
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Long-Li Lai | + 3406 word(s) | 3406 | 2021-08-24 10:23:46 | | | |
| 2 | Vivi Li | Meta information modification | 3406 | 2021-08-25 07:57:56 | | |
Video Upload Options
Most triazine-based liquid crystalline (LC) dendrimers reported thus far are the main-chain LC macromolecules with long flexible chains at their periphery and attached to internal rigid or semi-rigid frameworks. Their formation of mesogenic phases often depends on the intermolecular face-to-face π–π interactions between dendritic molecules, which are unusual. Their mesogenic phases can also be formed by incorporation of mesogenic units to the dendritic skeletons through long flexible chains, as most side-chain LC dendrimers, in which the peripheral mesogenic units generally play the important roles. For main-chain triazine-based dendrimers, their morphology is maintained by restricted freedom of rigid or semi-rigid connecting units, and their formations of LC phases are therefore not straightforward to be controlled.
1. Introduction
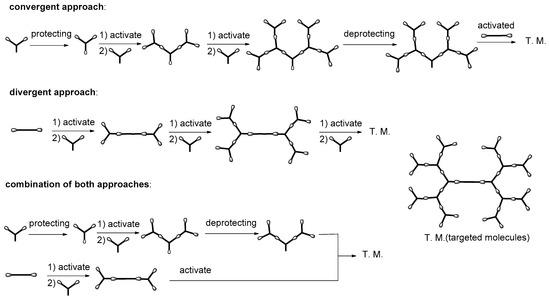
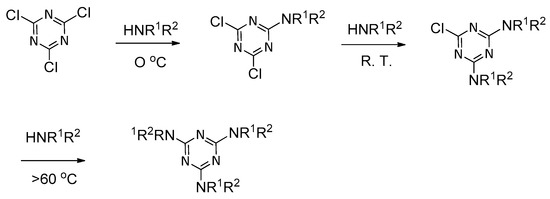
2. Formation of Triazine-Based LC Dendrimers by Manipulating Their Molecular Morphology
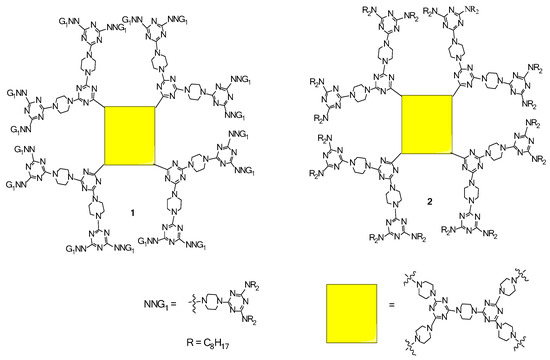
2.1. LC Dendrimers with C2 Symmetry
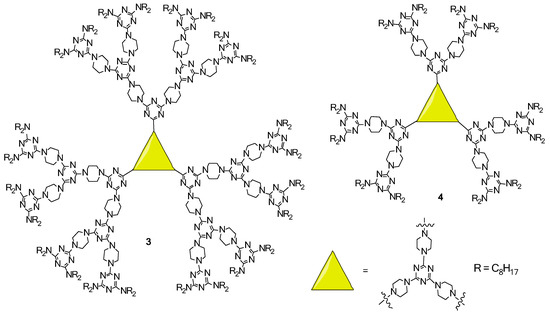
2.2. LC Dendrimers with C3 Symmetry
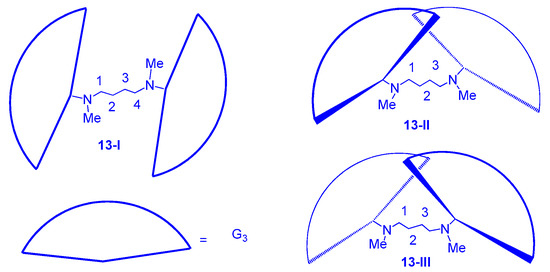
2.3. LC Dendrimers with the Loss of C2 Symmetry by Changing the Peripheral Groups
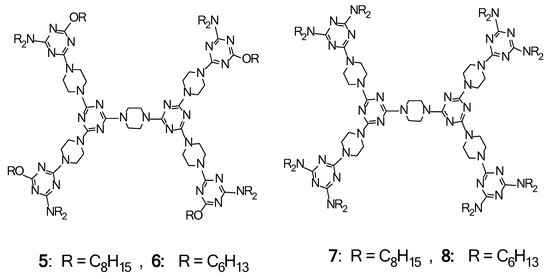
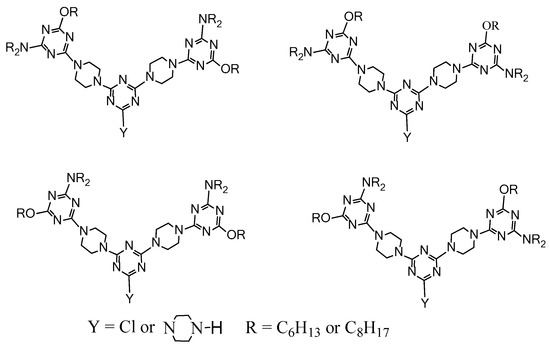
2.4. LC Dendrimers with the Loss of C2 Symmetry by Changing the Central Cores
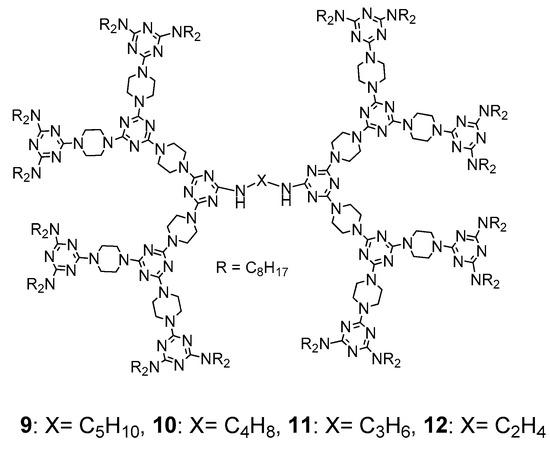
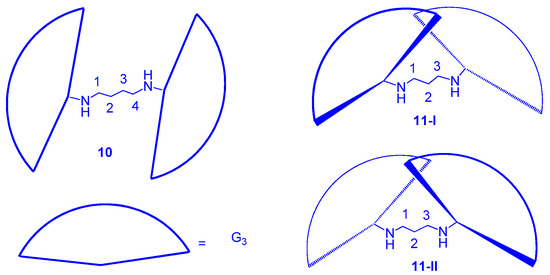
3. Formation of Triazine-Based LC Dendrimers by Adding Extra Components
3.1. Formation of LC Dendrimers by Intermolecular π–π Interaction
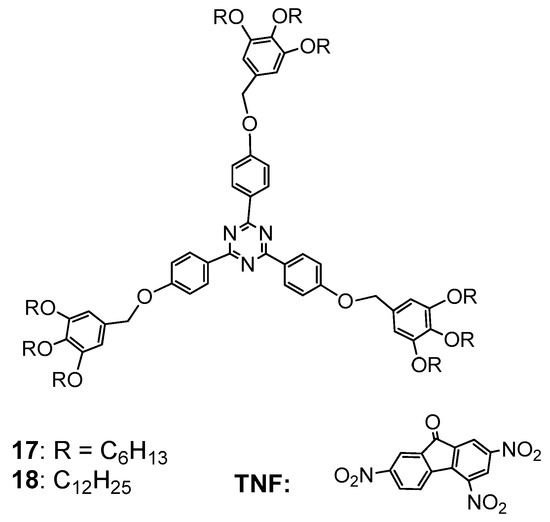
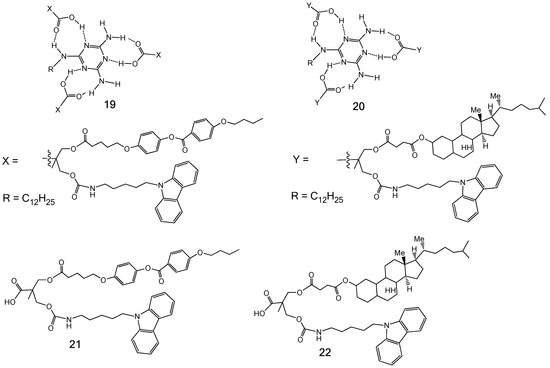
3.2. Formation of LC Dendrimers by Intermolecular H-Bond Interaction
References
- Tomalia, D.A.; Fréchet, J.M.J. Discovery of dendrimers and dendritic polymers: A brief historical perspective. J. Polym. Sci. A Polym. Chem. 2002, 40, 2719–2728.
- Caminade, A.-M.; Turrin, C.-O.; Laurent, R.; Ouali, A.; Delavaux-Nicot, B. Dendrimers: Towards Catalytic, Material and Biomedical Uses; Wiley: New York, NY, USA, 2011.
- Janaszewska, A.; Lazniewska, J.; Trzepiński, P.; Marcinkowska, M.; Klajnert-Maculewicz, B. Cytotoxicity of Dendrimers. Biomolecules 2019, 9, 330.
- Chauhan, A.S. Dendrimers for Drug Delivery. Molecules 2018, 23, 938.
- Sherje, A.P.; Jadhav, M.; Dravyakar, B.R.; Kadam, D. Dendrimers: A versatile nanocarrier for drug delivery and targeting. Int. J. Pharm. 2018, 548, 707–720.
- Kaur, D.; Jain, K.; Mehra, N.K.; Kesharwani, P.; Jain, N.K. A review on comparative study of PPI and PAMAM dendrimers. J. Nanopart. Res. 2016, 18, 146.
- Kesharwani, P.; Jain, K.; Jain, N.K. Dendrimer as nanocarrier for drug delivery. Prog. Polym. Sci. 2014, 39, 268–307.
- Madaan, K.; Kumar, S.; Poonia, N.; Lather, V.; Pandita, D. Dendrimers in drug delivery and targeting: Drug-dendrimer interactions and toxicity issues. J. Pharm. Bioallied Sci. 2014, 6, 139–150.
- Mignani, S.; El Kazzouli, S.; Bousmina, M.; Majoral, J.-P. Expand classical drug administration ways by emerging routes using dendrimer drug delivery systems: A concise overview. Adv. Drug Deliv. Rev. 2013, 65, 1316–1330.
- Liu, Y.; Lopes, R.P.; Lüdtke, T.; Di Silvio, D.; Moya, S.; Hamon, J.-R.; Astruc, D. “Click” dendrimer-Pd nanoparticle assemblies as enzyme mimics: Catalytic o-phenylenediamine oxidation and application in colorimetric H2O2 detection. Inorg. Chem. Front. 2021, 8, 3301–3307.
- Yamamoto, K.; Imaoka, T.; Tanabe, M.; Kambe, T. New Horizon of Nanoparticle and Cluster Catalysis with Dendrimers. Chem. Rev. 2020, 120, 1397–1437.
- Tang, Y.-H.; Cangiotti, M.; Kao, C.-L.; Ottaviani, M.F. EPR Characterization of Copper(II) Complexes of PAMAM-Py Dendrimers for Biocatalysis in the Absence and Presence of Reducing Agents and a Spin Trap. J. Phys. Chem. B 2017, 121, 10498–10507.
- Neumann, P.; Dib, H.; Caminade, A.-M.; Hey-Hawkins, E. Redox Control of a Dendritic Ferrocenyl-Based Homogeneous Catalyst. Angew. Chem. Int. Ed. 2015, 54, 311–314.
- Twyman, L.J.; King, A.S.H.; Martin, I.K. Catalysis inside dendrimers. Chem. Soc. Rev. 2002, 31, 69–82.
- Astruc, D.; Chardac, F. Dendritic Catalysts and Dendrimers in Catalysis. Chem. Rev. 2001, 101, 2991–3024.
- Crooks, R.M.; Zhao, M.; Sun, L.; Chechik, V.; Yeung, L.K. Dendrimer-Encapsulated Metal Nanoparticles: Synthesis, Characterization, and Applications to Catalysis. Acc. Chem. Res. 2001, 34, 181–190.
- Concellón, A.; Termine, R.; Golemme, A.; Romero, P.; Marcos, M.; Serrano, J.L. High hole mobility and light-harvesting in discotic nematic dendrimers prepared via ‘click’ chemistry. J. Mater. Chem. C 2019, 7, 2911–2918.
- Yim, D.; Sung, J.; Kim, S.; Oh, J.; Yoon, H.; Sung, Y.M.; Kim, D.; Jang, W.-D. Guest-Induced Modulation of the Energy Transfer Process in Porphyrin-Based Artificial Light Harvesting Dendrimers. J. Am. Chem. Soc. 2017, 139, 993–1002.
- Nantalaksakul, A.; Reddy, D.R.; Bardeen, C.J.; Thayumanavan, S. Light Harvesting Dendrimers. Photosyn. Res. 2006, 87, 133–150.
- Balzani, V.; Ceroni, P.; Maestri, M.; Vicinelli, V. Light-harvesting dendrimers. Curr. Opin. Chem. Biol. 2003, 7, 657–665.
- Adronov, A.; Fréchet, J.M.J. Light-harvesting dendrimers. Chem. Comm. 2000, 18, 1701–1710.
- Gilat, S.L.; Adronov, A.; Fréchet, J.M.J. Light Harvesting and Energy Transfer in Novel Convergently Constructed Dendrimers. Angew. Chem. Int. Ed. 1999, 38, 1422–1427.
- Wang, Y.; He, X.; Lu, Q. Polyamidoamine dendrimer functionalized cellulose nanocrystals for CO2 capture. Cellulose 2021, 28, 4241–4251.
- El Kadib, A.; Katir, N.; Bousmina, M.; Majoral, J.P. Dendrimer–Silica hybrid mesoporous materials. New J. Chem. 2012, 36, 241–255.
- Bonaccorsi, L.; Lombardo, D.; Longo, A.; Proverbio, E.; Triolo, A. Dendrimer Template Directed Self-Assembly during Zeolite Formation. Macromolecules 2009, 42, 1239–1243.
- Yoo, S.; Lunn, J.D.; Gonzalez, S.; Ristich, J.A.; Simanek, E.E.; Shantz, D.F. Engineering Nanospaces: OMS/Dendrimer Hybrids Possessing Controllable Chemistry and Porosity. Chem. Mater. 2006, 18, 2935–2942.
- Kriesel, J.W.; Tilley, T.D. Dendrimers as Building Blocks for Nanostructured Materials: Micro- and Mesoporosity in Dendrimer-Based Xerogels. Chem. Mater. 1999, 11, 1190–1193.
- Martínez, N.P.; Inostroza-Rivera, R.; Durán, B.; Molero, L.; Bonardd, S.; Ramírez, O.; Isaacs, M.; Díaz Díaz, D.; Leiva, A.; Saldías, C. Exploring the Effect of the Irradiation Time on Photosensitized Dendrimer-Based Nanoaggregates for Potential Applications in Light-Driven Water Photoreduction. Nanomaterials 2019, 9, 1316.
- Avila-Salas, F.; Pereira, A.; Rojas, M.A.; Saavedra-Torres, M.; Montecinos, R.; Bonardd, S.; Quezada, C.; Saldías, S.; Díaz Díaz, D.; Leiva, A.; et al. An experimental and theoretical comparative study of the entrapment and release of dexamethasone from micellar and vesicular aggregates of PAMAM-PCL dendrimers. Eur. Polym. J. 2017, 93, 507–520.
- Axenov, K.V.; Laschat, S. Thermotropic Ionic Liquid Crystals. Materials 2011, 4, 206–259.
- Rosen, B.M.; Wilson, C.J.; Wilson, D.A.; Peterca, M.; Imam, M.R.; Percec, V. Dendron-Mediated Self-Assembly, Disassembly, and Self-Organization of Complex Systems. Chem. Rev. 2009, 109, 6275–6540.
- Goodby, J.W.; Saez, I.M.; Cowling, S.J.; Görtz, V.; Draper, M.; Hall, A.W.; Sia, S.; Cosquer, G.; Lee, S.-E.; Raynes, E.P. Transmission and Amplification of Information and Properties in Nanostructured Liquid Crystals. Angew. Chem. Int. Ed. 2008, 47, 2754–2787.
- Kim, H.-J.; Jung, E.-Y.; Jin, L.Y.; Lee, M. Solution Behavior of Dendrimer-Coated Rodlike Coordination Polymers. Macromolecules 2008, 41, 6066–6072.
- Inoue, K. Functional dendrimers, hyperbranched and star polymers. Prog. Polym. Sci. 2000, 25, 453–571.
- Hernández-Ainsa, S.; Barberá, J. Fluorinated liquid crystalline dendrimers. J. Fluor. Chem 2015, 177, 37–45.
- Donnio, B.; Buathong, S.; Bury, I.; Guillon, D. Liquid crystalline dendrimers. Chem. Soc. Rev. 2007, 36, 1495–1513.
- Marcos, M.; Martín-Rapún, R.; Omenat, A.; Serrano, J.L. Highly congested liquid crystal structures: Dendrimers, dendrons, dendronized and hyperbranched polymers. Chem. Soc. Rev. 2007, 36, 1889–1901.
- Saez, I.M.; Goodby, J.W. Supermolecular liquid crystals. J. Mater. Chem. 2005, 15, 26–40.
- Deschenaux, R.; Donnio, B.; Guillon, D. Liquid-crystalline fullerodendrimers. New J. Chem. 2007, 31, 1064–1073.
- Gehringer, L.; Bourgogne, C.; Guillon, D.; Donnio, B. Liquid-Crystalline Octopus Dendrimers: Block Molecules with Unusual Mesophase Morphologies. J. Am. Chem. Soc. 2004, 126, 3856–3867.
- Gehringer, L.; Guillon, D.; Donnio, B. Liquid Crystalline Octopus: An Alternative Class of Mesomorphic Dendrimers. Macromolecules 2003, 36, 5593–5601.
- Meier, H.; Lehmann, M. Stilbenoid Dendrimers. Angew. Chem. Int. Ed. 1998, 37, 643–645.
- Pesak, D.J.; Moore, J.S. Columnar Liquid Crystals from Shape-Persistent Dendritic Molecules. Angew. Chem. Int. Ed. Engl. 1997, 36, 1636–1639.
- Li, J.-F.; Crandall, K.A.; Chu, P.; Percec, V.; Petschek, R.G.; Rosenblatt, C. Dendrimeric Liquid Crystals: Isotropic−Nematic Pretransitional Behavior. Macromolecules 1996, 29, 7813–7819.
- Percec, V.; Chu, P.; Ungar, G.; Zhou, J. Rational Design of the First Nonspherical Dendrimer Which Displays Calamitic Nematic and Smectic Thermotropic Liquid Crystalline Phases. J. Am. Chem. Soc. 1995, 117, 11441–11454.
- Goossens, K.; Lava, K.; Bielawski, C.W.; Binnemans, K. Ionic Liquid Crystals: Versatile Materials. Chem. Rev. 2016, 116, 4643–4807.
- Donnio, B. Liquid–crystalline metallodendrimers. Inorg. Chim. Acta 2014, 409, 53–67.
- Kato, T.; Mizoshita, N.; Kishimoto, K. Functional Liquid-Crystalline Assemblies: Self-Organized Soft Materials. Angew. Chem. Int. Ed. 2006, 45, 38–68.
- Schnabel, W.J.; Rätz, R.; Kober, E. The Synthesis of Substituted Melams. J. Org. Chem. 1962, 27, 2514–2519.
- Thurston, J.T.; Dudley, J.R.; Kaiser, D.W.; Hechenbleikner, I.; Schaefer, F.C.; Holm-Hansen, D. Cyanuric Chloride Derivatives. I. Aminochloro-s-triazines. J. Am. Chem. Soc. 1951, 73, 2981–2983.
- Takagi, K.; Hattori, T.; Kunisada, H.; Yuki, Y. Triazine dendrimers by divergent and convergent methods. J. Polym. Sci. A Polym. Chem. 2000, 38, 4385–4395.
- Lai, L.-L.; Wang, L.-Y.; Lee, C.-H.; Lin, Y.-C.; Cheng, K.-L. Nanomaterials of Triazine-Based Dendrons: Convergent Synthesis and Their Physical Studies. Org. Lett. 2006, 8, 1541–1544.
- Steffensen, M.B.; Hollink, E.; Kuschel, F.; Bauer, M.; Simanek, E.E. Dendrimers based on [1,3,5]-triazines. J. Polym. Sci. A Polym. Chem. 2006, 44, 3411–3433.
- Lai, L.-L.; Lee, C.-H.; Wang, L.-Y.; Cheng, K.-L.; Hsu, H.-F. Star-Shaped Mesogens of Triazine-Based Dendrons and Dendrimers as Unconventional Columnar Liquid Crystals. J. Org. Chem. 2008, 73, 485–490.
- Kotha, S.; Kashinath, D.; Kumar, S. Synthesis of liquid crystalline materials based on 1,3,5-triphenylbenzene and 2,4,6-triphenyl-1,3,5-s-triazine. Tetrahedron Lett. 2008, 49, 5419–5423.
- Thiebaut, O.; Bock, H.; Grelet, E. Face-on Oriented Bilayer of Two Discotic Columnar Liquid Crystals for Organic Donor−Acceptor Heterojunction. J. Am. Chem. Soc. 2010, 132, 6886–6887.
- Shearman, G.C.; Yahioglu, G.; Kirstein, J.; Milgrom, L.R.; Seddon, J.M. Synthesis and phase behaviour of β-octaalkyl porphyrins. J. Mater. Chem. 2009, 19, 598–604.
- Yang, C.-W.; Hsia, T.-H.; Chen, C.-C.; Lai, C.-K.; Liu, R.-S. Synthesis and Columnar Mesophase of Fluorescent Liquid Crystals Bearing a C2-Symmetric Chiral Core. Org. Lett. 2008, 10, 4069–4072.
- Li, Z.; Zhi, L.; Lucas, N.T.; Wang, Z. Triangle-shaped polycyclic aromatics based on tribenzocoronene: Facile synthesis and physical properties. Tetrahedron 2009, 65, 3417–3424.
- Lai, L.-L.; Hsu, S.-J.; Hsu, H.-C.; Wang, S.-W.; Cheng, K.-L.; Chen, C.-J.; Wang, T.-H.; Hsu, H.-F. Formation of Columnar Liquid Crystals on the Basis of Unconventional Triazine-Based Dendrimers by the C3-Symmetric Approach. Chem. Eur. J. 2012, 18, 6542–6547.
- Alfa Aesar. Research Chemicals, Metals and Materials; Alfa Aesar: Ward Hill, MA, USA, 2011; p. 124.
- Lai, L.L.; Wang, S.W.; Cheng, K.L.; Lee, J.J.; Wang, T.H.; Hsu, H.F. Induction of the columnar phase of unconventional dendrimers by breaking the C2 symmetry of molecules. Chem. Eur. J. 2012, 18, 15361–15367.
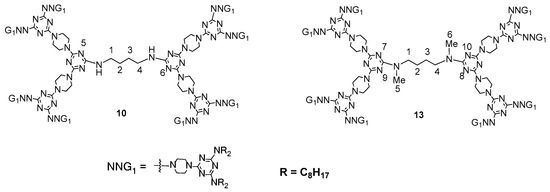
- Lai, L.-L.; Hsieh, J.-W.; Cheng, K.-L.; Liu, S.-H.; Lee, J.-J.; Hsu, H.-F. A Small Change in Central Linker Has a Profound Effect in Inducing Columnar Phases of Triazine-Based Unconventional Dendrimers. Chem. Eur. J. 2014, 20, 5160–5166.
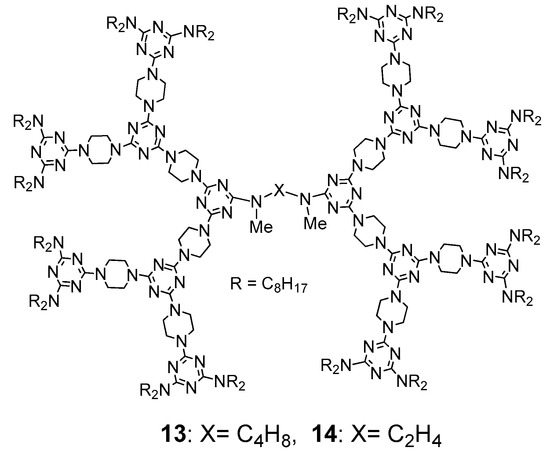
- Tsai, M.-J.; Hsieh, J.-W.; Lai, L.-L.; Cheng, K.-L.; Liu, S.-H.; Lee, J.-J.; Hsu, H.-F. Converting Nonliquid Crystals into Liquid Crystals by N-Methylation in the Central Linker of Triazine-Based Dendrimers. J. Org. Chem. 2016, 81, 5007–5013.
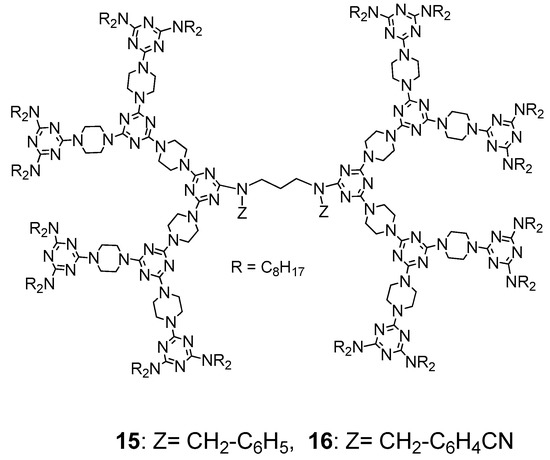
- Lee, C.-H.; Huang, C.-C.; Li, C.-Y.; Lai, L.-L.; Lee, J.-J.; Hsu, H.-F. Both increasing the Iso-to-Col transition and lowering the solidifying temperatures of a triazine-based dendrimer by introducing CN polar groups in the dendritic core. J. Mater. Chem. C 2019, 7, 14232–14238.
- Castelar, S.; Barberá, J.; Marcos, M.; Romero, P.; Serrano, J.-L.; Golemme, A.; Termine, R. Supramolecular dendrimers based on the self-assembly of carbazole-derived dendrons and triazine rings: Liquid crystal, photophysical and electrochemical properties. J. Mater. Chem. C 2013, 1, 7321–7332.





