
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Shih-Min Hsia | + 2599 word(s) | 2599 | 2021-08-12 04:35:42 | | | |
| 2 | Vivi Li | + 1 word(s) | 2600 | 2021-08-24 08:40:22 | | |
Video Upload Options
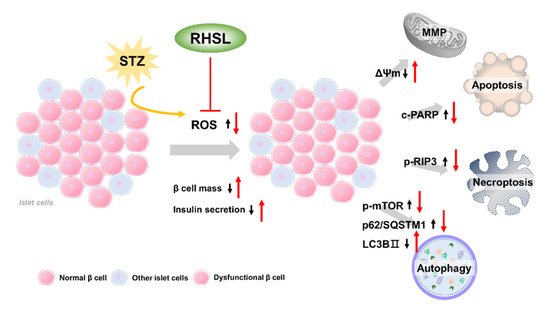
Type 2 diabetes mellitus is a complex multifactorial disease characterized by insulin resistance and dysfunction of pancreatic β-cells. Rice husk silica liquid (RHSL) is derived from rice husks and has not been explored in diabetes mellitus until now. Previous studies showed that rice husk is enriched with silica, and its silica nanoparticles are higher more biocompatible. To investigate the potential protective role of RHSL on pancreatic β cells, we utilized RIN-m5F pancreatic β cells and explored RHSL effect after streptozotocin (STZ)-stimulation. The recovery effects of RHSL were evaluated using flow cytometry, Western blotting, and immunofluorescence analysis. Results of our study showed that RHSL reversed the cell viability, insulin secretion, reactive oxygen species (ROS) production, and the change of mitochondria membrane potential (ΔΨm) in STZ-treated RIN-m5F cells. Moreover, the expression of phospho-receptor-interacting protein 3 (p-RIP3) and cleaved-poly (ADP-ribose) polymerase (PARP), phospho-mammalian target of rapamycin (p-mTOR), and sequestosome-1 (p62/SQSTM1) were significantly decreased, while the transition of light chain (LC)3-I to LC3-II was markedly increased after RHSL treatment in STZ-induced RIN-m5F cells. Interestingly, using autophagy inhibitors such as 3-methyladenine (3-MA) and chloroquine (CQ) both showed an increase in cleaved-PARP protein level, indicating apoptosis induction.
1. Introduction
2. Current Researches
2.1. The Effect of RHSL and STZ on RIN-m5F Cells Viability
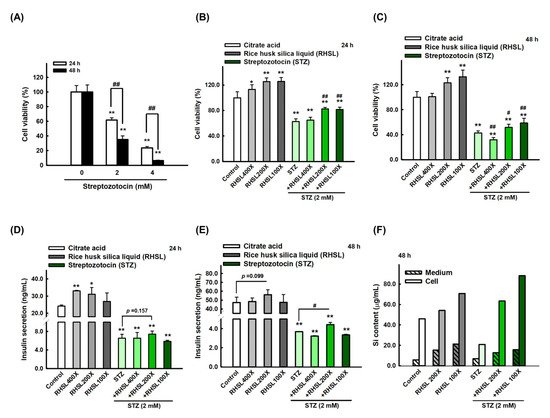
2.2. STZ Inhibits Insulin Secretion and Silicon Content in RIN-m5F Cells Which Is Restored by RHSL Co-Treatment
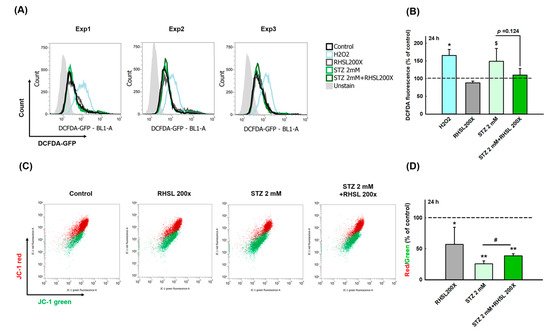
2.3. RHSL Protects RIN-m5F Cells Against STZ-induced ROS Production and Dysfunction of Mitochondria
3. Discussion
References
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98.
- Galicia-Garcia, U.; Benito-Vicente, A.; Jebari, S.; Larrea-Sebal, A.; Siddiqi, H.; Uribe, K.B.; Ostolaza, H.; Martín, C. Pathophysiology of type 2 diabetes mellitus. Int. J. Mol. Sci. 2020, 21, 6275.
- Bolzán, A.D.; Bianchi, M.S. Genotoxicity of streptozotocin. Mutat. Res. 2002, 512, 121–134.
- Goyal, S.N.; Reddy, N.M.; Patil, K.R.; Nakhate, K.T.; Ojha, S.; Patil, C.R.; Agrawal, Y.O. Challenges and issues with streptozotocin-induced diabetes-a clinically relevant animal model to understand the diabetes pathogenesis and evaluate therapeutics. Chem. Biol. Interact. 2016, 244, 49–63.
- Hayashi, K.; Kojima, R.; Ito, M. Strain differences in the diabetogenic activity of streptozotocin in mice. Biol. Pharm. Bull. 2006, 29, 1110–1119.
- Sandler, S.; Andersson, A.K.; Larsson, J.; Makeeva, N.; Olsen, T.; Arkhammar, P.O.G.; Hansen, J.B.; Karlsson, F.A.; Welsh, N. Possible role of an ischemic preconditioning-like response mechanism in katp channel opener-mediated protection against streptozotocin-induced suppression of rat pancreatic islet function. Biochem. Pharmacol. 2008, 76, 1748–1756.
- Reffitt, D.M.; Ogston, N.; Jugdaohsingh, R.; Cheung, H.F.; Evans, B.A.; Thompson, R.P.; Powell, J.J.; Hampson, G.N. Orthosilicic acid stimulates collagen type 1 synthesis and osteoblastic differentiation in human osteoblast-like cells in vitro. Bone 2003, 32, 127–135.
- Nielsen, F.H. Update on the possible nutritional importance of silicon. J. Trace Elem. Med. Biol. (GMS) 2014, 28, 379–382.
- Rico, H.; Gallego-Lago, J.L.; Hernández, E.R.; Villa, L.F.; Sanchez-Atrio, A.; Seco, C.; Gérvas, J.J. Effect of silicon supplement on osteopenia induced by ovariectomy in rats. Calcif. Tissue Int. 2000, 66, 53–55.
- Jugdaohsingh, R.; Tucker, K.L.; Qiao, N.; Cupples, L.A.; Kiel, D.P.; Powell, J.J. Dietary silicon intake is positively associated with bone mineral density in men and premenopausal women of the framingham offspring cohort. J. Bone Miner. Res. 2004, 19, 297–307.
- Maehira, F.; Ishimine, N.; Miyagi, I.; Eguchi, Y.; Shimada, K.; Kawaguchi, D.; Oshiro, Y. Anti-diabetic effects including diabetic nephropathy of anti-osteoporotic trace minerals on diabetic mice. Nutrition 2011, 27, 488–495.
- Ways, T.M.M.; Ng, K.W.; Lau, W.M.; Khutoryanskiy, V.V. Silica nanoparticles in transmucosal drug delivery. Pharmaceutics 2020, 12, 751.
- Eleazu, C.O.; Eleazu, K.C.; Chukwuma, S.; Essien, U.N. Review of the mechanism of cell death resulting from streptozotocin challenge in experimental animals, its practical use and potential risk to humans. J. Diabetes Metab. Disord. 2013, 12, 60.
- Turk, J.; Corbett, J.A.; Ramanadham, S.; Bohrer, A.; McDaniel, M.L. Biochemical evidence for nitric oxide formation from streptozotocin in isolated pancreatic islets. Biochem. Biophys. Res. Commun. 1993, 197, 1458–1464.
- Skelin, M.; Rupnik, M.; Cencic, A. Pancreatic beta cell lines and their applications in diabetes mellitus research. Altex 2010, 27, 105–113.
- Liu, Y.; Han, J.; Zhou, Z.; Li, D. Paeoniflorin protects pancreatic β cells from stz-induced damage through inhibition of the p38 mapk and jnk signaling pathways. Eur. J. Pharmacol. 2019, 853, 18–24.
- Wang, Y.; He, D.; Ni, C.; Zhou, H.; Wu, S.; Xue, Z.; Zhou, Z. Vitamin D induces autophagy of pancreatic β-cells and enhances insulin secretion. Mol. Med. Rep. 2016, 14, 2644–2650.
- Hu, S.; Kuwabara, R.; de Haan, B.J.; Smink, A.M.; de Vos, P. Acetate and butyrate improve β-cell metabolism and mitochondrial respiration under oxidative stress. Int. J. Mol. Sci. 2020, 21, 1542.
- AI-Nahdi, A.M.T.; John, A.; Raza, H. Elucidation of molecular mechanisms of streptozotocin-induced oxidative stress, apoptosis, and mitochondrial dysfunction in rin-5f pancreatic β-cells. Oxidative Med. Cell. Longev. 2017, 2017, 7054272.
- Sinha, K.; Das, J.; Pal, P.B.; Sil, P.C. Oxidative stress: The mitochondria-dependent and mitochondria-independent pathways of apoptosis. Arch. Toxicol. 2013, 87, 1157–1180.
- Sha, W.; Hu, F.; Bu, S. Mitochondrial dysfunction and pancreatic islet β-cell failure (review). Exp. Ther. Med. 2020, 20, 266.
- Al-Nahdi, A.M.T.; John, A.; Raza, H. Cytoprotective effects of n-acetylcysteine on streptozotocin-induced oxidative stress and apoptosis in rin-5f pancreatic β-cells. Cell Physiol. Biochem. 2018, 51, 201–216.
- Yu, W.C.; Chen, Y.L.; Hwang, P.A.; Chen, T.H.; Chou, T.C. Fucoidan ameliorates pancreatic β-cell death and impaired insulin synthesis in streptozotocin-treated β cells and mice via a sirt-1-dependent manner. Mol. Nutr. Food Res. 2017, 61, 1700136.
- Liang, H.; Pan, Y.; Teng, Y.; Yuan, S.; Wu, X.; Yang, H.; Zhou, P. A proteoglycan extract from ganoderma lucidum protects pancreatic beta-cells against stz-induced apoptosis. Biosci. Biotechnol. Biochem. 2020, 84, 2491–2498.
- Ye, Y.; Liu, J.; Xu, J.; Sun, L.; Chen, M.; Lan, M. Nano-sio2 induces apoptosis via activation of p53 and bax mediated by oxidative stress in human hepatic cell line. Toxicol. Vitr. Int. J. Publ. Assoc. BIBRA 2010, 24, 751–758.
- Kaczmarek, A.; Vandenabeele, P.; Krysko, D.V. Necroptosis: The release of damage-associated molecular patterns and its physiological relevance. Immunity 2013, 38, 209–223.
- Zhang, D.W.; Shao, J.; Lin, J.; Zhang, N.; Lu, B.J.; Lin, S.C.; Dong, M.Q.; Han, J. Rip3, an energy metabolism regulator that switches tnf-induced cell death from apoptosis to necrosis. Science 2009, 325, 332–336.
- Hsu, S.-K.; Chang, W.-T.; Lin, I.L.; Chen, Y.-F.; Padalwar, N.B.; Cheng, K.-C.; Teng, Y.-N.; Wang, C.-H.; Chiu, C.-C. The role of necroptosis in ros-mediated cancer therapies and its promising applications. Cancers 2020, 12, 2185.
- Yi, W.; OuYang, Q. Adiponectin improves diabetic nephropathy by inhibiting necrotic apoptosis. Arch. Med. Sci. AMS 2019, 15, 1321–1328.
- Marasco, M.R.; Linnemann, A.K. Β-cell autophagy in diabetes pathogenesis. Endocrinology 2018, 159, 2127–2141.
- Mazza, S.; Maffucci, T. Autophagy and pancreatic β-cells. Vitam. Horm. 2014, 95, 145–164.
- Jung, H.S.; Chung, K.W.; Won Kim, J.; Kim, J.; Komatsu, M.; Tanaka, K.; Nguyen, Y.H.; Kang, T.M.; Yoon, K.H.; Kim, J.W.; et al. Loss of autophagy diminishes pancreatic beta cell mass and function with resultant hyperglycemia. Cell Metab. 2008, 8, 318–324.
- Klionsky, D.J.; Abdelmohsen, K.; Abe, A.; Abedin, M.J.; Abeliovich, H.; Acevedo Arozena, A.; Adachi, H.; Adams, C.M.; Adams, P.D.; Adeli, K.; et al. Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 2016, 12, 1–222.
- Yang, Y.P.; Hu, L.F.; Zheng, H.F.; Mao, C.J.; Hu, W.D.; Xiong, K.P.; Wang, F.; Liu, C.F. Application and interpretation of current autophagy inhibitors and activators. Acta Pharmacol. Sin. 2013, 34, 625–635.
- Pasquier, B. Autophagy inhibitors. Cell. Mol. Life Sci. CMLS 2016, 73, 985–1001.
- Bahramsoltani, R.; Farzaei, M.H.; Sajadimajd, S.; Iranpanah, A.; Khazaei, M.; Pourjabar, Z.; Hajimahmoodi, M.; Rahimi, R. In vitro and in vivo antidiabetic activity of tamarix stricta boiss.: Role of autophagy. J. Ethnopharmacol. 2021, 269, 113692.
- Jugdaohsingh, R.; Anderson, S.H.; Tucker, K.L.; Elliott, H.; Kiel, D.P.; Thompson, R.P.; Powell, J.J. Dietary silicon intake and absorption. Am. J. Clin. Nutr. 2002, 75, 887–893.
- Robberecht, H.; Van Cauwenbergh, R.; Van Vlaslaer, V.; Hermans, N. Dietary silicon intake in belgium: Sources, availability from foods, and human serum levels. Sci. Total Environ. 2009, 407, 4777–4782.





