, X.H.; Wu, T.M.; Wu, Z.J.; Li, Z.Z.; Wang, J.H.; Gao, Q.X.; Yi, B.; Shi, Y.; et al. Whole-Transcriptome Sequencing Identifies Key Differentially Expressed mRNAs, miRNAs, lncRNAs, and circRNAs Associated with CHOL. Mol. Ther. Nucleic Acids 2020, 21, 592–603.
- Arita, T.; Ichikawa, D.; Konishi, H.; Komatsu, S.; Shiozaki, A.; Shoda, K.; Kawaguchi, T.; Hirajima, S.; Nagata, H.; Kubota, T.; et al. Circulating long non-coding RNAs in plasma of patients with gastric cancer. Anticancer Res. 2013, 33, 3185–3193.
- Ren, S.; Wang, F.; Shen, J.; Sun, Y.; Xu, W.; Lu, J.; Wei, M.; Xu, C.; Wu, C.; Zhang, Z.; et al. Long non-coding RNA metastasis associated in lung adenocarcinoma transcript 1 derived miniRNA as a novel plasma-based biomarker for diagnosing prostate cancer. Eur. J. Cancer 2013, 49, 2949–2959.
- Li, Q.; Shao, Y.; Zhang, X.; Zheng, T.; Miao, M.; Qin, L.; Wang, B.; Ye, G.; Xiao, B.; Guo, J. Plasma long noncoding RNA protected by exosomes as a potential stable biomarker for gastric cancer. Tumour Biol. 2015, 36, 2007–2012.
- Dong, L.; Lin, W.; Qi, P.; Xu, M.D.; Wu, X.; Ni, S.; Huang, D.; Weng, W.W.; Tan, C.; Sheng, W.; et al. Circulating Long RNAs in Serum Extracellular Vesicles: Their Characterization and Potential Application as Biomarkers for Diagnosis of Colorectal Cancer. Cancer Epidemiol. Biomark. Prev. 2016, 25, 1158–1166.
- Arroyo, J.D.; Chevillet, J.R.; Kroh, E.M.; Ruf, I.K.; Pritchard, C.C.; Gibson, D.F.; Mitchell, P.S.; Bennett, C.F.; Pogosova-Agadjanyan, E.L.; Stirewalt, D.L.; et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl. Acad. Sci. USA 2011, 108, 5003–5008.
- Redova, M.; Sana, J.; Slaby, O. Circulating miRNAs as new blood-based biomarkers for solid cancers. Future Oncol. 2013, 9, 387–402.
- Reis, E.M.; Verjovski-Almeida, S. Perspectives of Long Non-Coding RNAs in Cancer Diagnostics. Front. Genet. 2012, 3, 32.
- Pritchard, C.C.; Kroh, E.; Wood, B.; Arroyo, J.D.; Dougherty, K.J.; Miyaji, M.M.; Tait, J.F.; Tewari, M. Blood cell origin of circulating microRNAs: A cautionary note for cancer biomarker studies. Cancer Prev. Res. 2012, 5, 492–497.
- Qi, P.; Zhou, X.Y.; Du, X. Circulating long non-coding RNAs in cancer: Current status and future perspectives. Mol. Cancer 2016, 15, 39.
- Shi, T.; Gao, G.; Cao, Y. Long Noncoding RNAs as Novel Biomarkers Have a Promising Future in Cancer Diagnostics. Dis. Markers 2016, 2016, 9085195.
More
Information
Subjects:
Oncology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
690
Revisions:
2 times
(View History)
Update Date:
03 Sep 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





