
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Angela Milán-Tomás | + 2193 word(s) | 2193 | 2021-07-13 08:19:51 | | | |
| 2 | Lily Guo | Meta information modification | 2193 | 2021-08-19 08:54:15 | | | | |
| 3 | Jamir Pitton Rissardo | + 84 word(s) | 2277 | 2023-09-16 02:32:16 | | |
Video Upload Options
Lewy body dementias (LBDs) consist of dementia with Lewy bodies (DLB) and Parkinson’s disease dementia (PDD), which are clinically similar syndromes that share neuropathological findings with widespread cortical Lewy body deposition, often with a variable degree of concomitant Alzheimer pathology. The objective of this article is to provide an overview of the neuropathological and clinical features, current diagnostic criteria, biomarkers, and management of LBD. Literature research was performed using the PubMed database, and the most pertinent articles were read and are discussed in this paper. The diagnostic criteria for DLB have recently been updated, with the addition of indicative and supportive biomarker information. The time interval of dementia onset relative to parkinsonism remains the major distinction between DLB and PDD, underpinning controversy about whether they are the same illness in a different spectrum of the disease or two separate neurodegenerative disorders. The treatment for LBD is only symptomatic, but the expected progression and prognosis differ between the two entities. Diagnosis in prodromal stages should be of the utmost importance, because implementing early treatment might change the course of the illness if disease-modifying therapies are developed in the future. Thus, the identification of novel biomarkers constitutes an area of active research, with a special focus on α-synuclein markers.
1. Introduction
2. Current Diagnostic Criteria for Parkinson’s Disease Dementia and Lewy Body Dementia
| DLB 1 | PDD 2 | ||
|---|---|---|---|
| Central features | Essential for a diagnosis: Dementia, in early stages with memory impairment, may not necessarily occur but is usually evident with progression. Deficits in tests of attention, executive function, and visuoperceptual ability may be especially prominent and occur early. | Core Features (I) | Essential for a diagnosis (both must be present): Diagnosis of Parkinson disease according to Queen Square Brain Bank criteria and Dementia syndrome with impairment in more than one cognitive domain |
| Core clinical features | The first three typically occur early and may persist throughout the course:
|
Associated clinical features (II) |
|
|
PDD; lack of behavioral symptoms, however, does not exclude the diagnosis | ||
| Supportive clinical features | Severe sensitivity to antipsychotic agents; postural instability; repeated falls; syncope or other transient episodes of unresponsiveness; severe autonomic dysfunction, e.g., constipation, orthostatic hypotension, urinary incontinence; hypersomnia; hyposmia; hallucinations in other modalities; systematized delusions; apathy, anxiety, and depression. | None of the group (III) features present | Features which do not exclude PDD, but make the diagnosis uncertain:
|
| Indicative biomarkers |
|
None of the group (IV) features present | Features suggesting other conditions or diseases as cause of mental impairment, which, when present, make it impossible to reliably diagnose PDD: 1. Cognitive and behavioral symptoms appearing solely in the context of other conditions such as acute confusion due to (a.) systemic diseases or abnormalities (b.) drug intoxication 2. Major Depression according to DSM IV |
| Supportive biomarkers |
|
Supportive or indicative biomarkers |
No supportive or indicative biomarkers are needed for the diagnosis of PDD as per Emre et al. (2007) diagnostic criteria. |
| Diagnosis of probable or possible DLB |
Probable: (a) ≥2 core clinical features of DLB are present, with or without the presence of indicative biomarkers, OR (b). Only one core clinical feature is present, but with ≥1 indicative biomarkers. Probable DLB should not be diagnosed on the basis of biomarkers alone. Possible: (a). Only one core clinical feature of DLB is present, with no indicative biomarker evidence, OR (b). ≥1 indicative biomarkers is present but there are no core clinical features. |
Diagnosis of probable or possible PDD |
Probable: (a) Core features: Both must be present; (b). Associated clinical features: Typical profile of cognitive deficits and the presence of at least one behavioral symptom (lack of behavioral symptoms, however, does not exclude the diagnosis); (c) None of the group III features present; (d) None of the group IV features present. Possible: (a) Core features: Both must be present (b). Associated clinical features: Atypical profile of cognitive impairment in one or more domains (e.g., fluent aphasia, or pure storage-failure type amnesia) and behavioral symptoms may or may not be present; OR (c) One or more of the group III features present, (d) None of the group IV features present |
3. Diagnostic Approach
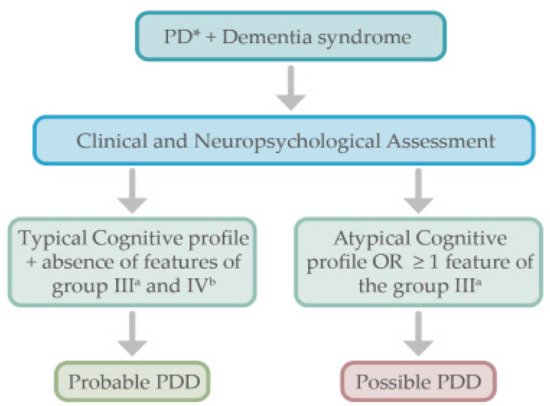
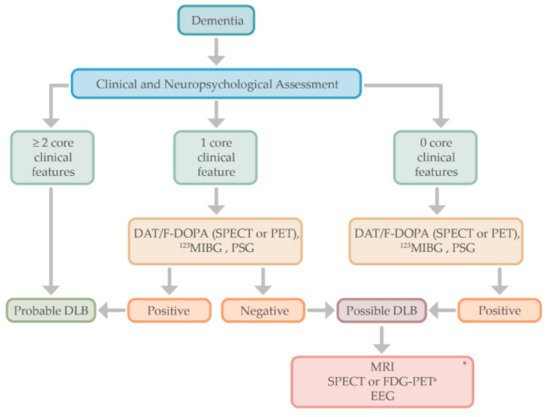
4. Behavioral and Neuropsychiatric Manifestations of Lewy Body Dementias
References
- Harding, A.J.; Halliday, G.M. Cortical Lewy body pathology in the diagnosis of dementia. Acta Neuropathol. 2001, 102, 355–363.
- Klein, J.C.; Eggers, C.; Kalbe, E.; Weisenbach, S.; Hohmann, C.; Vollmar, S.; Baudrexel, S.; Diederich, N.J.; Heiss, W.D.; Hilker, R. Neurotransmitter changes in dementia with Lewy bodies and Parkinson disease dementia in vivo. Neurology 2010, 74, 885–892.
- Zaccai, J.; McCracken, C.; Brayne, C. A systematic review of prevalence and incidence studies of dementia with Lewy bodies. Age Ageing 2005, 34, 561–566.
- Hogan, D.B.; Fiest, K.M.; Roberts, J.I.; Maxwell, C.J.; Dykeman, J.; Pringsheim, T.; Steeves, T.; Smith, E.E.; Pearson, D.; Jetté, N. The prevalence and incidence of dementia with lewy bodies: A systematic review. Can. J. Neurol. Sci. 2016, 43, S83–S95.
- Pfeiffer, R.F. Non-motor symptoms in Parkinson’s disease. Park. Relat. Disord. 2016, 22, S119–S122.
- Rodriguez-Oroz, M.C.; Jahanshahi, M.; Krack, P.; Litvan, I.; Macias, R.; Bezard, E.; Obeso, J.A. Initial clinical manifestations of Parkinson’s disease: Features and pathophysiological mechanisms. Lancet Neurol. 2009, 8, 1128–1139.
- Litvan, I.; Goldman, J.G.; Tröster, A.I.; Schmand, B.A.; Weintraub, D.; Petersen, R.C.; Mollenhauer, B.; Adler, C.H.; Marder, K.; Williams-Gray, C.H.; et al. Diagnostic criteria for mild cognitive impairment in Parkinson’s disease: Movement Disorder Society Task Force guidelines. Mov. Disord. 2012, 27, 349–356.
- Goldman, J.G.; Sieg, E. Cognitive impairment and dementia in Parkinson disease. Clin. Geriatr. Med. 2020, 36, 365–377.
- Nicoletti, A.; Luca, A.; Baschi, R.; Cicero, C.E.; Mostile, G.; Davì, M.; Pilati, L.; Restivo, V.; Zappia, M.; Monastero, R. Incidence of mild cognitive impairment and dementia in Parkinson’s disease: The Parkinson’s disease cognitive impairment study. Front. Aging Neurosci. 2019, 10, 1–12.
- Janvin, C.C.; Larsen, J.P.; Aarsland, D.; Hugdahl, K. Subtypes of mild cognitive impairment in Parkinson’s disease: Progression to dementia. Mov. Disord. 2006, 21, 1343–1349.
- Gasca-Salas, C.; Estanga, A.; Clavero, P.; Aguilar-Palacio, I.; González-Redondo, R.; Obeso, J.A.; Rodriguez-Oroz, M.C. Longitudinal assessment of the pattern of cognitive decline in non-demented patients with advanced Parkinson’s disease. J. Parkinson’s Dis. 2014, 4, 677–686.
- Litvan, I.; Aarsland, D.; Adler, C.H.; Goldman, J.G.; Kulisevsky, J.; Mollenhauer, B.; Rodriguez-Oroz, M.C.; Tröster, A.I.; Weintraub, D. MDS task force on mild cognitive impairment in Parkinson’s disease: Critical review of PD-MCI. Mov. Disord. 2011, 26, 1814–1824.
- Hely, M.A.; Reid, W.G.J.; Adena, M.A.; Halliday, G.M.; Morris, J.G.L. The Sydney Multicenter Study of Parkinson’s disease: The inevitability of dementia at 20 years. Mov. Disord. 2008, 23, 837–844.
- Stephen N. Gomperts; Lewy Body Dementias. Contin. Lifelong Learn. Neurol. 2016, 22, 435-463.
- Emre, M.; Aarsland, D.; Brown, R.; Burn, D.J.; Duyckaerts, C.; Mizuno, Y.; Broe, G.A.; Cummings, J.; Dickson, D.W.; Gauthier, S.; et al. Clinical diagnostic criteria for dementia associated with Parkinson’s disease. Mov. Disord. 2007, 22, 1689–1707.
- Thomas, A.J.; Donaghy, P.; Roberts, G.; Colloby, S.J.; Barnett, N.A.; Petrides, G.; Lloyd, J.; Olsen, K.; Taylor, J.P.; McKeith, I.; et al. Diagnostic accuracy of dopaminergic imaging in prodromal dementia with Lewy bodies. Psychol. Med. 2019, 49, 396–402.
- Yoshita, M.; Taki, J.; Yamada, M. A clinical role for [123I]MIBG myocardial scintigraphy in the distinction between dementia of the Alzheimer’s-type and dementia with Lewy bodies. J. Neurol. Neurosurg. Psychiatry 2001, 71, 583–588.
- Mckeith, I.G.; Sci, M.; Boeve, B.F.; Dickson, D.W.; Halliday, G.; Taylor, J.-P.; Psych, M.; Weintraub, D.; Aarsland, D.; Galvin, J.; et al. Diagnosis and management of dementia with Lewy bodies Fourth consensus report of the DLB Consortium. Neurology 2017, 89, 88–100.
- Jellinger, K.A.; Korczyn, A.D. Are dementia with Lewy bodies and Parkinson’s disease dementia the same disease? BMC Med. 2018, 16, 34.
- Lenka, A.; Pagonabarraga, J.; Pal, P.K.; Bejr-Kasem, H.; Kulisvesky, J. Minor hallucinations in Parkinson disease: A subtle symptom with major clinical implications. Neurology 2019, 93, 259–266.
- Martínez-Horta, S.; Riba, J.; de Bobadilla, R.F.; Pagonabarraga, J.; Pascual-Sedano, B.; Antonijoan, R.M.; Romero, S.; Mañanas, M.À.; García-Sanchez, C.; Kulisevsky, J. Apathy in parkinson’s disease: Neurophysiological evidence of impaired incentive processing. J. Neurosci. 2014, 34, 5918–5926.
- Weintraub, D.; Mamikonyan, E. The Neuropsychiatry of Parkinson disease: A perfect storm. Am. J. Geriatr. Psychiatry 2019, 27, 998–1018.
- Aarsland, D.; Ballard, C.; Larsen, J.P.; McKeith, I. A comparative study of psychiatric symptoms in dementia with Lewy bodies and Parkinson’s disease with and without dementia. Int. J. Geriatr. Psychiatry 2001, 16, 528–536.
- Marsh, L. Depression and Parkinson’s disease: Current knowledge. Curr. Neurol. Neurosci. Rep. 2013, 13, 409.
- Patterson, L.; Rushton, S.P.; Attems, J.; Thomas, A.J.; Morris, C.M. Degeneration of dopaminergic circuitry influences depressive symptoms in Lewy body disorders. Brain Pathol. 2019, 29, 544–557.
- Ishihara, L.; Brayne, C. A systematic review of depression and mental illness preceding Parkinson’s disease. Acta Neurol. Scand. 2006, 113, 211–220.
- McKeith, I.; Cummings, J. Behavioural changes and psychological symptoms in dementia disorders. Lancet Neurol. 2005, 4, 735–742.
- Kuring, J.K.; Mathias, J.L.; Ward, L. Prevalence of depression, anxiety and PTSD in people with dementia: A systematic review and meta-analysis. Neuropsychol. Rev. 2018, 28, 393–416.
- Fritze, F.; Ehrt, U.; Hortobagyi, T.; Ballard, C.; Aarsland, D. Depressive symptoms in Alzheimer’s disease and Lewy body dementia: A one-year follow-up study. Dement. Geriatr. Cogn. Disord. 2011, 32, 143–149.
- Aarsland, D.; Brønnick, K.; Ehrt, U.; De Deyn, P.P.; Tekin, S.; Emre, M.; Cummings, J.L. Neuropsychiatric symptoms in patients with Parkinson’s disease and dementia: Frequency, profile and associated care giver stress. J. Neurol. Neurosurg. Psychiatry 2007, 78, 36–42.
- Borroni, B.; Agosti, C.; Padovani, A. Behavioral and psychological symptoms in dementia with Lewy-bodies (DLB): Frequency and relationship with disease severity and motor impairment. Arch. Gerontol. Geriatr. 2008, 46, 101–106.
- Breitve, M.H.; Brønnick, K.; Chwiszczuk, L.J.; Hynninen, M.J.; Aarsland, D.; Rongve, A. Apathy is associated with faster global cognitive decline and early nursing home admission in dementia with Lewy bodies. Alzheimer’s Res. Ther. 2018, 10.
- Fields, J.A. Cognitive and neuropsychiatric features in Parkinson’s and Lewy body dementias. Arch. Clin. Neuropsychol. 2017, 32, 786–801.
- Hynninen, M.J.; Breitve, M.H.; Rongve, A.; Aarsland, D.; Nordhus, I.H. The frequency and correlates of anxiety in patients with first-time diagnosed mild dementia. Int. Psychogeriatr. 2012, 24, 1771–1778.
- Ffytche, D.H.; Creese, B.; Politis, M.; Chaudhuri, K.R.; Weintraub, D.; Ballard, C.; Aarsland, D. The psychosis spectrum in Parkinson disease. Nat. Rev. Neurol. 2017, 13, 81–95.
- Fénelon, G.; Mahieux, F.; Huon, R.; Ziégler, M. Hallucinations in Parkinson’s disease. Prevalence, phenomenology and risk factors. Brain 2000, 123, 733–745.
- Gomperts, S.N. Lewy body dementias: Dementia with lewy bodies and Parkinson disease dementia. Continuum 2016, 22, 435–463.
- State, D.; Events, S.L. Clinical features of auditory hallucinations in patients with dementia with lewy bodies: A soundtrack of visual hallucinations. J. Clin. Psychiatry 2014, 2012, 11–12.
- Pagonabarraga, J.; Martinez-Horta, S.; Fernández de Bobadilla, R.; Pérez, J.; Ribosa-Nogué, R.; Marín, J.; Pascual-Sedano, B.; García, C.; Gironell, A.; Kulisevsky, J. Minor hallucinations occur in drug-naive Parkinson’s disease patients, even from the premotor phase. Mov. Disord. 2016, 31, 45–52.
- Uchiyama, M.; Nishio, Y.; Yokoi, K.; Hirayama, K.; Imamura, T.; Shimomura, T.; Mori, E. Pareidolias: Complex visual illusions in dementia with Lewy bodies. Brain 2012, 135, 2458–2469.
- Uchiyama, M.; Nishio, Y.; Yokoi, K.; Hosokai, Y.; Takeda, A.; Mori, E. Pareidolia in Parkinson’s disease without dementia: A positron emission tomography study. Park. Relat. Disord. 2015, 21, 603–609.





