
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Maciej Miarka | + 1871 word(s) | 1871 | 2021-07-25 11:03:23 | | | |
| 2 | Beatrix Zheng | + 172 word(s) | 2043 | 2021-08-11 10:22:07 | | |
Video Upload Options
Optimizing patients’ condition before liver transplantation (LT) could potentially improve survival of LT patients. We focused on sarcopenia, as a common factor in liver transplant candidates that can impact their cardiopulmonary performance at the point of listing, morbidity, and mortality after LT. We performed a single-center cohort study on 98 consecutive patients with liver cirrhosis who were transplanted between March 2015 and December 2017. The third lumbar vertebra skeletal muscle index (L3SMI) was calculated using CT imaging to distinguish sarcopenia at listing for LT. Data regarding liver function, body mass index (BMI), cardiac biomarkers, the peak oxygen uptake (VO2) and LT outcome were collected and correlated to L3SMI. For data analysis the Dell Statistica (Version 13. Dell Inc., Rondrock, TX, USA) was used. In total, 98 cirrhotic patients were included. Fifty-five (56.1%) patients, mostly males, had sarcopenia according to L3SMI, with the lowest L3SMI in males with alcohol-related liver disease. Lower L3SMI correlated with lower BMI, lower VO2 peak, and higher NTproBNP (all p < 0.001) and revealed an essential correlation with prolonged ICU stay (r = −0.21, p < 0.05). 33 patients were unable to perform cardio-pulmonary exercise test, mostly sarcopenic (67%), with more advanced liver insufficiency (assessed with CPC and MELD scores) and longer stay at ICU after LT (all p < 0.001). Sarcopenia was common among LT recipients. It was associated with inferior result in cardio-pulmonary performance before LT and prolonged ICU stay after grafting.
1. Introduction
2. Analysis on Results
| Sarcopenic (n = 55) |
Non-Sarcopenic (n = 43) |
p Value | |
|---|---|---|---|
| Age | 55 (9) | 57 (9) | 0.244 |
| Gender (female) | 20% | 28% | 0.364 |
| Etiology HCV HCC ALD |
29% 20% 51% |
32.5% 35% 32.5% |
0.716 0.100 0.071 |
| BMI (kg/m2) | 24.8 (5) | 29.3 (5) | <0.001 |
| MELD (points) | 18 (14) | 16 (11) | 0.161 |
| CPC (points) | 10 (3) | 9 (4) | 0.289 |
| VO2 Peak (J. eq.) | 63.2 (33) | 79.3 (29) | 0.064 |
| TnI | 0.009 (0.02) | 0.010 (0.03) | 0.988 |
| NT-proBNP | 176 (263) | 74 (79) | 0.043 |
| L3 SMI | VO2 Peak (J. Eq.) |
TnI | NT-proBNP | |
|---|---|---|---|---|
| L3SMI | 0.592 ** | −0.095 | −0.531 ** | |
| VO2 Peak (J. eq.) | 0.592 ** | −0.098 | −0.354 | |
| TnI | −0.095 | −0.098 | 0.107 | |
| NT-proBNP | −0.531 ** | −0.354 | 0.107 | |
| BMI | 0.473 ** | 0.485 ** | 0.197 | −0.056 |
| MELD | −0.159 | −0.151 | 0.380 * | 0.266 |
| CPC | −0.159 | −0.142 | 0.344 * | 0.088 |
| ICU Stay | −0.208 * | −0.217 | 0.087 | 0.358 * |
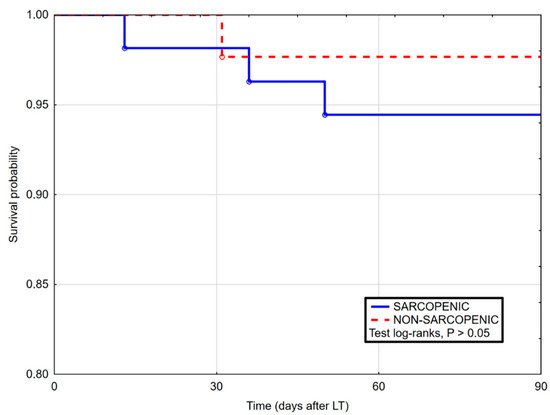
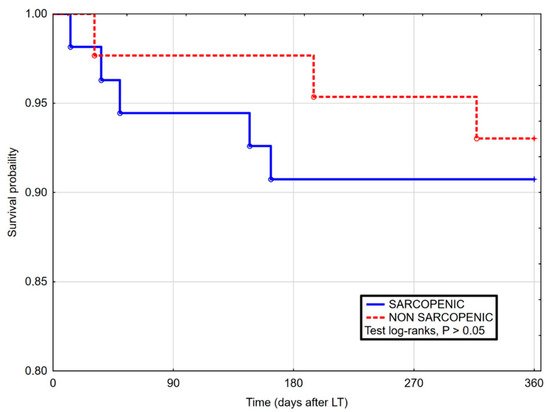
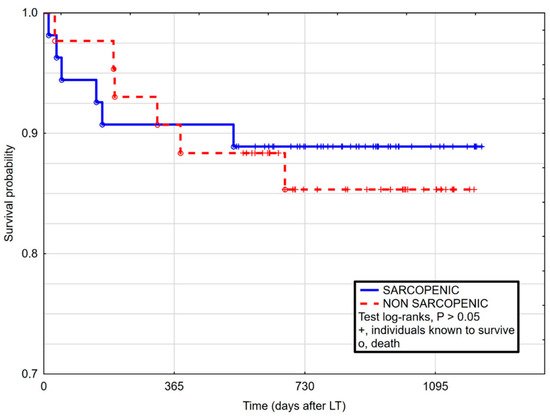
3. Current Insights
References
- Tandon, P.; Ismond, K.P.; Riess, K.; Duarte-Rojo, A.; Al-Judaibi, B.; Dunn, M.A.; Holman, J.; Howes, N.; Haykowsky, M.J.F.; Josbeno, D.A.; et al. Exercise in cirrhosis: Translating evidence and experience to practice. J. Hepatol. 2018, 69, 1164–1177.
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423.
- Englesbe, M.J.; Lee, J.S.; He, K.; Fan, L.; Schaubel, D.E.; Sheetz, K.H.; Harbaugh, C.M.; Holcombe, S.A.; Campbell, D.A., Jr.; Sonnenday, C.J.; et al. Analytic morphomics, core muscle size, and surgical outcomes. Ann. Surg. 2012, 256, 255–261.
- Streja, E.; Molnar, M.Z.; Kovesdy, C.P.; Bunnapradist, S.; Jing, J.; Nissenson, A.R.; Mucsi, I.; Danovitch, G.M.; Kalantar-Zadeh, K. Associations of pretransplant weight and muscle mass with mortality in renal transplant recipients. Clin. J. Am. Soc. Nephrol. 2011, 6, 1463–1473.
- Tandon, P.; Ney, M.; Irwin, I.; Ma, M.M.; Gramlich, L.; Bain, V.G.; Esfandiari, N.; Baracos, V.; Montano-Loza, A.J.; Myers, R.P. Severe muscle depletion in patients on the liver transplant wait list: Its prevalence and independent prognostic value. Liver Transplant. 2012, 18, 1209–1216.
- Durand, F.; Buyse, S.; Francoz, C.; Laouénan, C.; Bruno, O.; Belghiti, J.; Moreau, R.; Vilgrain, V.; Valla, D. Prognostic value of muscle atrophy in cirrhosis using psoas muscle thickness on computed tomography. J. Hepatol. 2014, 60, 1151–1157.
- Meza-Junco, J.; Montano-Loza, A.J.; Baracos, V.E.; Prado, C.M.; Bain, V.G.; Beaumont, C.; Esfandiari, N.; Lieffers, J.R.; Sawyer, M.B. Sarcopenia as a prognostic index of nutritional status in concurrent cirrhosis and hepatocellular carcinoma. J. Clin. Gastroenterol. 2013, 47, 861–870.
- Montano-Loza, A.J.; Meza-Junco, J.; Prado, C.M.; Lieffers, J.R.; Baracos, V.E.; Bain, V.G.; Sawyer, M.B. Muscle wasting is associated with mortality in patients with cirrhosis. Clin. Gastroenterol. Hepatol. 2012, 10, 166–173.e1.
- Bhanji, R.A.; Takahashi, N.; Moynagh, M.R.; Narayanan, P.; Angirekula, M.; Mara, K.C.; Dierkhising, R.A.; Watt, K.D. The evolution and impact of sarcopenia pre- and post-liver transplantation. Aliment. Pharmacol. Ther. 2019, 49, 807–813.
- Van Vugt, J.L.; Levolger, S.; de Bruin, R.W.; van Rosmalen, J.; Metselaar, H.J.; IJzermans, J.N.M. Systematic Review and Meta-Analysis of the Impact of Computed Tomography-Assessed Skeletal Muscle Mass on Outcome in Patients Awaiting or Undergoing Liver Transplantation. Am. J. Transplant. 2016, 16, 2277–2292.
- Krell, R.W.; Kaul, D.R.; Martin, A.R.; Englesbe, M.J.; Sonnenday, C.J.; Cai, S.; Malani, P.N. Association between sarcopenia and the risk of serious infection among adults undergoing liver transplantation. Liver Transplant. 2013, 19, 1396–1402.
- DiMartini, A.; Cruz, R.J., Jr.; Dew, M.A.; Myaskovsky, L.; Goodpaster, B.; Fox, K.; Kim, K.H.; Fontes, P. Muscle mass predicts outcomes following liver transplantation. Liver Transplant. 2013, 19, 1172–1180.
- Englesbe, M.J.; Patel, S.P.; He, K.; Lynch, R.J.; Schaubel, D.E.; Harbaugh, C.; Holcombe, S.A.; Wang, S.C.; Segev, D.L.; Sonnenday, C.J. Sarcopenia and mortality after liver transplantation. J. Am. Coll. Surg. 2010, 211, 271–278.
- Hamaguchi, Y.; Kaido, T.; Okumura, S.; Fujimoto, Y.; Ogawa, K.; Mori, A.; Hammad, A.; Tamai, Y.; Inagaki, N.; Uemoto, S. Impact of quality as well as quantity of skeletal muscle on outcomes after liver transplantation. Liver Transplant. 2014, 20, 1413–1419.
- Kalafateli, M.; Mantzoukis, K.; Choi Yau, Y.; Mohammad, A.O.; Arora, S.; Rodrigues, S.; de Vos, M.; Papadimitriou, K.; Thorburn, D.; O’Beirne, J.; et al. Malnutrition and sarcopenia predict post-liver transplantation outcomes independently of the Model for End-stage Liver Disease score. J. Cachexia Sarcopenia Muscle 2017, 8, 113–121.
- Masuda, T.; Shirabe, K.; Ikegami, T.; Harimoto, N.; Yoshizumi, T.; Soejima, Y.; Uchiyama, H.; Ikeda, T.; Baba, H.; Maehara, Y. Sarcopenia is a prognostic factor in living donor liver transplantation. Liver Transplant. 2014, 20, 401–407.
- Montano-Loza, A.J.; Meza-Junco, J.; Baracos, V.E.; Prado, C.M.; Ma, M.; Meeberg, G.; Beaumont, C.; Tandon, P.; Esfandiari, N.; Sawyer, M.B.; et al. Severe muscle depletion predicts postoperative length of stay but is not associated with survival after liver transplantation. Liver Transplant. 2014, 20, 640–648.
- D’Avola, D.; Cuervas-Mons, V.; Marti, J.; Ortiz de Urbina, J.; Lladó, L.; Jimenez, C.; Otero, E.; Suarez, F.; Rodrigo, J.M.; Gómez, M.A.; et al. Cardiovascular morbidity and mortality after liver transplantation: The protective role of mycophenolate mofetil. Liver Transplant. 2017, 23, 498–509.
- Lai, C.W.; Minto, G.; Challand, C.P.; Hosie, K.B.; Sneyd, J.R.; Creanor, S.; Struthers, R.A. Patients’ inability to perform a preoperative cardiopulmonary exercise test or demonstrate an anaerobic threshold is associated with inferior outcomes after major colorectal surgery. Br. J. Anaesth. 2013, 111, 607–611.
- Epstein, S.K.; Freeman, R.B.; Khayat, A.; Unterborn, J.N.; Pratt, D.S.; Kaplan, M.M. Aerobic capacity is associated with 100-day outcome after hepatic transplantation. Liver Transplant. 2004, 10, 418–424.
- Bhanji, R.A.; Narayanan, P.; Moynagh, M.R.; Takahashi, N.; Angirekula, M.; Kennedy, C.C.; Mara, K.C.; Dierkhising, R.A.; Watt, K.D. Differing Impact of Sarcopenia and Frailty in Nonalcoholic Steatohepatitis and Alcoholic Liver Disease. Liver Transplant. 2019, 25, 14–24.
- Bonet-Ponce, L.; Saez-Atienzar, S.; da Casa, C.; Flores-Bellver, M.; Barcia, J.M.; Sancho-Pelluz, J.; Romero, F.J.; Jordan, J.; Galindo, M.F. On the mechanism underlying ethanol-induced mitochondrial dynamic disruption and autophagy response. Biochim. Biophys. Acta 2015, 1852, 1400–1409.
- Nishikawa, H.; Shiraki, M.; Hiramatsu, A.; Moriya, K.; Hino, K.; Nishiguchi, S. Japan Society of Hepatology guidelines for sarcopenia in liver disease (1st edition): Recommendation from the working group for creation of sarcopenia assessment criteria. Hepatol. Res. 2016, 46, 951–963.
- Villalta, J.; Ballesca, J.L.; Nicolas, J.M.; Martinez de Osaba, M.J.; Antunez, E.; Pimentel, C. Testicular function in asymptomatic chronic alcoholics: Relation to ethanol intake. Alcohol. Clin. Exp. Res. 1997, 21, 128–133.
- Lang, C.H.; Frost, R.A.; Deshpande, N.; Kumar, V.; Vary, T.C.; Jefferson, L.S.; Kimball, S.R. Alcohol impairs leucine-mediated phosphorylation of 4E-BP1, S6K1, eIF4G, and mTOR in skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2003, 285, E1205–E1215.
- Benjamin, J.; Shasthry, V.; Kaal, C.R.; Anand, L.; Bhardwaj, A.; Pandit, V.; Arora, A.; Rajesh, S.; Pamecha, V.; Jain, V.; et al. Characterization of body composition and definition of sarcopenia in patients with alcoholic cirrhosis: A computed tomography based study. Liver Int. 2017, 37, 1668–1674.
- Kang, S.H.; Jeong, W.K.; Baik, S.K.; Cha, S.H.; Kim, M.Y. Impact of sarcopenia on prognostic value of cirrhosis: Going beyond the hepatic venous pressure gradient and MELD score. J. Cachexia Sarcopenia Muscle 2018, 9, 860–870.
- Montano-Loza, A.J.; Duarte-Rojo, A.; Meza-Junco, J.; Baracos, V.E.; Sawyer, M.B.; Pang, J.X.; Beaumont, C.; Esfandiari, N.; Myers, R.P. Inclusion of Sarcopenia Within MELD (MELD-Sarcopenia) and the Prediction of Mortality in Patients With Cirrhosis. Clin. Transl. Gastroenterol. 2015, 6, e102.
- Van Vugt, J.L.A.; Alferink, L.J.M.; Buettner, S.; Gaspersz, M.P.; Bot, D.; Darwish Murad, S.; Feshtali, S.; van Ooijen, P.M.A.; Polak, W.G.; Porte, R.J.; et al. A model including sarcopenia surpasses the MELD score in predicting waiting list mortality in cirrhotic liver transplant candidates: A competing risk analysis in a national cohort. J. Hepatol. 2017.
- Prentis, J.M.; Manas, D.M.; Trenell, M.I.; Hudson, M.; Jones, D.J.; Snowden, C.P. Submaximal cardiopulmonary exercise testing predicts 90-day survival after liver transplantation. Liver Transplant. 2012, 18, 152–159.
- Dharancy, S.; Lemyze, M.; Boleslawski, E.; Neviere, R.; Declerck, N.; Canva, V.; Wallaert, B.; Mathurin, P.; Pruvot, F.R. Impact of impaired aerobic capacity on liver transplant candidates. Transplantation 2008, 86, 1077–1083.
- Carey, E.J.; Steidley, D.E.; Aqel, B.A.; Byrne, T.J.; Mekeel, K.L.; Rakela, J.; Vargas, H.E.; Douglas, D.D. Six-minute walk distance predicts mortality in liver transplant candidates. Liver Transplant. 2010, 16, 1373–1378.
- Yadav, A.; Chang, Y.H.; Carpenter, S.; Silva, A.C.; Rakela, J.; Aqel, B.A.; Byrne, T.J.; Douglas, D.D.; Vargas, H.E.; Carey, E.J.; et al. Relationship between sarcopenia, six-minute walk distance and health-related quality of life in liver transplant candidates. Clin. Transplant. 2015, 29, 134–141.
- Das, S.R.; Drazner, M.H.; Dries, D.L.; Vega, G.L.; Stanek, H.G.; Abdullah, S.M.; Canham, R.M.; Chung, A.K.; Leonard, D.; Wians, F.H., Jr.; et al. Impact of body mass and body composition on circulating levels of natriuretic peptides: Results from the Dallas Heart Study. Circulation 2005, 112, 2163–2168.
- Zheng, L.H.; Wu, L.M.; Yao, Y.; Chen, W.S.; Bao, J.R.; Huang, W.; Shi, R.; Zhang, K.J.; Zhang, S. Impact of body mass index on plasma N-terminal ProB-type natriuretic peptides in Chinese atrial fibrillation patients without heart failure. PLoS ONE 2014, 9, e105249.
- Fulks, M.; Kaufman, V.; Clark, M.; Stout, R.L. NT-proBNP Predicts All-Cause Mortality in a Population of Insurance Applicants, Follow-up Analysis and Further Observations. J. Insur. Med. 2017, 47, 107–113.
- Martins, T.; Vitorino, R.; Moreira-Goncalves, D.; Amado, F.; Duarte, J.A.; Ferreira, R. Recent insights on the molecular mechanisms and therapeutic approaches for cardiac cachexia. Clin. Biochem. 2014, 47, 8–15.
- Ooi, P.H.; Hager, A.; Mazurak, V.C.; Dajani, K.; Bhargava, R.; Gilmour, S.M.; Mager, D.R. Sarcopenia in Chronic Liver Disease: Impact on Outcomes. Liver Transplant. 2019, 25, 1422–1438.





