
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Wenlong Yang | + 1556 word(s) | 1556 | 2021-08-18 08:17:46 | | | |
| 2 | Amina Yu | + 21 word(s) | 1577 | 2021-08-27 08:09:04 | | |
Video Upload Options
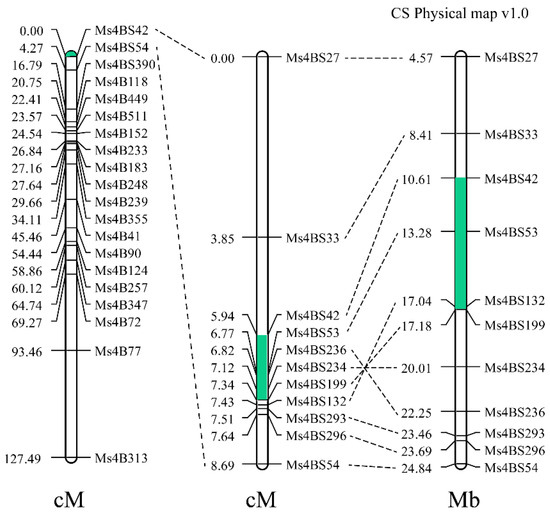
The utilization of heterosis is an important way to improve wheat yield, and the production of wheat hybrid seeds mainly relies on male-sterile lines. Male sterility in line 15 Fan 03 derived from a cross of 72,180 and Xiaoyan 6 is controlled by a single recessive gene. The gene was mapped to the distal region of chromosome 4BS in a genetic interval of 1.4 cM and physical distance of 6.57 Mb between SSR markers Ms4BS42 and Ms4BS199 using an F2 population with 1205 individuals. Sterile individuals had a deletion of 4.57 Mb in the region presumed to carry the Ms1 locus. The allele for sterility was therefore named ms1s. Three CAPS markers were developed and verified from the region upstream of the deleted fragment and can be used for ms1s marker-assisted selection in wheat hybrid breeding. This work will enrich the utilization of male sterility genetic resources.
1. Introduction
2. Discussion
References
- Deng, X.; Wang, H.; Tang, X.; Zhou, J.; Chen, H.; He, G.; Chen, L.; Xu, Z. Hybrid rice breeding welcomes a new era of molecular crop design. Sci. Sin. Vitae 2013, 43, 864–868.
- Chang, Z.; Chen, Z.; Wang, N.; Xie, G.; Lu, J.; Yan, W.; Zhou, J.; Tang, X.; Deng, X.W. Construction of a male sterility system for hybrid rice breeding and seed production using a nuclear male sterility gene. Proc. Natl. Acad. Sci. USA 2016, 113, 14145–14150.
- Wilson, J.A.; Ross, W.M. Cross breeding in wheat, Triticum aestivum L. II. hybrid seed set on a cytoplasmic male-sterile winter wheat composite subjected to cross-pollination. Crop. Sci. 1962, 2, 415–417.
- Singh, S.K.; Chatrath, R.; Mishra, B. Perspective of hybrid wheat research, A review. Indian J. Agric. Sci. 2010, 80, 1013–1027.
- Whitford, R.; Fleury, D.; Reif, J.C.; Garcia, M.; Okada, T.; Korzun, V.; Langridge, P. Hybrid breeding in wheat, technologies to improve hybrid wheat seed production. J. Exp. Bot. 2013, 64, 5411–5428.
- Pugsley, A.T.; Oram, R.N. Genic male sterility in wheat. Aust. Plant Breed. Genet. Newsl. 1959, 14, 10–11.
- Liu, B.H.; Deng, J.Y. A dominant gene for male sterility in wheat. Plant Breed. 1986, 97, 204–209.
- Franckowiak, J.D.; Maan, S.S.; Williams, N.D. A proposal for hybrid wheat utilizing Aegilops squarrosa L. cytoplasm. Crop. Sci. 1976, 16, 725–728.
- Maan, S.S.; Kianian, S.F. Third dominant male sterility gene in common wheat. Wheat Inf. Serv. 2001, 93, 27–31.
- Sasakuma, T.; Maan, S.S.; Williams, N.D. EMS-induced male-sterile mutants in euplasmic and alloplasmic common wheat. Crop. Sci. 1978, 18, 850–853.
- Klindworth, D.L.; Williams, N.D.; Maan, S.S. Chromosomal location of genetic male sterility genes in four mutants of hexaploid wheat. Crop. Sci. 2002, 42, 1447–1450.
- Pallotta, M.A.; Warner, P.; Kouidri, A.; Tucker, E.J.; Baes, M.; Suchecki, R.; Watson-Haigh, N.; Okada, T.; Garcia, M.; Sandhu, A.; et al. Wheat ms5 male-sterility is induced by recessive homoeologous A and D genome non-specific lipid transfer proteins. Plant J. 2019, 99, 673–685.
- Xia, C.; Zhang, L.; Zou, C.; Gu, Y.; Duan, J.; Zhao, G.; Wu, J.; Liu, Y.; Fang, X.; Gao, L.; et al. A TRIM insertion in the promoter of Ms2 causes male sterility in wheat. Nat. Commun. 2017, 8, 15407.
- Maan, S.S.; Carlson, K.M.; Williams, N.D.; Yang, T. Chromosomal arm location and gene-centromere distance of a dominant gene for male sterility in wheat. Crop. Sci. 1987, 27, 494–500.
- Suneson, C.A. Use of Pugsley’s sterile wheat in cross breeding. Crop. Sci. 1962, 2, 534–535.
- Zhou, K.; Wang, S.; Feng, Y.; Ji, W.; Wang, G. A new male sterile mutant LZ in wheat (Triticum aestivum L.). Euphytica 2008, 159, 403–410.
- Fossati, A.; Ingold, M. A male sterile mutant in Triticum aestivum. Wheat Inf. Serv. 1970, 30, 8–10.
- Driscoll, C.J. Registration of cornerstone male sterile wheat germplasm 1 (Reg. No.GP 74). Crop. Sci. 1977, 17, 190.
- Wang, Z.; Li, J.; Chen, S.; Heng, Y.; Chen, Z.; Yang, J.; Zhou, K.; Pei, J.; He, H.; Deng, X.W.; et al. Poaceae-specific MS1 encodes a phospholipid-binding protein for male fertility in bread wheat. Proc. Natl. Acad. Sci. USA 2017, 114, 12614–12619.
- Gill, B.S.; Anand, S.C. Genetic male sterility for hybrid seed production in wheat. Crop. Sci. 1970, 10, 385–386.
- Driscoll, C.J. XYZ system of producing hybrid wheat. Crop. Sci. 1972, 12, 516–517.
- Driscoll, C.J. Modified XYZ system of producing hybrid wheat. Crop. Sci. 1985, 25, 1115–1116.
- Huang, S.S.; Xue, C.P.; Xu, J.; Li, W.L. A preliminary report of selections and studies on male sterile-maintainable line of blue-grained wheat. Acta Bot. Boreali-Occident. Sin. 1988, 8, 162–166.
- Huang, S.S.; Li, W.L.; Xu, J.; Xue, C.P. The development of a blue marked nucleus male sterile line and its maintainer in bread wheat. Acta Agron. Sin. 1991, 17, 81–87.
- Zhou, K.; Wang, S.; Feng, Y.; Liu, Z.; Wang, G. The 4E-ms system of producing hybrid wheat. Crop. Sci. 2006, 46, 250–255.
- Tucker, E.J.; Baumann, U.; Kouidri, A.; Suchecki, R.; Baes, M.; Garcia, M.; Okada, T.; Dong, C.; Wu, Y.; Sandhu, A.; et al. Molecular identification of the wheat male fertility gene Ms1 and its prospects for hybrid breeding. Nat. Commun. 2017, 8, 869.
- Athwal, D.S.; Phul, P.S.; Minocha, J.L. Genetic male sterility in wheat. Euphytica 1967, 16, 354–360.
- Sun, L.; Yang, W.; Li, Y.; Shan, Q.; Ye, X.; Wang, D.; Yu, K.; Lu, W.; Xin, P.; Pei, Z.; et al. A wheat dominant dwarfing line with Rht12 which reduces stem cell length and affects GA synthesis, is a 5AL terminal deletion line. Plant J. 2019, 97, 887–900.





