| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mohsen Fallah Vostakola | + 2220 word(s) | 2220 | 2021-08-26 15:59:49 |
Video Upload Options
This review summarizes the current status, operating principles, and recent advances in high-temperature polymer electrolyte membranes (HT-PEMs), with a particular focus on the recent developments, technical challenges and commercial prospects of the HT-PEM fuel cells. A detailed review of the most recent research activities has been covered by this work, with a major focus on the state-of-the-art concepts describing the proton conductivity and degradation mechanisms of HT-PEMs. Also, the fuel cell performance and the lifetime of HT-PEM fuel cells as a function of operating conditions have been discussed. In addition, the review will highlight the important outcomes found in recent literature about the HT-PEM fuel cell. The main objectives of this review paper will be as follows: (1) the latest development of the HT-PEMs, primarily based on polybenzimidazole membranes, (2) the latest development of the fuel cell performance and the lifetime of the HT-PEMs.
1. Introduction
Sufficient access to clean energy sources is one of the ongoing key challenges for global development that directly impacts industrial development, economic growth, and human well-being. Historically, the energy sector is widely dominated based on fossil fuels (such as petroleum fuels, natural gas, coal, etc.) which are the primary source of carbon dioxide (CO2) and the other greenhouse gases emissions to the environment. This has fundamentally driven a global climate change that has been accelerated over the past few decades and hence needs significant and immediate actions in order to alter both the energy sources and energy conversion techniques. There is a growing movement by the research and manufacturing communities to alleviate the impact of the petroleum-based economy by developing clean energy sources for implementing an alternative hydrogen-based economy [1][2][3]. Given the current environmental challenges associated with fossil fuels, fuel cell technology has been introduced as a promising, cleaner, high energy density, and more efficient power generation system [2][4][5]. Lately, polymer electrolyte membrane (PEM) fuel cells, which directly convert the chemical energy of hydrogen into electrical energy, have been predominantly developed as the most common commercial fuel cell [6][7].Hydrogen gas (as a fuel with the highest energy density) and oxygen (usually taken from air) are being used in a PEM fuel cell over the surface of electrodes to produce water through a few electrochemical reactions that are associated with the electrical power generation [8].
2. Desirable properties of PEMs
The PEM is the heart of the membrane electrode assemblies (MEAs) that are used for the fabrication of the PEM fuel cell stacks [9]. The PEM has a vital role in a fuel cell assembly by performing various functions such as being a carrier path for proton transport from the anode to the cathode side, a dense separation layer to block mixing the reactants, and an electric insulation layer between the anode and cathode. Many studies in the past few years have reported the enhancement of various functions of PEMs [10][11][12][13]. One of the key objectives for the PEMs development is to reduce the cells' total fabrication cost and improve their electrochemical performance and durability [14][15]. A desirable cost-effective PEM should exhibit an acceptable thermochemical and thermomechanical stability, low permeability to fuel and oxidants, high proton conductivity, high compatibility with the electrodes in an MEA, long durability, and low electro-osmotic drag coefficient [16].
3. Improving proton conductivity of the membranes
3.1. Effects of dopants and additives
Several dopants have been used to improve the proton conductivity of PEMs, especially PBI. The proton conductivity of PBI is very low, and it requires the incorporation of dopants, additives, etc. [17]. Full or partial protonation or deprotonation of the polymers happens depending on the concentration and chemical nature of the dopant [18]. PBI is an amphoteric compound and has both proton acceptor and proton donor sites [19]. A wide variety of acids and bases, such as H2SO4 [20], H3PO4 [21], NaOH [22], KOH [23], etc., have been used as dopants. Compared to the low-temperature cells, HT-PEMs require high-boiling dopants which can operate at elevated operational temperatures. Dopant concentration is a key parameter in improving proton conductivity, however, up to a certain amount. Excessive doping may deteriorate the conductivity. For example, acid doping with a high concentration (>11 mol.L-1) decreases the proton conductivity [18].
3.2. Effect of molecular weight
Asensio et al. [24] reported that molecular weight (MW) has a vital role in improving mechanical strength. In order to prepare a highly conductive membrane, the polymer matrix with a comparably higher molecular weight should be applied. However, such an approach may not have a significant impact on the resulting proton conductivity of PEMs.
3.3. Polymer composites
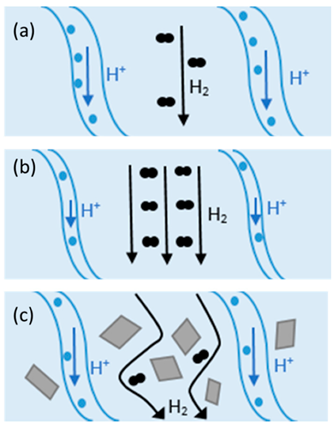
An effective approach to improve the proton conductivity of PEMs is to incorporate some additional components. Composite membranes showed promising potential to be used as HT-PEMFCs. Composite materials consist of two or more constituents with different chemical, mechanical, or physical properties [25]. Composite membranes have widely been used in PEMFCs such as PTFE/Nafion [25], metal-oxide-recast/Nafion [26], copper phthalocyanine tetrasulfonic acid tetrasodium salt (CuTSPc)/Nafion [27], calcium titanate/PBI [28], PBI/graphene oxide [29], etc. The change in H2 crossover and proton conductivity at different operating conditions with/without filler in Nafion is illustrated in Figure 1. As shown in Figure 1(a), the channels within the membrane are fully saturated with water and the mechanisms for proton conduction can be either “Grotthus” or diffusion. In this case, open circuit voltage (OCV) can be reduced due to the molecular H2 passing through the membrane (H2 crossover). At higher temperatures (Figure 1(b)), water begins evaporation and results in the shrinkage of the channels and decreasing proton conductivity. On the other hand, H2 crossover improves at elevated temperatures. Adding filler to the composite (Figure 1(c)) can decrease H2 crossover and increase H2 path to migrate from anode to cathode .[25]
Figure 1 Proton and H2 transport at (a) 80 °C, (b) at elevated temperatures, and (c) in Nafion composite at high temperatures [25]
Mixed-matrix membranes (MMMs) are a class of membranes comprised of a solid phase uniformly distributed in a polymer matrix. MMMs benefit from the advantages of the polymer membrane (high flexibility and ion exchange capacity) and inorganic constituents (high thermal and mechanical properties, water uptake, and proton conductivity) [30]. Among the inorganic materials, metal-organic frameworks such as Fe, Cr, Al, and Zr have high specific surface areas and offer higher proton conductivity. These MOFs can improve proton conductivity by defect engineering, post-synthetic modification, and impregnation with acidic molecules [31].
4. Proton conduction mechanism
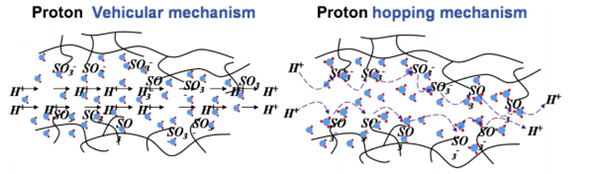
Numerous reports have been studied the proton conduction mechanism in PEM fuel cells; however, the exact mechanism of proton conduction in the PEMs has not been fully recognised and understood yet. Among various theories introduced so far, the ‘Vehicular’ and ‘Grotthuss’ mechanisms have been more commonly approved to explain the proton transfer mechanisms in PEMs [32][33]. Usually, the PEMS with activation energy below 14 KJ.mol-1 can be explained better with the ‘Vehicular’ mechanism, while those exhibit higher activation energies are better represented with the ‘Grotthus’ mechanism [34]. In fact, the proton transfer explained by both the ‘Vehicular’ and Grotthus mechanisms does not act independently in PEMs. Both theories can make helpful assistances to understand better the proton conduction mechanism in PEMs. The proton transfer happens with a ‘vehicle’ or proton solvent (for example, water molecules as hydrates in the PEM molecular structure) with providing a suitable diffusion rate in the ‘Vehicular’ mechanism [35]. In the ‘Grotthuss’ mechanism known as the ‘hopping’ mechanism, a vehicle or proton solvent is not essential. Protons could simply jump from one site to another alongside the proton acceptor sites in the PEMs backbone [36]. Figure 2 displays a schematic diagram illustrating both the ‘Vehicular’ and ‘hopping’ mechanisms for proton conduction paths in PEMs.
Figure 2 The Schematic design of the Vehicular Mechanism and hopping mechanism as proton conduction in PEMs [37][38].
5. Degradation and fuel cell performance
The degradation of the PA-doped PBI-based membrane can be initiated by the chemical degradation, which happens by H containing end-groups attacked with hydrogen peroxide and its radicals that finally results in a mechanical/thermal degradation [39]. The N-H bond attack in the imidazole ring causes a ring-opening for imidazole and ‘scission’ of the macromolecular chain, which can be studied by FTIR spectra. In the PA-doped PBI membranes, the PA has an inhibiting effect on the decomposition of H2O2 and hence can decrease the chemical degradation of the PBI membrane [39]. A research study in this field has reported that the PBI membrane displayed 15% weight loss after 20 h of exposure to a 3% H2O2, which was higher than that of the Nafion 117 membrane [40]. In another study, the stress at the break of the PBI membrane dropped from 52.9 MPa to 33.9 MPa due to the peroxide attack [41]. The physical properties can be considered as an essential factor in the degradation of the PEMs. Deformation of the membrane, which takes place due to the compressive forces of the bipolar plates in MEAs, leads to pinhole formation and can decline the chemical degradation of the PEMs [42]. The swelling and shrinking of the PBI membrane can cause mechanical stress to the membrane. From thermal stability results, there is no significant weight loss at 150 °C and 500 °C [43][44].
6. Experimental characterization
6.1. In-situ characterisation
The polarization curve named the I – V curve can be measured by less expensive electrochemical devices and methods. The method can be done using a potentiostat-galvanostat analyser over a range of temperatures and hydrogen/oxygen flow rates [45]. The impedance spectroscopy and cyclic voltammetry are known as more expensive methods compared with the polarization curve [46][47]. In this method, the electrical current is changed, and the equivalent voltage output is measured and plotted against the current density. The voltage output is an important parameter as well that shows the figure of merit for a fuel cell [48].
6.2. Ex-situ characterisation
Ex-situ characterization techniques can be used in complementarity with in-situ electrochemical techniques. These techniques significantly affect PEM fuel cells' progress, particularly to understand the morphology of their component and stability [49][50]. The Raman spectroscopy, infrared (IR), and nuclear magnetic resonance (NMR) spectra have been used to explore the PEMs and their protonation by different acids [29][39].
6.3. In-plane and through-plane techniques
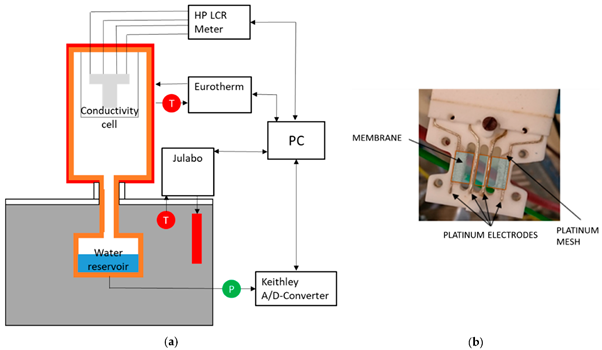
In-plane conductivity measurement is a technique in which two platinum plate electrodes and a sheet of the sample (membrane) are mounted on a Teflon cell, and two parallel Pt electrodes are placed on one side of the membrane. To measure the proton conductivity in liquid water or at relative humidity lower than 100%, the cell is placed in a distilled deionized water or a humid chamber with controlled humidity (thermos-controlled humid chamber), respectively [51].
Figure 3 Schematic of in-plane test set-up [52].
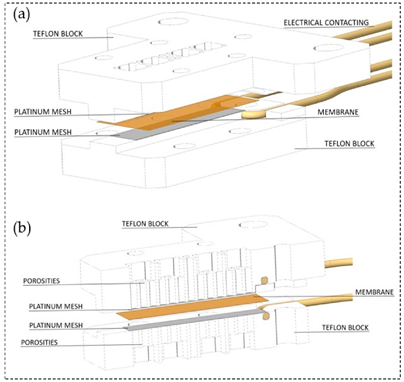
Another technique for measuring the proton conduction is through-plane conductivity. The test set-up is comprised of two PTFE blocks that build the framework and the membrane sheet is placed between two Pt electrodes. To determine the resistance, the high-frequency intercept of the impedance with the real axis should be used [52][51].
Figure 4 Schematic of through-plane test set-up [52]
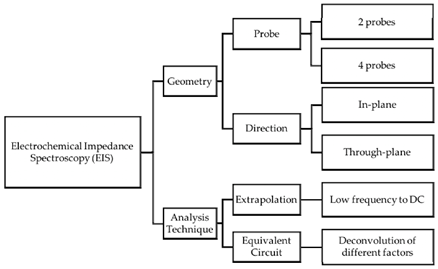
6.4. The 2-probe electrodes and 4-probe electrodes cells
In terms of the functional probe configuration, the EIS measurement can be classified into two major groups: 2-probe and 4-probe configurations [53]. In the 2-probe configuration, the electrodes operate under low frequencies and an alternating electric field. Before polarity reversal, some ions may reach the electrodes, and thereby the electric field in the membrane decreases (electrode blocking) [54]. In the 4-probe configuration, the charge build-up effect near the electrodes diminishes because of using different electrodes with sufficient distance from the charge build-up region. The distance between the probes in both configurations plays an important in measuring the conductivity [55]. It has been reported that the 2-probe configuration can be used for high-resistance materials because of the negligible impedance in the circuit. On the other hand, the 4-probe arrangement is the appropriate choice for the ionic conducting materials with low resistivity due to the diminished interfacial effects in these materials [55].
A summary of the different possible configurations (geometry and analysis techniques) for the EIS measurements is shown in Figure 5.
Figure 5 Possible configurations for EIS measurement [55].
6.5. Proton conductivity and PA doping level
The proton conductivity of PA-doped PBI membranes has been studied by several researchers. The researchers measured the anhydrous proton conductivity of different PA-doped PBI-based PEMs and proposed that the conductivity mechanism is dominated by the proton transfer from an -N site to a neighbouring vacant one by the phosphate counter-ion through a Grotthus mechanism. They confirmed counter-ion mobility by 1H and 31P NMR analysis [56]. The bound- and unbound-PA molecules and H2PO4− anion in PA-doped PBI-based membranes mainly participate in the proton conduction through the ‘Grotthus’ mechanism [57]. Generally, the PA doping level (PAdop) has been achieved by dipping the membranes into PA (85 wt.%) for five days [57]. In this method, the membranes were dried in a vacuum oven at 60 °C for 24 h, and the doping level was determined by measuring the absorbed acid weight by the membranes [58].
6.6. Thermal, chemical, and mechanical stability
The Fenton test has been widely used to study the chemical degradation of PBI-based PEMs [118]. The chemical stability of PEMs is of much concern to the lifetime of PEM fuel cells. In the Fenton test, ferrous ions (Fe2+/Fe3+) were used as the catalyst for H2O2 decomposition. The chemical stability of the PBI membrane (weight loss) was measured in H2O2 solutions having a trace amount of ferrous ions (Fe2+/Fe3+) [39]. The •OH or •OOH radicals form in situ from the decomposition of H2O2 and attack the polymer chain that has hydrogen bonding. The PEMs based on perfluorinated sulphonic acid such as Nafion display high chemical stability than those based on polyaromatic hydrocarbons [40].
References
- Changhee Song; Sanghoon Lee; Bonhyun Gu; Ikwhang Chang; Gu Young Cho; Jong Dae Baek; Suk Won Cha; A Study of Anode-Supported Solid Oxide Fuel Cell Modeling and Optimization Using Neural Network and Multi-Armed Bandit Algorithm. Energies 2020, 13, 1621, 10.3390/en13071621.
- Yun Wang; Daniela Fernanda Ruiz Diaz; Ken S. Chen; Zhe Wang; Xavier Cordobes Adroher; Materials, technological status, and fundamentals of PEM fuel cells – A review. Materials Today 2019, 32, 178-203, 10.1016/j.mattod.2019.06.005.
- Mohsen Fallah Vostakola; Bahman Amini Horri; Progress in Material Development for Low-Temperature Solid Oxide Fuel Cells: A Review. Energies 2021, 14, 1280, 10.3390/en14051280.
- B. Shri Prakash; R. Pavitra; S. Senthil Kumar; S.T. Aruna; Electrolyte bi-layering strategy to improve the performance of an intermediate temperature solid oxide fuel cell: A review. Journal of Power Sources 2018, 381, 136-155, 10.1016/j.jpowsour.2018.02.003.
- Fellipe Sartori da Silva; Teófilo Miguel Souza; Novel materials for solid oxide fuel cell technologies: A literature review. International Journal of Hydrogen Energy 2017, 42, 26020-26036, 10.1016/j.ijhydene.2017.08.105.
- Hossein Beydaghi; Ahmad Bagheri; Parisa Salarizadeh; Sepideh Kashefi; Khadijeh Hooshyari; Ali Amoozadeh; Taiebeh Shamsi; Francesco Bonaccorso; Vittorio Pellegrini; Enhancing the Performance of Poly(phthalazinone ether ketone)-Based Membranes Using a New Type of Functionalized TiO2 with Superior Proton Conductivity. Industrial & Engineering Chemistry Research 2020, 59, 6589-6599, 10.1021/acs.iecr.9b06813.
- Aniket Kumar; Jaewoon Hong; Yejin Yun; Aman Bhardwaj; Sun-Ju Song; The role of surface lattice defects of CeO2−δ nanoparticles as a scavenging redox catalyst in polymer electrolyte membrane fuel cells. Journal of Materials Chemistry A 2020, 8, 26023-26034, 10.1039/d0ta09397a.
- Khadijeh Hooshyari; Mehran Javanbakht; Parisa Salarizadeh; Ahmad Bageri; Advanced nanocomposite membranes based on sulfonated polyethersulfone: influence of nanoparticles on PEMFC performance. Journal of the Iranian Chemical Society 2019, 16, 1617-1629, 10.1007/s13738-019-01638-x.
- Abdol Mohammad Attaran; Fabrication and Characterization of Poly Vinyl Alcohol/ Poly Vinyl Pyrrolidone/MnTiO3 Nanocomposite Membranes for PEM Fuel Cells. Iranian Journal of Energy and Environment 2013, 2, 86-90, 10.5829/idosi.ijee.2013.04.02.02.
- Parisa Salarizadeh; Mohammad Bagher Askari; Marziyeh Mohammadi; Khadijeh Hooshyari; Electrocatalytic performance of CeO2-decorated rGO as an anode electrocatalyst for the methanol oxidation reaction. Journal of Physics and Chemistry of Solids 2020, 142, 109442, 10.1016/j.jpcs.2020.109442.
- Xiaobai Li; Peng Wang; Zhenchao Liu; Jinwu Peng; Chengying Shi; Wei Hu; Zhenhua Jiang; Baijun Liu; Arylether-type polybenzimidazoles bearing benzimidazolyl pendants for high-temperature proton exchange membrane fuel cells. Journal of Power Sources 2018, 393, 99-107, 10.1016/j.jpowsour.2018.05.011.
- Rizwan Haider; Yichan Wen; Zi-Feng Ma; David P. Wilkinson; Lei Zhang; Xianxia Yuan; Shuqin Song; Jiujun Zhang; High temperature proton exchange membrane fuel cells: progress in advanced materials and key technologies. Chemical Society Reviews 2020, 50, 1138-1187, 10.1039/d0cs00296h.
- Sangrae Lee; Kwangwon Seo; Ravindra V. Ghorpade; Ki-Ho Nam; Haksoo Han; High temperature anhydrous proton exchange membranes based on chemically-functionalized titanium/polybenzimidazole composites for fuel cells. Materials Letters 2019, 263, 127167, 10.1016/j.matlet.2019.127167.
- Parisa Hosseinabadi; Khadijeh Hooshyari; Mehran Javanbakht; Morteza Enhessari; Synthesis and optimization of nanocomposite membranes based on SPEEK and perovskite nanoparticles for polymer electrolyte membrane fuel cells. New Journal of Chemistry 2019, 43, 16232-16245, 10.1039/c9nj03980e.
- Parisa Salarizadeh; Mehran Javanbakht; Saeed Pourmahdian; Maryam Sabooni Asre Hazer; Khadijeh Hooshyari; Mohammad Bagher Askari; Novel proton exchange membranes based on proton conductive sulfonated PAMPS/PSSA-TiO2 hybrid nanoparticles and sulfonated poly (ether ether ketone) for PEMFC. International Journal of Hydrogen Energy 2018, 44, 3099-3114, 10.1016/j.ijhydene.2018.11.235.
- Abdol Mohammad Attaran; Mehran Javanbakht; Khadijeh Hooshyari; Morteza Enhessari; New proton conducting nanocomposite membranes based on poly vinyl alcohol/poly vinyl pyrrolidone/BaZrO3 for proton exchange membrane fuel cells. Solid State Ionics 2015, 269, 98-105, 10.1016/j.ssi.2014.11.003.
- Xinwei Sun; Stian Christopher Simonsen; Truls Norby; Athanasios Chatzitakis; Composite Membranes for High Temperature PEM Fuel Cells and Electrolysers: A Critical Review. Membranes 2019, 9, 83, 10.3390/membranes9070083.
- Jakobsen, Mark Tonny Dalsgaard; Jensen, Jens Oluf; Cleemann, Lars Nilausen; Li, Qingfeng. Durability Issues and Status of PBI-Based Fuel Cells; Springer International Publishing: Switzerland, 2016; pp. 487-509.
- Alexey Y. Leykin; Andrey A. Askadskii; Viktor G. Vasilev; Alexander L. Rusanov; Dependence of some properties of phosphoric acid doped PBIs on their chemical structure. Journal of Membrane Science 2010, 347, 69-74, 10.1016/j.memsci.2009.10.007.
- Yu Kang; Jing Zou; Zhaonan Sun; Fanghui Wang; Hong Zhu; Kefei Han; Wensheng Yang; Huaihe Song; Qinghan Meng; Polybenzimidazole containing ether units as electrolyte for high temperature proton exchange membrane fuel cells. International Journal of Hydrogen Energy 2013, 38, 6494-6502, 10.1016/j.ijhydene.2013.03.051.
- Mojtaba Farrokhi; Mahdi Abdollahi; Enhancing medium/high temperature proton conductivity of poly(benzimidazole)-based proton exchange membrane via blending with poly(vinyl imidazole-co-vinyl phosphonic acid) copolymer: Proton conductivity-copolymer microstructure relationship. European Polymer Journal 2020, 131, 109691, 10.1016/j.eurpolymj.2020.109691.
- Hongze Luo; Guntars Vaivars; Bolade Agboola; Shichun Mu; Mkhulu Mathe; Anion exchange membrane based on alkali doped poly(2,5-benzimidazole) for fuel cell. Solid State Ionics 2012, 208, 52-55, 10.1016/j.ssi.2011.11.029.
- Jingjing Lin; XiaoMing Yan; Gaohong He; Wanting Chen; Dongxing Zhen; Tiantian Li; Lin Ma; Xuemei Wu; Thermoplastic interpenetrating polymer networks based on polybenzimidazole and poly (1, 2-dimethy-3-allylimidazolium) for anion exchange membranes. Electrochimica Acta 2017, 257, 9-19, 10.1016/j.electacta.2017.09.126.
- Juan Antonio Asensio; Eduardo Sanchez; Pedro Gomez-Romero; Proton-conducting membranes based on benzimidazole polymers for high-temperature PEM fuel cells. A chemical quest. Chemical Society Reviews 2010, 39, 3210-3239, 10.1039/b922650h.
- Gabriele G. Gagliardi; Ahmed Ibrahim; Domenico Borello; Ahmad El-Kharouf; Composite Polymers Development and Application for Polymer Electrolyte Membrane Technologies—A Review. Molecules 2020, 25, 1712, 10.3390/molecules25071712.
- † Kevork T. Adjemian; † Raymond Dominey; † Lakshmi Krishnan; † Hitoshi Ota; † Paul Majsztrik; † Tao Zhang; † Jonathan Mann; † Brent Kirby; † Louis Gatto; † Melanie Velo-Simpson; et al.† Jacklyn Leahy† Supramanian Srinivasan⊥ And Jay B. BenzigerAndrew Bocarsly Function and Characterization of Metal Oxide−Nafion Composite Membranes for Elevated-Temperature H2/O2PEM Fuel Cells. Chemistry of Materials 2006, 18, 2238-2248, 10.1021/cm051781b.
- Yanan Wei; Tianhua Qian; Jiawen Liu; Xaojing Guo; Qiaojuan Gong; Zhaorong Liu; Binglun Tian; Jinli Qiao; Novel composite Nafion membranes modified with copper phthalocyanine tetrasulfonic acid tetrasodium salt for fuel cell application. Journal of Materiomics 2019, 5, 252-257, 10.1016/j.jmat.2019.01.006.
- P. Muthuraja; S. Prakash; V.M. Shanmugam; S. Radhakrsihnan; P. Manisankar; Novel perovskite structured calcium titanate-PBI composite membranes for high-temperature PEM fuel cells: Synthesis and characterizations. International Journal of Hydrogen Energy 2018, 43, 4763-4772, 10.1016/j.ijhydene.2017.12.010.
- Nurhan Üregen; Kübra Pehlivanoğlu; Yağmur Özdemir; Yilser Devrim; Development of polybenzimidazole/graphene oxide composite membranes for high temperature PEM fuel cells. International Journal of Hydrogen Energy 2017, 42, 2636-2647, 10.1016/j.ijhydene.2016.07.009.
- Erigene Bakangura; Liang Wu; Liang Ge; Zhengjin Yang; Tongwen Xu; Mixed matrix proton exchange membranes for fuel cells: State of the art and perspectives. Progress in Polymer Science 2016, 57, 103-152, 10.1016/j.progpolymsci.2015.11.004.
- Jorge Escorihuela; Riccardo Narducci; Vicente Compañ; Ferdinando Costantino; Proton Conductivity of Composite Polyelectrolyte Membranes with Metal‐Organic Frameworks for Fuel Cell Applications. Advanced Materials Interfaces 2019, 6, 1801146, 10.1002/admi.201801146.
- Qian Li; Qiang Yin; Ya-Shan Zheng; Zhi-Jun Sui; Xing-Gui Zhou; De Chen; Yi-An Zhu; Insights into Hydrogen Transport Behavior on Perovskite Surfaces: Transition from the Grotthuss Mechanism to the Vehicle Mechanism. Langmuir 2019, 35, 9962-9969, 10.1021/acs.langmuir.8b04138.
- Linas Vilciauskas; Klaus-Dieter Kreuer; Comment on “Mixed Grotthuss and Vehicle Transport Mechanism in Proton Conducting Polymers from Ab initio Molecular Dynamics Simulations”. Chemistry of Materials 2011, 23, 3377-3378, 10.1021/cm200865v.
- Shuvra Singha; Rambabu Koyilapu; Kausik Dana; Tushar Jana; Polybenzimidazole-Clay Nanocomposite Membrane for PEM fuel cell: Effect of organomodifier structure. Polymer 2019, 167, 13-20, 10.1016/j.polymer.2019.01.066.
- Haoqin Zhang; Wenjia Wu; Jingtao Wang; Tao Zhang; Benbing Shi; Jindun Liu; Shaokui Cao; Enhanced anhydrous proton conductivity of polymer electrolyte membrane enabled by facile ionic liquid-based hoping pathways. Journal of Membrane Science 2015, 476, 136-147, 10.1016/j.memsci.2014.11.033.
- Zhenyu Liu; Yu-Min Tsou; Gordon Calundann; Emory De Castro; New process for high temperature polybenzimidazole membrane production and its impact on the membrane and the membrane electrode assembly. Journal of Power Sources 2011, 196, 1055-1060, 10.1016/j.jpowsour.2010.08.026.
- W.H. J. Hogarth; Joao Carlos Diniz da Costa; Gaoqing (Max) Lu; Solid acid membranes for high temperature (¿140° C) proton exchange membrane fuel cells. Journal of Power Sources 2005, 142, 223-237, 10.1016/j.jpowsour.2004.11.020.
- Deuk Ju Kim; Min Jae Jo; Sang Yong Nam; A review of polymer–nanocomposite electrolyte membranes for fuel cell application. Journal of Industrial and Engineering Chemistry 2015, 21, 36-52, 10.1016/j.jiec.2014.04.030.
- Samuel Simon Araya; Fan Zhou; Vincenzo Liso; Simon Lennart Sahlin; Jakob Rabjerg Vang; Sobi Thomas; Xin Gao; Christian Jeppesen; Søren Knudsen Kær; A comprehensive review of PBI-based high temperature PEM fuel cells. International Journal of Hydrogen Energy 2016, 41, 21310-21344, 10.1016/j.ijhydene.2016.09.024.
- Qingfeng Li; Chao Pan; Jens Oluf Jensen; And Pernille Noyé; Niels Janniksen Bjerrum; Cross-Linked Polybenzimidazole Membranes for Fuel Cells. Chemistry of Materials 2007, 19, 350-352, 10.1021/cm0627793.
- Zhihong Chang; Hongting Pu; Decheng Wan; Lu Liu; Junjie Yuan; Zhenglong Yang; Chemical oxidative degradation of Polybenzimidazole in simulated environment of fuel cells. Polymer Degradation and Stability 2009, 94, 1206-1212, 10.1016/j.polymdegradstab.2009.04.026.
- LaConti, A. B.; Liu, H.; Mittelsteadt, C.; McDonald, R. C.; Polymer Electrolyte Membrane Degradation Mechanisms in Fuel Cells - Findings Over the Past 30 Years and Comparison with Electrolyzers. ECS Transactions 2006, 1, 199-219.
- S. R. Samms; S. Wasmus; Robert F Savinell; Thermal Stability of Proton Conducting Acid Doped Polybenzimidazole in Simulated Fuel Cell Environments. Journal of The Electrochemical Society 1996, 143, 1225-1232, 10.1149/1.1836621.
- Michael Jaffe; M. Ishaq Haider; Joseph Menczel; Joseph Rafalko; Thermal characterization of high performance PBI and 6F polymers and their alloys. Polymer Engineering & Science 1992, 32, 1236-1241, 10.1002/pen.760321709.
- Kug-Seung Lee; Byung-Seok Lee; Sung Jong Yoo; Soo-Kil Kim; Seung Jun Hwang; Hyung-Juhn Kim; Eunae Cho; Dirk Henkensmeier; Jeong Woo Yun; Suk Woo Nam; et al.Tae-Hoon LimJong Hyun Jang Development of a galvanostatic analysis technique as an in-situ diagnostic tool for PEMFC single cells and stacks. International Journal of Hydrogen Energy 2012, 37, 5891-5900, 10.1016/j.ijhydene.2011.12.152.
- Behzad Najafi; Paolo Bonomi; Andrea Casalegno; Fabio Rinaldi; Andrea Baricci; Rapid Fault Diagnosis of PEM Fuel Cells through Optimal Electrochemical Impedance Spectroscopy Tests. Energies 2020, 13, 3643, 10.3390/en13143643.
- Heli Wang; Clay Macomber; Jason Christ; Guido Bender; Bryan Pivovar; Huyen N. Dinh; Evaluating the Influence of PEMFC System Contaminants on the Performance of Pt Catalyst via Cyclic Voltammetry. Electrocatalysis 2013, 5, 62-67, 10.1007/s12678-013-0159-9.
- Thomas Dobbelaere; Philippe Vereecken; Christophe Detavernier; A USB-controlled potentiostat/galvanostat for thin-film battery characterization. HardwareX 2017, 2, 34-49, 10.1016/j.ohx.2017.08.001.
- Nadine Pilinski; Maren Rastedt; Peter Wagner; Investigation of Phosphoric Acid Distribution in PBI Based HT-PEM Fuel Cells. ECS Transactions 2015, 69, 323-335, 10.1149/06917.0323ecst.
- Yuka Oono; Atsuo Sounai; Michio Hori; Long-term cell degradation mechanism in high-temperature proton exchange membrane fuel cells. Journal of Power Sources 2012, 210, 366-373, 10.1016/j.jpowsour.2012.02.098.
- Olena Selyanchyn; Roman Selyanchyn; Stephen M. Lyth; A Review of Proton Conductivity in Cellulosic Materials. Frontiers in Energy Research 2020, 8, 596164, 10.3389/fenrg.2020.596164.
- Patrick Heimerdinger; Andreas Rosin; Michael Danzer; Thorsten Gerdes; A Novel Method for Humidity-Dependent Through-Plane Impedance Measurement for Proton Conducting Polymer Membranes. Membranes 2019, 9, 62, 10.3390/membranes9050062.
- G Alberti; Mario Casciola; L Massinelli; B Bauer; Polymeric proton conducting membranes for medium temperature fuel cells (110–160°C). Journal of Membrane Science 2001, 185, 73-81, 10.1016/s0376-7388(00)00635-9.
- Zhong Xie; Chaojie Song; Bernhard Andreaus; Titichai Navessin; Zhiqing Shi; Jiujun Zhang; Steven Holdcroft; Discrepancies in the Measurement of Ionic Conductivity of PEMs Using Two- and Four-Probe AC Impedance Spectroscopy. Journal of The Electrochemical Society 2006, 153, E173-E178, 10.1149/1.2258091.
- Angie L. Rangel-Cárdenas; Ger J. M Koper; Transport in Proton Exchange Membranes for Fuel Cell Applications—A Systematic Non-Equilibrium Approach. Materials 2017, 10, 576, 10.3390/ma10060576.
- T Dippel; K Kreuer; J Lassegues; D Rodriguez; Proton conductivity in fused phosphoric acid; A 1H/31P PFG-NMR and QNS study. Solid State Ionics 1993, 61, 41-46, 10.1016/0167-2738(93)90332-w.
- Y.-L. Ma; J. S. Wainright; M. H. Litt; R. F. Savinell; Conductivity of PBI Membranes for High-Temperature Polymer Electrolyte Fuel Cells. Journal of The Electrochemical Society 2004, 151, A8-A16, 10.1149/1.1630037.
- Sara Barati; Mahdi Abdollahi; Behnam Khoshandam; Mohsen Mehdipourghazi; Highly proton conductive porous membranes based on polybenzimidazole/ lignin blends for high temperatures proton exchange membranes: Preparation, characterization and morphology- proton conductivity relationship. International Journal of Hydrogen Energy 2018, 43, 19681-19690, 10.1016/j.ijhydene.2018.08.191.