
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Houda Ben Slama | + 2458 word(s) | 2458 | 2021-07-13 06:06:26 | | | |
| 2 | Nora Tang | + 24 word(s) | 2482 | 2021-08-06 04:09:29 | | |
Video Upload Options
Synthetic dyes are mostly derived from petrochemical compounds, they are commercialized in liquid, powder, pastes, or granule forms. They are endowed with multiple potentialities such as fast and consistent coloration with different classes of fabrics as mentioned in the above section, a wide range of color pigments and shades, facility of manipulation, stability over several external factors and economical energy consumption. Therefore, the majority of synthetic dyes cause harmful impacts when discharged in non-treated or partially treated forms in the environment.
1. Introduction
The worldwide developmental process influenced all fields of life by providing rapidity, efficacy and comfort. However, it has also engendered side effects related to biosphere pollution coming from uncontrolled pollutant discharge from all sorts of industries, especially those manipulating harmful and recalcitrant compounds [1]. Particularly, the dye industries generate huge amounts of hazardous wastewater routinely [2]. Dyes are used for the coloration of several materials such as textile fibers, paper, cosmetics, tannery, leather, food, pharmaceutical products, etc., [3][4]. Before 1856, dyes were derived from natural sources only. The increasing demand and excessive costs of natural dye extraction engendered the discovery of the first synthetic dye aniline (mauveine) in 1856 by a chemist named Perkin [5]. This purple dye gave a stable and uniformly distributed color when applied to silk [6]. Dying industries depended on synthetic dyes ever since and started to expand globally, attaining nearly 8 × 105 tons of synthetic dyes produced per year [7][8]. Notably, the textile industry accounts for ~75% of the global dyestuff market and involves around ten thousand different dyes used for printing and/or coloring multiple types of fabrics [9][10]. Textile industries are mostly located in developing countries such as India, Bangladesh, Sri Lanka and Vietnam, where they enhanced employment capacity, building the economy and foreign exchange earnings [11][12]. However, these countries do not fully respect effluent discharge norms because of their poor wastewater treatment systems. They often reject large quantities of untreated or partially treated dye effluents, eventually resulting in huge environmental pollution [13][14]. Thus, our study aims to give a comprehensive survey on textile synthetic dyes and the discharged effluents of textile industries. It focuses on introducing synthetic dyes and their classification in the first part. It delimits the impacts of these dyes on the ecosystem and human beings in the second part and discusses the available treatment methods of the released textile industry wastewater in the last part.
2. Classification of Dyes
They use dyes in different ways on textile fabric (Figure 1). Bleaching is the removal of dye color (decolorization) from textile fibers, and finishing comprises crosslinking, softening and waterproofing [7]. Natural dyes, which have been known ever since ancient times, are derived mainly from plants; and synthetic dyes are artificially synthesized from chemical compounds. These are cellulose fiber dyes, protein fiber dyes and synthetic fibers dyes [4][15].
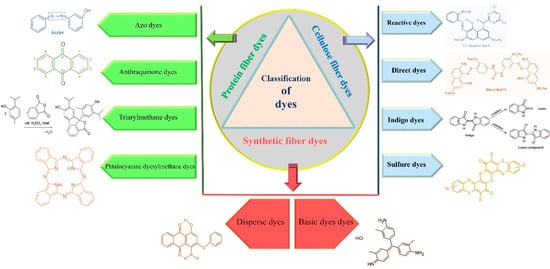
Cellulose fiber originates from plants such as linen, cotton, ramie, rayon, lyocell and hemp (Figure 1). These types of fabrics give perfect dyeing results with reactive dyes, direct dyes, indigo dyes and sulfur dyes [16].
Reactive dyes constitute the major class of cellulose fiber dyes and work well with some protein fibers (Figure 1). They are known for their high pigmentation, permanent effect, facility of manipulation under a wide temperature range and versatility due to diverse reactive groups able to form covalent bonds with multiple fibers [10][14].
Direct dyes are very affordable, yet tend to remain in an aqueous form rather than binding to cellulose fibers (they can be used with certain synthetic fibers as well). Thereby, they are combined with inorganic electrolytes and anionic salts in the form of sodium sulfate (Na2SO4) or sodium chloride (NaCl) to enhance their fabric binding capacities (Figure 1). Thus, it is recommended to wash them in a cold cycle and with fabrics of the same color [17].
The indigo or dark blue color belongs to the classification of vat dyes, which are originally not soluble in water but became soluble after an alkaline reduction (Figure 1). The textile dyeing process occurs with the water-soluble or leuco form of indigo, then this form oxidizes under air exposure and returns to its original insoluble or keto form to ensure a perfect bonding of the dye to the fabric. The indigo dyes are mostly used in blue denim dyeing, which explains their production in huge amounts around the world [18][19].
Sulfur dyes constitute a small, yet important class due to their excellent dyeing properties, ease of application and low cost (Figure 1). They have a complex structure with a disulfide (S–S) bridge. They belong to the vat dye classification; thus, they are reduced from the keto to the leuco form via sodium sulfide utilization. Leuco sulfur becomes soluble in water to achieve the dyeing purpose [20][21].
Protein fibers such as silk, cashmere, angora, mohair and wool originate from animal sources (Figure 1). They are susceptible to high pH levels; hence they are dyed using a water-soluble acid dyestuff to obtain a molecule of an insoluble dye on the fiber [22]. Acid dyes encompass azo dyes as the most important group followed by anthraquinone, triarylmethane and phtalocyanine dyes [23][24].
Azo dyes account for the largest category (60–70%) of the total synthetic dyes industry due to their versatility, cost-effectiveness, simplicity of utilization, high stability and high intensity of the color [25][26]. This dye has a prominent chromophore (-N = N-) structure, ensuring the solubility of the dyes in water and its attachment to the fiber [27][28]. Azo dyes are classified into three groups (mono, di and poly) depending on the number of azo groups in their structure (Figure 1). These groups are attached to an aromatic or heterocyclic compound on one side and an unsaturated heterocycle, carboxyl, sulphonyl, or aliphatic group on the other side [29][30].
The class of anthraquinone is extensively used in textile dyeing industries; the red dyestuff particularly has been used for a long time [31]. These dyes are known for their solubility in water, bright colors and excellent fastness properties (Figure 1). The antharaquinone structure could constitute junctions with azo dyes [32][33].
The triphenylmethane dyes are widely applied in the textile industry for either dyeing wool and silk protein fibers when formed of two groups of sulfonic acid (SO3H). They can be used as indicators if they contain only one sulfonic acid (SO3H) auxochrome in their chemical structure (Figure 1). These dyestuffs are known for their solubility in water and their wide and intense color range [16][34].
The phtalocyanine family of dyes is synthesized by a reaction between the 1,4-Dicyanobenzene compound with a metallic atom (Nickel, Cobalt, Copper, etc.) to produce green and blue shades (Figure 1). They have multiple inherent properties such as good colorfastness to light, resistance to oxidation, solubility in water and chemical stability [35][36].
Synthesized fibers are composed of spandex, polyester, acrylic, polyamide, polyoacetate, polypropylene, ingeo and acetate fabrics (Figure 1). They are used in 60% of global fiber production due to their wide application range. These fibers are dyed using direct dyes, basic dyes and disperse dyes [37][38].
Disperse dyes are the smallest molecules among all dyes. These dyes are insoluble in water but stable under high-temperature exposure (Figure 1). The high-temperature dyeing solution is a mixture between the dyestuff powder and the dispersing agent [39][40][41].
Basic dyes are also called cationic dyes because they transform into colorful cationic salts responsible for dyeing the anionic fiber textile [42]. These dyes are susceptible to light; thus, they are strictly used for dyeing paper nylon and modified polyesters. Their principal structures are cyanine, triarylmethane, anthraquinone, diarylmethane, diazahemicyanine, oxazine, hemicyanine, thiazine and hemicyanine [10] (Figure 1).
3. Characteristics and Impacts of Synthetic Dyes
Synthetic dyes are mostly derived from petrochemical compounds, they are commercialized in liquid, powder, pastes, or granule forms [15]. Therefore, the majority of synthetic dyes cause harmful impacts when discharged in non-treated or partially treated forms in the environment [43][44]. Hence, a huge volume of improper discharge is rejected continuously [45][46]. Dye effluents contain high biological and chemical oxygen demand (BOD and COD) and they are very rich in organic and inorganic pollutants such as chlorinated compounds, heavy metals, sulfur, nitrates, naphtol, soaps, chromium compounds, formaldehyde, benzidine, sequestering agents and dyes and pigments [47][48].
Liquid and solid wastes discharged from textile industries contain dyes, plastic, polyester, fibers, yarns and other hazardous materials as mentioned in the above section (Figure 2). These polymeric compounds have been responsible for the pollution of local landfill habitats and agricultural fields, especially in developing countries. This soil pollution engenders plant growth inhibition by causing oxidative stress, lowering the protein content, photosynthesis and CO2assimilating rates [7][49].
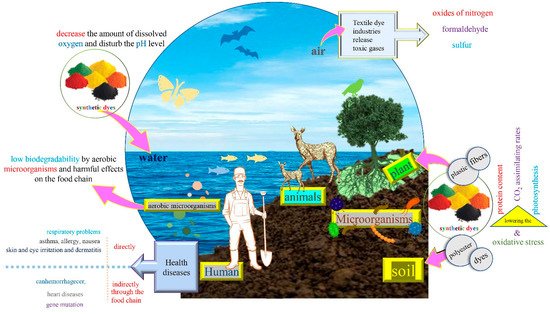
Textile dye industries release toxic gases like sulfur, formaldehyde, oxides of nitrogen, volatile compounds, particulate matter and dusts distinguished by an unpleasant smell (Figure 2). This air pollution could affect humans (workers and customers), animals, the final product and the environment [50][51][52].
The wastewater contains multiple toxic materials and its color result from the discharge of several dyes; it is noticeable and very recalcitrant even in low concentrations ( , yet the average concentration of textile effluent dye reaches about 300 mg/L [53]. These factors lead to several ecological impacts on the aquatic system such as the inhibition of photosynthesis in aquatic plants, low biodegradability by aerobic microorganisms and harmful effects on the food chain [54][55]. Water is highly susceptible to pollution compared to the other areas, and it is also hard to determine the pollution level in aquatic systems.
Dye products and by-products existing in wastewater discharge or the dust produced inside the textile industry pose serious damages and long-lasting health impacts to human beings (Figure 2). They affect several vital organs (brain, kidney, liver, heart) and systems (respiratory, immune, reproductive) of the human organism [56][57]. Diseases may occur either directly through inhalation such as respiratory problems, asthma, allergy, nausea, or skin and eye irritation and dermatitis, or indirectly through the food chain such as tuberculosis, cancer, hemorrhage, gene mutations, and heart disease [2][58].
4. Applied Strategies for Textile Dye Wastewater Treatment
Textile industries use huge amounts of water in the dyeing processes, making it hard to treat the enormous quantities of wastewater discharge (Figure 3). Therefore, several countries have imposed rules to reach a standard before the effluent is released into the ecosystem or is reused for other purposes. For this to happen, physical, chemical and biological technologies have been employed to protect the environment and human health from discharge problems. These strategies could be implemented solely or in combination to obtain effective results [13][29][59].
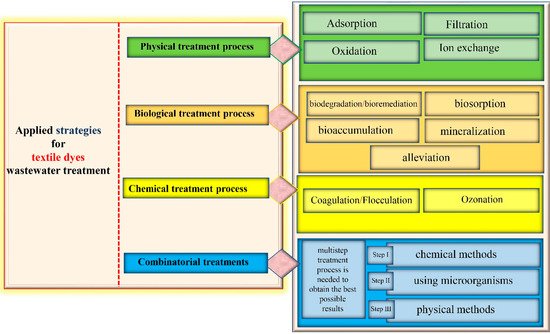
The physical treatment (Figure 3) of industrial wastewater involves conventional processes such as adsorption, filtration, ion exchange and oxidation [15].
The achievement of dyes decolorization by adsorption with activated carbon was extensively studied. For instance, malachite green was adsorbed with curcuma-based activated carbon [60], with tetraethylenepentamine activated carbon [61] and with carbon coated layered double hydroxide [62]. Rhodamine B was successfully adsorbed by ordered mesoporous carbon and commercial activated carbon and by treated rice-husk based activated carbon 4 adsorption occurred via the activated carbon.
Nanotechnology is the science of nanoscale materials (size ≤ 100 nm); it has attracted numerous researchers due to the great potentials of nanoparticles in removing textile dyes [63]. The shape, size, structure, purity and arrangement of nanomaterials matters in the process of decolorization [64]. Nanoparticles are mainly applied directly to wastewater to adsorb rejected dyes on their surface for further elimination [65]. Multiple scientific works covered dye degradation using nanocomposites including graphene oxide/zinc oxide (GO/ZnO)
Filtration is a membrane-based separation process (Figure 3) that involves popular techniques such as reverse osmosis, ultrafiltration and nanofiltration, to allow the acquisition of reusable water and recycled dyes [66][67][68]. The concept of these procedures consists of transporting industrial wastewater throughout several membranes differing by mesh size and separation mechanisms to finally obtain clean water [69][70]. Dye waste water treatment by the membrane filtration method was reported by [71][72][73][74][75][76][77][78][79][80][81][82][83][84][85][86][87][88][89][90][91][92][93][94], he used nano-membrane filtration. [95] coupled electro-fenton reaction with membrane filtration to achieve better results.
Ion-exchange adsorbents are introduced in the wastewater in either solid or liquid form, they are used to bind harmful anions or cations of the opposite charge, and in turn, release an equivalent amount of non-harmful hydrogen ions [96][97]. The review paper of Hassan and Carr [98] addressed strategies for reactive dye removal by applying the ion-exchange method [98][99][100]. Raghu and Basha (2007) combined the ion-exchange with chemical or electrochemical methods to remove dyes from textile wastewater [101]. This technique is great for the elimination of toxic and soluble pollutants (Figure 3) from effluent water; however, its use has been limited because of its high cost [102].
Advanced oxidation processes (AOPs) have been extensively applied for textile dye degradation due to their powerful ability to oxidize a wide range of synthetic dyes and other complex pollutants existing in textile effluents [103][104]. They include catalytic oxidation, which is the process of active radical production (hydroxyl or sulfate radicals) on a particular catalyst surface [105][106]. This reaction occurs in an acid medium (pH ~ 3) to allow the decomposition of the hydrogen peroxide into hydroxyl free radicals acting as strong oxidants [107]. The main drawbacks of the oxidation technique are the generation of hazardous by-products in cases of incomplete oxidation and the possibility of sludge formation [108][109].
The most common chemical treatment processes (Figure 3) are coagulation, flocculation and ozonation [53]; they are utilized for contaminant elimination and in particular, those released in textile wastewater [110].
Coagulation and flocculation (Figure 3) are the simplest chemical methods for the pretreatment of textile dye effluent [111][112]. This technique enables the elimination of suspended insoluble materials by adding charged chemical colloids (Aluminium sulfate (Al2(SO4)3), Iron (III) chloride (FeCl3), Iron (II) sulfate (FeSO4), alum, lime, etc.) provoking coagulation and settling with oppositely charged particles in the polluted water [113]. Acid red 73 was treated by coagulation [114] and
This approach uses ozone as a strong oxidizer, its cleaning properties allow it to eliminate toxic textile effluent compounds such as azo dyes [115][116]. Other benefits of ozone, when used in the gaseous state (Figure 3), are no fluctuation of the volume of wastewater and no generation of sludge [117]. In this respect, the literature has multiple examples on the degradation of dyes including Reactive red 120 [106] The main shortcomings of this process are that it is highly susceptible to many factors (pH, salts, temperature, etc.), it can release toxic compounds and its cost is elevated [113].
This process could also be induced on a laboratory scale by isolating and screening appropriate microorganisms, succeeded by a scale up to allow for textile effluent treatment and decolorization [3][118][119]. It is noteworthy to cite the role of extracellular laccase produced by multiple fungi [120] also mentioned the ability of few filamentous fungi in the treatment of crystal violet and methylene blue dyes [71][72][73][74][75][76][77][78][79][80][81][82][83][84][85][86][87][88][89][90][91][92][93][94][95][121][98][99][100][122][123][101][124][125][126][127][128][129][130][131][132][133][134][135][136][137][138][139][140][141][142][143][144][120][145][146][147][148][149][150][151]. proved to be useful in azo dyes degradation [120][152].Lysinibacillussp.
All the above-mentioned treatments proved effective in alleviating textile wastewater toxicity (Figure 3). It combines several treatment methods, starting with chemical methods to allow for the elimination of solid contaminants [16][153][154]. The secondary treatment is achieved using microorganisms to reduce the COD and BOD rates, to remove turbidity and to convert the generated sludge from the primary treatment into non-harmful products [155]. In the tertiary treatment, physical methods are applied to ensure the total decontamination of the textile wastewater and its safe reuse or release in the environment [156].
References
- Bharagava, R.N.; Saxena, G.; Mulla, S.I. Introduction to industrial wastes containing organic and inorganic pollutants and bioremediation approaches for environmental management. In Bioremediation of Industrial Waste for Environmental Safety; Springer: New York, NY, USA, 2020; pp. 1–18.
- Khan, S.; Malik, A. Toxicity Evaluation of Textile Effluents and Role of Native Soil Bacterium in Biodegradation of a Textile Dye. Environ. Sci. Pollut. Res. 2018, 25, 4446–4458.
- Celia, M.P.; Suruthi, S. Textile Dye Degradation Using Bacterial Strains Isolated from Textile Mill Effluent. Int. J. Appl. Res. 2016, 2, 337–341.
- Shindhal, T.; Rakholiya, P.; Varjani, S.; Pandey, A.; Ngo, H.H.; Guo, W.; Ng, H.Y.; Taherzadeh, M.J. A Critical Review on Advances in the Practices and Perspectives for the Treatment of Dye Industry Wastewater. Bioengineered 2021, 12, 70–87.
- Druding, S.C. (Ed.) Dye History from 2600 BC to the 20th Century. 1982. Available online: http://www.straw.com/sig/dyehist.html (accessed on 1 June 2021).
- Singh, L.; Singh, V.P. Textile dyes degradation: A microbial approach for biodegradation of pollutants. In Microbial Degradation of Synthetic Dyes in Wastewaters; Springer: New York, NY, USA, 2015; pp. 187–204.
- Bhatia, D.; Sharma, N.R.; Singh, J.; Kanwar, R.S. Biological Methods for Textile Dye Removal from Wastewater: A Review. Crit. Rev. Environ. Sci. Technol. 2017, 47, 1836–1876.
- Jamee, R.; Siddique, R. Biodegradation of Synthetic Dyes of Textile Effluent by Microorganisms: An Environmentally and Economically Sustainable Approach. Eur. J. Microbiol. Immunol. 2019, 9, 114–118.
- Madhav, S.; Ahamad, A.; Singh, P.; Mishra, P.K. A Review of Textile Industry: Wet Processing, Environmental Impacts, and Effluent Treatment Methods. Environ. Qual. Manag. 2018, 27, 31–41.
- Thakur, S.; Chauhan, M.S. Treatment of Dye Wastewater from Textile Industry by Electrocoagulation and Fenton Oxidation: A Review. In Proceedings of the Water Quality Management; Singh, V.P., Yadav, S., Yadava, R.N., Eds.; Springer: Singapore, 2018; pp. 117–129.
- Desore, A.; Narula, S.A. An Overview on Corporate Response towards Sustainability Issues in Textile Industry. Environ. Dev. Sustain. 2018, 20, 1439–1459.
- Lellis, B.; Fávaro-Polonio, C.Z.; Pamphile, J.A.; Polonio, J.C. Effects of Textile Dyes on Health and the Environment and Bioremediation Potential of Living Organisms. Biotechnol. Res. Innov. 2019, 3, 275–290.
- Hossen, M.Z.; Hussain, M.E.; Hakim, A.; Islam, K.; Uddin, M.N.; Azad, A.K. Biodegradation of Reactive Textile Dye Novacron Super Black G by Free Cells of Newly Isolated Alcaligenes Faecalis AZ26 and Bacillus Spp Obtained from Textile Effluents. Heliyon 2019, 5, e02068.
- Barathi, S.; Karthik, C.; Nadanasabapathi, S.; Padikasan, I.A. Biodegradation of Textile Dye Reactive Blue 160 by Bacillus Firmus (Bacillaceae: Bacillales) and Non-Target Toxicity Screening of Their Degraded Products. Toxicol. Rep. 2020, 7, 16–22.
- Gita, S.; Hussan, A.; Choudhury, T.G. Impact of Textile Dyes Waste on Aquatic Environments and Its Treatment. Environ. Ecol. 2017, 35, 2349–2353.
- Ghaly, A.E.; Ananthashankar, R.; Alhattab, M.; Ramakrishnan, V.V. Production, Characterization and Treatment of Textile Effluents: A Critical Review. J. Chem. Eng. Process Technol. 2014, 5, 1–18.
- Burkinshaw, S.M.; Salihu, G. The Role of Auxiliaries in the Immersion Dyeing of Textile Fibres: Part 5 Practical Aspects of the Role of Inorganic Electrolytes in Dyeing Cellulosic Fibres with Direct Dyes. Dyes Pigment. 2019, 161, 581–594.
- Paz, A.; Carballo, J.; Pérez, M.J.; Domínguez, J.M. Biological Treatment of Model Dyes and Textile Wastewaters. Chemosphere 2017, 181, 168–177.
- Chowdhury, M.F.; Khandaker, S.; Sarker, F.; Islam, A.; Rahman, M.T.; Awual, M.R. Current Treatment Technologies and Mechanisms for Removal of Indigo Carmine Dyes from Wastewater: A Review. J. Mol. Liq. 2020, 318, 114061.
- Nguyen, T.A.; Juang, R.S. Treatment of Waters and Wastewaters Containing Sulfur Dyes: A Review. Chem. Eng. J. 2013, 219, 109–117.
- Khattab, T.A.; Abdelrahman, M.S.; Rehan, M. Textile Dyeing Industry: Environmental Impacts and Remediation. Environ. Sci. Pollut. Res. 2020, 27, 3803–3818.
- dos Santos Pisoni, D.; de Abreu, M.P.; Petzhold, C.L.; Rodembusch, F.S.; Campo, L.F. Synthesis, Photophysical Study and BSA Association of Water-Insoluble Squaraine Dyes. J. Photochem. Photobiol. A Chem. 2013, 252, 77–83.
- Wan, Z.; Li, D.; Jiao, Y.; Ouyang, X.; Chang, L.; Wang, X. Bifunctional MoS2 Coated Melamine-Formaldehyde Sponges for Efficient Oil–Water Separation and Water-Soluble Dye Removal. Appl. Mater. Today 2017, 9, 551–559.
- Salem, M.Z.; Ibrahim, I.H.; Ali, H.M.; Helmy, H.M. Assessment of the Use of Natural Extracted Dyes and Pancreatin Enzyme for Dyeing of Four Natural Textiles: HPLC Analysis of Phytochemicals. Processes 2020, 8, 59.
- Li, W.; Mu, B.; Yang, Y. Feasibility of Industrial-Scale Treatment of Dye Wastewater via Bio-Adsorption Technology. Bioresour. Technol. 2019, 277, 157–170.
- Zhang, L.; Shao, Q.; Xu, C. Enhanced Azo Dye Removal from Wastewater by Coupling Sulfidated Zero-Valent Iron with a Chelator. J. Clean. Prod. 2019, 213, 753–761.
- Singh, R.L.; Singh, P.K.; Singh, R.P. Enzymatic Decolorization and Degradation of Azo Dyes—A Review. Int. Biodeterior. Biodegrad. 2015, 104, 21–31.
- Louati, I.; Elloumi-Mseddi, J.; Cheikhrouhou, W.; Hadrich, B.; Nasri, M.; Aifa, S.; Woodward, S.; Mechichi, T. Simultaneous Cleanup of Reactive Black 5 and Cadmium by a Desert Soil Bacterium. Ecotoxicol. Environ. Saf. 2020, 190, 110103.
- Al-Tohamy, R.; Sun, J.; Fareed, M.F.; Kenawy, E.R.; Ali, S.S. Ecofriendly Biodegradation of Reactive Black 5 by Newly Isolated Sterigmatomyces Halophilus SSA1575, Valued for Textile Azo Dye Wastewater Processing and Detoxification. Sci. Rep. 2020, 10, 12370.
- Liu, S.H.; Tsai, S.L.; Guo, P.Y.; Lin, C.W. Inducing Laccase Activity in White Rot Fungi Using Copper Ions and Improving the Efficiency of Azo Dye Treatment with Electricity Generation Using Microbial Fuel Cells. Chemosphere 2020, 243, 125304.
- Shahid, M.; Wertz, J.; Degano, I.; Aceto, M.; Khan, M.I.; Quye, A. Analytical Methods for Determination of Anthraquinone Dyes in Historical Textiles: A Review. Anal. Chim. Acta 2019, 1083, 58–87.
- Novotnỳ, Č.; Dias, N.; Kapanen, A.; Malachová, K.; Vándrovcová, M.; Itävaara, M.; Lima, N. Comparative Use of Bacterial, Algal and Protozoan Tests to Study Toxicity of Azo-and Anthraquinone Dyes. Chemosphere 2006, 63, 1436–1442.
- Gürses, A.; Açıkyıldız, M.; Güneş, K.; Gürses, M.S. Classification of Dye and Pigments. In Dyes and Pigments; Gürses, A., Açıkyıldız, M., Güneş, K., Gürses, M.S., Eds.; Springer Briefs in Molecular Science; Springer International Publishing: Cham, Switzerland, 2016; pp. 31–45. ISBN 978-3-319-33892-7.
- Cao, D.J.; Wang, J.J.; Zhang, Q.; Wen, Y.Z.; Dong, B.; Liu, R.J.; Yang, X.; Geng, G. Biodegradation of Triphenylmethane Dye Crystal Violet by Cedecea Davisae. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 210, 9–13.
- Silva, M.C.; Corrêa, A.D.; Amorim, M.T.; Parpot, P.; Torres, J.A.; Chagas, P.M. Decolorization of the Phthalocyanine Dye Reactive Blue 21 by Turnip Peroxidase and Assessment of Its Oxidation Products. J. Mol. Catal. B Enzym. 2012, 77, 9–14.
- Dindaş, G.B.; Şahin, Z.; Cengiz Yatmaz, H.; İşci, Ü. Cobalt Phthalocyanine-TiO2 Nanocomposites for Photocatalytic Remediation of Textile Dyes under Visible Light Irradiation. J. Porphyr. Phthalocyanines 2019, 23, 561–568.
- Cesa, F.S.; Turra, A.; Baruque-Ramos, J. Synthetic Fibers as Microplastics in the Marine Environment: A Review from Textile Perspective with a Focus on Domestic Washings. Sci. Total Environ. 2017, 598, 1116–1129.
- Almroth, B.M.; Åström, L.; Roslund, S.; Petersson, H.; Johansson, M.; Persson, N.K. Quantifying Shedding of Synthetic Fibers from Textiles; a Source of Microplastics Released into the Environment. Environ. Sci. Pollut. Res. 2018, 25, 1191–1199.
- Meireles, G.; Daam, M.A.; Sanches, A.L.; Zanoni, M.V.; Soares, A.M.; Gravato, C.; de Oliveira, D.P. Red Disperse Dyes (DR 60, DR 73 and DR 78) at Environmentally Realistic Concentrations Impact Biochemical Profile of Early Life Stages of Zebrafish (Danio Rerio). Chem.-Biol. Interact. 2018, 292, 94–100.
- Al-Etaibi, A.M.; El-Apasery, M.A. Dyeing Performance of Disperse Dyes on Polyester Fabrics Using Eco-Friendly Carrier and Their Antioxidant and Anticancer Activities. Int. J. Environ. Res. Public Health 2019, 16, 4603.
- Bayramoglu, G.; Kunduzcu, G.; Arica, M.Y. Preparation and Characterization of Strong Cation Exchange Terpolymer Resin as Effective Adsorbent for Removal of Disperse Dyes. Polym. Eng. Sci. 2020, 60, 192–201.
- Berradi, M.; Hsissou, R.; Khudhair, M.; Assouag, M.; Cherkaoui, O.; El Bachiri, A.; El Harfi, A. Textile Finishing Dyes and Their Impact on Aquatic Environs. Heliyon 2019, 5, e02711.
- Ito, T.; Adachi, Y.; Yamanashi, Y.; Shimada, Y. Long–Term Natural Remediation Process in Textile Dye–Polluted River Sediment Driven by Bacterial Community Changes. Water Res. 2016, 100, 458–465.
- Singh, L. Biodegradation of Synthetic Dyes: A Mycoremediation Approach for Degradation/Decolourization of Textile Dyes and Effluents. J. Appl. Biotechnol. Bioeng. 2017, 3, 430–435.
- Yang, C.; Li, L.; Shi, J.; Long, C.; Li, A. Advanced Treatment of Textile Dyeing Secondary Effluent Using Magnetic Anion Exchange Resin and Its Effect on Organic Fouling in Subsequent RO Membrane. J. Hazard. Mater. 2015, 284, 50–57.
- Yaseen, D.A.; Scholz, M. Shallow Pond Systems Planted with Lemna Minor Treating Azo Dyes. Ecol. Eng. 2016, 94, 295–305.
- Gürses, A.; Açıkyıldız, M.; Güneş, K.; Gürses, M.S. Colorants in health and environmental aspects. In Dyes and Pigments; Springer: New York, NY, USA, 2016; pp. 69–83.
- Donkadokula, N.Y.; Kola, A.K.; Naz, I.; Saroj, D. A Review on Advanced Physico-Chemical and Biological Textile Dye Wastewater Treatment Techniques. Rev. Environ. Sci. Biotechnol. 2020, 19, 543–560.
- Vikrant, K.; Giri, B.S.; Raza, N.; Roy, K.; Kim, K.H.; Rai, B.N.; Singh, R.S. Recent Advancements in Bioremediation of Dye: Current Status and Challenges. Bioresour. Technol. 2018, 253, 355–367.
- Aldalbahi, A.; El-Naggar, M.E.; El-Newehy, M.H.; Rahaman, M.; Hatshan, M.R.; Khattab, T.A. Effects of Technical Textiles and Synthetic Nanofibers on Environmental Pollution. Polymers 2021, 13, 155.
- Park, M.; Lee, K.S.; Shim, J.; Liu, Y.; Lee, C.; Cho, H.; Kim, M.J.; Park, S.J.; Yun, Y.J.; Kim, H.Y.; et al. Environment Friendly, Transparent Nanofiber Textiles Consolidated with High Efficiency PLEDs for Wearable Electronics. Org. Electron. 2016, 36, 89–96.
- Muthu, S.S. Introduction. In Sustainability in the Textile Industry; Muthu, S.S., Ed.; Textile Science and Clothing Technology; Springer: Singapore, 2017; pp. 1–8. ISBN 978-981-10-2639-3.
- Eslami, H.; Shariatifar, A.; Rafiee, E.; Shiranian, M.; Salehi, F.; Hosseini, S.S.; Eslami, G.; Ghanbari, R.; Ebrahimi, A.A. Decolorization and Biodegradation of Reactive Red 198 Azo Dye by a New Enterococcus Faecalis–Klebsiella Variicola Bacterial Consortium Isolated from Textile Wastewater Sludge. World J. Microbiol. Biotechnol. 2019, 35, 38.
- Imran, M.; Crowley, D.E.; Khalid, A.; Hussain, S.; Mumtaz, M.W.; Arshad, M. Microbial Biotechnology for Decolorization of Textile Wastewaters. Rev. Environ. Sci. Biotechnol. 2015, 14, 73–92.
- Sakib, A.A.; Masum, S.M.; Hoinkis, J.; Islam, R.; Molla, M.; Islam, A. Synthesis of CuO/ZnO Nanocomposites and Their Application in Photodegradation of Toxic Textile Dye. J. Compos. Sci. 2019, 3, 91.
- Kant, R. Textile Dyeing Industry an Environmental Hazard. Nat. Sci. 2011, 4, 17027.
- Rovira, J.; Domingo, J.L. Human Health Risks Due to Exposure to Inorganic and Organic Chemicals from Textiles: A Review. Environ. Res. 2019, 168, 62–69.
- Yadav, A.K.; Jain, C.K.; Malik, D.S. Toxic Characterization of Textile Dyes and Effluents in Relation to Human Health Hazards. J. Sustain. Environ. Res. 2014, 3, 95–102.
- Dai, Q.; Zhang, S.; Liu, H.; Huang, J.; Li, L. Sulfide-Mediated Azo Dye Degradation and Microbial Community Analysis in a Single-Chamber Air Cathode Microbial Fuel Cell. Bioelectrochemistry 2020, 131, 107349.
- Arora, C.; Kumar, P.; Soni, S.; Mittal, J.; Mittal, A.; Singh, B. Efficient Removal of Malachite Green Dye from Aqueous Solution Using Curcuma Caesia Based Activated Carbon. Desalination Water Treat. 2020, 195, 341–352.
- Ghasemi, M.; Mashhadi, S.; Asif, M.; Tyagi, I.; Agarwal, S.; Gupta, V.K. Microwave-Assisted Synthesis of Tetraethylenepentamine Functionalized Activated Carbon with High Adsorption Capacity for Malachite Green Dye. J. Mol. Liq. 2016, 213, 317–325.
- George, G.; Saravanakumar, M.P. Facile Synthesis of Carbon-Coated Layered Double Hydroxide and Its Comparative Characterisation with Zn–Al LDH: Application on Crystal Violet and Malachite Green Dye Adsorption—Isotherm, Kinetics and Box-Behnken Design. Environ. Sci. Pollut. Res. 2018, 25, 30236–30254.
- Chen, C.Y.; Kuo, J.T.; Cheng, C.Y.; Huang, Y.T.; Ho, I.H.; Chung, Y.C. Biological Decolorization of Dye Solution Containing Malachite Green by Pandoraea Pulmonicola YC32 Using a Batch and Continuous System. J. Hazard. Mater. 2009, 172, 1439–1445.
- Muthukumaran, C.; Sivakumar, V.M.; Thirumarimurugan, M. Adsorption Isotherms and Kinetic Studies of Crystal Violet Dye Removal from Aqueous Solution Using Surfactant Modified Magnetic Nanoadsorbent. J. Taiwan Inst. Chem. Eng. 2016, 63, 354–362.
- Fabryanty, R.; Valencia, C.; Soetaredjo, F.E.; Putro, J.N.; Santoso, S.P.; Kurniawan, A.; Ju, Y.H.; Ismadji, S. Removal of Crystal Violet Dye by Adsorption Using Bentonite–Alginate Composite. J. Environ. Chem. Eng. 2017, 5, 5677–5687.
- Lops, C.; Ancona, A.; Di Cesare, K.; Dumontel, B.; Garino, N.; Canavese, G.; Hérnandez, S.; Cauda, V. Sonophotocatalytic Degradation Mechanisms of Rhodamine B Dye via Radicals Generation by Micro-and Nano-Particles of ZnO. Appl. Catal. B Environ. 2019, 243, 629–640.
- Ding, L.; Zou, B.; Gao, W.; Liu, Q.; Wang, Z.; Guo, Y.; Wang, X.; Liu, Y. Adsorption of Rhodamine-B from Aqueous Solution Using Treated Rice Husk-Based Activated Carbon. Colloids Surf. A Physicochem. Eng. Asp. 2014, 446, 1–7.
- Machado, Ê.L.; de Sales Dambros, V.; Kist, L.T.; Lobo, E.A.; Tedesco, S.B.; Moro, C.C. Use of Ozonization for the Treatment of Dye Wastewaters Containing Rhodamine B in the Agate Industry. Water Air Soil Pollut. 2012, 223, 1753–1764.
- Khan, S.S.; Arunarani, A.; Chandran, P. Biodegradation of Basic Violet 3 and Acid Blue 93 by Pseudomonas Putida. CLEAN–Soil Air Water 2015, 43, 67–72.
- Deivasigamani, C.; Das, N. Biodegradation of Basic Violet 3 by Candida Krusei Isolated from Textile Wastewater. Biodegradation 2011, 22, 1169–1180.
- Galán, J.; Rodríguez, A.; Gómez, J.M.; Allen, S.J.; Walker, G.M. Reactive Dye Adsorption onto a Novel Mesoporous Carbon. Chem. Eng. J. 2013, 219, 62–68.
- Holkar, C.R.; Jadhav, A.J.; Pinjari, D.V.; Mahamuni, N.M.; Pandit, A.B. A Critical Review on Textile Wastewater Treatments: Possible Approaches. J. Environ. Manag. 2016, 182, 351–366.
- Rápó, E.; Aradi, L.E.; Szabó, Á.; Posta, K.; Szép, R.; Tonk, S. Adsorption of Remazol Brilliant Violet-5R Textile Dye from Aqueous Solutions by Using Eggshell Waste Biosorbent. Sci. Rep. 2020, 10, 8385.
- El Malah, T.; Nour, H.F.; Radwan, E.K.; Abdel Mageid, R.E.; Khattab, T.A.; Olson, M.A. A Bipyridinium-Based Polyhydrazone Adsorbent That Exhibits Ultrahigh Adsorption Capacity for the Anionic Azo Dye, Direct Blue 71. Chem. Eng. J. 2021, 409, 128195.
- Ruan, W.; Hu, J.; Qi, J.; Hou, Y.; Zhou, C.; Wei, X. Removal Of Dyes From Wastewater By Nanomaterials: A Review. Adv. Mater. Lett. 2019, 10, 9–20.
- Yogalakshmi, K.N.; Das, A.; Rani, G.; Jaswal, V.; Randhawa, J.S. Nano-bioremediation: A new age technology for the treatment of dyes in textile effluents. In Bioremediation of Industrial Waste for Environmental Safety; Springer: New York, NY, USA, 2020; pp. 313–347.
- Dhand, C.; Dwivedi, N.; Loh, X.J.; Ying, A.N.; Verma, N.K.; Beuerman, R.W.; Lakshminarayanan, R.; Ramakrishna, S. Methods and Strategies for the Synthesis of Diverse Nanoparticles and Their Applications: A Comprehensive Overview. Rsc Adv. 2015, 5, 105003–105037.
- Krishnan, S.K.; Subbiah, K.; Kandasamy, S.; Subramaniam, K. Application of Metal Nanoparticles for Textile Dye Remediation. In Proceedings of the Sustainable Development in Energy and Environment; Sivasubramanian, V., Pugazhendhi, A., Moorthy, I.G., Eds.; Springer: Singapore, 2020; pp. 217–223.
- Tara, N.; Siddiqui, S.I.; Rathi, G.; Chaudhry, S.A.; Asiri, A.M. Nano-Engineered Adsorbent for the Removal of Dyes from Water: A Review. Curr. Anal. Chem. 2020, 16, 14–40.
- Ali, Z.; Ahmad, R. Nanotechnology for Water Treatment. In Environmental Nanotechnology Volume 3; Dasgupta, N., Ranjan, S., Lichtfouse, E., Eds.; Environmental Chemistry for a Sustainable World; Springer International Publishing: Cham, Switzerland, 2020; pp. 143–163. ISBN 978-3-030-26672-1.
- Sadegh, H.; Ali, G.A.; Gupta, V.K.; Makhlouf, A.S.; Shahryari-Ghoshekandi, R.; Nadagouda, M.N.; Sillanpää, M.; Megiel, E. The Role of Nanomaterials as Effective Adsorbents and Their Applications in Wastewater Treatment. J. Nanostruct. Chem. 2017, 7, 1–14.
- Wang, T.; Lin, J.; Chen, Z.; Megharaj, M.; Naidu, R. Green Synthesized Iron Nanoparticles by Green Tea and Eucalyptus Leaves Extracts Used for Removal of Nitrate in Aqueous Solution. J. Clean. Prod. 2014, 83, 413–419.
- Nagajyothi, P.C.; Vattikuti, S.V.; Devarayapalli, K.C.; Yoo, K.; Shim, J.; Sreekanth, T.V. Green Synthesis: Photocatalytic Degradation of Textile Dyes Using Metal and Metal Oxide Nanoparticles-Latest Trends and Advancements. Crit. Rev. Environ. Sci. Technol. 2020, 50, 2617–2723.
- Pal, U.; Sandoval, A.; Madrid, S.I.; Corro, G.; Sharma, V.; Mohanty, P. Mixed Titanium, Silicon, and Aluminum Oxide Nanostructures as Novel Adsorbent for Removal of Rhodamine 6G and Methylene Blue as Cationic Dyes from Aqueous Solution. Chemosphere 2016, 163, 142–152.
- Kalia, A.; Singh, S. Myco-Decontamination of Azo Dyes: Nano-Augmentation Technologies. 3 Biotech 2020, 10, 384.
- Sahoo, T.; Sahu, J.R.; Panda, J.; Hembram, M.; Sahoo, S.K.; Sahu, R. Nanotechnology: An Efficient Technique of Contaminated Water Treatment. In Contaminants in Drinking and Wastewater Sources: Challenges and Reigning Technologies; Kumar, M., Snow, D.D., Honda, R., Mukherjee, S., Eds.; Springer Transactions in Civil and Environmental Engineering; Springer: Singapore, 2021; pp. 251–270. ISBN 9789811545993.
- Gupta, M.K.; Tandon, P.K.; Shukla, N. Nanotechnology: Environmentally Sustainable Solutions for Water Treatment. In Nanostructured Materials for Treating Aquatic Pollution; Springer: New York, NY, USA, 2019; pp. 225–242.
- Chu, T.P.; Nguyen, N.T.; Vu, T.L.; Dao, T.H.; Dinh, L.C.; Nguyen, H.L.; Hoang, T.H.; Le, T.S.; Pham, T.D. Synthesis, Characterization, and Modification of Alumina Nanoparticles for Cationic Dye Removal. Materials 2019, 12, 450.
- Sahinkaya, E.; Sahin, A.; Yurtsever, A.; Kitis, M. Concentrate Minimization and Water Recovery Enhancement Using Pellet Precipitator in a Reverse Osmosis Process Treating Textile Wastewater. J. Environ. Manag. 2018, 222, 420–427.
- Long, Q.; Zhang, Z.; Qi, G.; Wang, Z.; Chen, Y.; Liu, Z.Q. Fabrication of Chitosan Nanofiltration Membranes by the Film Casting Strategy for Effective Removal of Dyes/Salts in Textile Wastewater. ACS Sustain. Chem. Eng. 2020, 8, 2512–2522.
- Jin, P.; Zhu, J.; Yuan, S.; Zhang, G.; Volodine, A.; Tian, M.; Wang, J.; Luis, P.; Van der Bruggen, B. Erythritol-Based Polyester Loose Nanofiltration Membrane with Fast Water Transport for Efficient Dye/Salt Separation. Chem. Eng. J. 2021, 406, 126796.
- Yin, H.; Qiu, P.; Qian, Y.; Kong, Z.; Zheng, X.; Tang, Z.; Guo, H. Textile Wastewater Treatment for Water Reuse: A Case Study. Processes 2019, 7, 34.
- Ji, J.; Kulshreshtha, S.; Kakade, A.; Majeed, S.; Li, X.; Liu, P. Bioaugmentation of Membrane Bioreactor with Aeromonas Hydrophila LZ-MG14 for Enhanced Malachite Green and Hexavalent Chromium Removal in Textile Wastewater. Int. Biodeterior. Biodegrad. 2020, 150, 104939.
- Rashidi, H.R.; Sulaiman, N.M.; Hashim, N.A.; Hassan, C.R.; Ramli, M.R. Synthetic Reactive Dye Wastewater Treatment by Using Nano-Membrane Filtration. Desalination Water Treat. 2015, 55, 86–95.
- Liang, P.; Rivallin, M.; Cerneaux, S.; Lacour, S.; Petit, E.; Cretin, M. Coupling Cathodic Electro-Fenton Reaction to Membrane Filtration for AO7 Dye Degradation: A Successful Feasibility Study. J. Membr. Sci. 2016, 510, 182–190.
- Satapanajaru, T.; Chompuchan, C.; Suntornchot, P.; Pengthamkeerati, P. Enhancing Decolorization of Reactive Black 5 and Reactive Red 198 during Nano Zerovalent Iron Treatment. Desalination 2011, 266, 218–230.
- Nazari, P.; Setayesh, S.R. Effective Degradation of Reactive Red 195 via Heterogeneous Electro-Fenton Treatment: Theoretical Study and Optimization. Int. J. Environ. Sci. Technol. 2019, 16, 6329–6346.
- Hassan, M.M.; Carr, C.M. A Critical Review on Recent Advancements of the Removal of Reactive Dyes from Dyehouse Effluent by Ion-Exchange Adsorbents. Chemosphere 2018, 209, 201–219.
- Yurtsever, A.; Basaran, E.; Uçar, D. Process Optimization and Filtration Performance of an Anaerobic Dynamic Membrane Bioreactor Treating Textile Wastewaters. J. Environ. Manag. 2020, 273, 111114.
- Arslan, S.; Eyvaz, M.; Gürbulak, E.; Yüksel, E. A Review of State-of-the-Art Technologies in Dye-Containing Wastewater Treatment—The Textile Industry Case. Text. Wastewater Treat. 2016.
- Raghu, S.; Basha, C.A. Chemical or Electrochemical Techniques, Followed by Ion Exchange, for Recycle of Textile Dye Wastewater. J. Hazard. Mater. 2007, 149, 324–330.
- Sonai, G.G.; de Souza, S.M.; de Oliveira, D.; de Souza, A.A. The Application of Textile Sludge Adsorbents for the Removal of Reactive Red 2 Dye. J. Environ. Manag. 2016, 168, 149–156.
- Kalyani, D.C.; Telke, A.A.; Dhanve, R.S.; Jadhav, J.P. Ecofriendly Biodegradation and Detoxification of Reactive Red 2 Textile Dye by Newly Isolated Pseudomonas Sp. SUK1. J. Hazard. Mater. 2009, 163, 735–742.
- Wang, H.; Su, J.Q.; Zheng, X.W.; Tian, Y.; Xiong, X.J.; Zheng, T.L. Bacterial Decolorization and Degradation of the Reactive Dye Reactive Red 180 by Citrobacter Sp. CK3. Int. Biodeterior. Biodegrad. 2009, 63, 395–399.
- Moussavi, G.; Mahmoudi, M. Degradation and Biodegradability Improvement of the Reactive Red 198 Azo Dye Using Catalytic Ozonation with MgO Nanocrystals. Chem. Eng. J. 2009, 152, 1–7.
- Zhang, F.; Yediler, A.; Liang, X.; Kettrup, A. Effects of Dye Additives on the Ozonation Process and Oxidation By-Products: A Comparative Study Using Hydrolyzed CI Reactive Red 120. Dyes Pigments 2004, 60, 1–7.
- Saratale, R.G.; Saratale, G.D.; Chang, J.S.; Govindwar, S.P. Ecofriendly Degradation of Sulfonated Diazo Dye CI Reactive Green 19A Using Micrococcus Glutamicus NCIM-2168. Bioresour. Technol. 2009, 100, 3897–3905.
- Zuorro, A.; Lavecchia, R. Evaluation of UV/H2O2 Advanced Oxidation Process (AOP) for the Degradation of Diazo Dye Reactive Green 19 in Aqueous Solution. Desalination Water Treat. 2014, 52, 1571–1577.
- Palma-Goyes, R.E.; Silva-Agredo, J.; Vazquez-Arenas, J.; Romero-Ibarra, I.; Torres-Palma, R.A. The Effect of Different Operational Parameters on the Electrooxidation of Indigo Carmine on Ti/IrO2-SnO2-Sb2O3. J. Environ. Chem. Eng. 2018, 6, 3010–3017.
- Labiadh, L.; Barbucci, A.; Carpanese, M.P.; Gadri, A.; Ammar, S.; Panizza, M. Direct and Indirect Electrochemical Oxidation of Indigo Carmine Using PbO2 and TiRuSnO2. J. Solid State Electrochem. 2017, 21, 2167–2175.
- Ramesh, T.N.; Kirana, D.V.; Ashwini, A.; Manasa, T.R. Calcium Hydroxide as Low Cost Adsorbent for the Effective Removal of Indigo Carmine Dye in Water. J. Saudi Chem. Soc. 2017, 21, 165–171.
- Zapata-Castillo, P.; Villalonga-Santana, L.; Islas-Flores, I.; Rivera-Muñoz, G.; Ancona-Escalante, W.; Solís-Pereira, S. Synergistic Action of Laccases from Trametes Hirsuta Bm2 Improves Decolourization of Indigo Carmine. Lett. Appl. Microbiol. 2015, 61, 252–258.
- Li, H.; Zhang, R.; Tang, L.; Zhang, J.; Mao, Z. Manganese Peroxidase Production from Cassava Residue by Phanerochaete Chrysosporium in Solid State Fermentation and Its Decolorization of Indigo Carmine. Chin. J. Chem. Eng. 2015, 23, 227–233.
- Jorfi, S.; Barzegar, G.; Ahmadi, M.; Soltani, R.D.; Takdastan, A.; Saeedi, R.; Abtahi, M. Enhanced Coagulation-Photocatalytic Treatment of Acid Red 73 Dye and Real Textile Wastewater Using UVA/Synthesized MgO Nanoparticles. J. Environ. Manag. 2016, 177, 111–118.
- Lu, R.; Ma, L.; He, F.; Yu, D.; Fan, R.; Zhang, Y.; Long, Z.; Zhang, X.; Yang, Y. White-Rot Fungus Ganoderma Sp. En3 Had a Strong Ability to Decolorize and Tolerate the Anthraquinone, Indigo and Triphenylmethane Dye with High Concentrations. Bioprocess Biosyst. Eng. 2016, 39, 381–390.
- Adnan, L.A.; Hadibarata, T.; Sathishkumar, P.; Mohd Yusoff, A.R. Biodegradation Pathway of Acid Red 27 by White-Rot Fungus Armillaria Sp. F022 and Phytotoxicity Evaluation. CLEAN–Soil Air Water 2016, 44, 239–246.
- Jocic, D.; Vílchez, S.; Topalovic, T.; Molina, R.; Navarro, A.; Jovancic, P.; Julià, M.R.; Erra, P. Effect of Low-Temperature Plasma and Chitosan Treatment on Wool Dyeing with Acid Red 27. J. Appl. Polym. Sci. 2005, 97, 2204–2214.
- Bhole, B.D.; Ganguly, B.; Madhuram, A.; Deshpande, D.; Joshi, J. Biosorption of Methyl Violet, Basic Fuchsin and Their Mixture Using Dead Fungal Biomass. Curr. Sci. 2004, 86, 1641–1645.
- Durmus, Z.; Kurt, B.Z.; Durmus, A. Synthesis and Characterization of Graphene Oxide/Zinc Oxide (GO/ZnO) Nanocomposite and Its Utilization for Photocatalytic Degradation of Basic Fuchsin Dye. ChemistrySelect 2019, 4, 271–278.
- AI-Jawhari, I.F. Decolorization of Methylene Blue and Crystal Violet by Some Filamentous Fungi. Int. J. Environ. Bioremediat. Biodegrad. 2015, 3, 62–65.
- Liu, H.; Zhang, J.; Lu, M.; Liang, L.; Zhang, H.; Wei, J. Biosynthesis Based Membrane Filtration Coupled with Iron Nanoparticles Reduction Process in Removal of Dyes. Chem. Eng. J. 2020, 387, 124202.
- Marin, N.M.; Pascu, L.F.; Demba, A.; Nita-Lazar, M.; Badea, I.A.; Aboul-Enein, H.Y. Removal of the Acid Orange 10 by Ion Exchange and Microbiological Methods. Int. J. Environ. Sci. Technol. 2019, 16, 6357–6366.
- Joseph, J.; Radhakrishnan, R.C.; Johnson, J.K.; Joy, S.P.; Thomas, J. Ion-Exchange Mediated Removal of Cationic Dye-Stuffs from Water Using Ammonium Phosphomolybdate. Mater. Chem. Phys. 2020, 242, 122488.
- Cseri, L.; Topuz, F.; Abdulhamid, M.A.; Alammar, A.; Budd, P.M.; Szekely, G. Electrospun Adsorptive Nanofibrous Membranes from Ion Exchange Polymers to Snare Textile Dyes from Wastewater. Adv. Mater. Technol. 2021, 2000955.
- Babu, D.S.; Srivastava, V.; Nidheesh, P.V.; Kumar, M.S. Detoxification of Water and Wastewater by Advanced Oxidation Processes. Sci. Total Environ. 2019, 696, 133961.
- Al-Sakkaf, B.M.; Nasreen, S.; Ejaz, N. Degradation Pattern of Textile Effluent by Using Bio and Sono Chemical Reactor. Available online: https://www.hindawi.com/journals/jchem/2020/8965627/ (accessed on 10 February 2021).
- Nidheesh, P.V.; Rajan, R. Removal of Rhodamine B from a Water Medium Using Hydroxyl and Sulphate Radicals Generated by Iron Loaded Activated Carbon. RSC Adv. 2016, 6, 5330–5340.
- Javaid, R.; Qazi, U.Y. Catalytic Oxidation Process for the Degradation of Synthetic Dyes: An Overview. Int. J. Environ. Res. Public Health 2019, 16, 2066.
- Zhang, L.P.; Liu, Z.; Faraj, Y.; Zhao, Y.; Zhuang, R.; Xie, R.; Ju, X.J.; Wang, W.; Chu, L.Y. High-Flux Efficient Catalytic Membranes Incorporated with Iron-Based Fenton-like Catalysts for Degradation of Organic Pollutants. J. Membr. Sci. 2019, 573, 493–503.
- Lyu, L.; Han, M.; Cao, W.; Gao, Y.; Zeng, Q.; Yu, G.; Huang, X.; Hu, C. Efficient Fenton-like Process for Organic Pollutant Degradation on Cu-Doped Mesoporous Polyimide Nanocomposites. Environ. Sci. Nano 2019, 6, 798–808.
- Ameta, R.; Chohadia, A.K.; Jain, A.; Punjabi, P.B. Chapter 3—Fenton and Photo-Fenton Processes. In Advanced Oxidation Processes for Waste Water Treatment; Ameta, S.C., Ameta, R., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 49–87. ISBN 978-0-12-810499-6.
- Tarkwa, J.B.; Oturan, N.; Acayanka, E.; Laminsi, S.; Oturan, M.A. Photo-Fenton Oxidation of Orange G Azo Dye: Process Optimization and Mineralization Mechanism. Environ. Chem. Lett. 2019, 17, 473–479.
- Dotto, J.; Fagundes-Klen, M.R.; Veit, M.T.; Palácio, S.M.; Bergamasco, R. Performance of Different Coagulants in the Coagulation/Flocculation Process of Textile Wastewater. J. Clean. Prod. 2019, 208, 656–665.
- GilPavas, E.; Dobrosz-Gómez, I.; Gómez-García, M.Á. Coagulation-Flocculation Sequential with Fenton or Photo-Fenton Processes as an Alternative for the Industrial Textile Wastewater Treatment. J. Environ. Manag. 2017, 191, 189–197.
- Wang, J.; Chen, H.; Yuan, R.; Wang, F.; Ma, F.; Zhou, B. Intensified Degradation of Textile Wastewater Using a Novel Treatment of Hydrodynamic Cavitation with the Combination of Ozone. J. Environ. Chem. Eng. 2020, 8, 103959.
- Siddique, K.; Rizwan, M.; Shahid, M.J.; Ali, S.; Ahmad, R.; Rizvi, H. Textile Wastewater Treatment Options: A Critical Review. Enhancing Cleanup Environ. Pollut. 2017, 183–207.
- Su, Z.; Liu, T.; Yu, W.; Li, X.; Graham, N.J. Coagulation of Surface Water: Observations on the Significance of Biopolymers. Water Res. 2017, 126, 144–152.
- Miralles-Cuevas, S.; Oller, I.; Agüera, A.; Llorca, M.; Pérez, J.S.; Malato, S. Combination of Nanofiltration and Ozonation for the Remediation of Real Municipal Wastewater Effluents: Acute and Chronic Toxicity Assessment. J. Hazard. Mater. 2017, 323, 442–451.
- Yang, Y.C.; Zeng, S.S.; Ouyang, Y.; Sang, L.; Yang, S.Y.; Zhang, X.Q.; Huang, Y.Y.; Ye, J.; Xiao, M.T.; Zhang, N. An Intensified Ozonation System in a Tank Reactor with Foam Block Stirrer: Synthetic Textile Wastewater Treatment and Mass Transfer Modeling. Sep. Purif. Technol. 2021, 257, 117909.
- Powar, A.; Perwuelz, A.; Behary, N.; Hoang, L.V.; Aussenac, T.; Loghin, C.; Maier, S.S.; Guan, J.; Chen, G. Environmental Profile Study of Ozone Decolorization of Reactive Dyed Cotton Textiles by Utilizing Life Cycle Assessment. Sustainability 2021, 13, 1225.
- Kurade, M.B.; Waghmode, T.R.; Patil, S.M.; Jeon, B.H.; Govindwar, S.P. Monitoring the Gradual Biodegradation of Dyes in a Simulated Textile Effluent and Development of a Novel Triple Layered Fixed Bed Reactor Using a Bacterium-Yeast Consortium. Chem. Eng. J. 2017, 307, 1026–1036.
- Song, L.; Shao, Y.; Ning, S.; Tan, L. Performance of a Newly Isolated Salt-Tolerant Yeast Strain Pichia Occidentalis G1 for Degrading and Detoxifying Azo Dyes. Bioresour. Technol. 2017, 233, 21–29.
- Brahmbhatt, N.H.; Jasrai, R.T. The Role of Algae in Bioremediation of Textile Effluent. Methods 2016, 21, 28.
- Das, S.; Dash, H.R. Handbook of Metal-Microbe Interactions and Bioremediation; CRC Press: Boca Raton, FL, USA, 2017.
- Kaushik, P.; Malik, A. Mycoremediation of Synthetic Dyes: An Insight into the Mechanism, Process Optimization and Reactor Design. In Microbial Degradation of Synthetic Dyes in Wastewaters; Singh, S.N., Ed.; Environmental Science and Engineering; Springer International Publishing: Cham, Switzerland, 2015; pp. 1–25. ISBN 978-3-319-10942-8.
- Varjani, S.; Rakholiya, P.; Ng, H.Y.; You, S.; Teixeira, J.A. Microbial Degradation of Dyes: An Overview. Bioresour. Technol. 2020, 314, 123728.
- Rekik, H.; Zaraî Jaouadi, N.; Bouacem, K.; Zenati, B.; Kourdali, S.; Badis, A.; Annane, R.; Bouanane-Darenfed, A.; Bejar, S.; Jaouadi, B. Physical and Enzymatic Properties of a New Manganese Peroxidase from the White-Rot Fungus Trametes Pubescens Strain I8 for Lignin Biodegradation and Textile-Dyes Biodecolorization. Int. J. Biol. Macromol. 2019, 125, 514–525.
- Srinivasan, S.; Sadasivam, S.K.; Gunalan, S.; Shanmugam, G.; Kothandan, G. Application of Docking and Active Site Analysis for Enzyme Linked Biodegradation of Textile Dyes. Environ. Pollut. 2019, 248, 599–608.
- Giovanella, P.; Vieira, G.A.; Ramos Otero, I.V.; Pais Pellizzer, E.; de Jesus Fontes, B.; Sette, L.D. Metal and Organic Pollutants Bioremediation by Extremophile Microorganisms. J. Hazard. Mater. 2020, 382, 121024.
- Ji, D.; Xiao, C.; Zhao, J.; Chen, K.; Zhou, F.; Gao, Y.; Zhang, T.; Ling, H. Green Preparation of Polyvinylidene Fluoride Loose Nanofiltration Hollow Fiber Membranes with Multilayer Structure for Treating Textile Wastewater. Sci. Total Environ. 2021, 754, 141848.
- Khan, R.; Bhawana, P.; Fulekar, M.H. Microbial Decolorization and Degradation of Synthetic Dyes: A Review. Rev. Environ. Sci. Biotechnol. 2013, 12, 75–97.
- Maharani, V.; Vijayalakshmi, S.; Balasubramanian, T. Degradation and Detoxification of Reactive Azo Dyes by Native Bacterial Communities. Afr. J. Microbiol. Res. 2013, 7, 2274–2282.
- Abubakar, A.; Gaya, U. Box-Behnken-Optimized Cu (I) Photo-Fenton-like Degradation of Basic Violet 3. J. Mater. Environ. Sci. 2019, 10, 15–21.
- Ahmad, R.; Kumar, R. Conducting Polyaniline/Iron Oxide Composite: A Novel Adsorbent for the Removal of Amido Black 10B. J. Chem. Eng. Data 2010, 55, 3489–3493.
- Tanzifi, M.; Yaraki, M.T.; Kiadehi, A.D.; Hosseini, S.H.; Olazar, M.; Bharti, A.K.; Agarwal, S.; Gupta, V.K.; Kazemi, A. Adsorption of Amido Black 10B from Aqueous Solution Using Polyaniline/SiO2 Nanocomposite: Experimental Investigation and Artificial Neural Network Modeling. J. Colloid Interface Sci. 2018, 510, 246–261.
- Aragaw, T.A. Utilizations of Electro-Coagulated Sludge from Wastewater Treatment Plant Data as an Adsorbent for Direct Red 28 Dye Removal. Data Brief 2020, 28, 104848.





